b National Institute on Drug Dependence and Beijing Key Laboratory of Drug Dependence, Peking University, Beijing 100191, China;
c State Key Laboratory of Natural and Biomimetic Drugs, and Department of Chemical Biology, School of Pharmaceutical Sciences, Peking University, Beijing 100191, China;
d Peking-Tsinghua Center for Life Sciences and PKU-IDG/McGovern Institute for Brain Research, Academy for Advanced Interdisciplinary Studies Peking University, Beijing 100871, China;
e School of Basic Medicine, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430030, China
The number of methamphetamine (METH) abusers has rapidly grown worldwide in recent years [1]. The consecutive use of METH usually produces direct effects on the central nervous system, contributing to euphoria, intense emotions, alterations of self-esteem, and greater alertness, aggression, and sexual drive [2-4]. Methamphetamine use can also quickly lead to addiction, increasing the risk of overdose, which is a significant source of mortality across multiple populations. However, treatments for METH addiction are limited and mainly consist of behavioral and cognitive therapy [5], and no medications have been approved to date. Thus, more effective treatments are essential to prevent METH addiction and relieve associated symptoms.
Immunotherapy has been applied for the treatment of various types of diseases, including cancer [6]. Based on prominent results from exploiting the immune system, vaccines for METH addiction have also been investigated [7]. Methamphetamine-specific antibodies that are induced by such vaccines may neutralize circulating METH molecules and prevent their penetration into the brain. Due to the highly simplified structure of the METH molecule and its low immunogenicity, triggering a sufficient immune response to generate the necessary specific antibodies has been extremely difficult. Currently, the development of METH vaccines relies heavily on the conjugation of varied haptens to carrier proteins [7, 8], such as keyhole limpet hemocyanin (KLH) [9]. However, the introduction of foreign proteins elicits other concerns. First, the high immunogenicity and complexity of the carrier protein and requisite linker moiety may result in high titers of non-METH-specific antibodies, which would lead to immune suppression of the selected antigen [10]. Second, the conjugation of a small molecule to the large protein usually makes equivalency difficult to control, which may lead to issues with quality control. Third, heterogeneity of the carrier protein may cause systemic toxicity [10, 11]. Therefore, new strategies are needed toward the development of novel METH vaccines for clinical use. The present study describes our efforts to use a carrier protein-free strategy [12, 13] to construct and immunologically and behaviorally evaluate a fully synthetic METH vaccine.
Considering recent developments in multicomponent vaccines that contain haptens, T-helper (Th) epitopes, and immunological activators [14-17], we envisioned that the absence of carrier protein might be beneficial for improving immune specificity for the METH antigen and biosafety (Fig. 1A). Thus two three-component carrier protein-free constructs 1a and 1b were designed, each consisting of a ligand to the Toll-like receptor 2 (TLR2) [18, 19], a Th2 epitope-containing peptide sequence, and a METH hapten (Fig. 1B). Pam3Cys has been utilized in several vaccine candidates as a built-in adjuvant to activate the innate immune system [16, 18, 20], presumably as stimuli to the maturation of dendritic cells and subsequent activation of helper T cells [21]. To further induce the production of different subtypes of antibodies from plasma cells [22], the DRAHYNI peptide sequence, deriving from the E7 open reading frame protein of human papillomavirus (HPV) type 16, was incorporated to the construct as the Th epitope. With the METH hapten serving as a B cell epitope, we hoped that the designed construct could trigger an immune response, in which T-cell receptors (TCRs) on the T-helper cell could recognize the Th epitope that was presented by MHCII and assist with the differentiation and maturation of naive B cells into plasma cells [23, 24], leading to the production of specific anti-METH IgG antibodies by strengthening humoral immunity (Fig. 1A).

