b Department of Integrated Traditional Chinese & Western Medicine, The Second Xiangya Hospital, Central South University, Changsha 410011, China;
c National Engineering Research Center for Advanced Polymer Processing Technology, Zhengzhou University, Zhengzhou 450002, China
Alzheimer's disease (AD) is recognized as a progressive and fatal neurodegenerative condition. This disease is characterized by progressive and comprehensive cognitive impairment, including early and prominent memory loss that can encapsulate cognition, language, orientation, reasoning, judgment, and other dysfunctions as well as different degrees of personality and behavioral disorders which in turn affect daily life, work, and social interactions [1, 2]. Previous research has highlighted two characteristic pathological features of AD, senile plaques (SPs) formed by β-amyloid(Aβ) deposition and neurofibrillary tangles (NFTs) in neurons that result from the over-phosphorylation of Tau protein alongside [1-3]. Epidemiological studies have revealed that the prevalence of AD increases with age, manifesting in between 5% and 10% of individuals over 65 years of age, and seen in between 20% and 50% in individuals over 85 years of age [4, 5]. The cost of treating AD is therefore rising rapidly with the average population age, as is the burden on society. Indeed, as the global population ages, AD has gradually evolved into a major problem that can no longer be ignored [6]. Although the prevention and treatment of this condition has attracted widespread attention from governments and scientific communities around the world, currently approved drugs for the clinical treatment of AD (e.g., donepezil, memantine, livacimine, and galantamine) can only alleviate symptoms in patients and are accompanied by side effects such as nausea and diarrhea [7-9]. Several factors also limit the development of anti-central nervous system-related drugs, including difficulties in crossing the blood-brain barrier (BBB), short systemic circulation length and low bioavailability [10]. Drugs and drug delivery systems (DDS) that can cross the BBB and treat AD effectively are therefore urgently needed [11].
BBB is a special barrier to protect the central nervous system (CNS). Theoretically, BBB can exclude more than 98% of small molecule drugs and almost 100% of macromolecular drugs, regulating the homeostasis of the nervous system by strictly controlling the blood-brain pathways of these molecules [9, 10]. It only selectively transports molecules that are vital to the brain [12, 13]. Therefore, drug delivery across the BBB remains a major challenge in the treatment of CNS diseases. We must fully understand the structural characteristics of BBB and the mode of cross-cell transport, and select the appropriate drug and DDS, so as to carry out targeted and systematic treatment for AD [14, 15]. In recent years, DDS based on nanomaterials are emerging.
Nanotechnology is a rapidly emerging research field for the development of medical applications. Nanomaterials have attracted widespread attention in particular due to their high drug loading, low or no systemic toxicity, physicochemical stability, ameliorative drug performance and permeability. The potential use of nanomaterials in medicine can be attributed to their comparable size as nucleic acids, proteins and antibodies. Their intrinsic biomimetic and quantum properties as well as ability to carry and absorb other materials and biodegradability mean that nanomaterials are amongst the most powerful tools in biomedical fields. Nanomedicine are therefore been regard as a promising approach to significantly improve and enhance drug delivery, medical imaging, tissue regeneration, and diagnostic speed as well as contribute to other medical therapies. The risks involved in using nanomaterials are also much lower than traditional methods for treating brain diseases [16, 17]. Numerous researches have reported different nanomedicine delivery systems and have illustrated their superior performance. In this review, we discussed the latest developments in nanomaterial-based strategies for AD treatment, including several common applications for the delivery of therapeutic compounds and scaffolds for cell delivery [18].
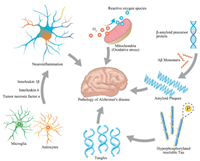
2. Overview of the pathogenesis of Alzheimer's diseaseThe underlying cause of AD remains unknown and many research directions remain ongoing regarding pathogenesis. It is certain that the pathogenesis of AD is complicated and involves multiple factors [19]; the main four hypotheses are reviewed in this section (Fig. 1).
|
Download:
|
| Fig. 1. Schematic representation of the pathology of AD. | |
The Aβ cascade hypothesis is one of the most widely accepted mainstream theory for AD pathogenesis and has dominated research for the past 30 years. One significant piece of evidence in support of this hypothesis is the excessive accumulation of Aβ peptides which eventually lead to the formation of extracellular SPs in the brain [2, 11, 19-21]. Aβ peptide is an amphiphilic polypeptide that comprises between 39 and 42 amino acids which has two main forms, Aβ1-40 and Aβ1-42. The first of these two is more common, but Aβ1-42 forms fibers faster and has strong neurotoxicity. Aggregates of Aβ1-42 are the main components of SPs and these peptides contain two hydrophobic residues, 16–20 and 30–40. It is also clear that Aβ1-42 peptides contain several charged amino acid residues (e.g., His, Glu, Asp, and Lys) and that hydrophobic and electrostatic interactions play key roles in their aggregation. We know that Aβ is a normal metabolite of the body, hydrolyzed from the β-amyloid precursor protein (APP). Thus, under normal physiological conditions, most APPs are cleaved by α-secretase and γ-secretase to produce soluble P3 fragments. At the same time, however, some APPs are cleaved into Aβ by β-secretase and γ-secretase in the cytoplasmic lysosome; production and degradation are in dynamic equilibrium under normal circumstances but when APP metabolism is abnormal, increased Aβ production and/or reduced degradation will lead to a large amount of Aβ deposition. As a result of the dynamic Aβ balance between fibrous, oligomeric, and monomeric forms, the oligomeric state of this peptide is the most toxic and so continuous deposition will cause structures to move more towards this form and have a toxic effect on neurons [3, 6].
2.2. Tau protein hyperphosphorylationThe over-phosphorylation of Tau protein is another possible neuropathological mechanism leading to AD [17]. Research has shown that Tau protein content in older patients with AD can be up to 300% higher than that in healthy counterparts; large proportions of these proteins aggregate in the brain tissue of AD patients [22]. Tau proteins are highly soluble and microtubule-associated; the main function of these compounds is to stabilize microtubules, especially axons, in phosphorylated form. The physiological effects of Tau phosphorylation include the regulation of microtubule dynamics, synapse growth and axonal transport. Thus, under normal physiological conditions, phosphorylation and dephosphorylation of Tau protein remain in dynamic equilibrium [23, 24], but in the case of AD, various factors interact and lead to hyperphosphorylation. Thus, hyperphosphorylated and insoluble Tau proteins tend to aggregate and assemble abnormally as paired spiral NFT filaments, and lose their ability to maintain normal morphologies and functions of microtubules, this process results in degenerative damage to neurons [25]. Hyperphosphorylated Tau proteins also abnormally occupy tubulin binding site, and make it difficult for driving proteins to return to the microtubule normally; this also eventually leads to the loss of microscopic function and irreversible damage to neurons [22, 26, 27].
2.3. Mitochondrial dysfunctionA large number of studies have shown that mitochondrial dysfunction plays an important role in the early occurrence of neurodegenerative diseases including AD. Mitochondria, organelles distributed in large numbers within neurons [28], have long been considered 'energy metabolism factories' that provide cells with adenosine triphosphate (ATP) to maintain energy balance via the oxidative phosphorylation pathway (OXPHOS) [29, 30]. In addition to participating in cell energy metabolism, mitochondria also play important physiological and pathological roles in regulating cell calcium and free radical homeostasis as well as apoptosis [31]. The basic characteristics of so-called mitochondrial dysfunction include the breakdown of mitochondrial oxidative respiratory chains, decreases in mitochondrial transmembrane potential (ΔΨ), extensive generation of reactive oxygen species (ROS), opening of the mitochondrial membrane permeability transition pore (MPTP) [32-34], and eventually cell oxidative stress. Mitochondrial dysfunction also causes the release of apoptotic factors such as cytochrome c, AIF, and Smac/DIABLO activate cysteine aspartate-specific protease (Caspase) also leading to apoptosis [35]. Research has shown that oxidative stress is widely involved in various pathophysiological processes including aging and the development of tumors. As the brain is rich in unsaturated fatty acids and is a high-oxygen-consuming organ, it is most susceptible to oxidative stress. Neuron cells, one type of hypermetabolic cells, maintain their basic functions depending on the energy provided by the mitochondrial respiratory chain and oxidative phosphorylation [36]. This means that when antioxidant capacity of a neuron decreases, defense functions decrease and physiological operation is difficult to maintain. Research has shown that excessive oxidative stress can lead to lipid peroxidation on nerve cell or organelle membranes, protein nitration, and the destruction of nucleic acids. These phenomena affect the synaptic abilities of nerve cells and can even lead to apoptosis [37-39].
2.4. NeuroinflammationThe Dutch scientists Eikelenboom and Stam [40] first reported in 1982 that numerous types of inflammation-related proteins are present in the brain of AD patients. These include complement proteins, immunoglobulins and acute-phase response proteins around Aβ plaques. This step forward meant that researchers began to realize that neuroinflammation might also play an important role in AD development. In subsequent work, McGeer et al. [41] revealed the presence of large numbers of activated microglia around the senile plaques of AD patients. These activated microglia release high proportions of proinflammatory factors including interleukin 1β (IL1β), interleukin 6 (IL-6) and tumor necrosis factor α (TNF-α) and further confirm that inflammation is one variable that cannot be ignored in AD pathological development [42, 43]. It is clear that the activation of microglia plays an important role in AD. A number of studies have also shown more recently that inflammation accompanies various stages of AD development; this response is interconnected with other mechanisms and jointly damages the nervous system, leading to AD development. Inflammation also plays an important role in AD pathogenesis, the essential factors are microglia, astrocytes, the complementary system and inflammatory factor responses. Microglial-mediated inflammation is therefore a significant pathological characteristics of AD patients' brains [44]. Microglia can induce the synthesis and secretion of pro-inflammatory factors once they are stimulated by Aβ, in other words, neuroinflammation can promote the deposition of Aβ, leading to neuronal loss and cognitive dysfunction [45-47].
2.5. Other AD pathogenesesA number of additional pathways are thought to also lead to AD, including insulin signaling pathway disorders [48, 49], cholinergic hypothesis [50, 51], autophagy dysfunction [52, 53], metal ion homeostasis imbalance [54] and other pathogeneses. None have, to date, been shown to be independent factors that cause AD and all seem to be related to one another. Although there has been significant progress in research on the etiology and pathogenesis of AD in recent years, the degeneration, apoptosis, and death of neurons, which are extremely complicated processes involving numerous interconnected factors, need to be studied further to understand the occurrence and development of AD.
