b Key Laboratory of Biomass Chemical Engineering of Ministry of Education, College of Chemical and Biological Engineering, Zhejiang University, Hangzhou 310027, China;
c College of Chemistry and Chemical Engineering, Liaocheng University, Liaocheng 252059, China;
d Ningbo Research Institute, Zhejiang University, Ningbo 315100, China
In recent years, over-reliance on antibiotics tetracycline (TC) in our life and overused Cr(Ⅵ) complex in industrial production have caused devastating damage to the ecological environment [1, 2]. As reported [3], large amounts of antibiotics and heavy metals (Cr(Ⅵ)) were both detected in the farmland. If not handled properly, it will further threaten our health. Among kinds of methods to resolve these pollution problems, photocatalytic technology has attracted widespread attention of scientists on account of its advantages such as green, economy, stability, and sustainability, providing a wide prospect for dealing with environmental pollution matter [4].
Among numerous traditional photocatalysts (NiO [5], ZnO [6], BiOBr [7] and BiWO6 [8]), BiOBr was widely used and synthesized because of its proper band gap (2.81 eV) and high photostability [9]. The photoinduced electrons of BiOBr could reduce Cr(Ⅵ), and the holes could be used to oxidize organic contaminants [10]. Nevertheless, the photocatalytic ability of BiOBr monomer was restrained because the light utilization ratio and separation efficiency of photoinduced charge carriers were low. Scientists have conducted many pieces of researches to improve intrinsic defects of BiOBr and found that constructing heterojunctions in semiconductors was a serviceable means, which could prompt the mobility of charge carriers and boost overall photocatalytic activity [11-14].
Up to now, there are many reports about constituting heterojunction in BiOBr and other semiconductors, such as TiO2/BiOBr [15], BiOCl/BiOBr [16] and BiOI/BiOBr [17]. Compared with the above mentioned materials, g-C3N4 is more popular with researchers on the score of its easy synthesis, abundant raw materials, high chemical and thermal stability [18-20]. Above all, g-C3N4 could form the staggered heterojunction structure with BiOBr as a result of their different band position, thus boosting the separation of photoinduced carriers and the photocatalytic ability [21]. However, the construction of binary heterojunctions still has certain limitations for inhibiting the recombination of photogenerated carriers due to the low transport ability of electrons. Hence, the conception for constructing Z-scheme heterojunction structure by loading a mediator between the two semiconductors is further carried out. Especially, with GO as the carrier, its large specific surface and high conductivity can offer the channels for charges, which greatly suppress the happening of photo-induced charge carriers recombining [22-24]. In this study, g-C3N4 nanosheets were firstly obtained via the calcination, then GO was loaded between g-C3N4 nanosheets and flower-like BiOBr by one step hydrothermal synthesis (Scheme S1 in Supporting information) to fabricate g-C3N4/GO/BiOBr ternary Z-scheme heterojunction. Taking into account the applicability of catalysts, our work is not limited to the photodegradation of single or homogenous simple pollutants like other reports [25, 26]. The photocatalytic performance of g-C3N4/GO/BiOBr ternary hybrid was appraised by tetracycline (TC) photodegradation and Cr(Ⅵ) photoreduction. Meanwhile, it is worth noting that this work not only discusses the synergistic effect among g-C3N4, GO and BiOBr but also illuminates the impact of combined contamination systems on photocatalyst properties. The specific experimental datils were recorded in the experimental section of Supporting information. Furthermore, the photoreaction mechanism of g-C3N4/GO/BiOBr ternary complex is shed light on in detail.
