b West China School of Stomatology, Sichuan University, Chengdu 610041, China
Hydrogels are soft biomaterials which are three-dimensionally crosslinked hydrophilic polymer networks that are formed through crosslinking of monomers or polymer chains via covalent bonds and/or noncovalent interactions such as hydrogen bonds, electrostatic interactions, host-guest complexations and their combinations [1, 2]. Cells can survive in hydrogel due to its biocompatibility [3, 4]. Owing to their extracellular matrix (ECM)-like structure, hydrogels can be delivered via injection to defect site, entrap growth factors, drugs and cells in the mesh and control the release of the materials as required [5]. Moreover, hydrogels demonstrate excellent integration with surrounding tissues, thereby avoiding the complexity of surgical removal and reducing the possibility of an inflammatory response [6].
Photo-crosslinking is one of the most well-explored methods of creating covalently crosslinked hydrogels, through the exposure of a photosensitive system composed of photo-crosslinkable hydrogels, photo-initiators and other compounds such as cells and therapeutic molecules, to ultraviolet (200–400 nm) or visible light (400–800 nm). By simply adjusting the light intensity, exposure time and illuminated area, photo-crosslinking provides an exceptional control over the spatiotemporal formation of the hydrogel and its network properties. Photo-crosslinkable hydrogels are attractive materials for tissue engineering applications as they provide fast crosslinking under cell-compatible conditions. Photo-crosslinkable hydrogels have advantage of easy filling of irregularly shaped defects, which leads to good contact between regenerating construct and surrounding native tissue. This review summarizes photo-crosslinkable hydrogels and their applications in tissue engineering [1, 7].
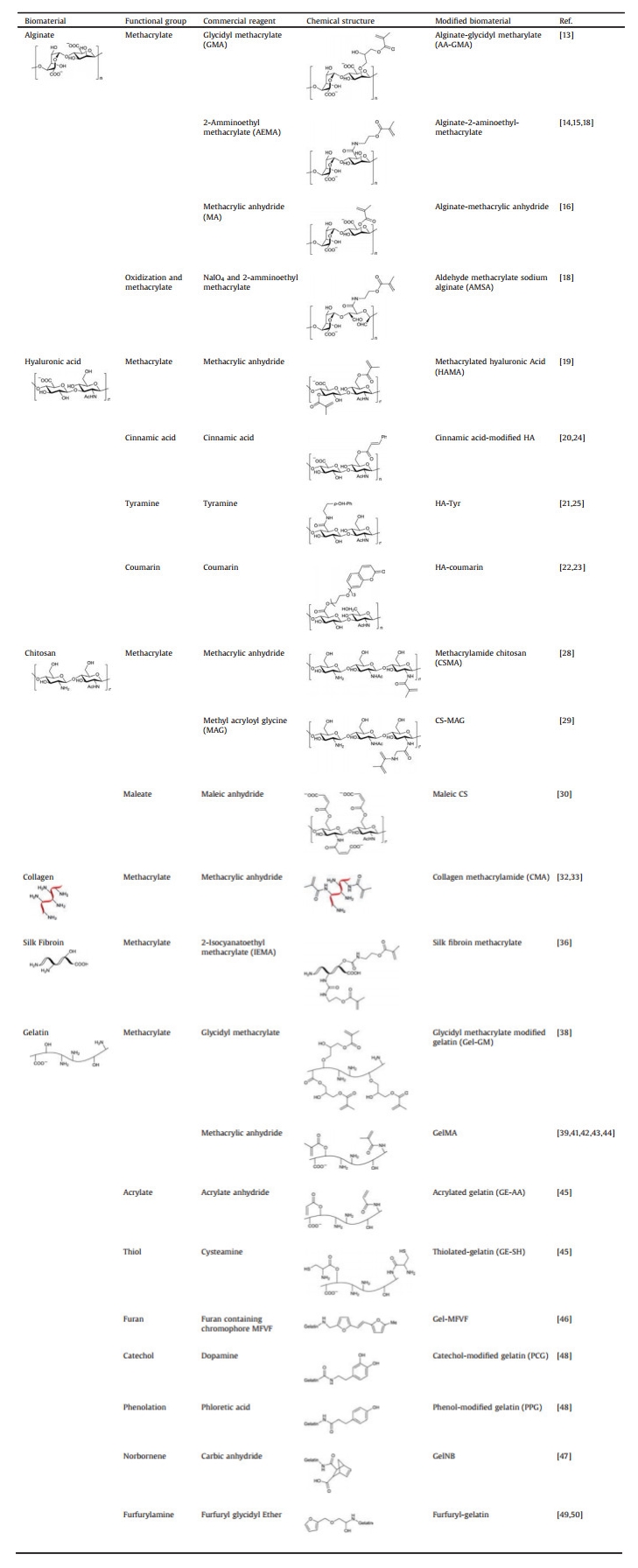
2. Biomaterials for photo-crosslinkable hydrogelsPhoto-crosslinkable hydrogels are water-soluble photo-crosslinkable polymers with two or more reactive groups. A variety of naturally derived and synthetic materials can be used to form photo-crosslinkable hydrogels. Representative naturally derived materials include alginate, hyaluronic acid, chitosan, collagen, silk fibroin and gelatin. Synthetic materials include Pluronic [8], poly(vinyl alcohol) (PVA), poly(acrylic acid) (PAA), polyethylene glycol (PEG) and polypeptides [4]. In this review, we focus on some representative naturally derived materials and their photo-crosslinkable hydrogels which are also summarized in Table 1.
|
|
Table 1 Recently reported photo-crosslinkable hydrogels derived from chemically modified natural biomaterials. |
Alginate (ALG) is a linear polysaccharide, which is sourced from brown algae. Structurally, it consists of co-polymers composed of the polysaccharides (1, 4)-linked β-D-mannuronic acid (M) and α-L-guluronic acid (G) [9]. Alginate hydrogels can be easily prepared through ionic crosslinking between carboxylate anion and divalent cations such as Ca2+ [4, 10-12].
To increase the stability and avoid dissolution of alginate hydrogels, photo-crosslinkable methacrylated alginate is synthesized by the introduction of methacrylate groups via modification of its carboxyl or hydroxyl side groups, which is more stable than Ca2+ crosslinked alginate. These different methacrylate groups include glycidyl methacrylate [13], 2-aminoethyl methacrylate [14, 15] and methacrylic anhydride [16]. As a result, three methacrylated alginate hydrogels are synthesized: alginate-glycidyl methacrylate, alginate-2-aminoethyl methacrylate and alginate-methacrylic anhydride [7]. Methacrylated alginate can be photo-crosslinked either by argon ion laser (514 nm) or ultraviolet (245 nm) irradiation in the presence of photo-initiating systems [17].
On the other hand, alginate can be composited with other types of hydrogels to achieve photo-crosslinking. Yuan et al. synthesized a double crosslinked network hydrogel from aldehyde methacrylate sodium alginate (AMSA) and ethylenediamine modified gelatin (AG). The AMSA was synthesized via a two-step process: oxidization to form an aldehyde sodium alginate, and then functionalization with methacrylate groups. Next, the primary network was formed through the reaction between amino groups in AG and aldehyde groups in AMSA via the Schiff-base reaction. The hydrogel structure with the double crosslinked network was formed through radical reaction of methacrylate groups in AMSA initiated by a 365 nm UV light [18].
2.1.2. Hyaluronic acidHyaluronic acid (HA) is an anionic and non-sulfated glycosaminoglycan, composed of alternating units of D-glucuronic acid and D-N-acetylglucosamine, linked via alternating β-1, 4 and β-1, 3 glycosidic bonds. HA is one of the major components of extracellular matrix of skin, cartilage and vitreous humor [1, 9, 11].
HA can be chemically modified with photo-crosslinkable functional groups such as methacrylic groups and glycidyl methacrylic groups, which enable feasible crosslinking of the resulting HA derivative via exposure to visible or ultraviolet light [19]. HA has also been extensively derivatized with photoactive moieties, including cinnamic acid [20], tyramine [21], coumarin [22, 23] and hexamethylenediamine. Cinnamic acid, as a photo-crosslinkable residue, undergoes photo-dimerization upon UV irradiation through [2+2] cycloaddition reactions with the formation of cyclobutane rings. Cinnamic acid-modified HA obtains a photo-crosslinkable derivative without an initiator, which results in a water-insoluble hydrogel with good biocompatibility [24]. Tyramine (Tyr) is a phenolic photoactive moiety. HA-Tyr hydrogel is synthetized via a photochemical process initiated by riboflavin through the formation of dityramines [25]. Visible light (500 nm) is also utilized to form covalent HA-Tyr networks [21]. Coumarin, as a photocleavable group, has been grafted onto HA chains to form photo/thermo-responsive nanogels [26]. For example, Beninatto et al. demonstrated that HA-coumarin could crosslink upon near-UV irradiation via a [2+2] photo-induced cycloaddition reaction without an initiator [23].
