Over the past two decades, numerous studies have presented that DNA not only stores genetic information [1], but also has greater potential applications. DNA-base hybrid catalysts comprised of double-stranded DNA and metal ions is a good example [2-6]. Due to its chirality and biocompatibility, DNA manifests excellent advantages in chiral catalysis [7]. Further researches have discussed that DNA-base hybrid catalysts have certain chemical effects and enantioselectivity [8]. Through a varied complexation of metal ions, DNA can participate in numerous catalytic reactions, such as Diels-Alder (D-A) [9] and Friedel-Crafts [10] reactions.
G-Quadruplex (G4) is different from the conventional double-strand. It is a secondary structure formed by superimposing multiple quadruple guanines [11]. Most research objects are induced by a single-strand DNA. A conventional G4 is generally classified as either parallel structure or anti-parallel structure. Based on a classification by means of glycosidic bond angles (GBA), antiparallel can be divided into structures comprising the same type of GBA guanine sequences and different types of guanine [12]. G4 has a variety of chain orientations and ring structures [13], and its internal cavity constitutes a good chiral conversion condition [14]. In this case, G4 catalysis has attracted the attention of many researchers. With a more regular and controllable structure, it is likely that G4 can be used as an applicable chiral template, and some metal ion cores can be added to enhance its catalytic effect and enantioselectivity [13, 15-17].
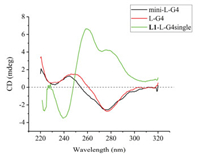
Chung et al. has discovered a 28-nt sequence L-G4 (sequence 5'-T(GGT)4TG(TGG)3TGTT-3'), which exhibits a distinct circular dichroism (CD) profile with a negative peak at ~275 nm and a positive peak at ~250 nm (Fig. 1), nearly inverted from that of all right-handed G4 topologies reported to date [18]. Recently, a second case of mini-L-G4 (sequence 5'-GT(GGT)3G-3') has been developed, which proves a very similar CD signal as L-G4 [19]. However, recently, studies on the function of L-G4 have not yet been reported.
|
Download:
|
| Fig. 1. CD spectra of different oligodeoxynucleotides. L-G4: L-G4 (10 μmol/L) with KCl (100 mmol/L) and NaCl (20 mmol/L) in MOPS buffer (20 mmol/L, pH 6.5); mini-L-G4: mini-L-G4 (10 μmol/L) with KCl (100 mmol/L) and NaCl (20 mmol/L) in MOPS buffer (20 mmol/L, pH 6.5); L1-L-G4single: L-G4-single (no G4 structure formed, 10 μmol/L), L1 (50 μmol/L), without any buffer, do not need anneal. | |
The D-A reaction is a significant carbon-carbon bond formation reaction in organic synthesis [20, 21]. Over the past few decades, the search for innovative catalytic strategies for controlling the establishment of new carbon-carbon bonds and stereo-centres has gained extensive attention [22-24]. Among these strategies, biomolecules and particularly nucleic acid materials have been regarded as promising catalysts due to their biocompatibility and mild reaction conditions [13, 15-17, 25, 26]. In this study, L-G4 is applied with a special conformation for D-A reaction. Initially, the D-A model reaction [27] between aza-chalcone (1) and cyclopentadiene (2) was selected to probe the catalytic performance of L-G4. It was found that L-G4 alone could promote D-A reactions with its distinct laevorotatory properties, while the enantiomeric excess of the endo isomer of product 3a was −6% (Table 1, entry 3), which was contrary to previous reports and has caught researchers' attention. The enantioselectivity of the D-A reaction as promoted by L-G4 was a little higher than that of the uncatalyzed reaction (Table 1, entry 3 vs. entry 1), indicating that L-G4 may be utilised as an enantioselective catalyst reaction of D-A.