The morphology of samples could be analyzed by scanning electron microscopy (SEM), transmission electron microscopy (TEM) and high resolution TEM (HRTEM) images. As depicted in Fig. 1a, it can be seen that flower-like BiOBr was successfully deposited at g-C3N4 nanosheets through the hydrothermal method. In Fig. 1b, the wrinkling on the surface of carbon nitride nanosheets was caused by the loading of GO, the GO existed between g-C3N4 nanosheets and flower-like BiOBr was benefit for the moving of photogenerated charge carriers [27, 28]. The TEM image (Fig. 1c) of 7%g-C3N4/10%GO/BiOBr also proved the position of GO was between g-C3N4 and BiOBr. The lattice spacing of 0.353 nm (Fig. S1a in Supporting information) was consistent with the crystallographic plane (101) of BiOBr crystal [29]. Meanwhile, the presence of C, N, O, Br and Bi elements was confirmed in the Energy Dispersive X-ray (EDX) spectrum (Fig. S1b in Supporting information). The element content of 7%g-C3N4/10%GO/BiOBr corresponding EDX spectrum was shown in Table S1 (Supporting information). The atomic fraction ratio (Table S1) of C and N was higher than that of pure g-C3N4, which demonstrated the extra carbon was from the addition of GO. Comparing the elemental mapping images of element O (Fig. 1d) with other elements (Figs. 1e–h), the distribution of certain O was consistent with C further verified the existence of GO. Besides, we used the FT-IR spectra to analyze the surface functional groups of materials, and the results were detailly explained in Fig. S2 (Supporting information). Thereinto, the characteristic peaks of GO at 1055 cm−1 (C-O stretching vibration) and 1732 cm−1 (C=O bond) [30] were present in 7%g-C3N4/10%GO/BiOBr composite, which also indicated the introduction of GO.

|
Download:
|
| Fig. 1. SEM images of 7%g-C3N4/BiOBr (a) and 7%g-C3N4/10%GO/BiOBr (b); TEM (c) and the corresponding elemental mappings (d-h) of 7%g-C3N4/10%GO/BiOBr. | |
The structure property of the as-prepared materials was studied by the nitrogen adsorption-desorption isotherm curve. From Fig. S3 (Supporting information), the nitrogen adsorption-desorption isotherm curve of samples belonged to type Ⅳ curves with H3 retention loops, reflecting the existence of pore structure [31]. Wherein, 7%g-C3N4/10%GO/BiOBr exhibited the highest absorption ability and the largest specific surface area, which could provide more active sites for the pollutants and be benefit for the photocatalytic reaction.
The crystal structures and element compositions of materials were characterized by X-ray diffraction (XRD) patterns (Fig. S4 in Supporting information). The peak at 10.6° was in according with the plane (001) of GO [32, 33]. For g-C3N4, the peaks value at 12.7° and 27.2° were designated to the (100) and (002) crystal planes, which was involved in the in-plane structure of tri-s-triazine units and interlayer stacking of its conjugated aromatic groups, respectively [34]. Whereas for BiOBr, the diffraction peaks at 10.9°, 21.9°, 25.2°, 31.7°, 32.3°, 39.4°, 46.2° and 57.1° were responded to the (001), (002), (101), (102), (110), (112), (200) and (212) crystal planes according to the card (JCPDS No. 09-0393) [35]. In the XRD patterns of composite, we could observe the characteristic peak resulted from the interlayer stacking of g-C3N4. However, the diffraction peak of GO could not be found in 7%g-C3N4/10%GO/BiOBr due to the low content [36]. Besides, there were no extra peaks observed in XRD patterns (Fig. S4), and one can conclude that 7%g-C3N4/10%GO/BiOBr has high purity [37].
X-ray photoelectron spectroscopy (XPS) measurements are often used as a means to analyze compositions and chemical states of as-prepared materials. The related elements (C, N, O, Br and Bi) of 7%g-C3N4/10%GO/BiOBr were all detected in the survey spectrum of XPS (Fig. S5a in Supporting information). As shown in Fig. S5b (Supporting information), the high-resolution C 1 s XPS spectrum was assigned to four peaks located at 284.6, 285.6, 286.6 and 288.6 eV, which was ascribed to sp2 C-C bonds, C-N bonding, C-O bonding, and C=O bonding, indicating the formation of GO and g-C3N4 [38-40]. There are three different peaks located at 399.0, 400.5 and 402.2 eV in the high-resolution N 1 s XPS spectrum (Fig. S5c in Supporting information), which were identified as the C=N-C, N-(C)3 and C-N-H, respectively [41, 42]. The high-resolution O 1 s XPS spectrum (Fig. S5d in Supporting information) was deconvoluted into several different peaks, in which the first peak at 530.2 eV was corresponded to the Bi-O bond, and other fitted peaks located at 531.6, 532.8 and 533.8 eV were designated to surface O-H, crystal water, and COOH, respectively [21, 43]. Furthermore, the two peaks (Fig. S5e in Supporting information) at the binding energies of 68.4 eV and 69.2 eV were classified as Br 3d5/2 and Br 3d3/2 [44]. Meanwhile, the XPS spectrum (Fig. S5f in Supporting information) of Bi 4f showed two peaks at 159.49 eV and 164.76 eV, corresponding to Bi 4f7/2 and Bi 4f5/2, which indicated that the element state of Bi was Bi3+ [45].