2.1.3. ChitosanChitosan is a linear polysaccharide produced from renewable sources such as shell of shellfish, crustacean shells, insect cuticles, mushrooms envelopes and the wastes of the seafood industry, however, commercially available chitosan is produced from deacetylation of chitin. Chitosan is a cationic hydrogel composed of randomly arranged β-(1, 4)-linked d-glucosamine and N-acetyl-d-glucosamine units, varying in composition, sequence and molecular chain length [9, 11, 27].
There are lots of methods to modify chitosan to obtain photo-crosslinkable chitosan. For example, methacrylamide chitosan (CSMA) is synthesized by reacting chitosan primary amine groups with methacrylic anhydride [28]. In another way, Qi et al. fabricated chitosan grafted methyl acryloyl glycine (MAG) photo-crosslinkable hydrogels [29]. Moreover, the photo-clickable thiol-ene hydrogels based chitosan were synthesized using photo-crosslinkable maleic chitosan (MCS) and thiol-terminated poly(vinyl alcohol) (TPVA) in the presence of a biocompatible photo-initiator [30].
2.2. Polypeptide-based photo-crosslinkable biomaterials 2.2.1. CollagenCollagen is a fibrous protein, the major component of ECM and presents different morphologies in different tissues. Inherent in the structure of collagen fibers are arginine-glycine-aspartic acid (RGD) amino acid sequences [4, 9]. Collagen can be crosslinked by temperature or pH change or even by using vitamin Riboflavin. Moreover, collagen can also be crosslinked via UV irradiation. Nagaraj et al. fabricated crosslinked collagen through visible light irradiation using erythrosine B (EB) and methylene blue (MB) as the photo-sensitizer in the presence of ammonium persulphate (APS) [31]. Collagen can also be modified with methacrylamide (MA) [32]. For example, Yang et al. fabricated photo-crosslinked methacrylamide-modified collagen hydrogel to control actin traction and subsequent contraction of the collagen matrix and differentiation of BMSCs toward chondrogenic lineage [33].
2.2.2. Silk fibroinSilk fibroin (SF) is a class of proteins produced from insects, spiders and worms, which are composed of fibroin (70%–80%) and sericin (20%–30%) [9]. In the same way, photo-crosslinking methods have also been applied for the fabrication of SF-based hydrogels. The first strategy to produce photo-crosslinkable SF accompanies by covalent bond formation between active photo-reaction groups of SFs, mainly tyrosine residues, in the presence of reducing agents. Whittaker et al. suggested the facile ruthenium-catalyzed photo-crosslinking method for the fabrication of photo-crosslinkable SF hydrogel. Under visible light and tris(2, 2-bipyridyl)dichlororuthenium(II) hexahydrate as a catalyst in SF solution, tyrosine radicals can be formed, thereby allowing formation of SF hydrogel through dityrosine links [34]. Another study suggested flavin-mononucleotide photo-initiator, a nontoxic water-soluble variant of riboflavin, could be used for SF hydrogel crosslinking [35]. The second strategy comprises techniques involving chemical modification of SF chains which transform reactive amino acid residues into reactive olefinic functional groups such as methacrylate and further subjected to photo-crosslinking [36].
2.2.3. GelatinGelatin, a denatured protein, is derived from collagen. It consists of 18 different amino acids characterized by a repetitive unit of glycine-X-Y, in which X and Y predominantly consist of proline and hydroxyproline which enables the formation of triple helices or physical crosslinks via interchain hydrogen bonds [4, 9, 37].
The carboxyl groups in aspartic acid and glutamic acid, hydroxyl groups of serine and threonine, amino groups of lysine can be used for a variety of modifications to incorporate photo-crosslinkable groups. The carboxyl and hydroxyl groups can react with glycidyl methacrylate to synthesize photo-crosslinkable gelatin derivatives [38]. Photo-crosslinkable methacrylated gelatin (GelMA) can be synthesized by forming covalent bonds between the lysine aminos of gelatin and methacrylic anhydride [39, 40]. The synthesized photo-crosslinkable GelMA can be crosslinked in the presence of a photo-initiator upon applying UV irradiation. Irgacure 2959 is the commonly used photo-initiator for the photo-crosslinking [41-44].
Besides GelMA, there are many other photo-crosslinked gelatins. For example, Li et al. provided a facile method to modify gelatin with acrylate anhydride and cysteamine by grafting free vinyl groups and thiol groups to the gelatin backbone to prepare acrylated gelatin (GE-AA) and thiolated gelatin (GE-SH) respectively. The mixtures of the two precursors were then cross-linked via a photoinitiated thiol-acrylate reaction [45]. García-Astrain et al. reported a method to produce a novel photo-crosslinked gelatin. In their research, gelatin was modified with a furan containing chromophore MFVF (5-[2-(5-methyl furylene vinylene)]furancarboxyaldehyde). Schiff base formation between the primary amino groups of gelatins and the aldehyde groups of MFVF was conducted, resulting in a photo-sensitive cross-linkable hydrogel by near UV-light irradiation in absence of initiators [46]. Moreover, Greene et al. have prepared highly tunable gelatin-based hydrogels using orthogonal thiol-norbornene photochemistry [47]. Besides these gelatin-based photo-crosslinkable hydrogels which do not need a photo-initiator, there are many other types that require different photo-initiators. Koshy et al. synthesized catechol-modified gelatin (PCG) and phenol-modified gelatin (PPG) via EDC/NHS chemistry which crosslinks via ruthenium-based photochemistry [48]. Moreover, AnilKumar et al. used furfuryl-gelatin as a novel, visible-light crosslinkable hydrogel. Either rose bengal or riboflavin was used as a visible-light photo-initiator [49, 50].
3. Crosslinking mechanisms of photo-crosslinkable hydrogelsHydrogel crosslinking (gelation) is an important aspect in preserving the shape of a fabrication construct thereby minimizing structure collapse. The different gelation mechanisms can be subdivided into two categories: physical and chemical crosslinking. Chemical crosslinked hydrogels are formed through chemical reactions and the formation of covalent bonds that constitute the network. To induce chemical crosslinking, typical methods include UV irradiation, dehydrothermal treatment, and addition of crosslinkers. Physically crosslinked hydrogels have their networks held together by molecular entanglements and/or secondary forces like ionic, hydrogen bonds and hydrophobic interactions [1]. Although photo-crosslinking method belongs to chemical crosslinking, physical crosslinking still exists in photo-crosslinkable hydrogels. For example, thermal gelation for collagen [51] and ultrasonic crosslinking for silk fibroin [52].
Photo-crosslinking is one of the chemical crosslinking methods of creating hydrogels, through the exposure of a photosensitive system composed of unsaturated prepolymers, photo-initiators, and other compounds such as cells and therapeutic molecules to ultraviolet or visible light. Photo-crosslinking used for the synthesis of bio-compatible hydrogels can be classified into free-radical-initiated chain polymerization and bio-orthogonal click reactions.
3.1. Free-radical photopolymerizationFree-radical photopolymerization, based on (meth)acrylate functionalized prepolymers and occurring through a chain-growth mechanism, is the most popular method for producing hydrogels. The incident light energy (photons) which is absorbed by the photo-initiators triggers the formation of free radicals that react with vinyl bonds present in the prepolymer, promoting the establishment of chemical crosslinks between polymer chains. However, radical-initiated chain-growth polymerization has several limitations, including: (1) relatively poor control over the crosslinking kinetics, (2) oxygen inhibition, (3) the presence of unreacted double bonds that might potentially react with biological substances, and (4) the generation of heterogeneities within the polymer network, due to the random chain polymerization.
3.2. Bio-orthogonal click reactionsBio-orthogonal click reactions are characterized by orthogonal reactivity and the step-growth mechanism of the polymerization reaction, enabling the fabrication of structurally uniform hydrogels with minimal network defects. The thiol-norbornene (thiol-ene) photo-click reaction has emerged as an important method among the bio-orthogonal click schemes. This reaction involves light-mediated orthogonal reactions between multi-functionalized macromers that are end-capped with norbornene functionalities and sulfhydryl-containing linkers, in the presence of low amounts of photo-initiator. Under UV or visible light irradiation, the thiol-ene reaction promotes the rapid radical-mediated addition of thiols to carbon-carbon double bonds within functionalized prepolymers, yielding thioether bonds. In addition, click chemistry is insensitive to water and oxygen, and can proceed under mild reaction conditions with higher efficiency, selectivity, and faster kinetics when compared to free-radical polymerization [53].