|
|
Table 1 Diels–Alder reaction catalyzed by L-G4-based catalysts.a |
A complex of Cu(NO3)2 and L-G4 was assembled to test the ability of (L-G4-Cu2+) to catalyse D-A reaction enantioselectivity. L-G4-Cu2+ provides a significant reaction rate enhancement (Table 1, entry 4), and L-G4 (Table 1, entry 3) or Cu(NO3)2 (Table 1, entry 2) have been observed. An excellent diastereoselectivity (endo/exo 96:4) and good enantioselectivity (−52% ee) of product 3a has also been observed. These results indicate that L-G4-Cu2+ can be utilised as an effective catalyst to provide stereoselectivity and increase reaction rate. To further understand L-G4-Cu2+, the influences of the different cations (Table S1 in Supporting information) in divalent metal salts are assessed. In all cases, these complexes in the D-A reaction, lower conversion and enantioselectivity are obtained (Fig. S9 and Table S1 in Supporting information). With the presence of G4 ligand, catalytic ability and enantioselectivity of L-G4-Cu2+ have been further improved (Table 1, entries 5 and 6). Over the course of this research, the second case of mini-L-G4 was reported [19]. A study was then performed on its catalytic performance, eventually determining that the catalytic effect of mini-L-G4 (Table 1, entries 7-9) was not as good as that of the first L-G4. It was speculated that the configuration and enantioselectivity of the absolute product rely on G4DNA conformation.
To validate the catalytic activity of L-G4-Cu2+, the initial rate (Vinit) of the D–A reaction catalysed by L-G4, Cu2+ and L-G4-Cu2+ (Fig. S2 in Supporting information) was measured. At a fixed concentration of 1a, VinitL-G4-Cu was greater than VinitCu2+ and VinitL-G4 (Fig. S2), signifying that L-G4 and Cu2+ assemble into a whole L-G4-Cu2+ complex. Such little difference between Vinit L-G4 and Vinit un-catalysing that the catalytic function of L-G4 was weak. The apparent second-order rate constant (kapp) was then estimated from the initial rate of the D–A reaction [3, 27]. Cu2+ and L-G4 increased kapp by 3.1 times and 3.8 times, respectively (Table 2, entries 2 and 3), while L-G4-Cu2+ caused a rate acceleration of up to 11.9 times (Table 2, entry 4). Compared with Cu2+ as a catalyst only, L-G4-Cu2+ has a moderate 4-fold acceleration in the D–A reaction rate, which was probably because of the coordination of L-G4 with Cu2+ and the substrate. When L-G4, Cu2+, and 1a were mixed in a certain proportion, the color of the solution changed obviously, from nearly colorless to obvious yellow.
|
|
Table 2 Kinetic parameters for L-G4, Cu2+ and L-G4-Cu2+.a |
In this case, a research on the combination of L-G4, Cu2+ and 1a was conducted. First, using a job plot (Fig. S5 in Supporting information), two sets of separate substrates 1a and L-G4 CD were made; measuring the signal value of 280 nm. It was found that the two were not combined. Another group entails 2.5% equiv. Cu2+ being added; in this case, the signal has changed. The calculated difference was linear, implying that the addition of Cu2+ was combined. The UV spectrum was measured after annealing L-G4 and Cu2+ (Fig. S6 in Supporting information), taking the signal value of 260 nm, and calculating the molar ratio as approximately L-G4:Cu2+ = 1:12. In the case of fixed L-G4:Cu2+=1:12, the combination of L-G4 and 1a (Fig. S8 in Supporting information) was explored; a rough combination of L-G4:Cu2+: 1a=1:12:45 was then obtained. To rule out that 2 can be combined with L-G4 and Cu2+, another experiment (Fig. S7 in Supporting information) was performed, which eventually finds that 2 was not combined. At the same time, it can be deduced from the UV–vis method that the addition of Cu2+ promotes a better binding than other ions (Fig. S9 in Supporting information), which was consistent with the catalytic results (Table S1).
Whilst optical terms have depicted that the structure of L-G4 and mini-L-G4 are similar, there are remarkable differences in catalytic effects (Table 1), which further signify that a complete G4 structure highlights the significance of enantioselectivity. At the same time, to achieve a better catalytic efficiency, the G4 ligand [28], was used through the screening of ligands L1 [29] and L2 [30] (Supporting information) to stabilise G4 and to coordinate with Cu2+ and 1a, so as to obtain better results (Table 1). Through G4 ligands, one of them can speculate that 1a and Cu2+ may interact with L-G4. For the mini-L-G4, the same strategy was used to implement the above experiment, in which it was observed that the catalytic effect has become better, but yet not as good as L-G4. Taken together, this work proposes an approach based on the currently known asymmetric catalysis of left-handed G4.