The UV–vis diffuse reflectance spectra were used to analyze the optical property of as-prepared samples. In Fig. S6a (Supporting information), the g-C3N4 exhibited the light absorption characteristic in both ultraviolet and visible light regions, while the absorption scope of BiOBr mainly focused in the ultraviolet region. After BiOBr coupling with g-C3N4, the absorption edge of the curve for g-C3N4/BiOBr showed a significant red shift as compared to BiOBr monomer. Moreover, it could be found that the ternary hybrid displayed a stronger light response than g-C3N4/BiOBr, indicating a synergy effect among GO, BiOBr and g-C3N4 [18]. This phenomenon demonstrated ternary complex could adsorb more light, thus improving the efficiency of the photocatalytic reaction. The diagram of band gap value can be obtained by plotting (αhν)1/2 on hv and extending the tangent to coordinate axis on band gap site [46]. The band gap (Fig. S6b in Supporting information) of g-C3N4, BiOBr, 7%g-C3N4/BiOBr, 7%g-C3N4/5%GO/BiOBr, 7%g-C3N4/10%GO/BiOBr, and 7%g-C3N4/15% GO/BiOBr were measured to be 2.7, 2.95, 2.84, 2.82, 2.76 and 2.74 eV, respectively.
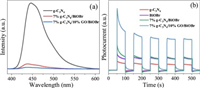
The Photoluminescence (PL) spectra were devoted to study the separation, transfer, and trapping capacity of photogenerated charge carriers in the as-synthesized samples [47]. Besides, the intensity of PL signal could reflect the recombination condition of photoinduced charge carriers [48]. All the samples were excited at 350 nm. As demonstrated in Fig. 2a, it is evident that the PL intensity of 7%g-C3N4/10%GO/BiOBr was the lowest among all investigated samples. Thus, one could conclude that the separation of photogenerated electrons and holes was effectively promoted in 7%g-C3N4/10%GO/BiOBr ternary composite. To further inquire the separation efficiency of samples, the photocurrent experiments and electrochemical impedance spectroscopy (EIS) of the as-prepared samples were performed. As clarified in Fig. 2b, all the samples maintained outstanding repeatability and stability after five intermittent irradiation cycles. And it can be broadly found that the 7%g-C3N4/10%GO/BiOBr displayed the highest photocurrent intensity than the g-C3N4, BiOBr and 7%g-C3N4/BiOBr, declaring separation efficiency of charge carriers was advanced. Similarly, the arc radius of Nyquist plot in 7%g-C3N4/10%GO/BiOBr (Fig. S7 in supporting information) was the smallest among all the samples, indicating the surface charge transferring was fastest [28].