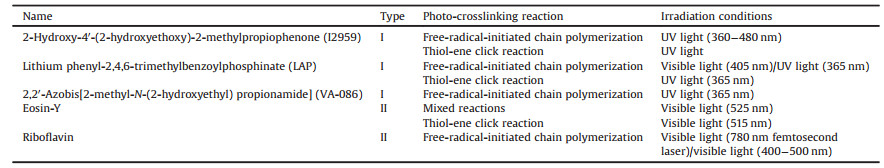
Currently available photo-initiators can be divided into two main categories: radical or cationic. Radical photo-initiators are the most used, due to their superior biocompatibility. On the other hand, the use of cationic photo-initiators results in the formation of protonic acids, making these photo-initiators less useful in biomedical applications. According to the mechanism involved in the generation of free radicals, radical photo-initiators can be further categorized as photo-cleavable photo-initiators (type I) or bimolecular photo-initiators (type II). Upon light exposure, type I photo-initiators (e.g., benzoin derivatives, benzylketal, acetophenone derivatives, hydroxyalkylphenones and acylphosphine oxides) absorb incident photons and decompose into two primary radicals that initiate the crosslinking, whereas type II photo-initiators (e.g., benzophenone, camphor quinone and thioxanthone) abstract the hydrogen from a co-initiator to generate secondary radicals for cross-linking. Some common photo-initiators are listed in Table 2.
|
|
Table 2 Summary of the most common photo-initiators of photo-crosslinking system. |
The definition of biofabrication is that the automated generation of biologically functional products with structural organization from living cells, bioactive molecules, biomaterials, cell aggregates such as micro-tissues, or hybrid cell-material constructs, through bioprinting or bio-assembly and subsequent tissue maturation processes [54-58]. Here we focus on the most important and best-established techniques for biofabrication of photo-crosslinkable hydrogels.
4.1. Traditional biofabrication methods for photo-crosslinkable hydrogelsTraditional biofabrication methods for photo-crosslinkable hydrogels usually crosslink hydrogels through ordinary light source. The representative technology is 3D bioprinting [59]. The process of 3D bioprinting can be divided into three steps: (1) preprocessing for preparation of the bio-ink and generation of the computer-aided design (CAD) "blueprint"; (2) the processing step, which typically involves the printing of the 3D structure; and (3) post-processing, such as photo-crosslinking [60]. To date, several 3D bioprinting methods have been developed, among them, inkjet 3D bioprinting and extrusion 3D bioprinting techniques can be used to biofabricate photo-crosslinking hydrogels [61].
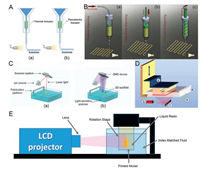
4.1.1. Inkjet 3D bioprintingInkjet 3D bioprinting, also referred to as drop-by-drop 3D bioprinting, which printing the 3D constructs layer by layer by depositing ink drops on successive layers. Inkjet bioprinter uses thermal or piezoelectric forces to eject droplets of photo-crosslinkable hydrogels from a print head nozzle to receiving platform (Fig. 1A) [62]. The hydrogels will crosslink instantly upon its exposure to a light source [60, 63, 64]. However, the limiting factors of inkjet are the demand for low viscosity inks and higher extrusion flow and smaller nozzles which induce damage due to shear stress on cell membranes [59].
|
Download:
|
| Fig. 1. Biofabrication techniques for photo-crosslinkable hydrogels. (A) Inkjet 3D bioprinting: (a) Thermal actuators, (b) piezoelectric actuators. Reproduced with permission [62]. Copyright 2018, Elsevier. (B) Extrusion 3D bioprinting: (a) Pneumatic, (b) Piston-driven, (c) screw-driven. Reproduced with permission [62]. Copyright 2018, Elsevier. (C) Stereolithography: (a) Laser-writing, (b) mask-based writing. Reproduced with permission [53]. Copyright 2015, Elsevier. (D) Digital light processing consisting of (a) vat filled with photopolymer resin, (b) light source, (c) micromirror array, (d) vertically movable building platform and (e) tilting device to replenish the uncured bottom layer. Reproduced with permission [77]. Copyright 2017, American Chemical Society. (E) Computed axial lithography. Reproduced with permission [80]. Copyright 2020, MDPI. | |
Extrusion 3D bioprinting is defined as a process wherein the hydrogel is extruded through a nozzle with the aid of mechanical force (stepper motor, piston, pneumatic devices or actuating rollers [65]), which results in production of well-defined filaments (diameters in a range of 150–300 μm [66]) [67-71] (Fig. 1B). The extruded filaments must necessarily exhibit structural integrity and possess the ability of self-support, in order to prevent ink spreading or structural collapsing [62]. To address this challenge, photo-crosslinking is usually required to improve the mechanical robustness and structural fidelity of 3D printed objects [63, 72]. Crossl-inking can be triggered before (pre-crosslink), after (post-crosslink) or during (in situ crosslink) extrusion. However, this method has limitations as well. First of all, cell damage and death as well as cell sedimentation and aggregation exist due to the shear stress and small orifice diameter of the nozzles used to deliver the bio-ink. Also, the printing resolution is limited by the physical confinement of the nozzles and it may occur potential nozzle clogging [73].
4.2. Laser-assisted biofabrication methods for photo-crosslinkable hydrogels 4.2.1. Stereolithography (SLA)Stereolithography is a solid freeform fabrication technique that employs a reservoir containing photo-crosslinkable hydrogel pre-solution, a laser with X-Y control and a stage or fabrication platform with vertical control. The curing reaction is triggered by the incidence of light with an appropriate wavelength (UV, visible or near-infrared), intensity and duration. Fabrication occurs at the surface of the structure and the stages lowers incrementally, allowing layers to be crosslinked on top of each other, thus creating 3D structures in a bottom-up fashion. Stereolithographic processes produce 3D structures in a layer-by-layer fashion using two fundamental irradiation approaches: direct or laser writing and mask-based writing (Fig. 1C). The first employs a focused laser beam to selectively induce the polymerization of a liquid photopolymer, while the second transfers an entire image to a liquid prepolymer by irradiating through a patterned mask that contains transparent zones corresponding to the sections of the model to be built. The major advantage of the mask-based approach lies in the fast processing and the low-density flux of light over the prepolymer, which avoid undesirable polymerizations [53]. One special type of SLA system is two-photon polymerization (TPP), which utilizes a focused near-infrared femtosecond laser with a wavelength of 800 nm to induce a nonlinear optical effect. Although the object size is limited, TPP offers the possibility to produce constructs with spatial resolutions as small as 100 nm [5, 74, 75]. While SLA possesses numerous advantages, such as good biocompatibility, high resolution and great efficiency, there still remain some challenges to be addressed [76]. For example, it will take a long time to manufacture large structures [59]. To solve this problem, a new version of top-down SLA using digital light projectors that has recently attracted attention is continuous liquid interface production (CLIP). CLIP utilizes an oxygen-permeable window delivering oxygen to the glass-hydrogel interface. The oxygen inhibits the polymerization reaction, creating a persistent liquid interface, allowing in combination with precise process timing print speeds to be further increased [5].
4.2.2. Digital light processing (DLP)Unlike SLA, which uses a point light source to pattern only one voxel at a time and print patterns with its movement, DLP can solidify an entire pattern of one layer by using micro-mirror array devices or dynamic liquid-crystal masks that can directly form a mask pattern (Fig. 1D) [77]. The throughput of the SLA process is slow due to the point-by-point scanning nature of the direct-write of the laser system while the DLP exploits a digital micro mirror-array device (DMD), to produce a dynamic digital mask: An entire part cross section can be cured at one time, resulting in a faster process than scanning a laser beam [78, 79]. Initially, the illumination of the photo-crosslinkable hydrogels was conducted from the top of the vat, while the resulting model was immersed in a liquid composition. In other words, it created 3D structures in an up-bottom fashion. However, this approach required significant amounts of photosetting material, and significantly limited the height of the print.
4.2.3. Computed axial lithography (CAL)Recently, a novel volumetric additive manufacturing method named CAL was developed which can achieve the concurrent printing of all points within a 3D object by illuminating a rotating volume of photosensitive material with a dynamically evolving light pattern (Fig. 1E). Unlike conventional vat photopolymerization that prints products layer by layer, CAL delivered light energy to the material volume as a series of 2D images [80]. Each image projection propagates through the material from a different angle. The superposition of light energy results in the solidification of the whole entity according to the designed geometries at one time. This volumetric additive manufacturing method is several orders of magnitude faster than the layer-by-layer method [81, 82].
5. Biological applications of photo-crosslinkable hydrogelsBiofabrication has the potential to engineer heterogeneous tissue structures, including shape-based tissues such as bone, cartilage, skin and cornea; organized tissues such as skeletal muscle and neural tissues; composite tissues such as osteochondral and musculotendinous tissues; and whole organs with vasculature and functional inner structures [83-86]. In the following, we will introduce some representative applications of photo-crosslinkable hydrogels in tissue engineering.
