b Beijing National Laboratory for Molecular Sciences, Institute of Chemistry, Chinese Academy of Sciences, Beijing 100190, China;
c College of Metallurgy and Materials Engineering, Hunan University of Technology, Zhuzhou 412000, China;
d Max Planck Institute for Polymer Research, Ackermannweg 10, 55128 Mainz, Germany
Because of various advantages, such as low cost, easy fabrication, large area and flexibility [1-3], organic solar cells (OSCs) have attracted numerous attentions and rapid progress has been achieved after decades of development. The power conversion efficiency (PCE) of single bulk heterojunction (BHJ) has raised to over 18%via materials innovation, device engineering and interface processing [4-6]. Nevertheless, relatively narrow absorption spectra of binary OSCs still restricts the value of short-circuit current density (Jsc) [7, 8]. To make full use of the sunlight, the tandem OSCs have been researched and fabricated [9, 10]. Through the complementary absorption of each active layer, tandem OSCs can exhibit a wide absorption spectrum in the visible-near infrared region (vis-NIR) [11-13]. Chen and his colleagues reported tandem OSCs with a record-breaking PCE of 17.3% with the absorption onset of ~1050 nm [9]. Unfortunately, the fabrication of tandem OSCs based on solution processing requires complicated technical processes, giving rise to low productivity and high costs, which might hinder further industrial production of tandem OSCs [14, 15]. On the contrary, the active layer of ternary OSCs is composed of two donors and one acceptor, or one donor and two acceptors as a blend to cover the solar spectrum in a broad wavelength range, which makes the fabrication of ternary OSCs easier with a lower cost [16].
It has been demonstrated that a wider absorption spectrum for ternary OSCs is beneficial to reach higher Jsc [17]. However, the photovoltaic performance of OSCs is simultaneously determined by Jsc, open circuit voltage (Voc) and fill factor (FF), which is related to the absorption spectrum, molecular energy level and morphology of active layers [4, 18, 19]. The energy levels of three materials in ternary OSCs affect Voc via the establishment of cascade energy level structures, which can eventually influence the charge dissociation [20]. In addition, the film morphology can also have a significant impact on charge transport and collection, which is linked to FF for the device [21, 22]. Unlike binary OSCs, ternary OSCs generally propose more complex morphology and more recombination centers, therefore, the compatibility of three materials is particularly important for the morphology of active layers [23, 24]. Therefore, the careful selection of materials is a prerequisite for high performance ternary OSCs [25, 26]. Additionally, to obtain the maximum value of Voc, Jsc and FF, device engineering to fine film morphology is essential [27, 28]. Until now, research of ternary OSCs has made great progress, and the reported efficiency currently exceeding 17% [20, 29].
From our previous work, the PCE of binary OSCs based on OFQx-T: PC71BM is 7.59% [30]. In this work, we selected a medium band gap polymer donor PTB7-Th and a medium band gap non-fullerene acceptor ITIC as the third component to prepare ternary OSCs to improve the photovoltaic performance of OFQx-T: PC71BM blends. The optimal PCE of two ternary OSCs reaches 8.17% and 8.72% with ITIC and PTB7-Th as the third component, respectively. The improvement of PCE is mostly ascribed to the enhanced Jsc, resulting from the improved photon harvesting, more balanced mobility and weaker bimolecular recombination.
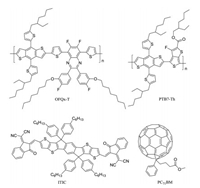
The chemical structures of OFQx-T, PTB7-Th, ITIC, PC71BM are illustrated in Fig. 1. Normalized ultraviolet-visible (UV–vis) absorption spectra of neat OFQx-T, ITIC, PTB7-Th, PC71BM films are shown in Fig. S1 (Supporting information). The normalized absorption spectrum of blend films OFQx-T: PC71BM, OFQx-T: PC71BM: ITIC and PTB7-Th: OFQx-T: PC71BM are shown in Fig. 2a. The absorption onset (λonset) of OFQx-T, ITIC, PTB7-Th is 727 nm, 776 nm and 759 nm, respectively. From Fig. 2a, it is apparent that addition of ITIC or PTB7-Th can effectively extend the absorption spectrum of OFQx-T: PC71BM blend films.