3. Nanomaterials applied to target AD pathogenesisIn recent decades, nanomaterials have been widely utilized in many fields of disease treatment because of their unique characteristics with great value in biological applications. Various types of nanomaterials have been developed in succession, including inorganic nanoparticles (NPs), organic NPs, carbon nanotubes, quantum dots, micelles, liposomes, dendrimers and other types of nanomaterials (Fig. 2). The advantages of nanomaterials include good stability, good biocompatibility, good biodegradability, low cytotoxicity, targeting and controlled drug release [14]. These excellent properties make nanomaterials attractive candidates for drug delivery across the BBB.

|
Download:
|
| Fig. 2. Several typical nanomaterial models. Reproduced with permission [16]. Copyright 2018, Royal Society of Chemistry. | |
The pathological mechanism of AD and the physicochemical properties of BBB determine the targeting strategy of DDS. Drugs can enter the brain through nanocarriers via several different delivery systems, including carrier-mediated transport, chemical and active DDS, receptor-mediated transport and endocytosis, temporary interruption of the BBB and intranasal delivery. Drug-loaded nanoparticles can be covalently bonded, encapsulated, or adsorbed [9, 16]. Moreover, many properties of nanomaterials, such as particle size, composition, hydrophobicity, hydrophilicity, surface charge, and dissociation, can affect the transport capacity of BBB, providing a broad space for researchers to develop more promising nanomaterial-based BBB crossing strategies [14, 55, 56].
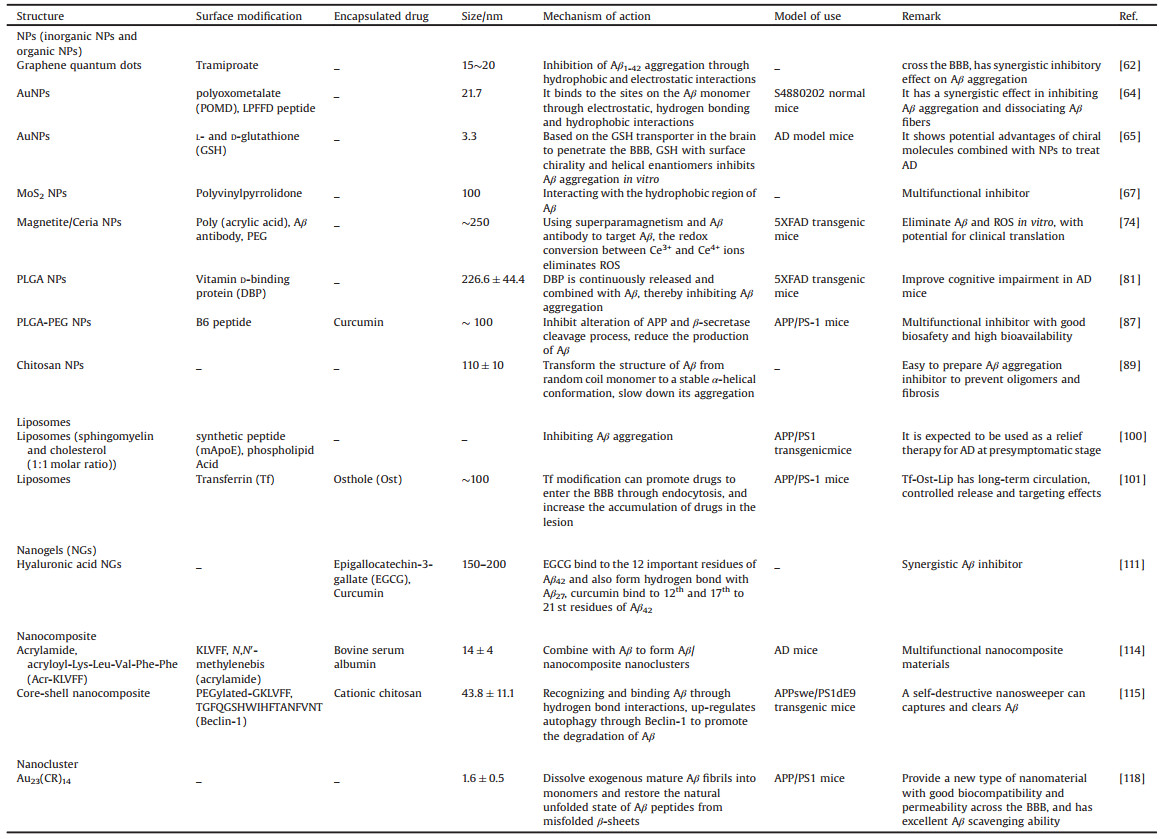
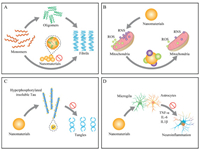
3.1. Nanotherapeutic approaches targeting the deposition of AβSince Aβ is associated with the mechanism of most cases of AD, and the aggregation of Aβ is the key pathogenic factor leading to AD, there has been extensive research on Aβ clearance. In particular, it is clear that soluble oligomers of Aβ (AβO) are strong neurotoxic to brain cells and so clearing Aβ aggregates, preventing or interfering with Aβ aggregation are the two main therapeutic directions for the treatment of AD [3] (Fig. 3A). Numerous Aβ aggregation inhibitors are considered as potential drug candidates. Traditional inhibitors mainly comprise peptides or peptide compounds as well as small organic molecules. The BBB permeability of these compounds, however, remains poor. Although both in vivo stability and inefficiency hinder the application of these compounds, nanomaterials nevertheless do have huge advantages when targeting Aβ aggregation [10, 19] (Table 1).
|
Download:
|
| Fig. 3. Nanotherapy for different pathogenesis of AD. (A) Aβ-based AD treatment strategy. (B) Tau-based AD treatment strategy. (C) AD treatment strategies based on mitochondrial oxidative stress. (D) AD treatment strategies based on neuroinflammation. | |
|
|
Table 1 Nanomaterials mainly for the Aβ hypothesis. |
A range of studies have been conducted on the effects of NPs on Aβ aggregation, most often gold, selenium, cerium and other inorganic forms as well as polymers. A combination of drugs and NPs can improve treatment effectiveness.
Inorganic NPs, without a doubt, bear the brunt. Numerous previous studies have reported inorganic NP carriers for drug delivery, imaging and therapeutic purposes, including gold and selenium NPs as well as carbon quantum dots, etc. Indeed, compared with organic NP carriers, inorganic forms have the advantages of good biocompatibility, stability, and hydrophilicity as well as low toxicity. A nanodepletor composed of ultra-large mesoporous silica and anti-Aβ single-chain variable fragments can target and eliminate aggregated Aβ monomers [57]. Similarly, Mahmoudi and co-workers reported that GO tablets can delay Aβ fibrillation by adsorbing amyloid monomers [58]. It is clear that carbon nanotubes inhibit the formation of a large number of oligomers in the amyloid peptides (16–22) of AD [59]; the interaction between NPs and Aβ is mainly based on electrostatic attraction and high surface ratio effects. Carbon-based NPs are widely used in AD research due to their non-toxicity, high biocompatibility, unique optical properties, and multi-functional surface [60].
In a previous study, Liu et al. [61] reported that graphene quantum dots (GQDs) can cross the BBB as a highly effective and low cytotoxic Aβ inhibitor. Studies have shown that the inhibitory effect of GQDs on Aβ aggregation is significantly related to surface charge; inhibitory effects increase as the surface charge decreases. It has also been suggested that hydrophobic and electrostatic interactions are two important factors that inhibit Aβ1-42 aggregation. Then, they combined two Aβ aggregation inhibitors, GQDs and tramiproate, to obtain GQD-T, which has a synergistic inhibitory effect on Aβ aggregation [62].
Due to its good biocompatibility, easy functionalization and potential ability to cross the BBB, Au NPs are widely used in nano-drug delivery systems [63]. Modify peptides and other substances on its surface to achieve the effect of treating AD. In a promising study, Gao and co-workers developed a multifunctional Aβ inhibitor based on AuNPs [64]. In this approach, a polyoxometalate with a Wells-Dawson structure (POMD) was coupled to a LPFFD peptide which was used as aβ-sheet blocker and then placed on the surface of AuNPs. The resultant AuNPs@POMD-pep exhibits the synergistic effect of inhibiting Aβ aggregation, dissociating Aβ fibers, and reducing Aβ-mediated peroxidase activity as well as Aβ-induced cytotoxicity in vitro. A recent study showed that gold NPs with surface modification of L-glutathione and D-glutathione (denoted as L3.3 and D3.3, respectively). Both chiral NPs can cross the BBB and inhibit the aggregation of Aβ42 without obvious toxicity. Compared with the enantiomer L3.3, D3.3 has stronger binding affinity to Aβ42 and higher brain biodistribution, and has a stronger inhibitory effect on Aβ42 fibrillation in AD mice [65]. Other inorganic NPs such as Se NPs [66] or MoS2 NPs [67] can also achieve a certain inhibitory effect on Aβ aggregation by surface functional modification.
Magnetic NPs (MNPs) are also widely used in the diagnosis and treatment of nanomedicine [68]. They can accurately accumulate in the ideal position under the condition of an external magnetic field, so they are very suitable for the targeted delivery of genes and drugs [69]. Among the magnetic materials, iron oxide is the most widely used magnetic material in biomedical applications due to its biodegradability, biocompatibility and superparamagnetic properties [68-71]. At present, many studies have reported iron oxide NPs as a new strategy for drug delivery in AD [72].
In one study, Fluorescent carboxyl magnetic Nile red particles (FMNPs) cross the BBB and enter the brain under the action of a functionalized magnetic field (FMF). Under the same FMF conditions, dextran-coated Fe3O4 magnetic NPs loaded with osmotin (OMNPs) were also transported to the brain of the mice treated with Aβ1–42. Compared with natural osmotin, OMNP can effectively attenuate synaptic deletion caused by Aβ1–42, Aβ accumulation, BACE-1 expression and Tau protein hyperphosphorylation. This magnetic drug delivery method is expected to make progress in preclinical and clinical applications [73]. Kim et al. [74] used multifunctional magnetite/cerium NPs (MCNAs) with a core/shell structure and combined with Aβ antibody to purify blood in vitro, that is, through the action of an external magnetic field to specifically capture Aβ peptides in the blood. The cerium NPs in the shell of MCNAs can eliminate ROS generated by the body's immune response through the redox conversion between Ce3+ and Ce4+ ions, so as to reduce oxidative stress and prevent inflammation. This in vitro blood Aβ clearance strategy does not require evaluation of in vivo activity and has a certain potential for clinical translation.