|
Download:
|
| Fig. 2. The PL spectra (a) of g-C3N4, 7%g-C3N4/BiOBr, and 7%g-C3N4/10%GO/BiOBr and the photocurrent test (b) of g-C3N4, BiOBr, 7%g-C3N4/BiOBr and 7%g-C3N4/10%GO/BiOBr. | |
The photocatalytic performance of samples was estimated by photodegradation TC (10mg/L) solution under the visible light, and the consequence was shown in Fig. 3a. As seen in Fig. 3a, the change in concentration of pollution without photocatalyst could be negligible. However, 7%g-C3N4/BiOBr binary composite showed better photocatalytic degradation performance than the g-C3N4 and BiOBr, which indicated that the fabricated heterojunction in complex prompted the separation of photogenerated charge carriers. Interestingly, the photodegradation rate of TC by 7%g-C3N4/BiOBr was higher than the ternary complex in 60 min under the illumination, while the photodegradation effect was less durable. Oppositely, g-C3N4/GO/BiOBr ternary composite could continue to photodegrade after 60 min, and the photodegradation ability of 7%g-C3N4/10%GO/BiOBr for TC was the best. The above test results demonstrated the large specific area of composite could provide more reaction sites for the pollutant, and the GO could act as the channel of electrons to facilitate the separation of photoinduced carriers in the Z-scheme heterojunction structure [33], while the excessive GO nanosheets might have a negative effect on the performance of photocatalyst.

|
Download:
|
| Fig. 3. (a) Photodegradation of TC by g-C3N4, BiOBr, 7%g-C3N4/BiOBr and g-C3N4/GO/BiOBr series. (b) Simultaneous photocatalytic degradation of TC and reduction Cr(Ⅵ) by 7%g-C3N4/10%GO/BiOBr. | |
Given that the composition of wastewater is not unitary in most cases, antibiotics and heavy metals could exist together [3, 49]. The photocatalyst will have a broad prospect if it can simultaneously deal with multiple pollutants. The removal of Cr(Ⅵ) is different from photodegrading TC, which depend on the photoreduction ability of the catalyst [35]. The photocatalytic experimental results were shown in Fig. 3b. The blank experiments (without catalyst) indicated that the photodegradation of TC and reduction of Cr(Ⅵ) in the single pollution system or their combined pollution system could be neglected. Of note, the combined contamination system prompted the photoreduction of Cr(Ⅵ), but weakened the photodegradation of TC. Comparing the dark absorption curves of 7%g-C3N4/10%GO/BiOBr in the combined pollution system, it can be seen that the Cr(Ⅵ) absorption was much higher than the absorption of TC, thus the Cr(Ⅵ) may occupy more surface-active sites than TC. As we know, the reduction of Cr(Ⅵ) mainly depends on the photogenerated electrons. Although the Cr(Ⅵ) could acquire electrons alone in the single pollutant system, the recombination efficiency of photogenerated charge carriers in a single pollutant was much higher than in the compound system due to the lack of competition with electrons [50]. Therefore, the photoreduction efficiency of Cr(Ⅵ) was enhanced than before. Under the visible light, Cr(Ⅵ) was competed with O2 for electrons to change into Cr(Ⅲ), resulting in lessening the number of O2·−. As a result, the photodegradation ability of TC was weakened. Be that as it may, the photodegradation efficiency of TC could reach about 80%. By this token, the photocatalyst has an excellent ability to simultaneously degrade TC and reduce Cr(Ⅵ) under the visible light.
Furthermore, the recycling experiments of 7%g-C3N4/10%GO/BiOBr in the single pollution system of Cr(Ⅵ) and TC, as well as their combined contaminants were also carried out. (Figs. S8-S10 in Supporting information). After four cycling experiments, the photocatalytic ability of material showed a negligible decrease in the above pollutant system. Meanwhile, the sample also kept the initial morphology and crystal structure, which indicated the samples had high stability and reusability.
The active species experimental results of photodegrading TC by 7%g-C3N4/10%GO/BiOBr was shown in Fig. S11 (Supporting information). Thereinto, EDTA-2Na [51], isopropanol [52] and l-ascorbic acid [52], as well as potassium bromate [53] were used as trapping agents for h+, ·OH, O2·− and e−, respectively. The photodegradation rate of TC by 7%g-C3N4/10%GO/BiOBr was obviously suppressed after adding EDTA-2Na and l-ascorbic acid, while the addition of isopropanol and potassium bromate had a slender effect on the photodegradation ability. On the basics of the above experimental results, it could be concluded that the main active species are O2·− and h+.