|
Download:
|
| Fig. 1. (A) Proposed mechanism by which the conjugate induces humoral immunity. (B) Design of the lipopeptide-based methamphetamine vaccine. | |
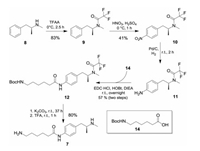
The synthesis of the designed vaccine was initiated by preparing the METH hapten with a proper linker used to connect the Th2 epitope-containing peptide. Accordingly, the synthesis of compound 7 was carried out by first derivatize D-methamphetamine (8) [9], whose secondary amine was protected with a trifluoro acetyl (TFA) group using TFAA (Scheme 1), leading to the generation of compound 9 in an 83% yield. Nitration reaction with concentrated nitric acid gave rise to compound 10 (41%). Hydrogenolysis of the obtained nitro-substituted METH derivative, followed by amide coupling with N-Boc-6-aminohexanoic acid, led to the formation of 12 in a decent yield (57%) over two steps. After the removal of both protecting groups, a properly functionalized hapten METH derivative 7 was obtained, appending a linker that is ready for connecting to the C-termini of the peptides. On the other hand, Pam3Cys-OH (13) was synthesized based on the previously reported methods using racemic glycidol as the starting materials [25, 26], as it was demonstrated that the stereochemical configuration of the glycerol in a synthetic MUC1 antitumor vaccine was inconsequential to its immunogenicity [26].
|
Download:
|
| Scheme 1. Synthesis of hapten 7. | |
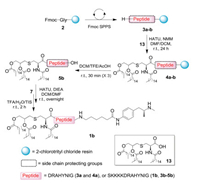
Two Th2 epitope-containing peptide sequences, DRAHYNI and SKKKKDRAHYNI (Scheme 2), were synthesized in a straightforward manner using Fmoc-based solid-phase peptide synthesis (SPPS). Notably, a glycine residue was installed at the peptide C-terminus to avoid possible epimerization in the later coupling step for connecting the hapten. Moreover, 2-chlorotrityl chloride resin was utilized for peptide synthesis, which allowed the cleavage from resin without removal of all other protecting groups, and ensured the selective conjugation of compound7 to the C-terminus of lipopeptide 5. In practice, amide coupling between amine 4 and Pam3Cys-OH (13) on the resin was achieved using HATU and NMM, followed by cleavage from the resin using a DCM/TFE/AcOH cocktail to give compound 5 with only the C-terminal carboxylate exposed for further reactions. After amidation with hapten 7, global deprotection using the TFA cocktail (TFA/TIS/water, 95:2.5:2.5) afforded the crude vaccine construct. It was observed that crude 1a displayed poor solubility and dissolved only in DMSO, which made it impossible to be purified using reverse-phase high-performance liquid chromatography (HPLC). In contrast, construct 1b with extra amino acids SKKKK in the sequence exhibits significantly improved solubility and could be purified on HPLC, affording the desired product in good yield (21%). These results indicate that the introduction of multiple lysines in the sequence is necessary for the preparation of pure synthetic vaccines with hydrophobic hapten and lipid chains [27].
|
Download:
|
| Scheme 2. Synthesis of vaccine 1b. | |
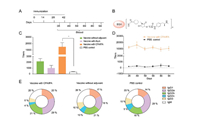
With the pure vaccine construct in hand, we evaluated the immunogenic capability of the vaccine in mice and the possible combination with suitable adjuvants. Two widely studied immunostimulants, Alum, and Freund's adjuvant were chosen in our study, as they have been utilized in the developments of vaccines to drugs of abuse and addiction [28]. Female mice (6- to 7-week-old BALB/c) were divided into four groups (n=6/group) and subcutaneously injected with different sample solutions (30 μg of vaccine 1b with phosphate-buffered saline (PBS), the vaccine formulated with Alum, the vaccine with Freund's adjuvant [29], and PBS control) on days 0, 14, 28 and 42. Serum was collected on days 35, 49, 59, 69, 80 and 94 (Fig. 2A). Antibody titers were analyzed using an enzyme-linked immunosorbent assay (ELISA) coated with a BSA-METH hapten conjugate [9] (Fig. 2B). The experimental results showed that the synthetic vaccine itself was able to stimulate the secretion of IgG antibodies, where the IgG titers elicited were tenfold higher than that of the PBS control group (Fig. 2C). This observation is in accord with the previous studies on the synthetic vaccine constructs containing Pam3Cys, which induce strong humoral immunity without external adjuvant existed [18, 30]. This data further confirmed the self-adjuvanting role of Pam3Cys in the synthetic vaccine. On the other hand, the vaccine with Freund's adjuvant produced significantly higher METH-specific antibody titers (1:10000 to 1:25000), whereas the vaccine with Alum or PBS alone produced anti-METH antibodies at low titers, indicating that Freund's adjuvant may play an important role in the process of the immune response induced by this METH vaccine. By monitoring the mice immunized with vaccine 1b and Freund's adjuvant, the assay showed that METH-specific antibody titers reached the highest level after the fourth immunization and remained relatively stable for at least one and half months (Fig. 2D). The persistent antibodies generated could be beneficial for preventing addictive behaviors and relapse, as they may sequester METH in the blood and reduce its distribution to the brain [31].