Although inorganic NPs have many excellent properties that can provide ideas and space for the development of DDS, the clinical application of inorganic nanomaterials still cannot achieve breakthroughs because of their clearance routes and long-term potential toxicity.
Meanwhile, polymer NPs have become a major research hotspot due to their advantages of high biocompatibility, small volume, high drug loading, good water solubility, low toxicity, biodegradability and easy modification [75]. For example, poly(lactic-co-glycolic acid) (PLGA), polyethylene glycol (PEG), polycaprolactone (PCL), chitosan, hyaluronic acid, and dendrimers have also been applied to anti-AD drug development [76, 77].
PLGA is a biodegradable and biocompatible material approved by the Food and Drug Administration (FDA). The half-life and release rate of the PLGA loaded material can be adjusted by the ratio of monomers or functional groups that make up the end of the PLGA [78]. Therefore, PLGA NPs are widely used in the fields of diagnosis, imaging and drug delivery [79, 80].
Jeon [81] and his team used PLGA as a carrier to load vitamin D-binding protein (DBP) into PLGA NPs and obtained DBP-PLGA NPs with an average particle size of 226.6±44.4 nm. Because 5XFAD mice destroy BBB during the onset of AD, intravenously injected DBP-PLGA NPs can enter the brain through the disrupted BBB, and DEP is continuously released and combined with Aβ, thereby inhibiting Aβ accumulation in the peripheral and CNS. Further studies using NPs tracers are warranted to determine the pharmacokinetics of NPs. Phytol-coated PLGA NPs have also been developed to regulate Aβ aggregation in AD [82]. These NPs can prevent the accumulation of Aβ, and exhibit anti-cholinesterase and antioxidant properties but exhibit no cytotoxicity in nerve 2a cells. Data from in vivo experiments suggest phytol-PLGA NPs can penetrate the BBB.
Curcumin (Cur) exhibits a number of functions including anti-oxidation, anti-inflammatory, anti-tumor, anti-virus, and anti-bacterial effects [83]. Numerous studies have shown that Cur can reduce the production and aggregation of Aβ, Tau hyperphosphorylation and the formation of neurofibrillary tangles [84]. Combining Cur with a nanometer DDS can overcome the limitations of its low bioavailability and enhance its applicability. In earlier work, Doggui et al. [85] reported that encapsulation of Cur in PLGA NPs can increase bioavailability; compared with free Cur, Cur-containing PLGA NPs exhibit enhanced uptake capacity in SHSY5Y cells and were, therefore, able to cross the BBB. Research performed by Khalil and co-workers revealed that PLGA NPs and PLGA-PEG NPs were 15.6 times and 55.4 times more bioavailable than Cur aqueous suspension, respectively [86]. Similarly, Fan and co-workers reported that Cur-loaded PLGA-PEG NPs bind to B6 peptide (PLGA-PEG-B6/Cur) [87]. Results reveal that PLGA-PEG can be used to increase bioavailability while the B6 peptide targets the TFR highly expressed in brain capillary endothelial cells and neurons to increase the permeability of BBB. The NPs can increase cell uptake, and have good blood compatibility. Experiments both in vitro and in vivo show that PLGA-PEG-B6/Cur can reduce the formation and deposition of Aβ in the hippocampus and can also inhibit excessive Tau phosphorylation.
Chitosan is an easily surface modified cationic polymer extracted from chitin. Developing these NPs is of great significance in terms of crossing the BBB and treating AD [88]. Indeed, Jha A et al. [89] reported the dual effects of two chitosan-based naked chitosan NPs and chitosan-PLGA NPs on the inhibition of Aβ1-42 peptides and the decomposition of pre-formed soluble and the inhibition of insoluble aggregates. In another research, Jiang and colleagues systematically evaluated the self-assembly of a series of chitosan-hyaluronic acid (CH) NPs bearing different surface charges [90]. Both positive and negative CH NPs inhibit the Aβ aggregation and this inhibitory effect increased in concert with surface charge density. Studies across different pH values have further confirmed the importance of electrostatic interactions in Aβ aggregation and show that the key role of CH NPs is due to the change in Aβ charge properties in concert with pH. This research provides new insights into surface charge effects on Aβ aggregation.
Lys-Leu-Val-Phe-Phe (KLVFF) is a peptide sequence of Aβ fragment, which is considered to be the main driving factor of Aβ fibrosis. This fragment has been shown to be a key fragment that monitors Aβ aggregates and co-assembles through strong hydrogen bonds to inhibit its fibrosis [91], many studies have used this peptide sequence in nanomaterials to achieve the purpose of treating AD [92, 93]. In earlier work, Kim and co-workers reported that fullerenes can specifically interact with KLVFF, the central hydrophobic motif of Aβ, and therefore inhibit Aβ aggregation at early stages [94]. Recently, Liu et al. [95] reported two KLVFF and FMOC-KLVFF self-assembled fluorescent NPs named KNP and FKNP, which can detect and distinguish different concentrations and forms of Aβ42, and bind to Aβ to inhibit its aggregation. These two NPs have potential applications for the diagnosis, monitoring disease progression and prediction of AD outcome.
3.1.2. LiposomesIt is clear that liposomes possess a number of advantages including amphiphilicity, biodegradability, and enhanced bioavailability. They are also broadly adaptability for encapsulating drugs. In terms of drug dissolution characteristics, molecules are captured inside the liposome, inserted into the lipid bilayer or adsorbed directly onto the surface; this approach can improve drug stability and facilitate slow release. Modified liposomes have targeted specificity and can become potential multifunctional AD nano-platforms [96-99].
Mancini et al. [100] designed a bifunctional liposome (mApoE-PA-LIP), which consists of a synthetic peptide (mApoE) containing the apolipoprotein-E receptor binding domain and a phospholipid acid (PA) that can bind to Aβ, used to target the BBB and inhibit Aβ aggregation. In another study, Kong et al. [101] wrapped osthole (Ost) in a transferrin (Tf)-modified liposome to obtain Tf-Ost-Lip. Among them, Tf mediates the transport of Ost-Lip through the blood-brain barrier through TfRs on the surface of cerebral capillary endothelial cells, thus increasing the accumulation of Ost in the brain. The average particle size of the liposome was about 100 nm, the encapsulation rate was over 90%, and the drug load was 3.71% ± 0.038%. Studies in vivo and in vitro have shown that Tf-Ost-Lip can protect APP-SH-SY5Y cells from neurotoxicity, reduce the deposition of Aβ, and inhibit the apoptosis, oxidative stress and neuroinflammation of APP/PS-1 mice. Furthermore, studies have shown that liposomes modified by Cur or Cur derivatives have high Aβ affinity, and have a certain inhibitory effect on Aβ aggregation and induced cytotoxicity [97, 102, 103].
3.1.3. Nanogels (NGs)Earlier work has shown that NGs are hydrogel systems composed of natural or synthetic cross-linked polymers. They possess enhanced water absorption properties as well as controlled release, targeting, biodegradability, biocompatibility, drug loading capacity and permeability [104, 105]. As a result of particle sizes and surface characteristics, active and passive drug release can be achieved using NGs; their high loading capacity means that targeted or continuous drug release can be achieved and so NGs have broad prospects for biomedical applications [106-108].
In one key piece of recent work, Ikeda and colleagues reported on a biocompatible NG that comprises a polysaccharide amylopectin backbone with a hydrophobic cholesterol moiety (CHP) [109]. These CHP NGs can be combined with Aβ; this approach changes the conformation of Aβ from a random coil to a stable α-helical structure, and inhibits the aggregation of Aβ1-42. Indeed, NGs composed of amino-modified CHP (CHPNH2) are positively charged under physiological conditions and have an enhanced inhibitory effect when compared to CHP NGs. This result indicates that electrostatic interactions between CHPNH2 and Aβ are very important for inhibiting fibril formation.
Results presented by Jiang et al. [110] show that self-assembled Cur-HA NGs can inhibit Aβ aggregation and cytotoxicity. Based on this, they designed and prepared a synergistic inhibitor HA NGs with the double modification of EGCG and Cur (CEHA). These two Aβ inhibitors have synergistic effects when presented in NGs; their differences in hydrophobicity and appropriate SD provide a good nanostructure for Aβ penetration and separation with respect to one another. The results showed that the inhibition rate of CEHA was 69% and 55% higher than that of EHA and CHA, respectively. Cytotoxicity test showed that SH-SY5Y cells incubated with Aβ and CEHA had 28% higher inhibition rate than the mixture of Aβ and HA. This dual inhibitor is expected to be used to develop effective agents against Aβ aggregation and cytotoxicity [111].
3.1.4. Multifunctional nanocompositesA key study reported results relating to a GO nanocomposite; when it was combined with AuNPs prepared by pulsed laser ablation (PLA), it turned out to play a key role in regulating Aβ aggregation and inhibiting cytotoxicity applications [112]. In other work, Ahmad and co-workers reported a graphene oxide-iron oxide (GOIO) nanocomposite and demonstrated its ability to regulate Aβ aggregation [113].
A peptide-polymer nanomaterial is another new kind of functional material which exhibits good biodegradability, biocompatibility, and low toxicity. Once this polymer material has been modified by targeting polypeptide residues with different biological effects, a single material can effectively achieve multiple biological functions and thereby improves drug efficacy. Zhao [114] and co-workers developed a spherical nanocomposite material (NC-KLVFF) with an average diameter of 14±4 nm. It was prepared by encapsulating bovine serum albumin (BSA) on a polymer layer containing cross-linked KLVFF synthesized by in situ polymerization. Results show that NC-KLVFF can combine with Aβ monomers to form Aβ/NC-KLVFF nanoclusters, thereby preventing Aβ from forming toxic oligomers and fibers, reducing Aβ-induced neuronal damage, inhibiting inflammation, and restore the ability of intracranial microglia to engulf Aβ. This nanomaterial provides a feasible method for the treatment of AD.