|
Download:
|
| Fig. 1. The chemical structures of OFQx-T, PTB7-Th, ITIC, PC71BM. | |

|
Download:
|
| Fig. 2. (a) Absorption spectrum of OFQx-T: PC71BM, OFQx-T: PC71BM: ITIC and PTB7-Th: OFQx-T: PC71BM blend films in thin film; (b) Energy level diagrams of OFQx-T, PTB7-Th, ITIC, PC71BM; (c) J-V characteristics of binary and ternary OSCs under illumination of AM 1.5 G at 100 mW/cm2; (d) EQE curves of binary and ternary OSCs. | |
The electrochemical properties of four materials were measured by cyclic voltammetry (CV), which are shown in Fig. S2 (Supporting information). The energy level diagrams of four materials can be estimated by the corresponding onsets of oxidation and reduction potentials. Fig. 2b shows the highest occupied molecular orbital (HOMO) and the lowest unoccupied molecular orbital (LUMO) energy levels of these used materials in the active layer. The detailed optical and electrochemical data are listed in Tables S1 and S2 (Supporting information). In OFQx-T: PC71BM: ITIC blend films, it is observed that the cascade energy level structures are built during three contents, which provides more charge transfer channels. Similar effects is anticipated for the PTB7-Th: OFQx-T: PC71BM blend films.
The photovoltaic properties of binary and ternary OSCs were investigated with the traditional structure of ITO/PEDOT: PSS/active layer/ZrAcac/Al. And all OSCs were characterized under AM 1.5 G simulated solar light with an intensity of 100 mW/cm2. The current density-voltage (J-V) curves and optimal photovoltaic parameters of binary and ternary OSCs are shown in Fig. 2c and summarized in Table 1, respectively. And photovoltaic parameters of binary and ternary OSCs with different conditions are listed in Table S2. The PCE of binary OFQx-T: PC71BM (1:1.5) blend films is 7.59%, with a Voc of 0.86 V, aJsc of 12.26 mA/cm2 and FF of 72%. With the simple ternary strategy, the PCE of the as cast OSCs were 4.54% and 4.15% by doping with 50 wt% ITIC or 50 wt% PTB7-Th. As seen in Table S3 (Supporting information), we tried to adjust the proportion of three components, the amount of 1, 8-diiodooctane (DIO) and thermal annealing to optimize the photovoltaic characteristics of ternary OSCs. The content of solvent additive DIO has a huge effect on the film forming process of active layers, and finally affects the surface morphology of the blend films. By optimizing the amount of DIO [31], Jsc of two ternary OSCs is greatly improved. The OFQx-T: PC71BM: ITIC blend films reaches the optimal PCE of 8.17% with a Voc of 0.89 V, Jsc of 14.35 mA/cm2 and FF of 64% by 1% DIO treatment. Similarly, optimal PCE of devices based on PTB7-Th: OFQx-T: PC71BM is 8.72%, with a Voc of 0.82 V, Jsc of 14.77 mA/cm2 and FF of 72% after 1% DIO treatment.
|
|
Table 1 Photovoltaic parameters of binary and ternary OSCs with optimal conditions. |
The external quantum efficiency (EQE) curves of the binary and ternary OSCs are presented in Fig. 2d. Binary OSCs shows a strong response in 300–750 nm, the addition of ITIC and PTB7-Th broadens the photo response spectrum about 50 nm in the NIR region. Compared with binary OSCs, the light absorption intensity of ternary OSCs is greatly enhanced, and maximum EQE value of PTB7-Th: OFQx-T: PC71BM blend films reaches 73%. The calculated Jsc of binary OSCs based on OFQx-T: PC71BM of 11.90 mA/cm2 is enhanced to ternary OSCs of 13.90 mA/cm2 for OFQx-T: PC71BM: ITIC and 14.34 mA/cm2 for PTB7-Th: OFQx-T: PC71BM respectively. Compared with J-V curves, the calculated Jsc shows the error less than 3%, proving the reliability of Jsc.