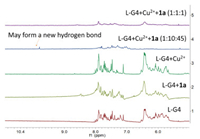
Using the preliminary catalytic results, the substrate specificity of L-G4-Cu2+ has been assessed with various substituted aza-chalcones (1a–d) (Table 3). L-G4-Cu2+, L1 and L2 are the active catalysts for all tested substrates in the D-A reaction. Likewise, the ee values of 3c and 3d have changed from negative to positive (Table 1, entries 3 and 4). It was speculated that there was a coordination with the methoxy group and the nitro group to the ring part of L-G4. Nevertheless, after adding a G4 ligand, the catalytic effect can be significantly enhanced. Related experiments on mini-L-G4 have also been conducted. Unfortunately, the results are unsatisfactory (Table S2 in Supporting information). The coordination structure of the metal ion or metal complex with the host macromolecule in the metalloenzyme was highly vital for catalytic performance, especially in an enantioselective reaction. To explore the location of Cu2+ in an L-G4-Cu2+ complex, the method of proton nuclear magnetic resonance (1H NMR) was used for investigation. This result suggests that, in the G4 loop ring, it can coordinate with Cu2+ (Fig. 2). After adding 1a, a remarkable result emerges, and a new hydrogen bond may be formed. Specifically, it was believed that L-G4-Cu2+-1a can reduce the LUMO of 1a, and the kinetic instability of ligand substitution may exist in a carbon-forming system within a rapid equilibrium between the electrons [31]. Significantly, this analysis determines the attractive prospect that L-G4-Cu2+ may function as enantioselective catalysts.
|
|
Table 3 Diels–Alder reaction catalyzed by L-G4-based catalysts.a |
|
Download:
|
| Fig. 2. 1H NMR of L-G4 in different conditions. 1: L-G4 (200 μmol/L) with KCl (100 mmol/L) and NaCl (20 mmol/L); 2: L-G4 (200 μmol/L) and 1a (200 μmol/L) with KCl (100 mmol/L) and NaCl (20 mmol/L); 3: L-G4 (200 μmol/L) and Cu2+ (200 μmol/L) with KCl (100 mmol/L) and NaCl (20 mmol/L). 4: L-G4 (200 μmol/L), Cu2+ (2 mmol/L) and 1a (9 mmol/L) with KCl (100 mmol/L) and NaCl (20 mmol/L); 5: L-G4 (200 μmol/L), Cu2+ (200 μmol/L) and 1a (200 μmol/L) with KCl (100 mmol/L) and NaCl (20 mmol/L). | |
To summarise, it was found that the various enantioselective D-A reactions can be attained using a catalyst based on L-G4. Furthermore, the enantioselectivity of the reaction can be increased by adding a G4 ligand (L1 and L2). By comparing the unsatisfactory effects of mini-L-G4, it was determined that a complete G4 structure was a necessary condition to ensure smooth catalysis. This special kind of L-G4 may have other applications in chemical synthesis and biology. More in-depth research on this type of G4 will be conducted in due course.
Declaration of competing interestThe authors report no declarations of interest.
AcknowledgmentsThis work was supported by the National Natural Science Foundation of China (Nos. 21432008, 91753201, and 21721005). We thank the large-scale instrument and equipment sharing foundation of Wuhan University.
Supplementary material related to this article can be found, in the online version, at doi: https://doi.org/10.1016/j.cclet.2020.12.047.
| [1] |
M. Frank-Kamenetskii, Nature 328 (1987) 17-18. DOI:10.1038/328017a0 |
| [2] |
A.J. Boersma, B.L. Feringa, G. Roelfes, Org. Lett. 9 (2007) 3647-3650. DOI:10.1021/ol7015274 |
| [3] |
(a) A.J. Boersma, J.E. Klijn, B.L. Feringa, G. Roelfes, J. Am. Chem. Soc. 130 (2008) 11783-11790. (b) Y.F. Wang, X. Zhang, C.C. Liu, X. Zhou, Acta Chim. Sin. 75 (2017) 692-698. |
| [4] |
A.J. Boersma, D. Coquiere, D. Geerdink, et al., Nat. Chem. 2 (2010) 991-995. DOI:10.1038/nchem.819 |
| [5] |
S. Park, H. Sugiyama, Angew. Chem. Int. Ed. 49 (2010) 3870-3878. DOI:10.1002/anie.200905382 |
| [6] |
S. Park, H. Sugiyama, Molecules 17 (2012) 12792-12803. DOI:10.3390/molecules171112792 |
| [7] |
A.J. Boersma, R.P. Megens, B.L. Feringa, G. Roelfes, Chem. Soc. Rev. 39 (2010) 2083-2092. DOI:10.1039/b811349c |
| [8] |
G. Roelfes, B.L. Feringa, Angew. Chem. Int. Ed. 44 (2005) 3230-3232. DOI:10.1002/anie.200500298 |
| [9] |
N.S. Oltra, G. Roelfes, Acc. Chem. Res. (2008) 6039-6041. |
| [10] |
E.W. Dijk, B.L. Feringa, G. Roelfes, Tetrahedron: Asymmetry 19 (2008) 2374-2377. DOI:10.1016/j.tetasy.2008.10.004 |
| [11] |
J.T. Davis, G.P. Spada, Chem. Soc. Rev. 36 (2007) 296-313. DOI:10.1039/B600282J |
| [12] |
A.I. Karsisiotis, N.M. Hessari, E. Novellino, et al., Angew. Chem. Int. Ed. 50 (2011) 10645-10648. DOI:10.1002/anie.201105193 |
| [13] |
L.X. Wang, J.F. Xiang, Y.L. Tang, Adv. Synth. Catal. 357 (2015) 13-20. DOI:10.1002/adsc.201400818 |