A new research result by Luo et al. [115] also used KLVFF peptides that can bind to Aβ. They designed a self-destructive nanosweeper with a core-shell structure, that is, modified KLVFF and Beclin-1 (TGFQGSHWIHFTANFVNT) on a cationic chitosan and then self-assembles into nanospheres. This nanosweeper recognizes and binds Aβ through hydrogen bond interactions, as well as up-regulates autophagy through Beclin-1 to promote the degradation of Aβ, thereby achieving the effect of synergistic treatment of AD.
Stimulus-responsive nanomaterials have also received much attention in biomedical applications. They respond to specific functions by receiving stimulus signals from the external environment, and have unique advantages in the diagnosis and treatment of AD [116]. In a recent study, Zhang's group designed and prepared a multifunctional nanoparticle system by integrating high Aβ binding affinity, stimulus-responsive drug release, and photothermal degradation characteristics [117]. In the system, the near-infrared absorbing conjugated polymer PDPP3T-O14 is used as the photothermographic core, and the near-infrared response stimulation of the outer layer is based on the thermal response polymer 1, 2-dipalmitoyl-sn-glycerol-3-phosphocholine (DPPC), the innermost is wrapped Cur, 5-poly β-fragmentation peptide Leu-Pro-Phe-Phe-Asp (LPFFD) is further connected to the surface of PEG lipid shell to realize the Aβ targeting. As expected, this nanocomposite has good Aβ targeting and significant photothermal dissociation effect, and has the ability of Aβ aggregation-dependent fluorescence detection. Under near-infrared laser irradiation, Cur was effectively released from the NPs effecting the decomposition of Aβ.
3.1.5. NanoclustersIn recent work, Sun and colleagues reported a gold nanocluster Au23(CR)14 containing a cysteine-arginine (CR) dipeptide as a ligand [118]. Experiments both in vivo and in vitro confirmed that Au23(CR)14 has the ability to dissolve Aβ fibers and reduce them to non-toxic unfolded monomers. This result not only expands the application of gold nanomaterials into biomedicine, but also provides a new path for nanomaterial-based drug development for AD diagnosis and treatment.
Obviously, many Aβ-targeting nanomaterials are also involved in the treatment of other AD pathological pathways, that is, have multiple therapeutic effects, which mainly due to the close connection between Aβ and other AD pathological processes.
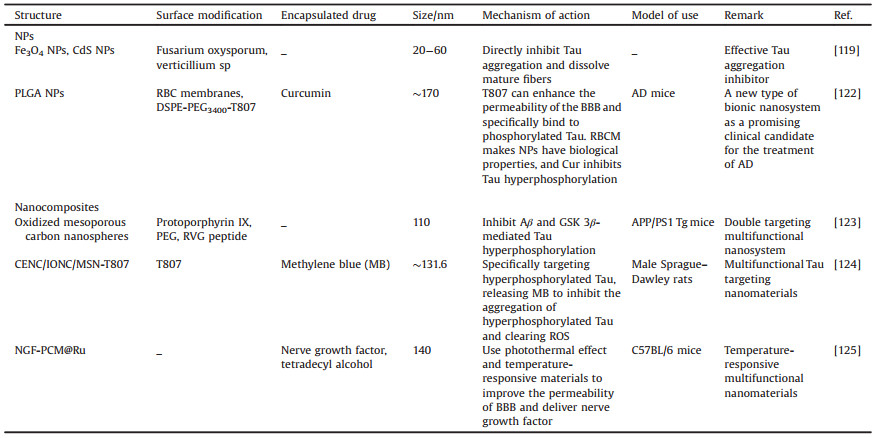
3.2. Nanotherapeutic approaches targeting the hyperphosphorylation of TauTau proteins are the most abundant microtubule-related proteins in the human body, particularly common in the peripheral and CNS (Fig. 3B). Research has shown that the pathological development of Tau proteins is closely related to the degree of AD cognitive impairment and thus provides a potential therapeutic target (Table 2).
|
|
Table 2 Nanomaterials mainly for Tau hypothesis. |
In an earlier study, Shweta [119] and co-workers reported on two kinds of protein-coated (PC) metal NPs (PC-Fe3O4 and PC-CdS) that have the ability to inhibit Tau aggregation in vitro. It was proved that these PC metal NPs, especially PC-Fe3O4 NPs, exhibited little affect the viability of neuroblastoma cells. In addition, PC-CdS NPs show dual properties of Tau inhibition and depolymerization. In the same year, Behafarid et al. [120] prepared folic acid-functionalized gold NPs (FA-Au NPs) and gold-Fe3O4 core-shell NPs (Au-Fe NPs), which showed binding affinity to tubulin and Tau.
As inorganic NPs still have problems such as difficulty in biodegradation in vivo and adverse reactions. In addition, Cur is being tested as a Tau targeting agent in phase II clinical trials [121]. Based on this, Gao [122] and his team developed Cur-loaded biomimetic PLGA NPs. The surface of the NPs is coated with a red blood cell membrane (RBCM), and the surface of the membrane is connected with T807 molecules. The obtained T807/RPCNP NPs have good biocompatibility and low toxicity, long-term in vivo circulation, can cross the BBB and locate p-tau in neurons, reduce intracellular p-tau levels, inhibit neuronal cell death, effectively eliminate ROS, and ameliorate AD progression in vivo and in vitro. The development of the biomimetic nanosystem demonstrates a novel targeted therapeutic strategy, which has the potential to become a clinical candidate system.
3.2.2. NanocompositesIn a noteworthy study, Xu and colleagues evaluated the use of protoporphyrin IX (PX) modified oxidized mesoporous carbon nanospheres (OMCN) [123]. In this context, PX@OMCN@PEG(OP)@RVGs comprises a novel type of multi-targeted AD multifunctional nano-grating that can effectively inhibit Tau phosphorylation. The application of focused ultrasound to PX also causes the generation of ROS and significantly inhibits the accumulation of Aβ. These methods both significantly improved the cognitive level of APP/PS1 transgenic mice and achieved dual-target AD inhibition. In addition, via the modification of the RVG peptide, PX was shown to be both safe and effective for in vivo and in vitro transmission through the BBB. The enhanced photothermal effects of NPs improve the BBB permeability of PX@OP@RVGs under near-infrared irradiation. These results show that this new type of PX@OP@RVG multifunctional nanodrug has dual-targeted therapeutic ability against AD.
An additional recent study reported on a novel MB-containing mesoporous silicon-mediated nanocomposite CeNC/IONC/MSN-T807 which uses okadaic acid (OA) as a tool to induce Tau protein phosphorylation at the cell and animal level [124]. The aim of this approach is to achieve the integration of targeted tau protein diagnosis and treatment of AD. In this context, in vitro experiments have confirmed that this nanocomposite combined with CeNC and MB can effectively inhibit oxidative stress in mitochondria, target highly phosphorylated Tau proteins, and inhibit the aggregation of phosphorylated forms; Animal experiments have also confirmed that this nanocomposite can protect neurons by relieving inflammation and thus improve AD symptoms. This nanocomposite therefore provides a new direction for exploration and research ideas for the Tau-targeted treatment of AD.
Liu et al. [125] reported a nanocomposite (NGF-PCM@Ru NPs) with high stability and good biocompatibility, which use flower-shaped hollow nano-ruthenium (Ru NPs) as a carrier, loaded with nerve growth factor (NGF) and temperature-responsive material tetradecyl alcohol (PCM). The nanocomposite has a good photothermal effect, under near-infrared (NIR) irradiation, it can effectively penetrate BBB, respond to phase changes in the injured area, release NGF, inhibit Tau hyperphosphorylation, and reduce oxidative stress restores nerve damage, thereby significantly improving the learning and memory ability of AD mice.
3.3. Nanotherapeutic approaches targeting mitochondrial oxidative stressMitochondrial dysfunction results in abnormal ROS production and causes oxidative stress damage. This oxidative stress response causes cell damage by directly oxidizing cell membranes, proteins, and nucleic acids, and well as inducing apoptosis. This stimulates strong neuroinflammation and leads to AD development (Fig. 3C). A large number of low-molecular-weight compounds and monomer components from traditional Chinese medicine have been shown to exert significant mitochondrial protective effects against nerve cell oxidative stress damage, including coenzyme Q10 (CoQ10), Cur, quercetin, and Ginkgo biloba extract. These components play a role in resisting AD physiological processes and should be further investigated using basic and subclinical research [36, 126, 127]. In-depth further research on the regulatory mechanisms of oxidative stress in AD as well as the development of drugs with specific targeting will provide more effective and broader approaches for clinical treatment.
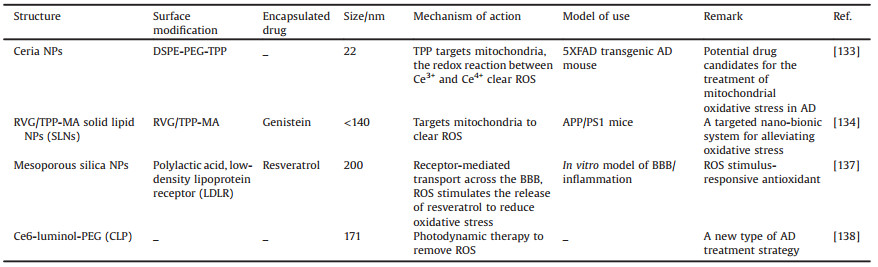
3.3.1. NPsCombining mitochondrial targeting ligands with drug-loaded nanomaterials is an effective way to achieve mitochondrial-targeted drug delivery (Table 3). Commonly used mitochondrial targeting ligands include triphenylphosphine (TPP), desquamium chloride (DQA), F16, guanidinium salts, rhodamine 19, rhodamine 123, and mitochondrial penetration peptides (MPPs). The bulk of these approaches comprise delocalized lipophiliccations (DLCs). The lipid solubility of these ligands allows them to pass through cell and mitochondrial membranes, while their positive charge is beneficial to overcome the mitochondrial membrane potential and enter the mitochondrial matrix, therefore providing a targeting effect [128]. As the mitochondria of cancer and other transformed cells have higher transmembrane potentials than normal ones, delocalized lipophilic cations can preferentially accumulate in cancer cell mitochondria. The advantage of directly binding a targeting ligand to a drug is that it enables targeted and more precise delivery to the mitochondria. These compounds generally have a problem, however, due to their poor water solubility, while some other mitochondrial targeting ligands are cytotoxic. The direct binding of mitochondrial targeting ligands to drugs may also affect chemical activity [129], so combining them with nanomaterials can mitigate some shortcomings such as poor solubility, low biocompatibility, and cytotoxicity [130].