|
Download:
|
| Fig. 2. Immunogenic capability of the vaccine. (A) Timeline of active immunization. (B) BSA-linked methamphetamine immunogen used as the coating antigen in ELISA. (C) Mean antibody titers on day 49. (D) Mean antibody titers at different time points after the third booster immunization. (E) Antibody isotypes and subtypes of the conjugate vaccine. The data are expressed as mean±SEM. *P < 0.05, n = 6/group. | |
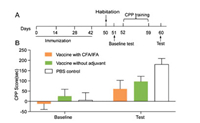
In order to obtain more information on the possible types of activation for different vaccine formulations, we further investigated the isotypes and subtypes of the generated antibodies (Fig. 2E). In the cases of the mice vaccinated with Freund's adjuvant, a higher level of IgG2a antibodies were elicited than that of the IgG1 type (IgG2a≥IgG1), indicating that FcγRIIb might be activated [32]. In contrast, the levels of IgG subtypes in the group vaccinated without adjuvant were approximately the same (IgG2a=IgG1), suggesting that the FcγRIIa signaling pathway was likely activated, thus enhancing antigen processing and presentation and inducing a T-cell response [32-34]. These results are also in accord with the literature precedent showing the use of Freund's adjuvant promotes an increase in the level of IgG2a [35]. Moreover, the IgG1 and IgG2 isotypes are believed to play crucial roles in antibody-dependent humoral immunity [23, 36, 37]. Therefore, the utilized Th2 epitope seemed to be critical for our vaccine for a class switch from other immunoglobulins to IgG antibodies, consistent with previous studies of carbohydrate vaccines without Th epitopes that led to no or low titers of IgG antibodies [38-40]. To investigate the effects of this carrier protein-free vaccine on METH-induced behaviors, conditioned place preference (CPP) and locomotor sensitization models were utilized to assess efficiency in mice in vivo. After active immunization, the mice underwent CPP training (0.5mg/kg METH, s.c.) for 8 days and then were tested (Fig. 3A) [41]. The repeated-measures analysis of variance (ANOVA) of CPP scores revealed a significant main effect of test phase (F1, 35=21.17, P < 0.0001). The mice that received the emulsion of vaccine and Freund's adjuvant had the lowest CPP scores during test (Fig. 3B), suggesting that higher anti-METH antibody titers may have slowed or reduced the entry of METH molecules into the brain, thus demonstrating better efficacy in preventing METH addiction. These results indicate that this vaccine that was formulated with Freund's adjuvant prevented the acquisition of METH-induced CPP by neutralizing the ability of METH to support contextual conditioning and decreasing its rewarding effects.