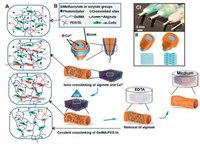
5.1. VascularThe incorporation of vascular networks in biofabricated constructs is crucial for whole organ bioprinting, which can provide cells with sufficient nutrition, growth factors, oxygen and remove waste [87]. To create the lumen of the vasculature network, one method is using a sacrificial material to create the 3D inter-connecting network, which can be removed after building the surrounding construct, leaving hollow channels for the perfusion of endothelial cells for endothelialization of the blood vessels [64]. The most commonly used sacrificial bioink is Pluronic. Once the constructs were fabricated and photo-crosslinked, the temperature was lowered to 4 ℃ to liquefy and remove the Pluronic, leaving behind open vascular channels. The resulting channels were then perfused with human umbilical vein endothelial cells (HUVECs), which attached to the channel walls, thereby resulting in vascularization of the complex bioprinted construct [60, 88]. Moreover, Jia et al. combined GelMA, sodium alginate and 4-arm poly(ethylene glycol)-tetra-acrylate (PEGTA) to fabricate complex multilayered 3D perfusable hollow vascular structures through using a multilayered coaxial extrusion system (Fig. 2). Among them, sodium alginate, which is sacrificial compound, can be removed by EDTA. This blend bioink displayed favorable biological characteristics that supported the spreading and proliferation of encapsulated endothelial and stem cells [89]. However, this tissue is limited in size owing to difficulties associated with direct perfusion [90]. Another method to build vasculature system is using novel biofabrication method directly without any sacrificial material. Zhu et al. created pre-vascularized tissues with complex three-dimensional (3D) microarchitectures using a rapid bioprinting method-microscale continuous optical bioprinting (μCOB) with a mixed GM-HA and GelMA bioink [91]. Despite significant improvements of current technologies to create three-dimensional (3D) blood vessels, the formation of a functional engineered vascular system with multiscale vessel networks from capillaries to large vessels has remained challenging in this field [92].
|
Download:
|
| Fig. 2. (A) Two independent crosslinking processes of the bioink, where alginate, GelMA and 4-arm PEGTA are ionically and covalently crosslinked, respectively, upon exposure to CaCl2 solution and UV light. (B) The procedure of bioprinting perfusable hollow tubes with the cell-encapsulating blend bioink and subsequent vascular formation. (C) The designed multilayered coaxial nozzles and biofabrication of perfusable hollow tubes with constant diameters and changeable sizes. Copied with permission [89]. Copyright 2016, Elsevier. | |
So far, heart transplantation is the only therapy choice for patients with heart failure at the end stage. However, given the fact that the number of available cardiac donors is quite limited, it is of huge demand to develop novel strategies to regenerate/repair infarcted hearts, thereby reducing the excessive dependence on cardiac donors. Increasing attentions have been paid to biofabrication, a very promising method to create customized cardiac valves, patches or even whole heart. Engineering cardiac tissues and organ models remains a great challenge due to the hierarchical structure of the native myocardium [93].
In some preliminary research, photo-crosslinkable hydrogels are mixed together to form a cell-carrying structure like heart tissue. Noshadi et al. used GelMA hydrogel for myocardial repair via visible-light-initiated photo-crosslinking [94]. On this basis Anil Kumar et al. added fibrin to a photo-crosslinkable furfuryl-gelatin hydrogel to fabricate cardiac cell-laden constructs seeded with human induced pluripotent stem cell-derived cardiomyocytes (iPS-CM) or CM cell lines with cardiac fibroblasts (CF) [95]. Gao et al. used multiphoton-excited 3D printing to generate a human-induced pluripotent stem cell-derived cardiac muscle patch (hCMP) which is based on GelMA hydrogels and seeded with cardiomyocytes, smooth muscle cells, and endothelial cells [96].
Moreover, other photo-crosslinkable hydrogels which can fabricate the structure with rhythmic contractions, same as heart, have also been developed. Zhang et al. developed endothelialized myocardial tissues through composite alginate-GelMA bioink seeded with cardiomyocytes, inducing the formation of myocardium with improved alignment capable of spontaneous and synchronous contraction [97]. Annabi et al. combined methacrylated tropoelastin (MeTro) hydrogels with microfabrication techniques with the aim of developing highly elastic micro-engineered hydrogels for cardiac tissue engineering applications. Furthermore, they found that the system is compatible with electrical stimulation, which is used to modulate the contractile properties of CMs seeded on these MeTro hydrogels [98].
5.3. NerveNerve repair and regeneration remain challenging problems worldwide, largely because of the extremely weak inherent regenerative capacity and accompanying fibrosis of native nerves. And large synthetic tissues will need to integrate with the host nervous system, biofabrication may be a means to generate new nervous tissue or to enhance the innervation of tissue engineered constructs [88]. Until now, there is less research on nerve tissue engineering, which needs more in-depth research urgently. Neural stem cells (NSCs) were employed as the primary cell source for nervous tissue engineer because of their ability to terminally differentiate into a variety of cell types including neurons, astrocytes, and oligodendrocytes, which is promising for advancing nerve repair and regeneration in the future [99]. Koffler et al. reported the use of a microscale continuous projection printing method (μCPP) to create a complex CNS structure which was biofabricated by PEGDA-GelMA hydrogels for regenerative medicine applications in the spinal cord [100].
5.4. Skin and wound dressingSkin tissue engineering is one of the most promising examples of biofabrication for several applications, such as development of topical drugs, wound healing studies and dermal toxicology research [93, 101]. Currently, natural hydrogels used to support epidermal regeneration are mainly collagen-or gelatin-based, which mimic the natural dermal extracellular matrix but often suffer from insufficient and uncontrollable mechanical and degradation properties [102]. In order to solve these problems, some photo-crosslinkable hydrogels have been developed to biofabricate skin tissues. Boddupalli et al. fabricated photo-crosslinked methacrylated alginate (ALGMA) hydrogels, which have the ability to tune collagen organization. It was proved that ALGMA can be ideal hydrogels ranging from corneal wound healing to epithelial wound dressing [103]. Eke et al. proved that GelMA/HAMA hydrogels loaded with adipose derived stem cells (ADSC) have the desired proliferative and angiogenic properties, which essential to wound healing and improve survival of tissue engineered skin [104].
In addition to hydrogels, biofabrication methods have also been improved. Several research have proved the potential of inkjet 3D bio-printing for engineering skin tissue has been demonstrated [60]. Moreover, Kim et al. fabricated a bi-layer scaffold composed of human hair keratin/chitosan nanofiber mat and gelatin methacrylate (GelMA) hydrogel with fibroblasts and HaCaT cells by using electrospinning and photopolymerization, to mimic dermis and epidermis of skin tissue [105, 106].
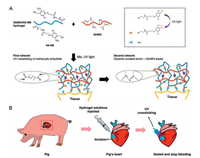
The 'ideal' wound dressing should absorb excess exudate and toxins, keep a good moisture between the wound and the dressing, preserve the wound from external sources of infection, prevent excess heat at the wound, have good permeability to gases, be supplied completely sterile and be easy to remove without further trauma to the wound [2]. Hydrogels hold exciting potential for wound healing applications in maintaining a moist environment conducive of accelerated healing with high biocompatibility to facilitate cell migration, proliferation and re-epithelialization [107]. Furthermore, their ability to encapsulate cells, growth factors and anti-bacterial holds tremendous promise for the bioprinting of customizable wound dressings [108, 109]. Hong et al. reported a photo-crosslinked hydrogels used as heart wound dressings, which composed of GelMA, N-(2-aminoethyl)-4-(4-(hydroxymethyl)-2-methoxy-5-nitrosophenoxy) butanamide (NB) linked to the glycosaminoglycan hyaluronic acid (HA-NB) with photo-initiator LAP (Fig. 3) [110]. Moreover, Chang et al. investigated the photo-crosslinkable hydrogel (GE-BTHE mixture) containing GE-BTHE, PEGDA, chitosan and methylene blue (MB), to protect wounds from infection that can serve as a convenient excisional wound dressing with antibacterial efficacy. GE-BTHE was synthesized by gelatin and a conjugate formed from the 3, 3′, 4, 4′-benzophenone tetracarboxylic dianhydride (BTDA) and the 2-hydroxyethyl methacrylate (HEMA). This composited hydrogel was found to have fast film-forming property attributed to the photochemical reaction between GE-BTHE and PEGDA, as well as the antibacterial activity in vitro attributed to the ingredients of chitosan and MB. Therefore, it has the potential to treat nonhealing wounds, deep burns, diabetic ulcers and a variety of mucosal wounds [111].
|
Download:
|
| Fig. 3. Heart wound dressing: (A) Constituent chemical structures and a schematic diagram illustrating the formation of the photo-crosslinked hydrogel. (B) Hemostatic properties of the matrix gel in a pig cardiac puncture injury model. Reproduced with permission [110]. Copyright 2019, Springer Nature. | |
Bone defects have become one of the leading causes of disability among people worldwide. Although autografting is regarded as the gold standard for bone defect repair, it is limited by the donor morbidity and uncertain adverse effects. Therefore, bone tissue engineering has attracted considerable attention from researchers as a promising strategy for repairing bone defects without the limitations and short comings of using bone autografts, allografts, or xenografts. The overarching goal in the area of bone engineering is to develop tough and native-like 3D biomaterials. Inspired by the combination of the soft and hard components of skeletal tissues, mineral-based nanocomposite hydrogels have recently emerged as promising biomaterials for skeletal-tissue engineering. Thus, it is anticipated that future advancements in skeletal-tissue engineering will rely on 3D-culturing systems containing mineral-based nanomaterials, such as tricalcium phosphate, nano-hydroxyapatite and bioactive glass [112, 113].