|
|
Table 3 NPs mainly for oxidative stress. |
Triphenylphosphine ion (TPP) derivatives are amongst the most widely studied mitochondrial targeting molecules [131, 132]. A study has discussed a mitochondrial-targeted TPP-ceria NPs and the surface of ceria NPs is modified by the conjugate of 3-CTPP and DSPE-PEG-NH2 (DSPE-PEG-TPP) [133]. In this example, the carboxyl group was obtained by combining it with the amine group in DSPE-PEG-NH2. Ceria NPs therefore also have great potential in the areas of nerve and cardio protection, wound healing, chronic inflammation, cancer and eye diseases. In particular, smaller ceria NPs (less than 5 nm) exhibit enhanced treatment efficiency because of their larger surface-to-volume ratio. Indeed, TPP is a lipophilic cation that can target mitochondria using negative mitochondrial membrane potential; this means that TPP-ceria NPs can reduce the reactive glial cell and mitochondrial morphological damage seen in mouse models. Data indicate that TPP-ceria NPs also provide a potential treatment for mitochondrial oxidative stress injuries. In a recent study, Han et al. [134] linked rabies virus glycoprotein (RVG 29) and TPP to the surface of macrophage (MA) membranes to prepare Genistein (GS) encapsulated and membrane-coated solid lipid NPs (RVG/TPP-MASLNs-GS). MA membranes can camouflage the NPs, improve the escape ability of the reticuloendothelial system (RES) and extend the circulation of the nanosystem. RVG 29 gives it the ability to cross the BBB and selective targeting of neurons. After entering the central nervous system neurons, TPP further guides DDS to mitochondria and releases GS to eliminate ROS. Experimental data shows that the designed biomimetic nanosystem delays the progression of AD through neuronal mitochondrial targeted delivery and it is a promising treatment option.
ROS-responsive DDS have become a research hotspot in the treatment of oxidative stress injury diseases including AD, and they have the potential to improve the therapeutic effect of drugs on excessive ROS. Generally, these nanocarriers contain polymers functionalized with chemical groups that react with ROS. Since these nanocarriers can release drugs in response to abnormally increased levels of ROS, ROS-responsive nanocarriers can potentially minimize the side effects of drugs on the whole body [135, 136]. The ROS responsive smart DDS has been proven to have the specificity and accuracy of targeted drug release therapy. Shen et al. [137] reported a polylactic acid (PLA)-coated mesoporous silica NPs (MSNPs) encapsulating resveratrol (RSV), which is functionalized with low-density lipoprotein receptor (LDLR). This delivery system can actively cross the BBB through receptor-mediated intracellular transport and release antioxidants RSV under ROS stimulation to treat AD caused by oxidative stress. One recent study reported that a NP self-assembled from an amphiphilic polymer with a luminescent donor (lumino) and a fluorescent acceptor [chlorinated olefin 6 (Ce6)] can be used for luminescence imaging as well as deep tissue power therapy photoluminescence [138]. Myeloperoxidase and ROS produced in an inflammatory site or tumor microenvironment can trigger the biofluorescence resonance energy transfer of NPs and singlet oxygen (1O2) production. This approach also makes cancer treatment possible. These self-illuminated NPs have good in vivo imaging capabilities as well as suitable tissue penetration and resolution in a variety of animal inflammation models.
3.4. Nanotherapeutic approaches targeting neuroinflammationMicroglia and astrocytes are known to perform a dual role in AD development (Fig. 3D). On the one hand, microglia can reduce AD cognitive impairment by removing excess β-amyloid from the brain. At the same time, excessive microglial cell activation increases the release of pro-inflammatory factors such as TNF-α and IL-1β which promote AD development [139, 140]. The use of the microglia inflammatory release pathway as a drug research target and developing therapies that selectively cut off the release of pro-inflammatory factors from microglia presents one way to reduce AD brain neuroinflammation [47, 139, 141].
3.4.1. NPsGold NPs have been studied in detail due to their ability to inhibit NF-κB expression and subsequent inflammation. A number of studies have shown that gold NPs blocked NF-κB activation by interacting with cys-179 of IKK-β and inhibiting the production of pro-inflammatory cytokines such as TNF-α and IL-1β [142, 143]. Gold NPs have also been shown to have anti-inflammatory and antioxidant effects in skin and muscle injury models [144].
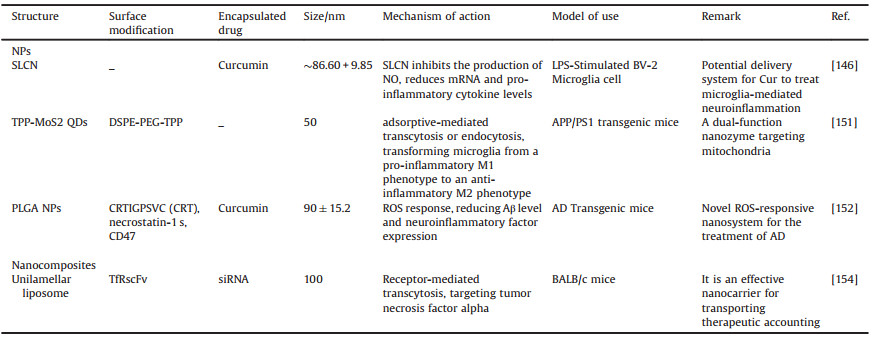
There is a plethora of research on the encapsulation of natural active small molecules with anti-inflammatory effects into DDS (Table 4). For instance, Gabriella and co-workers showed that loading quercetin into β-cyclodextrin-dodecyl carbonate NPs leads to a significantly stronger anti-inflammatory effect than the use of free quercetin [145]. In another work, Cur-loaded solid lipid NPs (SLCN) were effectively developed into NPs at a size of approximately 86 nm without inducing any toxicity in endothelial cells. The effect of Cur-loaded solid lipid NPs (SLCN) on BV-2 microglia when subject to LPS-induced neuro-inflammation was also investigated; results showed that, compared with traditional Cur, SLCN exerts a stronger inhibitory effect on nitric oxide (NO) production and it is also dose-dependent. In contrast to free Cur, mRNA and pro-inflammatory cytokine levels also decreased in a dose-dependent manner [146].
|
|
Table 4 Nanomaterials mainly for neuroinflammation. |
Studies have shown that microglia have two opposing forms of pro-inflammatory M1 phenotype and anti-inflammatory M2 phenotype [147, 148]. Endogenous stimuli, including ROS, inflammatory factors, and aggregated Aβ, can activate the pro-inflammatory M1 microglia, eventually leading to irreversible neuronal loss. On the contrary, activated anti-inflammatory M2 microglia promote the clearance of Aβ through the phagocytosis of Aβ, and reduce brain inflammation and Aβ toxicity [149, 150]. Based on this, Ren et al. [151] designed TPP-MoS2 QDs with SOD and CAT activity and effective mitochondrial targeting, which can stimulate microglia from an inflammatory M1 phenotype to an anti-inflammatory M2 Phenotypic differentiation, eliminate ROS, down-regulate pro-inflammatory cytokines IL-1β, IL-6 and TNF-α as well as up-regulate transforming growth factor-β to achieve direct neuron protection. This study provides a new therapeutic idea that can reduce AD pathology by targeting nanozymes to mitochondria and polarization of M1/M2 microglia.
Liu et al. [152] developed a novel PLGA NPs that conjugated CD47 extracellular domains via ROS-responsive phenylborate ester bonds, showing a "don't eat me" signal, and encapsulated BBB penetrating peptide CRTIGPSVC (CRT) and microglial modulator Nec-1s. The high level of ROS signaling in the rat brain promotes the release of CD47 from NPs, while NPs without CD47 can be effectively engulfed by microglia cells. The phagocytic NPs containing Nec-1s can regulate pathological microglia to a beneficial state, reduce microglia and astrocyte hyperplasia, and reduce cytokine production and oxidative stress in the brain of AD mice, resulting in a reduction of cognitive impairment and synaptic loss.
3.4.2. LiposomesResearch has shown that PEGylated NLs can improve P2×7/caspase-1/IL-1β signal-dependent microglial activation and thus mitigate neurovascular damage. This is therefore a promising brain-targeted therapeutic biomaterial that can inhibit p2×7-dependent neuroinflammation [153]. In previous research, Kim and colleagues used a receptor-mediated transcytosis mechanism to transport cationic NLs (scL) containing either oligonucleotides or siRNA to the brain. The small size and high stability of single-stranded fragment (TfRscFv) allowed its transport from the variable region of the TfR mAb to the brain [154]. Subsequent to intravenous injection into mice, these TfRscFv-scL nanocomplexes targeted ligands and themselves contained fluorescent oligonucleotides which entered the brain rapidly within six hours. Fluorescence intensity was also significantly higher than seen in a nanocomposite following injected of the untargeted portion. These workers were able to verify that TfR-targeted liposomes containing TNFα-targeting siRNA exert therapeutic effects in an acute neuro-inflammation model induced by lipopolysaccharide injection.
4. Future perspectivesAlthough successful improvements in AD therapy have been achieved in preclinical research using various nanomedicine, none of these systems has yet been translated for use with patients. The main reason is that those unwanted cytotoxic consequences of all candidates cannot be ignored in the highly complex biochemical environment of the CNS [155]. Certain metallic nanocarriers have been reported to facilitate Aβ fibrillation mediated AD progression and have also been involved in bio-accumulation mediated neurotoxicity [156-158]. Indeed, such NPs might disrupt organelles within macrophages including mitochondria, the endoplasmic reticulum and lysosomes and lead to the production of excess ROS and the release of pro-inflammatory mediators [159]. There is increasing interest in shifting research from targeting a single mechanism to multi-target therapy, which increases the possibility of treatment and expands the space for the development of nanomaterial-based DDS. In addition, as research continues to deepen, more potential AD case markers have also been unearthed [160]. Recent studies have shown, for example, that vascular dysfunction might be a pathological factor and therefore could be a potential therapeutic AD target [161]; this means that developing of NPs with multi-targeting and multi-therapeutic effects will likely be a promising strategy [162], the pathological mechanism associated with AD is far more complex than we thought. In comparison with pure drugs, direct brain delivery via loaded nanomaterials is currently limited and so our future research focus should be on overcoming the limitations of nanomaterials and developing more complete systems to accurately deliver large quantities of AD-related drugs to target region.