In order to investigate the charge recombination in binary and ternary OSCs, we measured the current density (Jsc) as a function of light intensity (Plight). The relationship of Jsc and Plight follows the formula of Jsc∝Plightα, when exponential factor α approaches 1, bimolecular type recombination in devices plays only a minor role for energy losses [32]. As shown in Fig. 3a, the α value of OFQx-T: PC71BM based devices is 0.94. The α of OFQx-T: PC71BM: ITIC and PTB7-Th: OFQx-T: PC71BM ternary OSCs are 0.95 and 0.96, respectively. This result demonstrates that two ternary OSCs have a higher α compared to binary devices, indicating weaker bimolecular recombination in ternary devices.

|
Download:
|
| Fig. 3. (a) Light intensity dependence of Jsc of the binary and ternary OSCs; (b) Jph-Veff characteristics of the binary and ternary OSCs; (c) The electron mobilities of binary and ternary OSCs; (d) The hole mobilities of binary and ternary OSCs. | |
To further explore the charge generation and collection efficiency in devices, we measured the variation curves of the photocurrent density (Jph) with effective voltage (Veff), as shown in Fig. 3b. Jph is described as JD-JL, where JD and JL is the current densities under standard simulated dark and light conditions, respectively. And Veff is considered to be in V0 -Vappl, in which V0 is the voltage when JD = JL, and Vappl is the applied voltage in devices. As is known, Jph will achieve a saturation value with the increase of Veff, namely saturation current density (Jsat). The Jph/Jsat ratio indicates the efficiency of charge dissociation and collection under the maximum power output condition [33]. The Jph/Jsat ratio of OFQx-T: PC71BM blend films is 89.58% at Veff = ~ 0.2 V. By contrast, Jph/Jsat ratio of OFQx-T: PC71BM: ITIC and PTB7-Th: OFQx-T: PC71BM blend films are 86.95% and 98.32%, respectively. Exciton dissociation and collection are not ideal in OFQx-T: PC71BM: ITIC blend films, which reduces the FF. The devices based on PTB7-Th: OFQx-T: PC71BM exhibit a higher exciton dissociation and collection efficiency, resulting in higher Jsc.
The hole and electron mobility of binary and ternary OSCs were investigated through space-charge limited current (SCLC) method based on single carrier devices [34]. The device structure of the hole-only and electron-only is ITO/PEDOT: PSS/active layer/Au and ITO/ZnO/active layer/PDINO/Al, respectively. Figs. 3c and d show the J1/2-V curves of binary and ternary OSCs. And detailed data for electron mobility (μe) and hole mobility (μh) are provided in Table S4 (Supporting information). The μe and μh of binary OSCs are 1.69×10−4 cm2 V-1 s-1 and 1.09×10−4 cm2 V-1 s-1, with the mobility ratio μe/μh of 1.55. The OFQx-T: PC71BM: ITIC blend films show the μe of 1.59×10−4 cm2 V-1 s-1, μh of 1.34×10−4 cm2 V-1 s-1 with μe/μh of 1.19. And PTB7-Th: OFQx-T: PC71BM blend films exhibit the μe of 2.78×10−4 cm2 V-1 s-1, μh of 2.06×10−4 cm2 V-1 s-1 with μe/μh of 1.35. The ternary OSCs have a more balanced mobility, which is beneficial for higher Jsc [35]. Moreover, higher and more balanced mobility facilitates higher Jsc for PTB7-Th-based ternary OSCs.
We further considered the blends morphology of binary and ternary OSCs through atomic force microscopy (AFM) and transmission electron microscopy (TEM). The AFM and TEM images are shown in Fig. 4. The root-mean-square (RMS) of OFQx-T: PC71BM blend films is 5.14 nm. The RMS values of ternary OSCs with ITIC and PTB7-Th are 18.90 nm and 2.33 nm, respectively. It indicates that for OFQx-T: PC71BM blend films, PTB7-Th with a smaller value has better compatibility. The effect of ITIC and PTB7-Th on blends morphology can be observed from TEM images, and the bright and dark zones are donor-rich and acceptor-rich domain, respectively [36]. Fig. 4a' shows a good blending between OFQx-T and PC71BM, resulting in OFQx-T: PC71BM minor phase separation. Although small separation size is conducive to excitons dissociation, it also limits the carrier transport to a certain extent. As seen, two ternary OSCs blend films exhibit a proper phase separation, especially a clear network structure was observed in PTB7-Th: OFQx-T: PC71BM blends. It suggests that smooth surface morphology and nanoscale network interpenetrating structure are beneficial to efficient exciton dissociation and charge transport. This work also illustrates the importance of a suitable third content to device morphology.