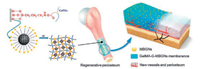
Nano-hydroxyapatite (nHA) is a naturally occurring ceramic that largely constitutes the inorganic phase of bone. A large number of photo-crosslinkable hydrogels have therefore involved the use of nHA to closely mimic the structural and elemental framework of natural bone [114, 115]. The composite hydrogel consisted of GelMA and nHA presented a lower swelling ratio, higher mechanical moduli and better biocompatibility when compared to the pure GelMA hydrogel [116-118]. Despite the extensive use of nHA in skeletal tissue engineering their slow dissolution has always been a practical concern [117, 119, 120]. Thus, more readily degradable calcium phosphate bioactive minerals likeβ-tricalcium phosphate (β-TCP) and biphasic calcium phosphate (BCP) (a mix of hydroxyapatite and β-TCP) have been sought-out to treat bone-related injuries [121]. Furthermore, mesoporous bioactive glass nanoparticles (MBGNs) chemically modified with GelMA were further incorporated into GelMA to fabricate an organic/inorganic co-cross-linked hydrogel membrane (GelMA-G-MBGNs). The GelMA-G-MBGNs hydrogel membrane displayed better mechanical property, durable degradation time, pH stable, biomineralization and long-term ion release (Fig. 4) [122].
|
Download:
|
| Fig. 4. Inorganic strengthened hydrogel membrane for regenerative periosteum using GelMA, sodium alginate and 4-arm poly(ethylene glycol)-tetra-acrylate (PEGTA) composited hydrogels. Copied with permission [122]. Copyright 2017, American Chemical Society. | |
Overall, a combination of strong mechanical properties, good biocompatible, not prone to immune rejection and high osteo-conductivity renders ceramics as promising nanofillers in hydrogel-based orthopedic therapies. In addition, these materials also maintain a high physical integrity during the healing process and can merge systemically with surrounding hard tissues. In order to extend the clinical applicability of nanocomposite-based hydrogel grafts, more research efforts are needed to address the challenges associated with their suboptimal mechanical strength, degradability and elasticity.
5.6. CartilageCartilage is a smooth elastic connective tissue as the structural and functional component of many specific body parts such as ear, nose and articular cartilage. Cartilage appears as a relatively simple tissue: It has no blood vessels or nerves. Cartilage tissue has no self-repair capacity and once cartilage defect emerges, it will finally cause the degenerative and osteoarthritic changes [93].
GelMA hydrogels have been used for 3D culture of chondrocytes for cartilage tissue engineering [81, 123, 124]. Actually, common hydrogels used in cartilage engineering are composited hydrogel which usually including photo-crosslinkable hydrogels, growth factors and other materials. Abbadessa et al. showed a composited hydrogel used for cartilage tissue formation in vitro. This UV cross-linked hydrogel based on chondroitin sulfate (CS), hyaluronic acid (HA), both were methacrylated, and thermosensitive methacrylated pHPMA-lac-PEG triblock copolymer, was laden with equine chondrocytes [125]. Sridhar et al. designed a hybrid bio-synthetic system consisting of poly(ethylene glycol) (PEG) end capped with thiols and crosslinked by norbornene-functionalized gelatin via a thiol-ene photopolymerization. The results indicated the potential of a hybrid PEG-gelatin hydrogel to permit chondrocyte-mediated remodeling and promote articular cartilage matrix production [126]. Zhou et al. firstly synthesized the photo-crosslinkable materials with vinyl groups, i.e., maleilated chitosan (MCS) and methacrylated silk fibroin (MSF) micro/nanoparticles. The results demonstrated that the micro/nanocomposite hydrogels with TGF-β1 was biocompatible to mouse articular chondrocytes, indicated their potential as tissue engineering scaffolds for cartilage repair [127].
5.7. Cancer modelTo implement basic cancer research and develop more effective cancer therapy, new methodologies are needed prior to clinical trials, for example, establishment of various in vivo cancer-bearing animal models and in vitro cancer models. The cancer models can provide promising cancer research platforms with no need for the animal models, thus being able to largely reduce the use of animals in cancer research; however, it is still a challenge to create fully functional cancer models in imitation/replication of the anatomical structure, biological feature and drug response of human solid tumors. Biofabrication is a powerful way to tackle this issue through creating 3D tissue engineered bio-constructs that are able to mimic/recapitulate the heterogeneity, vascular networks, multicellular spheroids and native ECM of solid tumor tissues [60, 128, 129].
For example, a breast cancer model can be created by the direct bioprinting of cell spheroids composed of breast cancer cells in the core, mammary fibroblasts and adipose cells into multi-well plates. To mimic the structure of breast tumor microenvironment, a novel double-network poly(Nε-acryloyl-l-lysine)/hyaluronic acid (pLysAAm/HA) hydrogels were fabricated by a two-step photo-crosslinking process for in vitro three-dimensional (3D) cell culture which can provide a 3D microenvironment for MCF-7 cells and is more representative of the in vivo breast cancer [130]. Furthermore, Zhou et al. developed a biomimetic bone matrix using tabletop stereolithography to investigate the interaction between breast cancer (BrCa) cells and bone stromal cells (fetal osteoblasts and human bone marrow mesenchymal stem cells (MSCs)). A series of bone matrices were consisted of osteoblasts or MSCs encapsulated in GelMA hydrogel with nano hydroxyapatite (nHA). The results demonstrate that the biofabricated matrix provides a suitable model with which to study the interactive effects of cells in the context of an artificial bone microenvironment and thus may serve as a valuable tool for the investigation of post metastatic breast cancer progression in bone [131].
There are other more tumor models that are being studied. Shih et al. utilized this biomimetic modular gelation system, which consisted of norbornene-functionalized gelatin (GelNB) or thiolated hyaluronic acid (THA) and EY to formulate artificial tumor niche and to study the effects of cell density and gel modulus on the formation of pancreatic ductal adenocarcinoma (PDAC) spheroids [132]. Cao et al. recently developed a tumor-on-a-chip with 3D bioprinted blood and lymphatic vessel pair (TOC-BBL) in a 3D hydrogel matrix based on GelMA [133].
6. Challenges and prospectsDespite all the progress over the years in tissue engineering, many challenges still remain unsolved. Challenges fall into two main categories: (1) biomanufacturing which involves 3D fabrication of the cells and biomaterials and (2) in vivo integration which involves post-implantation functionality and integration. One challenge in fabrication process is nozzle clogging in nozzle-based fabrication methods and needing long time to fabricate multiple materials. Another challenge is that the 3D constructs (such as hard tissue repair) need to be sufficiently stable and mechanically rigid to ensure successful transplantation. If the scaffold is not capable of maintaining its structure and provide mechanical support, any newly formed tissue will probably fail as a result of scaffold deformation. In terms of cell activity in photo-crosslinked hydrogels, biofabrication methods and addition agents both may do harm for cell. For example, the potential deleterious effects of light irradiation (the short wavelengths of UV light) for cell, which could also lead to local temperature increases, subsequently damage neighboring cells and tissue. Moreover, cell damage and death as well as cell sedimentation and aggregation exist due to the shear stress and the small orifice diameter of the nozzles used to deliver the hydrogel [60]. Cytotoxicity of photo-crosslinkable hydrogels also generated by the dissociation of photo-initiators, local inflammation due to unreacted double bonds, lack of control over the crosslinking kinetic. In addition, because of the limitation of the poor penetrability of UV or visible light, the internal applications of the photo-crosslinkable hydrogels will be inhibited in minimally invasive surgery. Nowadays, researches are working to overcome these detriments through the selection of appropriate photo-initiators, concentration, light wavelengths, intensity and irradiation time [134].
However, as more research is done on biofabrication techniques, biofabrication methods and quality of photo-crosslinkable hydrogels will eventually improve, providing capability to fabricate more complex 3D constructs. By further advancement of biofabrication methods, biofabrication of complex constructs accurately mimicking natural organs becomes practical, due to the capability to load different type of cells in different zones and on compatible photo-crosslinkable hydrogels. In addition, the photo-initiator of photo-crosslinking system will also overcome its cytotoxicity and gain higher biocompatibility. Enabled by the biofabrication technology of photo-crosslinkable hydrogels, customized tissue/organ grafts and drug delivery systems holds great promise for patient-specific healthcare, personalized medicine and precision medicine.