5. ConclusionsThis review article has summarized some recent advances in the use of nanomaterials for AD treatment from the perspective of different pathogeneses. Nanomaterial-based strategies have been shown to provide revolutionary therapeutic approach for AD treatment. Indeed, the use of nanomaterials makes it possible for numerous therapeutic agents to be delivered across the BBB and treat AD, a remarkable accomplishment and an advance on most existing treatments. Versatile functionalities of nanomaterials can be achieved by manipulating their physicochemical properties as well as via surface engineering and will no doubt lead to more personalized AD treatments. However, although promising, nanomaterial applications in clinical neuroscience remain in their infancy stage. Doubts about AD are still waiting to be explored, further research in using nanomaterials for the treatment of AD is warranted and numerous significant issues remain to be addressed.
Declaration of competing interestThe authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
AcknowledgmentsThis work was financially supported by Hunan Provincial Key Laboratory of Micro & Nano Materials Interface Science, the National Natural Science Foundation of China (Nos. 81603670, 81873169, 21773311, 21972169) and Hunan Provincial Natural Science Foundation of China (Nos. 2017JJ3459, 2020JJ4803) and the Hunan Provincial Science and Technology Plan Project, China (No. 2019TP1001).
| [1] |
A.S. Association, Alzheimers Dement. 16 (2020) 391-460. DOI:10.1002/alz.12068 |
| [2] |
R. Jakob-Roetne, H. Jacobsen, Angew. Chem. Int. Ed. 48 (2009) 3030-3059. DOI:10.1002/anie.200802808 |
| [3] |
J. Hardy, D.J. Selkoe, Science 297 (2002) 353-356. DOI:10.1126/science.1072994 |
| [4] |
M.M. Corrada, R. Brookmeyer, A. Paganini-Hill, D. Berlau, C.H. Kawas, Ann. Neurol. 67 (2010) 114-121. DOI:10.1002/ana.21915 |
| [5] |
C. Qiu, M. Kivipelto, E. von Strauss, Dialogues Clin. Neurosci. 11 (2009) 111-128. DOI:10.31887/DCNS.2009.11.2/cqiu |
| [6] |
M. Goedert, M.G. Spillantini, Science 314 (2006) 777-781. DOI:10.1126/science.1132814 |
| [7] |
J.L. Cummings, T. Morstorf, K. Zhong, Alzheimers Res. Ther. 6 (2014) 37. DOI:10.1186/alzrt269 |
| [8] |
J.Y.Y. Szeto, S.J.G. Lewis, Curr. Neuropharmacol. 14 (2016) 326-338. DOI:10.2174/1570159X14666151208112754 |
| [9] |
K.H. Wong, M.K. Riaz, Y. Xie, et al., Int. J. Mol. Sci. 20 (2019) 381-406. DOI:10.3390/ijms20020381 |
| [10] |
D. Furtado, M. Bjornmalm, S. Ayton, et al., Adv. Mater. 30 (2018) 1801362. DOI:10.1002/adma.201801362 |
| [11] |
S. Tiwari, V. Atluri, A. Kaushik, A. Yndart, M. Nair, Int. J. Nanomed. 14 (2019) 5541-5554. DOI:10.2147/IJN.S200490 |
| [12] |
Z. Redzic, Fluids Barriers CNS 8 (2011) 3. DOI:10.1186/2045-8118-8-3 |
| [13] |
W.A. Banks, BMC Neurol. 9 (2009) S3-S5. |
| [14] |
J. Xie, Z. Shen, Y. Anraku, K. Kataoka, X. Chen, Biomaterials 224 (2019) 11949. |
| [15] |
Y.H. Tsou, X.Q. Zhang, H. Zhu, S. Syed, X. Xu, Small 13 (2017) 1701921. DOI:10.1002/smll.201701921 |
| [16] |
N. Poovaiah, Z. Davoudi, H. Peng, et al., Nanoscale 10 (2018) 16962-16983. DOI:10.1039/C8NR04073G |
| [17] |
M.M. Patel, B.M. Patel, CNS Drugs 31 (2017) 109-133. DOI:10.1007/s40263-016-0405-9 |
| [18] |
R. Martín-Rapun, L. De Matteis, A. Ambrosone, et al., Curr. Pharm. Des. 23 (2017) 1927-1952. DOI:10.2174/1381612822666161226151011 |
| [19] |
H. Derakhshankhah, S. Sajadimajd, S. Jafari, et al., Nanomedicine 24 (2020) 102149. DOI:10.1016/j.nano.2020.102149 |
| [20] |
E. Karran, M. Mercken, B. De Strooper, Nat. Rev. Drug Discov. 10 (2011) 698-712. DOI:10.1038/nrd3505 |
| [21] |
S.H. Barage, K.D. Sonawane, Neuropeptides 52 (2015) 1-18. DOI:10.1016/j.npep.2015.06.008 |
| [22] |
S. Muralidar, S.V. Ambi, S. Sekaran, D. Thirumalai, B. Palaniappan, Int. J. Biol. Macromol. 163 (2020) 1599-1617. DOI:10.1016/j.ijbiomac.2020.07.327 |
| [23] |
K. Mi, G.V.W. Johnson, Curr. Alzheimer Res. 3 (2006) 449-463. DOI:10.2174/156720506779025279 |
| [24] |
M.J. Metcalfe, M.E. Figueiredo-Pereira, Mt. Sinai J. Med. 77 (2010) 50-58. DOI:10.1002/msj.20163 |
| [25] |
K.R. Brunden, J.Q. Trojanowski, V.M.Y. Lee, Nat. Rev. Drug Discov. 8 (2009) 783-793. DOI:10.1038/nrd2959 |
| [26] |
H. Braak, K. Del Tredici, Alzheimers Dement. 8 (2012) 227-233. DOI:10.1016/j.jalz.2012.01.011 |
| [27] |
D.R. Thal, M. Faendrich, Acta Neuropathol. 129 (2015) 163-165. DOI:10.1007/s00401-015-1387-2 |
| [28] |
R.K. Chaturvedi, M. Flint Beal, Free Radic. Biol. Med. 63 (2013) 1-29. DOI:10.1016/j.freeradbiomed.2013.03.018 |
| [29] |
M.T. Lin, M.F. Beal, Nature 443 (2006) 787-795. DOI:10.1038/nature05292 |
| [30] |
C. Guo, L. Sun, X. Chen, D. Zhang, Neural Regen. Res. 8 (2013) 2003-2014. |
| [31] |
R.H. Swerdlow, Biochim. Biophys. Acta: Mol. Basis Dis. 1812 (2011) 1630-1639. DOI:10.1016/j.bbadis.2011.08.012 |
| [32] |
K. Friedland-Leuner, C. Stockburger, I. Denzer, G.P. Eckert, W.E. Müller, Prog. Mol. Biol. Transl. Sci. 127 (2014) 183-210. |
| [33] |
R.H. Swerdlow, J. Alzheimers Dis. 62 (2018) 1403-1416. DOI:10.3233/JAD-170585 |
| [34] |
A. Johri, M.F. Beal, J. Pharmacol. Exp. Ther. 342 (2012) 619-630. DOI:10.1124/jpet.112.192138 |
| [35] |
X.M. Gu, H.C. Huang, Z.F. Jiang, Neurosci. Bull. 28 (2012) 631-640. DOI:10.1007/s12264-012-1270-2 |
| [36] |
C. Cheignon, M. Tomas, . Bonnefont-RousselotD, et al., Redox Biol. 14 (2018) 450-464. DOI:10.1016/j.redox.2017.10.014 |
| [37] |
P.I. Moreira, M.S. Santos, R. Selca, C.R. Oliveira, J.Neurol.Sci. 257 (2007) 206-214. DOI:10.1016/j.jns.2007.01.017 |
| [38] |
V. Van Giau, S.S.A. An, J.P. Hulme, J. Neurol. Sci. 395 (2018) 62-70. DOI:10.1016/j.jns.2018.09.033 |
| [39] |
M. Ankarcrona, F. Mangialasche, B. Winblad, J. Alzheimers Dis. 20 (2010) S579-S590. |
| [40] |
P. Eikelenboom, F.C. Stam, Acta Neuropathol. 57 (1982) 239-242. DOI:10.1007/BF00685397 |
| [41] |
P.L. McGeer, S. Itagaki, H. Tago, E.G. McGeer, Neurosci. Lett. 79 (1987) 195-200. DOI:10.1016/0304-3940(87)90696-3 |
| [42] |
H. Lian, A. Litvinchuk, A.C.A. Chiang, et al., J. Neurosci. 36 (2016) 577-589. DOI:10.1523/JNEUROSCI.2117-15.2016 |
| [43] |
F. Regen, J. Hellmann-Regen, E. Costantini, M. Reale, Curr. Alzheimer Res. 14 (2017) 1140-1148. |
| [44] |
Q. Alam, M.Z. Alam, G. Mushtaq, et al., Curr. Pharm. Des. 22 (2016) 541-548. DOI:10.2174/1381612822666151125000300 |
| [45] |
J.W. Kinney, S.M. Bemiller, A.S. Murtishaw, et al., Alzheimers Dement. 4 (2018) 575-590. |
| [46] |
W.W. Chen, X. Zhang, W.J. Huang, Mol. Med. Rep. 13 (2016) 3391-3396. DOI:10.3892/mmr.2016.4948 |
| [47] |
H. Wang, Y. Shen, H. Chuang, et al., Curr. Alzheimer Res. 16 (2019) 659-674. DOI:10.2174/1567205016666190503151648 |
| [48] |