|
Download:
|
| Fig. 3. Immunization with the lipopeptide-based vaccine inhibited methamphetamine-induced conditional place preference (CPP). (A) Timeline of immunization, CPP training, and test. (B) Conditioned place preference scores during baseline and test sessions. The data are expressed as mean±SEM. n=12-13/group. | |
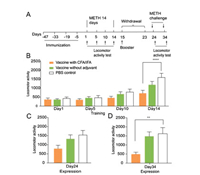
A locomotor sensitization model was then used to evaluate the effect of the vaccine on the psychomotor stimulant effect of METH. The procedure for locomotor sensitization was based on our previous studies [42], consisting of three phases: initiation, withdrawal, and expression. During initiation, the mice received METH injections (1 mg/kg, i.p.) for 14 consecutive days, and locomotor activity was measured on days 1, 5, 10 and 14 (Fig. 4A). The repeated-measures ANOVA of locomotor activity revealed significant effects of vaccine (F2, 40 = 3.455, P < 0.05) and test phase (F3, 120=34.67, P < 0.0001) and a significant vaccine × test phase interaction (F6, 120=3.25, P < 0.01; Fig. 4B). The post hoc analysis showed that control mice exhibited a significant increase in locomotor activity after the METH injection compared with the group that was vaccinated with Freund's adjuvant on the last day of the initiation phase (P < 0.0001). The group that was vaccinated without the adjuvant exhibited a tendency toward a decrease in locomotor activity compared with the control group. After 10 days of withdrawal, locomotor activity was tested following a METH challenge (1 mg/kg, i.p.) on days 24 and 34 (Figs. 4C and D). The one-way ANOVA revealed a significant main effect of vaccine (day 34: F2, 36 = 5.906, P < 0.01). The post hoc analysis showed a significant difference between the PBS control and adjuvant-vaccinated groups, indicating that the vaccine with Freund' adjuvant inhibited the initiation, expression, and maintenance of METH-induced locomotor sensitization. These behavioural results suggest that our tri-component lipopeptide-based vaccine effectively prevented the rewarding and psychoactive effects of METH.
|
Download:
|
| Fig. 4. Immunization with the lipopeptide-based vaccine prevented the initiation and expression of methamphetamine-induced locomotor sensitization. (A) Timeline of immunization and locomotor activity test. (B) Locomotor activity during the training of locomotor sensitization. (C) Locomotor activity after the methamphetamine challenge on day 24. (D) Locomotor activity after the methamphetamine challenge on day 34. The data are expressed as mean±SEM. **P < 0.01, ****P < 0.0001, n=12-14/group. | |
In the animal study, we observed no significant difference in animal states between mice with the non-adjuvanted vaccine and the ones in the control group, such as their weight, vital signs, and the capability of motion after immunization. The survival rate of the vaccinated group is 100%. Moreover, none of the animals with the non-adjuvanted vaccine experienced local reactogenicity or adverse events in the following behavioral test, indicating that this METH vaccine is generally safe for the animal. These observations are in accord with the previous studies showing the safety of full synthetic three-component vaccine and lipopeptide [43-45].
In summary, we have developed a METH vaccine candidate that was composed of the TLR2 ligand Pam3Cys, a Th2 epitope, and a METH hapten. Immunological and behavioural analyses of this vaccine in combination with Freund's adjuvant revealed that a robust humoral immune response was induced, and behavioural effects of METH were counteracted in mice. This carrier protein-free strategy may be beneficial for amplifying the immunogenicity of a hapten with a simple structure and minimizing an undesirable immune response. The preparation of this construct was also convenient and easily characterized, thus ensuring homogeneity of the vaccine and demonstrating an advantage with regard to quality control. Our findings provide valuable information for the further development of anti-METH vaccines, which may also be of use to the design of new vaccines for other drugs of abuse that could be promising for the treatment of addiction.
Declaration of competing interestThe authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
AcknowledgmentsThis work was supported in part by the National Natural Science Foundation of China (Nos. 81821092 and 81701312), Beijing Municipal Science and Technology Commission (Nos. Z181100001518005 and JQ18024), and National Key Research and Development Program of China (Nos. 2017YFC0803608 and 2019YFC0118604). All of the experiments were conducted at Peking University in accordance with National Institutes of Health guidelines and were approved by the Institutional Animal Care and Use Committee. We thank Drs. Xiaohui Zhang, Yuan Wang, and Xiaomeng Shi (Peking University) for spectroscopic assistance.