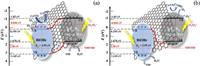
Generally, the values of valence band and conduction band were calculated based on the below equations: EVB = X – Ee + 0.5Eg (1), ECB = EVB − Eg (2), where the EVB represents the valence band, and the conduction band is expressed by ECB, and Ee is free electrons energy released to hydrogen scale (about 4.5 eV); The X values of g-C3N4 and BiOBr are 4.82 eV and 6.17 eV, which stands their electronegativity [54]; Eg is the band gap, and the Eg values of g-C3N4 and BiOBr are 2.70 eV and 2.95 eV according to the UV–vis diffuse reflectance spectra. The conduction band values of g-C3N4 and BiOBr were calculated to be -1.03 eV and 0.195 eV, respectively, and the valence band values of them were 1.67 eV and 3.145 eV. Given the above analysis, the traditional model (Fig. 4a) and Z-scheme heterojunction model (Fig. 4b) of g-C3N4/GO/BiOBr ternary complex were given. The function of GO was acted as a mediator to provide channels for photogenerated electrons or holes. According to the traditional scheme (Fig. 4a), the photoelectrons from g-C3N4 would transfer to the conduction band of BiOBr via GO. Meanwhile, the holes originated from BiOBr would move to the valence band of g-C3N4. Since the conduction band value of BiOBr was less negative than the −0.046 eV vs. NHE (O2 + e− = O2·−) [55], the valence band value of g-C3N4 was less positive than the 2.4 eV vs. NHE (standard redox potential of ·OH) [5]. O2·− cod not be obtained by the reaction, which was opposed to the results that O2·− was the major active species. Oppositely, the photogenerated electrons on conduction band of BiOBr would transfer to the valence band of g-C3N4 via GO based on Z-scheme heterojunction (Fig. 4b). The GO served as a mediator, which could effectively facilitate the separation of photogenerated charge carriers due to its satisfactory transmission ability. Furthermore, since the conduction band of g-C3N4 was more negative than the potential of E0 (O2/O2·−), the photogenerated electrons produced via g-C3N4 nanosheet would reduce Cr(Ⅵ) to Cr(Ⅲ), and the O2 would change into O2·−. Meanwhile, the ·OH produced by a small number of hole and O2·− would take part in the photodegradation of TC. In a word, the Z-scheme charge transfer way was the most feasible way, which could promote the photocatalytic performance through the effective separation of photogenerated charges and keep high oxidation and reduction ability [56].
|
Download:
|
| Fig. 4. The photodegradation mechanism of g-C3N4/GO/BiOBr: Traditional heterojunction (a) and Z-scheme heterojunction (b). | |
In conclusion, an innovative g-C3N4/GO/BiOBr photocatalyst was prepared by a simple, economical, and green method. Acting as a transport medium, the GO located between g-C3N4 nanosheets and flower-like BiOBr constructing g-C3N4/GO/BiOBr ternary Z-scheme heterojunction composite with satisfactory photocatalytic performance. According to the active species trapping experiments and the band-gap energy value, the transfer of photogenerated electrons and holes in g-C3N4/GO/BiOBr was accorded with the Z-scheme mechanism. The electrons from the conduction band of BiOBr moved to the valence band of g-C3N4 via GO. Furthermore, the mass ratio of g-C3N4 and GO as well as BiOBr also have an effect on the separation efficiency of photogenerated charge carriers and light absorption ability of photocatalyst. The optimal loadings amount of g-C3N4 and GO were determined to be 7% and 10%, respectively. The 7%g-C3N4/10%GO/BiOBr can not only deal with the single contaminant of TC or Cr(Ⅵ) but also has the ability to treat the mixed pollutants of TC and Cr(Ⅵ). Moreover, the g-C3N4/GO/BiOBr showed remarkable reusability and stability after four successive cycling experiments. Therefore, the developed ternary hybrid in this study holds great promise in removing harmful pollutants in the water.