|
Download:
|
| Fig. 4. (a-c) AFM images (5×5 μm2) and (a'-c') TEM images of binary and ternary OSCs. (a, a') OFQx-T: PC71BM blend films; (b, b') OFQx-T: PC71BM: ITIC blend films; (c, c') PTB7-Th: OFQx-T: PC71BM blend films. | |
In conclusion, ternary OSCs with ITIC or PTB7-Th as the third component have been fabricated successfully. Compared with the binary OSCs based on OFQx-T: PC71BM blends with efficiency of 7.59%, PCE of ternary OSCs increases to 8.17% with 50 wt% ITIC as a complementary acceptor. Meanwhile, efficiency of the optimized ternary OSCs with 50 wt% PTB7-Th into donors can be enhanced to 8.72% with a relative of 13% improvement. The additional absorption of the third component in ternary OSCs can enhance light harvesting and Jsc. The third component can also give rise to a more balance charge carrier mobility and weaker bimolecular recombination, which can also contribute to higher Jsc. Moreover, the PTB7-Th: OFQx-T: PC71BM blend films shows a clear network interpenetrating structure, giving rise to higher exciton dissociation and collection efficiency. This work suggests that ternary OSCs with an appropriate third component can be useful to improve device performance of binary donor-acceptor OSCs.
Declaration of competing interestThe authors report no declarations of interest.
AcknowledgmentsThis study has been supported by the National Natural Science Foundation of China (No. 21506258) and Natural Science Foundation of Hunan Province (Nos. 2016JJ3134, 2017JJ2325).
Appendix A. Supplementary dataSupplementary material related to this article can be found, in the online version, at doi:https://doi.org/10.1016/j.cclet.2020.09.032.
| [1] |
Q. Wei, W. Liu, M. Leclerc, et al., Sci. China Chem. 63 (2020) 1352-1366. DOI:10.1007/s11426-020-9799-4 |
| [2] |
L. Lu, T. Zheng, Q. Wu, et al., Chem. Rev. 115 (2015) 12666-12731. DOI:10.1021/acs.chemrev.5b00098 |
| [3] |
J. Yuan, Y. Zhang, L. Zhou, et al., Joule 3 (2019) 1140-1151. DOI:10.1016/j.joule.2019.01.004 |
| [4] |
G. Zhang, J. Zhao, P.C.Y. Chow, et al., Chem. Rev. 118 (2018) 3447-3507. DOI:10.1021/acs.chemrev.7b00535 |
| [5] |
X. Xu, Z. Bi, W. Ma, et al., Adv. Mater. 29 (2017) 1704271. DOI:10.1002/adma.201704271 |
| [6] |
Q. Liu, J. Qin, J. Xu, et al., Sci. Bull. 65 (2020) 272-275. DOI:10.1016/j.scib.2020.01.001 |
| [7] |
W. Huang, P. Cheng, Y.M. Yang, et al., Adv. Mater. 30 (2018) 1705706. DOI:10.1002/adma.201705706 |
| [8] |
W. Zhao, S. Li, H. Yao, et al., J. Am. Chem. Soc. 139 (2017) 7148-7151. DOI:10.1021/jacs.7b02677 |
| [9] |
L. Meng, Y. Zhang, X. Wan, et al., Science 361 (2018) 1094-1098. DOI:10.1126/science.aat2612 |
| [10] |
Y. Cui, B. Gao, Y. Qin, et al., J. Am. Chem. Soc. 139 (2017) 7302-7309. DOI:10.1021/jacs.7b01493 |
| [11] |