Declaration of competing interestThe authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
AcknowledgmentsThis work was supported by the National Natural Science Foundation of China (Nos. 81601613, 81771122, 81970985, 81970984, 81901060), Key Research Program of Sichuan Science and Technology Department (Nos. 2018SZ0037, 2019YFS0142, 19YYJC2625).
| [1] |
G. Chen, N. Kawazoe, Y. Ito, Photo-crosslinkable hydrogels for tissue engineering applications, in: Y. Ito (Ed. ), Photochemistry for Biomedical Applications: From Device Fabrication to Diagnosis and Therapy, Springer Singapore, Singapore, 2018, pp. 277-300.
|
| [2] |
E. Caló, V.V. Khutoryanskiy, Eur. Polym. J. 65 (2015) 252-267. DOI:10.1016/j.eurpolymj.2014.11.024 |
| [3] |
Y. Wang, Biomaterials 178 (2018) 663-680. DOI:10.1016/j.biomaterials.2018.03.008 |
| [4] |
A. Skardal, A. Atala, Ann. Biomed. Eng. 43 (2015) 730-746. DOI:10.1007/s10439-014-1207-1 |
| [5] |
T. Jungst, W. Smolan, K. Schacht, T. Scheibel, J. Groll, Chem. Rev. 116 (2016) 1496-1539. DOI:10.1021/acs.chemrev.5b00303 |
| [6] |
M. Qasim, D.S. Chae, N.Y. Lee, Int. J. Nanomed. 14 (2019) 4333-4351. DOI:10.2147/IJN.S209431 |
| [7] |
F. Araiza-Verduzco, E. Rodriguez-Velazquez, H. Cruz, et al., Materials 13 (2020) 534. DOI:10.3390/ma13030534 |
| [8] |
G. Liu, Z. Bao, J. Wu, Chin. Chem. Lett. 31 (2020) 1817-1821. DOI:10.1016/j.cclet.2020.03.005 |
| [9] |
S. Pina, J.M. Oliveira, R.L. Reis, Adv. Mater. 27 (2015) 1143-1169. DOI:10.1002/adma.201403354 |
| [10] |
T.H. Jovic, G. Kungwengwe, A.C. Mills, I.S. Whitaker, Front. Mech. Eng. 5(2019), doi: http://dx.doi.org/10.3389/fmech.2019.00019.
|
| [11] |
J. Yang, Y.S. Zhang, K. Yue, A. Khademhosseini, Acta Biomater. 57 (2017) 1-25. DOI:10.1016/j.actbio.2017.01.036 |
| [12] |
M. Szekalska, A. Puciłowska, E. Szymanska, P. Ciosek, K. Winnicka, Int. J. Polym. Sci. 2016 (2016) 1-17. DOI:10.1155/2016/7697031 |
| [13] |
X. Wang, T. Hao, J. Qu, C. Wang, H. Chen, J. Nanomater. 2015 (2015) 970619. DOI:10.2166/wst.2015.095 |
| [14] |
J. Oju, K.H. Bouhadir, M.M. Joseph, A. Eben, Biomaterials 30 (2009) 2724-2734. DOI:10.1016/j.biomaterials.2009.01.034 |
| [15] |
A. Garcia-Lizarribar, X. Fernandez-Garibay, F. Velasco-Mallorqui, et al., Macromol. Biosci. 18(2018) 1800167.
|
| [16] |
A. Mignon, D. Devisscher, G.J. Graulus, et al., Carbohydr. Polym. 155 (2017) 448-455. DOI:10.1016/j.carbpol.2016.08.102 |
| [17] |
X. Chen, H. Yan, W. Sun, et al., Polym. Bull. 72 (2015) 3097-3117. DOI:10.1007/s00289-015-1455-8 |
| [18] |
L. Yuan, Y. Wu, Q.S. Gu, et al., Int. J. Biol. Macromol. 96 (2017) 569-577. DOI:10.1016/j.ijbiomac.2016.12.058 |
| [19] |
S.L. Fenn, R.A. Oldinski, J. Biomed, Mater. Res. B 104 (2016) 1229-1236. DOI:10.1002/jbm.b.33476 |
| [20] |
T. Bobula, J. Betak, R. Buffa, et al., Carbohydr. Polym. 125 (2015) 153-160. DOI:10.1016/j.carbpol.2015.02.027 |
| [21] |
C. Loebel, S.E. Szczesny, B.D. Cosgrove, et al., Biomacromolecules 18 (2017) 855-864. DOI:10.1021/acs.biomac.6b01740 |
| [22] |
T.F. Stefanello, B. Couturaud, A. Szarpak-Jankowska, et al., Nanoscale 9 (2017) 12150-12162. DOI:10.1039/C7NR03964F |
| [23] |
R. Beninatto, C. Barbera, O. De Lucchi, et al., Mater. Sci. Eng. C: Biomimetic Supramol. Syst. 96 (2019) 625-634. DOI:10.1016/j.msec.2018.11.052 |
| [24] |
A.C. Fonseca, M.S. Lima, A.F. Sousa, et al., Polym. Chem. 10 (2019) 1696-1723. DOI:10.1039/C9PY00121B |
| [25] |
S. Grenier, P.E. Donnelly, J. Gittens, P.A. Torzilli, J. Biomech. 48 (2015) 122-129. DOI:10.1016/j.jbiomech.2014.10.023 |
| [26] |
Q. Liu, H. Wang, G. Li, et al., Chin. Chem. Lett. 30 (2019) 485-488. DOI:10.1016/j.cclet.2018.06.009 |
| [27] |
N. Bhattarai, J. Gunn, M. Zhang, Adv. Drug Deliv. Rev. 62 (2010) 83-99. DOI:10.1016/j.addr.2009.07.019 |
| [28] |
J. Liu, Y. Xiao, X. Wang, et al., Int. J. Biol. Macromol. 122 (2019) 19-28. DOI:10.1016/j.ijbiomac.2018.09.202 |
| [29] |
Z. Qi, J. Xu, Z. Wang, J. Nie, G. Ma, Int. J. Biol. Macromol. 53 (2013) 144-149. DOI:10.1016/j.ijbiomac.2012.10.021 |
| [30] |
Y. Zhou, S. Zhao, C. Zhang, et al., Carbohydr. Polym. 184 (2018) 383-389. DOI:10.1016/j.carbpol.2018.01.009 |
| [31] |
S.S. Nagaraj, S. Easwaramoorthi, J.R. Rao, P. Thanikaivelan, Int. J. Biol. Macromol. 131 (2019) 779-786. DOI:10.1016/j.ijbiomac.2019.03.109 |
| [32] |
K.E. Drzewiecki, J.N. Malavade, I. Ahmed, C.J. Lowe, D.I. Shreiber, Technology 5 (2017) 185-195. DOI:10.1142/S2339547817500091 |
| [33] |
K. Yang, J. Sun, Z. Guo, et al., J. Mat. Chem. B 6 (2018) 7543-7555. DOI:10.1039/C8TB02314J |
| [34] |
J.L. Whittaker, N.K. Dutta, A. Zannettino, N.R. Choudhury, J. Mat, Chem. B 4 (2016) 5519-5533. |
| [35] |
M.B. Applegate, B.P. Partlow, J. Coburn, et al., Adv. Mater. 28 (2016) 2417-2420. DOI:10.1002/adma.201504527 |
| [36] |
H.H. Kim, J.W. Kim, J. Choi, Y.H. Park, C.S. Ki, Polymer 153 (2018) 232-240. DOI:10.1016/j.polymer.2018.08.019 |
| [37] |
Y.S. Zhang, K. Yue, J. Aleman, et al., Ann. Biomed. Eng. 45 (2017) 148-163. DOI:10.1007/s10439-016-1612-8 |
| [38] |
X. Huang, Y. Zhang, F. Li, et al., J. Polym. Sci. 58 (2020) 1439-1449. DOI:10.1002/pol.20200138 |
| [39] |
B.B. Van Den Bulcke, N. De Rooze, H.E. Schacht, C. Maria, H. Berghmans, Biomacromolecules 1 (2000) 31-38. DOI:10.1021/bm990017d |
| [40] |
J. Huang, L. Chen, Z. Gu, J. Wu, J. Biomed, Nanotechnol. 15 (2019) 1357-1370. |
| [41] |
A.J. Berger, K.M. Linsmeier, P.K. Kreeger, K.S. Masters, Biomaterials 141 (2017) 125-135. DOI:10.1016/j.biomaterials.2017.06.039 |
| [42] |
J. Van Hoorick, P. Gruber, M. Markovic, et al., Biomacromolecules 18 (2017) 3260-3272. DOI:10.1021/acs.biomac.7b00905 |