Appendix A. Supplementary dataSupplementary material related to this article can be found, in the online version, at doi:https://doi.org/10.1016/j.cclet.2020.10.038.
| [1] |
A. Pabst, J.C. Castillo-Duque, A. Mayer, M. Klinghuber, R. Werkmeister, Dent. J. 5 (2017) 29. DOI:10.3390/dj5040029 |
| [2] |
A. Elkashef, F. Vocci, G. Hanson, et al., Subst. Abus. 29 (2008) 31-49. DOI:10.1080/08897070802218554 |
| [3] |
C.C. Cruickshank, K.R. Dyer, Addiction 104 (2009) 1085-1099. DOI:10.1111/j.1360-0443.2009.02564.x |
| [4] |
D.E. Rusyniak, Neurol. Clin. 29 (2011) 641-655. DOI:10.1016/j.ncl.2011.05.004 |
| [5] |
T. Brackins, N.C. Brahm, J.C. Kissack, J. Pharm. Pract. 24 (2011) 541-550. DOI:10.1177/0897190011426557 |
| [6] |
U. Sahin, O. Tureci, Science 359 (2018) 1355-1360. DOI:10.1126/science.aar7112 |
| [7] |
P.T.A. Trends Guide Bremer, K.D. Janda, Pharmacol. Rev. 69 (2017) 298-315. DOI:10.1124/pr.117.013904 |
| [8] |
R. Arora, C.N. Haile, T.A. Kosten, et al., Am. J. Addict. 28 (2019) 119-126. DOI:10.1111/ajad.12867 |
| [9] |
A.Y. Moreno, A.V. Mayorov, K.D. Janda, J. Am. Chem. Soc. 133 (2011) 6587-6595. DOI:10.1021/ja108807j |
| [10] |
P.W. Anderson, M.E. Pichichero, E.C. Stein, et al., J. Immunol. 142 (1989) 2464-2468. |
| [11] |
T. Buskas, Y. Li, G.J. Boons, Chem. Eur. J. 10 (2004) 3517-3524. DOI:10.1002/chem.200400074 |
| [12] |
A.S. Cordeiro, M.J. Alonso, Pharm. Pat. Anal. 5 (2016) 49-73. DOI:10.4155/ppa.15.38 |
| [13] |
L. Zeng, Z. Liao, W. Li, et al., Chin. Chem. Lett. 31 (2020) 1162-1164. DOI:10.1016/j.cclet.2019.10.015 |
| [14] |
H. Cai, Z.Y. Sun, M.S. Chen, et al., Angew. Chem. Int. Ed. 53 (2014) 1699-1703. DOI:10.1002/anie.201308875 |
| [15] |
T. Buskas, P. Thompson, G.J. Boons, Chem. Commun. (Camb.) (2009) 5335-5349. |
| [16] |
S. Dziadek, A. Hobel, E. Schmitt, H. Kunz, Angew. Chem. Int. Ed. 44 (2005) 7630-7635. DOI:10.1002/anie.200501594 |
| [17] |
A.S. Ashhurst, D.M. McDonald, C.C. Hanna, et al., J. Med. Chem. 62 (2019) 8080-8089. DOI:10.1021/acs.jmedchem.9b00832 |
| [18] |
B.L. Wilkinson, S. Day, L.R. Malins, V. Apostolopoulos, R.J. Payne, Angew. Chem. Int. Ed. 50 (2011) 1635-1639. DOI:10.1002/anie.201006115 |
| [19] |
A.O. Aliprantis, R.B. Yang, M.R. Mark, et al., Science 285 (1999) 736. DOI:10.1126/science.285.5428.736 |
| [20] |
Z. Zhou, H. Lin, C. Li, Z. Wu, Chin. Chem. Lett. 29 (2018) 19-26. DOI:10.1016/j.cclet.2017.09.047 |
| [21] |
T. Buskas, S. Ingale, G.J. Boons, Angew. Chem. Int. Ed. 44 (2005) 5985-5988. DOI:10.1002/anie.200501818 |
| [22] |
R.W. Tindle, G.J.P. Fernando, J.C. Sterling, I.H. Frazer, Proc. Natl. Acad. Sci. U. S. A. 88 (1991) 5887-5891. DOI:10.1073/pnas.88.13.5887 |