T.Y. Li, T. Meyer, Z. Ma, et al., J. Am. Chem. Soc. 139 (2017) 13636-13639. DOI:10.1021/jacs.7b07887 |
| [12] |
K. Zhang, K. Gao, R. Xia, et al., Adv. Mater. 28 (2016) 4817-4823. DOI:10.1002/adma.201506270 |
| [13] |
X. Lia, K. Li, S. Dan, et al., Chin. Chem. Lett. 31 (2020) 1243-1247. DOI:10.1016/j.cclet.2019.10.029 |
| [14] |
P. Cheng, C. Yan, Y. Wu, et al., Adv. Mater. 28 (2016) 8021-8028. DOI:10.1002/adma.201602067 |
| [15] |
Q. An, F. Zhang, J. Zhang, et al., Energy Environ. Sci. 9 (2016) 281-322. DOI:10.1039/C5EE02641E |
| [16] |
H. Huang, B. Sharma, J. Mater. Chem. A 5 (2017) 11501. DOI:10.1039/C7TA00887B |
| [17] |
X. Ma, M. Luo, W. Gao, et al., J. Mater. Chem. A 7 (2019) 7843-7851. DOI:10.1039/C9TA01497G |
| [18] |
M. Luo, C. Zhu, J. Yuan, et al., Chin. Chem. Lett. 30 (2019) 2343-2346. DOI:10.1016/j.cclet.2019.07.023 |
| [19] |
A. Wadsworth, M. Moser, A. Marks, et al., Chem. Soc. Rev. 48 (2019) 1596-1625. DOI:10.1039/C7CS00892A |
| [20] |
L. Zhan, S. Li, T.K. Lau, et al., Energy Environ. Sci. 13 (2020) 635-645. DOI:10.1039/C9EE03710A |
| [21] |
R. Yu, H. Yao, Y. Cui, et al., Adv. Mater. 31 (2019) 1902302. DOI:10.1002/adma.201902302 |
| [22] |
W. Gao, C. Zhong, G. Zhang, et al., ACS Energy Lett. 3 (2018) 1760-1768. DOI:10.1021/acsenergylett.8b00825 |
| [23] |
Y. Xie, F. Yang, Y. Li, et al., Adv. Mater. 30 (2018) 1803045. DOI:10.1002/adma.201803045 |
| [24] |
K. Weng, P. Bi, H. Ryu, et al., J. Mater. Chem. A 7 (2019) 3552. DOI:10.1039/C8TA12034J |
| [25] |
X. Ma, Y. Mi, F. Zhang, et al., Adv. Energy Mater. 8 (2018) 1702854. DOI:10.1002/aenm.201702854 |
| [26] |
H. Hu, L. Ye, M. Ghasemi, et al., Adv. Mater. 31 (2019) 1808279. DOI:10.1002/adma.201808279 |
| [27] |
J. Wang, W. Gao, Q. An, et al., J. Mater. Chem. A 6 (2018) 11751-11758. DOI:10.1039/C8TA03453B |
| [28] |
Y. Cho, S. Jeong, S. Lee, et al., Nano Energy 75 (2020) 104896. DOI:10.1016/j.nanoen.2020.104896 |
| [29] |
X. Du, L. Zhou, H. Lin, et al., Adv. Funct. Mater. 30 (2020) 1909837. DOI:10.1002/adfm.201909837 |
| [30] |
S. Xu, L. Feng, J. Yuan, et al., Org. Electron. 50 (2017) 7-15. DOI:10.1016/j.orgel.2017.07.005 |
| [31] |
S. Xie, J. Wang, R. Wang, et al., Chin. Chem. Lett. 30 (2019) 217-221. DOI:10.1016/j.cclet.2018.04.001 |
| [32] |
M. Zhang, F. Zhang, Q. An, et al., Nano Energy 22 (2016) 241-254. DOI:10.1016/j.nanoen.2016.02.032 |
| [33] |
L. Huo, T. Liu, X. Sun, et al., Adv. Mater. 27 (2015) 2938-2944. DOI:10.1002/adma.201500647 |
| [34] |
D.F. Barbe, J. Phys. D: Appl. Phys. 4 (1971) 1812. DOI:10.1088/0022-3727/4/11/427 |
| [35] |
S.S.M. Stolterfoht, A. Armin, H. Jin, et al., Adv. Mater. 7 (2017) 1601379. |
| [36] |
Y. Zhong, M.T. Trinh, R. Chen, et al., Nat. Commun. 6 (2015) 8242. DOI:10.1038/ncomms9242 |
 2021, Vol. 32
2021, Vol. 32 