| [43] |
L. Rebers, T. Granse, G.E.M. Tovar, A. Southan, K. Borchers, Gels 5 (2019) 4. DOI:10.3390/gels5010004 |
| [44] |
J. Van Hoorick, L. Tytgat, A. Dobos, et al., Acta Biomater. 97 (2019) 46-73. DOI:10.1016/j.actbio.2019.07.035 |
| [45] |
L. Li, C. Lu, L. Wang, et al., ACS Appl. Mater. Interfaces 10 (2018) 13283-13292. DOI:10.1021/acsami.7b17054 |
| [46] |
C. García-Astrain, C. Peña-Rodriguez, A. Retegi, et al., Mater. Lett. 160 (2015) 142-145. DOI:10.1016/j.matlet.2015.07.096 |
| [47] |
T. Greene, C.C. Lin, ACS Biomater. Sci. Eng. 1 (2015) 1314-1323. DOI:10.1021/acsbiomaterials.5b00436 |
| [48] |
Y. Liu, S. Cheong Ng, J. Yu, W.B. Tsai, Colloid Surf. B: Biointerfaces 174 (2019) 316-323. DOI:10.1016/j.colsurfb.2018.10.077 |
| [49] |
S. AnilKumar, S.C. Allen, N. Tasnim, et al., J. Biomed. Mater. Res. B 107 (2019) 314-323. DOI:10.1002/jbm.b.34123 |
| [50] |
C. García-Astrain, A. Gandini, C. Peña, et al., RSC Adv. 4 (2014) 35578-35587. DOI:10.1039/C4RA06122E |
| [51] |
S. Korpayev, G. Kaygusuz, M. Sen, et al., Int. J. Biol. Macromol. 156 (2020) 681-690. DOI:10.1016/j.ijbiomac.2020.04.109 |
| [52] |
J.B. Costa, J. Silva-Correia, J.M. Oliveira, R.L. Reis, Adv. Healthc. Mater. 6 (2017) 1701021. DOI:10.1002/adhm.201701021 |
| [53] |
R.F. Pereira, P.J. Bártolo, Engineering 1 (2015) 090-112. DOI:10.15302/J-ENG-2015015 |
| [54] |
N. Ashammakhi, S. Ahadian, F. Zengjie, et al., Biotechnol. J. 13 (2018) e1800148. DOI:10.1002/biot.201800148 |
| [55] |
S. Guven, P. Chen, F. Inci, et al., Trends Biotechnol. 33 (2015) 269-279. DOI:10.1016/j.tibtech.2015.02.003 |
| [56] |
J. Groll, T. Boland, T. Blunk, et al., Biofabrication 8 (2016) 013001. DOI:10.1088/1758-5090/8/1/013001 |
| [57] |
W. Gao, Y. Zhang, D. Ramanujan, et al., Computer-Aided Design 69 (2015) 65-89. DOI:10.1016/j.cad.2015.04.001 |
| [58] |
C. Mota, D. Puppi, F. Chiellini, E. Chiellini, J. Tissue Eng, Regen. Med. 9 (2015) 174-190. DOI:10.1002/term.1635 |
| [59] |
L. Moroni, T. Boland, J.A. Burdick, et al., Trends Biotechnol. 36 (2018) 384-402. DOI:10.1016/j.tibtech.2017.10.015 |
| [60] |
F. Pati, J. Gantelius, H.A. Svahn, Angew. Chem. Int. Ed. 55 (2016) 4650-4665. DOI:10.1002/anie.201505062 |
| [61] |
L. Zhang, G. Yang, B.N. Johnson, X. Jia, Acta Biomater. 84 (2019) 16-33. DOI:10.1016/j.actbio.2018.11.039 |
| [62] |
S. Derakhshanfar, R. Mbeleck, K. Xu, et al., Bioact. Mater. 3 (2018) 144-156. DOI:10.1016/j.bioactmat.2017.11.008 |
| [63] |
M. Nadgorny, A. Ameli, ACS Appl. Mater. Interfaces 10 (2018) 17489-17507. DOI:10.1021/acsami.8b01786 |
| [64] |
W. Zhu, X. Ma, M. Gou, et al., Curr. Opin. Biotechnol. 40 (2016) 103-112. DOI:10.1016/j.copbio.2016.03.014 |
| [65] |
B.N. Johnson, K.Z. Lancaster, G. Zhen, et al., Adv. Funct. Mater. 25 (2015) 6205-6217. DOI:10.1002/adfm.201501760 |
| [66] |
P. Scarfato, L. Di Maio, L. Incarnato, J. Appl. Polym. Sci. 13 (2015) 42458. DOI:10.1002/app.42597 |
| [67] |
U. Jammalamadaka, K. Tappa, J. Funct. Biomater. 9 (2018) 22. DOI:10.3390/jfb9010022 |
| [68] |
S. Ji, M. Guvendiren, Front. Bioeng. Biotechnol. 5 (2017) 23. DOI:10.3389/fbioe.2017.00023 |
| [69] |
A.V. Berezkin, Y.V. Kudryavtsev, Langmuir 31 (2015) 12279-12290. DOI:10.1021/acs.langmuir.5b03031 |
| [70] |
J. Yin, M. Yan, Y. Wang, J. Fu, H. Suo, ACS Appl. Mater. Interfaces 10 (2018) 6849-6857. DOI:10.1021/acsami.7b16059 |
| [71] |
A. Skardal, M. Devarasetty, H.W. Kang, et al., Acta Biomater. 25 (2015) 24-34. DOI:10.1016/j.actbio.2015.07.030 |
| [72] |
I.T. Ozbolat, M. Hospodiuk, Biomaterials 76 (2016) 321-343. DOI:10.1016/j.biomaterials.2015.10.076 |
| [73] |
K. Holzl, S. Lin, L. Tytgat, et al., Biofabrication 8 (2016) 032002. DOI:10.1088/1758-5090/8/3/032002 |
| [74] |
L. Brigo, A. Urciuolo, S. Giulitti, et al., Acta Biomater. 55 (2017) 373-384. DOI:10.1016/j.actbio.2017.03.036 |
| [75] |
E.D. Lemma, B. Spagnolo, M. De Vittorio, F. Pisanello, Trends Biotechnol. 37 (2019) 358-372. DOI:10.1016/j.tibtech.2018.09.008 |
| [76] |
S. Adepu, N. Dhiman, A. Laha, et al., Curr. Opin. Biomed. Eng. 2 (2017) 22-28. DOI:10.1016/j.cobme.2017.03.005 |
| [77] |
S.C. Ligon, R. Liska, J. Stampfl, M. Gurr, R. Mulhaupt, Chem. Rev. 117 (2017) 10212-10290. DOI:10.1021/acs.chemrev.7b00074 |
| [78] |
E. Fantino, A. Chiappone, F. Calignano, et al., Materials 9 (2016) 589. DOI:10.3390/ma9070589 |
| [79] |
F. Li, X. Ji, Z. Wu, et al., Mater. Lett. 276 (2020) 128037. DOI:10.1016/j.matlet.2020.128037 |
| [80] |
P. Fiedor, J. Ortyl, Materials 13 (2020) 2951. DOI:10.3390/ma13132951 |
| [81] |
P.N. Bernal, P. Delrot, D. Loterie, et al., Adv. Mater. 31 (2019) 1904209. DOI:10.1002/adma.201904209 |
| [82] |
B.E. Kelly, I. Bhattacharya, H. Heidari, et al., Science 363 (2019) 1075-1079. DOI:10.1126/science.aau7114 |
| [83] |
X. Wang, M. Jiang, Z. Zhou, J. Gou, D. Hui, Compos. B: Eng. 110 (2017) 442-458. DOI:10.1016/j.compositesb.2016.11.034 |
| [84] |
C.C. Li, M. Kharaziha, C. Min, R. Maas, M. Nikkhah, Adv. Exp. Med. Biol. 811 (2015) 15-31. DOI:10.1007/978-3-319-22345-2_2 |
| [85] |
C. McBeth, J. Lauer, M. Ottersbach, et al., Biofabrication 9 (2017) 015009. DOI:10.1088/1758-5090/aa53bd |
| [86] |
J.A. Hunt, R. Chen, T. van Veen, N. Bryan, J. Mat. Chem. B 2 (2014) 5319-5338. DOI:10.1039/C4TB00775A |
| [87] |
D.B. Kolesky, K.A. Homan, M.A. Skylar-Scott, J.A. Lewis, Proc. Natl. Acad. Sci. U. S. A. 113 (2016) 3179-3184. DOI:10.1073/pnas.1521342113 |
| [88] |
C. Mandrycky, Z. Wang, K. Kim, D.H. Kim, Biotechnol. Adv. 34 (2016) 422-434. DOI:10.1016/j.biotechadv.2015.12.011 |