| [23] |
G. Grødeland, E. Fossum, B. Bogen, Front. Immunol. 6 (2015) 367. |
| [24] |
R.N. Germain, Cell 76 (1994) 287-299. DOI:10.1016/0092-8674(94)90336-0 |
| [25] |
F. Reichel, A.M. Roelofsen, H.P.M. Geurts, et al., J. Am. Chem. Soc. 121 (1999) 7989-7997. DOI:10.1021/ja991319q |
| [26] |
L. Shi, H. Cai, Z.H. Huang, et al., ChemBioChem 17 (2016) 1412-1415. DOI:10.1002/cbic.201600206 |
| [27] |
W. Zeng, S. Ghosh, Y.F. Lau, L.E. Brown, D.C. Jackson, J. Immunol. 169 (2002) 4905. DOI:10.4049/jimmunol.169.9.4905 |
| [28] |
C.R. Alving, G.R. Matyas, O. Torres, R. Jalah, Z. Beck, Vaccine 32 (2014) 5382-5389. DOI:10.1016/j.vaccine.2014.07.085 |
| [29] |
H. Cai, M.S. Chen, Z.Y. Sun, et al., Angew. Chem. Int. Ed. 52 (2013) 6106-6110. DOI:10.1002/anie.201300390 |
| [30] |
H. Cai, Z.Y. Sun, Z.H. Huang, et al., Chem. Eur. J. 19 (2012) 1962-1970. |
| [31] |
M. Gooyit, P.O. Miranda, C.J. Wenthur, A. Ducime, K.D. Janda, ACS Chem. Neurosci. 8 (2017) 468-472. DOI:10.1021/acschemneuro.6b00389 |
| [32] |
R.J. Pleass, J.M. Woof, Trends Parasitol. 17 (2001) 545-551. DOI:10.1016/S1471-4922(01)02086-4 |
| [33] |
K.G. Smith, M.R. Clatworthy, Nature Rev. Immunol. 10 (2010) 328-343. DOI:10.1038/nri2762 |
| [34] |
S. Bournazos, J.V. Ravetch, Immunity 47 (2017) 224-233. DOI:10.1016/j.immuni.2017.07.009 |
| [35] |
J.A. Fontes, J.G. Barin, M.V. Talor, et al., Immun. Inflamm. Dis. 5 (2017) 163-176. DOI:10.1002/iid3.155 |
| [36] |
K.B. Lorvik, C. Hammarström, M. Fauskanger, et al., Cancer Res. 76 (2016) 6864. DOI:10.1158/0008-5472.CAN-16-1219 |
| [37] |
B. Pulendran, D. Artis, Science 337 (2012) 431. DOI:10.1126/science.1221064 |
| [38] |
T. Toyokuni, B. Dean, S.P. Cai, et al., J. Am. Chem. Soc. 116 (1994) 395-396. DOI:10.1021/ja00080a055 |
| [39] |
S.J. Danishefsky, J.R. Allen, Angew. Chem. Int. Ed. 39 (2000) 836-863. DOI:10.1002/(SICI)1521-3773(20000303)39:5<836::AID-ANIE836>3.0.CO;2-I |
| [40] |
S.F. Slovin, G. Ragupathi, C. Musselli, et al., J. Clin. Oncol. 21 (2003) 4292-4298. DOI:10.1200/JCO.2003.04.112 |
| [41] |
C.N. Haile, T.A. Kosten, X.Y. Shen, et al., Am. J. Addict. 24 (2015) 748-755. DOI:10.1111/ajad.12307 |
| [42] |
C.M. Xu, J. Wang, P. Wu, et al., J. Neurochem. 118 (2011) 126-139. DOI:10.1111/j.1471-4159.2011.07281.x |
| [43] |
W.H. Li, Y.M. Li, Chem. Rev. 120 (2020) 11420-11478. DOI:10.1021/acs.chemrev.9b00833 |
| [44] |
C. Durier, O. Launay, V. Meiffrédy, et al., AIDS 20 (2006) 1039-1049. DOI:10.1097/01.aids.0000222077.68243.22 |
| [45] |
F. Steinhagen, T. Kinjo, C. Bode, D.M. Klinman, Vaccine 29 (2011) 3341-3355. DOI:10.1016/j.vaccine.2010.08.002 |
 2021, Vol. 32
2021, Vol. 32 