K. Talbot, H.Y. Wang, H. Kazi, et al., J. Clin. Invest. 122 (2012) 1316-1338. DOI:10.1172/JCI59903 |
| [49] |
Y. Liu, F. Liu, I. Grundke-Iqbal, K. Iqbal, C.X. Gong, J. Pathol. 225 (2011) 54-62. DOI:10.1002/path.2912 |
| [50] |
H. Hampel, M.M. Mesulam, A.C. Cuello, et al., Brain 141 (2018) 1917-1933. DOI:10.1093/brain/awy132 |
| [51] |
X. Du, X. Wang, M. Geng, Transl. Neurodegener. 7 (2018) 2-8. DOI:10.1186/s40035-018-0107-y |
| [52] |
A. Di Meco, M.E. Curtis, E. Lauretti, D. Praticò, Biol. Psychiatry 87 (2020) 797-807. DOI:10.1016/j.biopsych.2019.05.008 |
| [53] |
M.S. Uddin, A. Stachowiak, A.A. Mamun, et al., Front. Aging Neurosci. 10 (2018) 4-21. DOI:10.3389/fnagi.2018.00004 |
| [54] |
A. Budimir, Acta Pharm. 61 (2011) 1-14. DOI:10.2478/v10007-011-0006-6 |
| [55] |
M.J. Hajipour, M.R. Santoso, F. Rezaee, et al., Trends Biotechnol. 35 (2017) 937-953. DOI:10.1016/j.tibtech.2017.06.002 |
| [56] |
K. Rajpoot, Curr. Drug Targets 21 (2020) 819-836. DOI:10.2174/1389450121666200106105633 |
| [57] |
H. Jung, Y.J. Chung, R. Wilton, et al., Adv. Funct. Mater. 30 (2020) 1910475. DOI:10.1002/adfm.201910475 |
| [58] |
M. Mahmoudi, O. Akhavan, M. Ghavami, F. Rezaee, S.M.A. Ghiasi, Nanoscale 4 (2012) 7322-7325. DOI:10.1039/c2nr31657a |
| [59] |
H. Li, Y. Luo, P. Derreumaux, G. Wei, Biophys. J. 101 (2011) 2267-2276. DOI:10.1016/j.bpj.2011.09.046 |
| [60] |
Q.L. Yan, M. Gozin, F.Q. Zhao, A. Cohen, S.P. Pang, Nanoscale 8 (2016) 4799-4851. DOI:10.1039/C5NR07855E |
| [61] |
Y. Liu, L.P. Xu, W. Dai, H. Dong, Y. Wen, X. Zhang, Nanoscale 7 (2015) 19060-19065. DOI:10.1039/C5NR06282A |
| [62] |
Y. Liu, L.P. Xu, Q. Wang, B. Yang, X. Zhang, ACS Chem. Neurosci. 9 (2018) 817-823. DOI:10.1021/acschemneuro.7b00439 |
| [63] |
Y.H. Liao, Y.J. Chang, Y. Yoshiike, Y.C. Chang, Y.R. Chen, Small 8 (2012) 3631-3639. DOI:10.1002/smll.201201068 |
| [64] |
N. Gao, H. Sun, K. Dong, J. Ren, X. Qu, Chemistry 21 (2015) 829-835. DOI:10.1002/chem.201404562 |
| [65] |
K. Hou, J. Zhao, H. Wang, et al., Nat. Commun. 11 (2020) 4790. DOI:10.1038/s41467-020-18525-2 |
| [66] |
Y. Qi, P. Yi, T. He, et al., Colloids Surf. A: Physicochem. Eng. Asp. 602 (2020) 125058-125066. DOI:10.1016/j.colsurfa.2020.125058 |
| [67] |
Q. Han, S. Cai, L. Yang, et al., ACS Appl. Mater. Interfaces 9 (2017) 21116-21123. DOI:10.1021/acsami.7b03816 |
| [68] |
A.H. Lu, E.L. Salabas, F. Schüth, Angew. Chem. Int. Ed. 46 (2007) 1222-1244. DOI:10.1002/anie.200602866 |
| [69] |
X. Li, J. Wei, K.E. Aifantis, et al., J. Biomed. Mater. Res. A 104 (2016) 1285-1296. DOI:10.1002/jbm.a.35654 |
| [70] |
S.M. Silva, R. Tavallaie, L. Sandiford, R.D. Tilley, J.J. Gooding, Chem. Commun. 52 (2016) 7528-7540. DOI:10.1039/C6CC03225G |
| [71] |
A.G. Roca, R. Costo, A.F. Rebolledo, et al., J. Phys. D: Appl. Phys. 42 (2009) 224002-224012. DOI:10.1088/0022-3727/42/22/224002 |
| [72] |
F. Dilnawaz, S.K. Sahoo, Drug Discov. Today 20 (2015) 1256-1264. DOI:10.1016/j.drudis.2015.06.008 |
| [73] |
F.U. Amin, A.K. Hoshiar, T.D. Do, et al., Nanoscale 9 (2017) 10619-10632. DOI:10.1039/C7NR00772H |
| [74] |
D. Kim, H.J. Kwon, T. Hyeon, Adv. Mater. 31 (2019) 1807965. DOI:10.1002/adma.201807965 |
| [75] |
S.D. Hettiarachchi, Y. Zhou, E. Seven, et al., J. Control. Release 314 (2019) 125-140. DOI:10.1016/j.jconrel.2019.10.034 |
| [76] |
A.S. Chauhan, Molecules 23 (2018) 938-946. DOI:10.3390/molecules23040938 |
| [77] |
N. Kamaly, B. Yameen, J. Wu, O.C. Farokhzad, Chem. Rev. 116 (2016) 2602-2663. DOI:10.1021/acs.chemrev.5b00346 |
| [78] |
C.E. Astete, C.M. Sabliov, J. Biomater. Sci. Polym. Ed. 17 (2006) 247-289. DOI:10.1163/156856206775997322 |
| [79] |
F. Danhier, E. Ansorena, J.M. Silva, et al., J. Control. Release 161 (2012) 505-522. DOI:10.1016/j.jconrel.2012.01.043 |
| [80] |
M. Mir, N. Ahmed, A.U. Rehman, Colloids Surf. B: Biointerfaces 159 (2017) 217-231. DOI:10.1016/j.colsurfb.2017.07.038 |
| [81] |
S.G. Jeon, M.Y. Cha, J.I. Kim, et al., Nanomedicine 17 (2019) 297-307. DOI:10.1016/j.nano.2019.02.004 |
| [82] |
S. Sathya, B. Shanmuganathan, S. Saranya, et al., Artif. Cells Nanomed. Biotechnol. 46 (2018) 1719-1730. |
| [83] |
Z. Stanic, Plant Foods Hum. Nutr. 72 (2017) 1-12. DOI:10.1007/s11130-016-0590-1 |
| [84] |
M. Tang, C. Taghibiglou, J. Alzheimers Dis. 58 (2017) 1003-1016. DOI:10.3233/JAD-170188 |
| [85] |
S.K. Tiwari, S. Agarwal, B. Seth, et al., ACS Nano 8 (2014) 76-103. DOI:10.1021/nn405077y |
| [86] |
N.M. Khalil, T.C. Frabel do Nascimento, D.M. Casa, et al., Colloids Surf. B: Biointerfaces 101 (2013) 353-360. DOI:10.1016/j.colsurfb.2012.06.024 |
| [87] |
S. Fan, Y. Zheng, X. Liu, et al., Drug Deliv. 25 (2018) 1091-1102. DOI:10.1080/10717544.2018.1461955 |
| [88] |
J. Sarvaiya, Y.K. Agrawal, Int. J. Biol. Macromol. 72 (2015) 454-465. DOI:10.1016/j.ijbiomac.2014.08.052 |
| [89] |
A. Jha, V. Ghormade, H. Kolge, K.M. Paknikar, J. Mater. Chem. B 7 (2019) 3362-3373. DOI:10.1039/C9TB00162J |
| [90] |
Z. Jiang, X. Dong, Y. Sun, Carbohydr. Res. 461 (2018) 11-18. DOI:10.1016/j.carres.2018.03.001 |
| [91] |
P. Li, Y. Chen, Y. Liu, Chin. Chem. Lett. 30 (2019) 1190-1197. DOI:10.1016/j.cclet.2019.03.035 |
| [92] |
A. Qu, F. Huang, A. Li, et al., Chem. Commun. 53 (2017) 1289-1292. DOI:10.1039/C6CC07803F |
| [93] |
P. Yang, C. Yang, K. Zhang, L. Wang, H. Wang, Chin. Chem. Lett. 29 (2018) 1811-1814. DOI:10.1016/j.cclet.2018.10.003 |
| [94] |
J.E. Kim, M. Lee, Biochem. Biophys. Res. Commun. 303 (2003) 576-579. DOI:10.1016/S0006-291X(03)00393-0 |
| [95] |
D. Liu, D. Fu, L. Zhang, L. Sun, Chin. Chem. Lett. 32 (2021) 1066-1070. DOI:10.1016/j.cclet.2020.09.009 |
| [96] |
L. Ordóñez-Gutiérrez, F. Wandosell, Front. Synaptic Neurosci. 12 (2020) 20-29. DOI:10.3389/fnsyn.2020.00020 |
| [97] |
S.G. Antimisiaris, Clin. Lipidol. 9 (2014) 477-481. DOI:10.2217/clp.14.33 |
| [98] |
S. Mourtas, A.N. Lazar, E. Markoutsa, C. Duyckaerts, S.G. Antimisiaris, Eur. J. Med. Chem. 80 (2014) 175-183. DOI:10.1016/j.ejmech.2014.04.050 |
| [99] |
C. Ross, M. Taylor, N. Fullwood, D. Allsop, Int. J. Nanomed. 13 (2018) 8507-8522. DOI:10.2147/IJN.S183117 |
| [100] |
S. Mancini, C. Balducci, E. Micotti, et al., J. Control. Release 258 (2017) 121-129. DOI:10.1016/j.jconrel.2017.05.013 |
| [101] |
L. Kong, X.T. Li, Y.N. Ni, et al., Int. J. Nanomed. 15 (2020) 2841-2858. DOI:10.2147/IJN.S239608 |
| [102] |
S. Mourtas, M. Canovi, C. Zona, et al., Biomaterials 32 (2011) 1635-1645. DOI:10.1016/j.biomaterials.2010.10.027 |
| [103] |
A.N. Lazar, S. Mourtas, I. Youssef, et al., Nanomedicine 9 (2013) 712-721. DOI:10.1016/j.nano.2012.11.004 |
| [104] |
J. Zheng, R. Fan, H. Wu, et al., Nat. Commun. 10 (2019) 1604-1615. DOI:10.1038/s41467-019-09601-3 |
| [105] |
H. Wu, Y. Lei, X. Song, et al., Colloids Surf. A: Physicochem. Eng. Asp. 575 (2019) 111-117. DOI:10.1016/j.colsurfa.2019.05.002 |