| [89] |
W. Jia, P.S. Gungor-Ozkerim, Y.S. Zhang, et al., Biomaterials 106 (2016) 58-68. DOI:10.1016/j.biomaterials.2016.07.038 |
| [90] |
L. Moroni, J.A. Burdick, C. Highley, et al., Nat. Rev. Mater. 3 (2018) 21-37. DOI:10.1038/s41578-018-0006-y |
| [91] |
W. Zhu, X. Qu, J. Zhu, et al., Biomaterials 124 (2017) 106-115. DOI:10.1016/j.biomaterials.2017.01.042 |
| [92] |
S.J. Paulsen, J.S. Miller, Dev. Dyn. 244 (2015) 629-640. DOI:10.1002/dvdy.24254 |
| [93] |
J. Li, C. Wu, P.K. Chu, M. Gelinsky, Mater. Sci. Eng. R 140 (2020) 100543. DOI:10.1016/j.mser.2020.100543 |
| [94] |
I. Noshadi, S. Hong, K.E. Sullivan, et al., Biomater. Sci. 5 (2017) 2093-2105. DOI:10.1039/C7BM00110J |
| [95] |
S. Anil Kumar, M. Alonzo, S.C. Allen, et al., ACS Biomater. Sci. Eng. 5 (2019) 4551-4563. DOI:10.1021/acsbiomaterials.9b00505 |
| [96] |
L. Gao, M.E. Kupfer, J.P. Jung, et al., Circ. Res. 120 (2017) 1318-1325. DOI:10.1161/CIRCRESAHA.116.310277 |
| [97] |
Y.S. Zhang, A. Arneri, S. Bersini, et al., Biomaterials 110 (2016) 45-59. DOI:10.1016/j.biomaterials.2016.09.003 |
| [98] |
N. Annabi, K. Tsang, S.M. Mithieux, et al., Adv. Funct. Mater. 23 (2013) 1300570. DOI:10.1002/adfm.201370199 |
| [99] |
X. Zhou, H. Cui, M. Nowicki, et al., ACS Appl. Mater. Interfaces 10 (2018) 8993-9001. DOI:10.1021/acsami.7b18197 |
| [100] |
J. Koffler, W. Zhu, X. Qu, et al., Nat. Med. 25 (2019) 263-269. DOI:10.1038/s41591-018-0296-z |
| [101] |
A. Arslan-Yildiz, R.E. Assal, P. Chen, et al., Biofabrication 8 (2016) 014103. DOI:10.1088/1758-5090/8/1/014103 |
| [102] |
X. Zhao, Q. Lang, L. Yildirimer, et al., Adv. Healthc. Mater. 5 (2016) 108-118. DOI:10.1002/adhm.201500005 |
| [103] |
A. Boddupalli, K.M. Bratlie, Biomater. Sci. 7 (2019) 1188-1199. DOI:10.1039/C8BM01535J |
| [104] |
G. Eke, N. Mangir, N. Hasirci, S. MacNeil, V. Hasirci, Biomaterials 129 (2017) 188-198. DOI:10.1016/j.biomaterials.2017.03.021 |
| [105] |
J.W. Kim, M.J. Kim, C.S. Ki, H.J. Kim, Y.H. Park, Int. J. Biol. Macromol. 105 (2017) 541-548. DOI:10.1016/j.ijbiomac.2017.07.067 |
| [106] |
X. Sun, Q. Lang, H. Zhang, et al., Adv. Funct. Mater. 27 (2017) 1604617. DOI:10.1002/adfm.201604617 |
| [107] |
A. Sun, X. He, L. Li, et al., NPG Asia Mater. 12 (2020) 25. DOI:10.1038/s41427-020-0206-y |
| [108] |
I.C. Carvalho, H.S. Mansur, Mater. Sci. Eng. C: Biomimetic Supramol. Syst. 78 (2017) 690-705. DOI:10.1016/j.msec.2017.04.126 |
| [109] |
J. Liu, G. Chinga-Carrasco, F. Cheng, et al., Cellulose 23 (2016) 3129-3143. DOI:10.1007/s10570-016-1038-3 |
| [110] |
Y. Hong, F. Zhou, Y. Hua, et al., Nat. Commun. 10 (2019) 2060. DOI:10.1038/s41467-019-10004-7 |
| [111] |
M.H. Chang, Y.P. Hsiao, C.Y. Hsu, P.S. Lai, Nanomaterials 8 (2018) 791. DOI:10.3390/nano8100791 |
| [112] |
T.T. Demirtas, G. Irmak, M. Gumusderelioglu, Biofabrication 9 (2017) 035003. DOI:10.1088/1758-5090/aa7b1d |
| [113] |
Z. Wan, P. Zhang, Y. Liu, L. Lv, Y. Zhou, Acta Biomater. 101 (2020) 29-42. |
| [114] |
A.C. Daly, F.E. Freeman, T. Gonzalez-Fernandez, et al., Adv. Healthc. Mater. 6 (2017) 1700298. DOI:10.1002/adhm.201700298 |
| [115] |
C. Liu, J. Wu, D. Gan, et al., J. Biomed. Mater. Res. B 108 (2020) 1814-1825. DOI:10.1002/jbm.b.34524 |
| [116] |
Y. Zuo, X. Liu, D. Wei, et al., ACS Appl. Mater. Interfaces 7 (2015) 10386-10394. DOI:10.1021/acsami.5b01433 |
| [117] |
M. Sadat-Shojai, M.T. Khorasani, A. Jamshidi, Chem. Eng. J. 289 (2016) 38-47. DOI:10.1016/j.cej.2015.12.079 |
| [118] |
P. Comeau, T. Willett, Macromol. Mater. Eng. 304 (2019) 1900142. DOI:10.1002/mame.201900142 |
| [119] |
K. Yue, G. Trujillo-de Santiago, M.M. Alvarez, et al., Biomaterials 73 (2015) 254-271. DOI:10.1016/j.biomaterials.2015.08.045 |
| [120] |
T. Thakur, J.R. Xavier, L. Cross, et al., J. Biomed. Mater. Res. A 104 (2016) 879-888. DOI:10.1002/jbm.a.35621 |
| [121] |
A.A. Egorov, A.Y. Fedotov, A.V. Mironov, et al., Beilstein J. Nanotechnol. 7 (2016) 1794-1799. DOI:10.3762/bjnano.7.172 |
| [122] |
T. Xin, Y. Gu, R. Cheng, et al., ACSAppl. Mater. Interfaces 9 (2017) 41168-41180. DOI:10.1021/acsami.7b13167 |
| [123] |
X. Li, S. Chen, J. Li, et al., Polymers 8 (2016) 269. DOI:10.3390/polym8080269 |
| [124] |
R. Levato, W.R. Webb, I.A. Otto, et al., Acta Biomater. 61 (2017) 41-53. DOI:10.1016/j.actbio.2017.08.005 |
| [125] |
A. Abbadessa, V.H.M. Mouser, M.M. Blokzijl, et al., Biomacromolecules 17 (2016) 2137-2147. DOI:10.1021/acs.biomac.6b00366 |
| [126] |
B.V. Sridhar, E.A. Dailing, J.L. Brock, et al., Regen.Eng. Transl.Med. 1 (2015) 11-21. DOI:10.1007/s40883-015-0002-3 |
| [127] |
Y. Zhou, K. Liang, S. Zhao, et al., Int. J. Biol. Macromol. 108 (2018) 383-390. DOI:10.1016/j.ijbiomac.2017.12.032 |
| [128] |
L.B. Weiswald, D. Bellet, V. Dangles-Marie, Neoplasia 17 (2015) 1-15. DOI:10.1016/j.neo.2014.12.004 |
| [129] |
Y. Chen, Y. Hao, Y. Huang, et al., J. Biomed. Nanotechnol. 15 (2019) 1923-1936. DOI:10.1166/jbn.2019.2821 |
| [130] |
W. Xu, J. Qian, Y. Zhang, et al., Acta Biomater. 33 (2016) 131-141. DOI:10.1016/j.actbio.2016.01.027 |
| [131] |
X. Zhou, W. Zhu, M. Nowicki, et al., ACS Appl. Mater. Interfaces 8 (2016) 30017-30026. DOI:10.1021/acsami.6b10673 |
| [132] |
H. Shih, T. Greene, M. Korc, C.C. Lin, Biomacromolecules 17 (2016) 3872-3882. DOI:10.1021/acs.biomac.6b00931 |
| [133] |
X. Cao, R. Ashfaq, F. Cheng, et al., Adv. Funct. Mater. 29 (2019) 1807173. DOI:10.1002/adfm.201807173 |
| [134] |
T.U. Nguyen, K.E. Watkins, V. Kishore, J. Biomed. Mater. Res. A 107 (2019) 1541-1550. DOI:10.1002/jbm.a.36668 |
 2021, Vol. 32
2021, Vol. 32