Declaration of competing interestThe authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
AcknowledgmentsThis work was financially supported by the National Natural Science Foundation of China (Nos. 21667019, 22066017, and 22002057), the Key Project of Natural Science Foundation of Jiangxi Province (No. 20171ACB20016), the Jiangxi Province Major Academic and Technical Leaders Cultivating Object Program (No. 20172BCB22014), the Science and Technology Department of Jiangxi Province (Nos. 20181BCB18003 and 20181ACG70025), the Key Laboratory of Photochemical Conversion and Optoelectronic Materials, TIPC, CSA (No. PCOM201906), the Key Project of Science and Technology Research of the Jiangxi Provincial Department of Education (Nos. DA201602063 and GJJ191044), the Aviation Science Foundation of China (No. 2017ZF56020), and Fujian Key Laboratory of Measurement and Control System for of Shore Environment (No. S1-KF1703), the Doctor's Start-up Fund of Nanchang Hangkong University (No. EA201902286). Y. Hou is grateful to the National Natural Science Foundation of China (Nos. 51702284, 21878270 and 21922811), Zhejiang Provincial Natural Science Foundation of China (No. LR19B060002), the Fundamental Research Funds for the Central Universities, and the Startup Foundation for Hundred-Talent Program of Zhejiang University.
Appendix A. Supplementary dataSupplementary material related to this article can be found, in the online version, at doi:https://doi.org/10.1016/j.cclet.2020.12.010.
| [1] |
M.H. Khan, H. Bae, J.Y. Jung, J. Hazard. Mater. 181 (2010) 659-665. DOI:10.1016/j.jhazmat.2010.05.063 |
| [2] |
K. Li, Z. Huang, S. Zhu, et al., Appl. Catal. B: Environ. 243 (2019) 386-396. DOI:10.1016/j.apcatb.2018.10.052 |
| [3] |
D. Wei, B. Li, H. Huang, et al., Chemosphere 197 (2018) 165-180. DOI:10.1016/j.chemosphere.2017.12.193 |
| [4] |
X. Wang, K. Maeda, A. Thomas, et al., Nat. Mater. 8 (2009) 76-80. DOI:10.1038/nmat2317 |
| [5] |
F.A. Harraz, R.M. Mohamed, A. Shawky, I.A. Ibrahim, J. Alloys Compd. 508 (2010) 133-140. DOI:10.1016/j.jallcom.2010.08.027 |
| [6] |
M.J. Height, S.E. Pratsinis, O. Mekasuwandumrong, P. Praserthdam, Appl. Catal. B: Environ. 63 (2006) 305-312. DOI:10.1016/j.apcatb.2005.10.018 |
| [7] |
J. Zhang, F. Shi, J. Lin, et al., Chem. Mater. 20 (2008) 2937-2941. DOI:10.1021/cm7031898 |
| [8] |
Q. Zhao, L. Liu, S. Li, R. Liu, Appl. Surf. Sci. 465 (2019) 164-171. DOI:10.1016/j.apsusc.2018.09.168 |
| [9] |
M. Zhang, C. Lai, B. Li, et al., J. Catal. 369 (2019) 469-481. DOI:10.1016/j.jcat.2018.11.029 |
| [10] |
Q. Huang, Y. Liu, T. Cai, X. Xia, J. Photochem. Photobiol. A: Chem. 375 (2019) 201-208. DOI:10.1016/j.jphotochem.2019.02.026 |
| [11] |