| [14] |
S. Neidle, S. Balasubramanian, Quadruplex Nucleic Acids, The Royal Society of Chemistry, London, 2006, pp. 1-30.
|
| [15] |
C. Wang, G. Jia, J. Zhou, et al., Angew. Chem. Int. Ed. 51 (2012) 9352-9355. DOI:10.1002/anie.201204850 |
| [16] |
C. Wang, Y. Li, G. Jia, et al., Chem. Commun. 48 (2012) 6232-6234. DOI:10.1039/c2cc31320k |
| [17] |
Y. Li, M. Cheng, J. Hao, et al., Chem. Sci. 6 (2015) 5578-5585. DOI:10.1039/C5SC01381J |
| [18] |
W.J. Chung, B. Heddi, E. Schmitt, et al., Proc. Natl. Acad. Sci. U. S. A. 112 (2015) 2729-2733. DOI:10.1073/pnas.1418718112 |
| [19] |
B. Bakalar, B. Heddi, E. Schmitt, Y. Mechulam, A.T. Phan, Angew. Chem. Int. Ed. 58 (2019) 2331-2335. DOI:10.1002/anie.201812628 |
| [20] |
V.E. Gouverneur, K. Houk, B. de Pascual-Teresa, et al., Science 262 (1993) 204-208. DOI:10.1126/science.8211138 |
| [21] |
T.M. Tarasow, S.L. Tarasow, B.E. Eaton, Nature 389 (1997) 54. DOI:10.1038/37950 |
| [22] |
T. Ose, K. Watanabe, T. Mie, Nature 422 (2003) 185. DOI:10.1038/nature01454 |
| [23] |
A. Serganov, S. Keiper, L. Malinina, Nat. Struct. Mol. Biol. 12 (2005) 218-224. DOI:10.1038/nsmb906 |
| [24] |
M.T. Reetz, N. Jiao, Angew. Chem. Int. Ed. 45 (2006) 2416-2419. DOI:10.1002/anie.200504561 |
| [25] |
A.J. Boersma, J.E. Klijn, B.L. Feringa, G. Roelfes, J. Am. Chem. Soc. 130 (2008) 11783-11790. DOI:10.1021/ja803170m |
| [26] |
X. Xu, W. Mao, F. Lin, Catal. Commun. 74 (2016) 16-18. DOI:10.1016/j.catcom.2015.09.012 |
| [27] |
S. Otto, F. Bertoncin, J.B. Engberts, J. Am. Chem. Soc. 118 (1996) 7702-7707. DOI:10.1021/ja960318k |
| [28] |
P. Murat, Y. Singh, E. Defrancq, Chem. Soc. Rev. 40 (2011) 5293-5307. DOI:10.1039/c1cs15117g |
| [29] |
B.S. Fu, J.G. Huang, Y.Q. Chen, et al., Chem. Commun. 52 (2016) 10052-10055. DOI:10.1039/C6CC04866H |
| [30] |
S. Muller, S. Kumari, R. Rodriguez, S. Balasubramanian, Nat. Chem. 2 (2010) 1095-1098. DOI:10.1038/nchem.842 |
| [31] |
K.A. Ahrendt, C.J. Borths, D.W.C. Macmillan, J. Am. Chem. Soc. 122 (2000) 4243-4244. DOI:10.1021/ja000092s |
 2021, Vol. 32
2021, Vol. 32 