| [106] |
M. Kaur, K. Sudhakar, V. Mishra, Int. J. Polym. Mater. 68 (2019) 287-296. DOI:10.1080/00914037.2018.1445629 |
| [107] |
A.V. Kabanov, S.V. Vinogradov, Angew. Chem. Int. Ed. 48 (2009) 5418-5429. DOI:10.1002/anie.200900441 |
| [108] |
M. Suhail, J.M. Rosenholm, M.U. Minhas, et al., Ther. Deliv. 10 (2019) 697-717. DOI:10.4155/tde-2019-0010 |
| [109] |
K. Ikeda, T. Okada, S. Sawada, K. Akiyoshi, K. Matsuzaki, FEBS Lett. 580 (2006) 6587-6595. DOI:10.1016/j.febslet.2006.11.009 |
| [110] |
Z. Jiang, X. Dong, H. Liu, et al., React. Funct. Polym. 104 (2016) 22-29. DOI:10.1016/j.reactfunctpolym.2016.04.019 |
| [111] |
Z. Jiang, X. Dong, X. Yan, et al., Sci. Rep. 8 (2018) 3505-3515. DOI:10.1038/s41598-018-21933-6 |
| [112] |
J. Li, Q. Han, X. Wang, et al., Small 10 (2014) 4386-4394. |
| [113] |
I. Ahmad, A. Mozhi, L. Yang, et al., Colloids Surf. B: Biointerfaces 159 (2017) 540-545. DOI:10.1016/j.colsurfb.2017.08.020 |
| [114] |
Y. Zhao, J. Cai, Z. Liu, et al., Nano Lett. 19 (2019) 674-683. DOI:10.1021/acs.nanolett.8b03644 |
| [115] |
Q. Luo, Y.X. Lin, P.P. Yang, et al., Nat. Commun. 9 (2018) 1802-1813. DOI:10.1038/s41467-018-04255-z |
| [116] |
S. Mura, J. Nicolas, P. Couvreur, Nat. Mater. 12 (2013) 991-1003. DOI:10.1038/nmat3776 |
| [117] |
Y. Lai, Y. Zhu, Z. Xu, et al., Adv. Funct. Mater. 30 (2020) 1908473. DOI:10.1002/adfm.201908473 |
| [118] |
W. Zhang, G. Gao, Z. Ma, et al., Nat. Sci. Rev. 7 (2020) 763-774. DOI:10.1093/nsr/nwz215 |
| [119] |
S.K. Sonawane, A. Ahmad, S. Chinnathambi, ACS Omega 4 (2019) 12833-12840. DOI:10.1021/acsomega.9b01411 |
| [120] |
B. Ghalandari, K. Asadollahi, A. Shakerizadeh, et al., J. Photochem. Photobiol. B 192 (2019) 131-140. DOI:10.1016/j.jphotobiol.2019.01.012 |
| [121] |
G.W. Small, P. Siddarth, Z. Li, et al., Am. J. Geriatr. Psychiatry 26 (2018) 266-277. DOI:10.1016/j.jagp.2017.10.010 |
| [122] |
C. Gao, X. Chu, W. Gong, et al., J. Nanobiotechnol. 18 (2020) 71-93. DOI:10.1186/s12951-020-00626-1 |
| [123] |
M. Xu, H. Zhou, Y. Liu, et al., ACS Appl. Mater. Interfaces 10 (2018) 32965-32980. DOI:10.1021/acsami.8b08230 |
| [124] |
Q. Chen, Y. Du, K. Zhang, et al., ACS Nano 12 (2018) 1321-1338. DOI:10.1021/acsnano.7b07625 |
| [125] |
H. Zhou, Y. Gong, Y. Liu, et al., Biomaterials 237 (2020) 119822. DOI:10.1016/j.biomaterials.2020.119822 |
| [126] |
C. Cervellati, E. Cremonini, C. Bosi, et al., Curr. Alzheimer Res. 10 (2013) 365-372. DOI:10.2174/1567205011310040003 |
| [127] |
G. Caruso, S.F. Spampinato, V. Cardaci, et al., Curr. Pharm. Des. 25 (2019) 4771-4781. |
| [128] |
F. Wang, M.A. Ogasawara, P. Huang, Mol. Aspects Med. 31 (2010) 75-92. DOI:10.1016/j.mam.2009.12.003 |
| [129] |
G. Battogtokh, Y.S. Choi, D.S. Kang, et al., Acta Pharm. Sin. B 8 (2018) 862-880. DOI:10.1016/j.apsb.2018.05.006 |
| [130] |
Z. Wang, W. Guo, X. Kuang, S. Hou, H. Liu, Asian J. Pharm. Sci. 12 (2017) 498-508. DOI:10.1016/j.ajps.2017.05.006 |
| [131] |
D. Guzman-Villanueva, M.R. Mendiola, H.X. Nguyen, et al., Methods Mol. Biol. (2019) 183-189 2000. |
| [132] |
J. Zielonka, J. Joseph, A. Sikora, et al., Chem. Rev. 117 (2017) 10043-10120. DOI:10.1021/acs.chemrev.7b00042 |
| [133] |
H.J. Kwon, M.Y. Cha, D. Kim, et al., ACS Nano 10 (2016) 2860-2870. DOI:10.1021/acsnano.5b08045 |
| [134] |
Y. Han, C. Gao, H. Wang, et al., Bioact. Mater. 6 (2021) 529-542. DOI:10.1016/j.bioactmat.2020.08.017 |
| [135] |
W.C. Ballance, E.C. Qin, H.J. Chung, M.U. Gillette, H. Kong, Biomaterials 217 (2019) 119292. DOI:10.1016/j.biomaterials.2019.119292 |
| [136] |
J. Liu, Y. Li, S. Chen, et al., Front. Chem. 8 (2020) 838-861. DOI:10.3389/fchem.2020.00838 |
| [137] |
Y. Shen, B. Cao, N.R. Snyder, et al., J. Nanobiotechnol. 16 (2018) 13-29. DOI:10.1186/s12951-018-0340-7 |
| [138] |
X. Xu, H. An, D. Zhang, et al., Sci. Adv. 5 (2019) eaat2953. DOI:10.1126/sciadv.aat2953 |
| [139] |
W.Y. Wang, M.S. Tan, J.T. Yu, L. Tan, Ann. Transl. Med. 3 (2015) 136-150. |
| [140] |
Y. Dong, X. Li, J. Cheng, L. Hou, Int. J. Mol. Sci. 20 (2019) 558-581. DOI:10.3390/ijms20030558 |
| [141] |
S. Hong, V.F. Beja-Glasser, B.M. Nfonoyim, et al., Science 352 (2016) 712-716. DOI:10.1126/science.aad8373 |
| [142] |
A.P. Muller, G.K. Ferreira, A.J. Pires, et al., Mater. Sci. Eng. C: Mater. Biol. Appl. 77 (2017) 476-483. DOI:10.1016/j.msec.2017.03.283 |
| [143] |
K.I. Jeon, M.S. Byun, D.M. Jue, Exp. Mol. Med. 35 (2003) 61-66. DOI:10.1038/emm.2003.9 |
| [144] |
E.G. Victor, P.C. Silveira, J.C. Possato, et al., J. Nanobiotechnol. 10 (2012) 11-19. DOI:10.1186/1477-3155-10-11 |
| [145] |
G. Testa, P. Gamba, U. Badilli, et al., PLoS One 9 (2014) e96795. DOI:10.1371/journal.pone.0096795 |
| [146] |
P. Ganesan, B. Kim, P. Ramalaingam, et al., Molecules 24 (2019) 1170-1180. DOI:10.3390/molecules24061170 |
| [147] |
X. Lan, X. Han, Q. Li, Q.W. Yang, J. Wang, Nat. Rev. Neurol. 13 (2017) 420-433. DOI:10.1038/nrneurol.2017.69 |
| [148] |
F. Zeng, Y. Wu, X. Li, et al., Angew. Chem. Int. Ed. 57 (2018) 5808-5812. DOI:10.1002/anie.201802309 |
| [149] |
W.M. Song, M. Colonna, Nat. Immunol. 19 (2018) 1048-1058. DOI:10.1038/s41590-018-0212-1 |
| [150] |
Y. Lu, Z. Guo, Y. Zhang, et al., Adv. Sci. 6 (2019) 1801586. DOI:10.1002/advs.201801586 |
| [151] |
C. Ren, D. Li, Q. Zhou, X. Hu, Biomaterials 232 (2020) 119752. DOI:10.1016/j.biomaterials.2019.119752 |
| [152] |
L. Zhang, X.G. Liu, D.Q. Liu, et al., Adv. Funct. Mater. 30 (2020) 1910691. DOI:10.1002/adfm.201910691 |
| [153] |
J.Y. Huang, Y.M. Lu, H. Wang, et al., Biomaterials 34 (2013) 7960-7970. DOI:10.1016/j.biomaterials.2013.07.009 |
| [154] |
S.S. Kim, A. Rait, E.R. Garrido-Sanabria, et al., Mol. Ther. 26 (2018) 84-94. DOI:10.1016/j.ymthe.2017.10.003 |
| [155] |
B. Formicola, A. Cox, R. Dal Magro, M. Masserini, F. Re, J. Biomed. Nanotechnol. 15 (2019) 1997-2024. DOI:10.1166/jbn.2019.2837 |
| [156] |
W.H. Wu, X. Sun, Y.P. Yu, et al., Biochem. Biophys. Res. Commun. 373 (2008) 315-318. DOI:10.1016/j.bbrc.2008.06.035 |
| [157] |
X. Feng, A. Chen, Y. Zhang, et al., Int. J. Nanomed. 10 (2015) 4321-4340. |
| [158] |
T. Wu, M. Tang, Nanomedicine 13 (2018) 233-249. DOI:10.2217/nnm-2017-0270 |
| [159] |
A. Radomska, J. Leszczyszyn, M.W. Radomski, Adv. Clin. Exp. Med. 25 (2016) 151-162. DOI:10.17219/acem/60879 |
| [160] |
M.D. Sweeney, A.P. Sagare, B.V. Zlokovic, Nat. Rev. Neurol. 14 (2018) 133-150. |
| [161] |
K. Govindpani, C. Vinnakota, H.J. Waldvogel, R.L. Faull, A. Kwakowsky, Neural Regen. Res. 15 (2020) 1030-1032. DOI:10.4103/1673-5374.270306 |
| [162] |
A. Kumar, R.K. Chaudhary, R. Singh, et al., Front. Neurosci. 14 (2020) 305-315. DOI:10.3389/fnins.2020.00305 |
 2021, Vol. 32
2021, Vol. 32