L. Kong, Z. Jiang, H.H. Lai, et al., J. Catal. 293 (2012) 116-125. DOI:10.1016/j.jcat.2012.06.011 |
| [12] |
Y. Hou, F. Zuo, A. Dagg, P. Feng, Angew. Chem. Int. Ed. 52 (2013) 1248-1252. DOI:10.1002/anie.201207578 |
| [13] |
Y. Hou, F. Zuo, A. Dagg, P. Feng, Nano Lett. 12 (2012) 6464-6473. DOI:10.1021/nl303961c |
| [14] |
Y. Hong, Y. Jiang, C. Li, et al., Appl. Catal. B: Environ. 180 (2016) 663-673. DOI:10.1016/j.apcatb.2015.06.057 |
| [15] |
X. Wang, W. Yang, F. Li, et al., J. Hazard. Mater. 292 (2015) 126-136. DOI:10.1016/j.jhazmat.2015.03.030 |
| [16] |
X. Chang, M.A. Gondal, Al-Saadi A.A., et al., J. Colloid Interface Sci. 377 (2012) 291-298. DOI:10.1016/j.jcis.2012.03.021 |
| [17] |
J. Cao, B. Xu, B. Luo, H. Lin, S. Chen, Catal. Commun. 13 (2011) 63-68. DOI:10.1016/j.catcom.2011.06.019 |
| [18] |
Y. Zhou, J. Li, C. Liu, P. Huo, H. Wang, Appl. Surf. Sci. 458 (2018) 586-596. DOI:10.1016/j.apsusc.2018.07.121 |
| [19] |
J. Liu, Y. Liu, N. Liu, et al., Science 347 (2015) 970-974. DOI:10.1126/science.aaa3145 |
| [20] |
J. Ke, M. Adnan Younis, Y. Kong, et al., Nano-Micro Lett. 10 (2018) 69. DOI:10.1007/s40820-018-0222-4 |
| [21] |
L. Ye, J. Liu, Z. Jiang, et al., Appl. Catal. B: Environ. 142 (2013) 1-7. |
| [22] |
D. Fu, G. Han, Y. Chang, J. Dong, Mater. Chem. Phys. 132 (2012) 673-681. DOI:10.1016/j.matchemphys.2011.11.085 |
| [23] |
Y. Hou, Z. Wen, S. Cui, X. Feng, J. Chen, Nano Lett. 16 (2016) 2268-2277. DOI:10.1021/acs.nanolett.5b04496 |
| [24] |
Y. Hou, Z. Wen, S. Cui, X. Guo, J. Chen, Adv. Mater. 25 (2013) 6291-6297. DOI:10.1002/adma.201303116 |
| [25] |
X. Yu, P. Wu, C. Qi, et al., J. Alloys Compd. 729 (2017) 162-170. DOI:10.1016/j.jallcom.2017.09.175 |
| [26] |
Y. Bao, K. Chen, Appl. Surf. Sci. 437 (2018) 51-61. DOI:10.1016/j.apsusc.2017.12.075 |
| [27] |
J. Chen, B. Yao, C. Li, et al., Carbon N. Y. 64 (2013) 225-229. DOI:10.1016/j.carbon.2013.07.055 |
| [28] |
D. Xu, B. Cheng, S. Cao, et al., Appl. Catal. B: Environ. 164 (2015) 380-388. DOI:10.1016/j.apcatb.2014.09.051 |
| [29] |
J. Wu, Y. Xie, Y. Ling, et al., Chem. Eng. J. 400 (2020) 125944. DOI:10.1016/j.cej.2020.125944 |
| [30] |
M. Thommes, K. Kaneko, A.V. Neimark, et al., Pure Appl. Chem. 87 (2015) 1051-1069. DOI:10.1515/pac-2014-1117 |
| [31] |
J. Yan, Z. Song, X. Wang, et al., Appl. Surf. Sci. 466 (2019) 70-77. DOI:10.1016/j.apsusc.2018.09.234 |
| [32] |
S. Dong, J. Sun, Y. Li, C. Yu, Y. Li, Appl. Catal. B: Environ. 144 (2014) 386-393. DOI:10.1016/j.apcatb.2013.07.043 |
| [33] |
Y. Liu, W. Jin, Y. Zhao, G. Zhang, W. Zhang, Appl. Catal. B: Environ. 206 (2017) 642-652. DOI:10.1016/j.apcatb.2017.01.075 |
| [34] |
Y.P. Zhu, M. Li, Y.L. Liu, T.Z. Ren, Z.Y. Yuan, J. Phys. Chem. C 118 (2014) 10963-10971. DOI:10.1021/jp502677h |
| [35] |
M. Li, C. Song, Y. Wu, et al., Appl. Surf. Sci. 478 (2019) 744-753. DOI:10.1016/j.apsusc.2019.02.017 |
| [36] |
D.A. Giannakoudakis, J.A. Arcibar-Orozco, T.J. Bandosz, Appl. Catal. B: Environ. 183 (2016) 37-46. DOI:10.1016/j.apcatb.2015.10.014 |
| [37] |
J. Xia, J. Di, H. Li, et al., Appl. Catal. B: Environ. 181 (2016) 260-269. DOI:10.1016/j.apcatb.2015.07.035 |
| [38] |
W.-K. Jo, S. Kumar, M.A. Isaacs, et al., Appl. Catal. B: Environ. 201 (2017) 159-168. DOI:10.1016/j.apcatb.2016.08.022 |
| [39] |
Z. Zhang, S. Zhai, M. Wang, et al., J. Alloys Compd. 659 (2016) 101-111. DOI:10.1016/j.jallcom.2015.11.027 |
| [40] |
Y. Ren, J. Zhang, Y. Liu, et al., ACS Appl. Mater. Interfaces 4 (2012) 4776-4780. DOI:10.1021/am301131h |
| [41] |
J. Lv, K. Dai, J. Zhang, et al., Appl. Surf. Sci. 358 (2015) 377-384. DOI:10.1016/j.apsusc.2015.06.183 |
| [42] |
Y. Deng, L. Tang, C. Feng, et al., Appl. Catal. B: Environ. 235 (2018) 225-237. DOI:10.1016/j.apcatb.2018.04.075 |
| [43] |
S. Qu, Y. Xiong, J. Zhang, Y. Tian, Q. Zhang, J. Colloid InterfaceSci. 527 (2018) 78-86. DOI:10.1016/j.jcis.2018.05.038 |
| [44] |
C. Zhao, W. Li, Y. Liang, et al., Appl. Catal. A:Gen. 527 (2016) 127-136. DOI:10.1016/j.apcata.2016.09.005 |
| [45] |
J. Di, J. Xia, M. Ji, et al., Appl. Catal. B: Environ. 183 (2016) 254-262. DOI:10.1016/j.apcatb.2015.10.036 |
| [46] |
Q. Liu, J. Shen, X. Yu, et al., Appl. Catal. B: Environ. 248 (2019) 84-94. DOI:10.1016/j.apcatb.2019.02.020 |
| [47] |
W.-J. Ong, L.-L. Tan, S.-P. Chai, S.-T. Yong, Nano Energy 13 (2015) 757-770. DOI:10.1016/j.nanoen.2015.03.014 |
| [48] |
H.F. Li, A.Q. Ma, D. Zhang, Y.Q. Gao, Y.H. Dong, RSC Adv. 10 (2020) 4681-4689. DOI:10.1039/C9RA10146B |
| [49] |
H. Che, G. Che, P. Zhou, C. Liu, H. Dong, J. Colloid Interface Sci. 546 (2019) 262-275. DOI:10.1016/j.jcis.2019.03.080 |
| [50] |
W. Zhao, J. Li, B. Dai, et al., Chem. Eng. J. 369 (2019) 716-725. DOI:10.1016/j.cej.2019.03.115 |
| [51] |
H. Huang, X. Han, X. Li, ACS Appl. Mater. Interfaces 7 (2015) 482-492. DOI:10.1021/am5065409 |
| [52] |
J. Lv, K. Dai, J. Zhang, et al., Sep. Purif. Technol. 178 (2017) 6-17. DOI:10.1016/j.seppur.2017.01.019 |
| [53] |
A. Syoufian, K. Nakashima, J. Colloid Interface Sci. 317 (2008) 507-512. DOI:10.1016/j.jcis.2007.09.092 |
| [54] |
Z. Yang, J. Li, F. Cheng, Z. Chen, X. Dong, J. Alloys Compd. 634 (2015) 215-222. DOI:10.1016/j.jallcom.2015.02.103 |
| [55] |
Y. Yang, C. Zhang, D. Huang, et al., Appl. Catal. B: Environ. 245 (2019) 87-99. DOI:10.1016/j.apcatb.2018.12.049 |
| [56] |
F. Chen, Q. Yang, J. Sun, et al., ACS Appl. Mater. Interfaces 8 (2016) 32887-32329. DOI:10.1021/acsami.6b12278 |
 2021, Vol. 32
2021, Vol. 32 

