b Department of Pharmacy, Zhongnan Hospital of Wuhan University, Wuhan 430071, China;
c School of Life Sciences, Northwestern Polytechnical University, Xi'an 710072, China;
d Institute for Advanced Study, Tongji University, Shanghai 200092, China
Oral delivery has been considered as a preferred administration and widely applied into clinic due to the low learning cost, ease of administration, patient compliance and feasibility for solid formulations [1-5].However, owing to the complicated gastrointestinal environment, oral delivery of small chemotherapeutics [6, 7] and bioactive molecules (DNA [8, 9], RNA [10], and protein [11], etc.) still face a series of challenges. Briefly, the physical barrier (gastrointestinal mucus, etc.), the chemical barrier (pH, enzyme, etc.), and the biological barrier (ATP-dependent efflux protein, etc.) could degrade cargos and/or decrease its oral absorption [12-14]. In addition, human intestine had a large surface area for absorption (approximately 400 m2) [15-18] and had the potential to directly absorb substances of smaller size [19-21]. Therefore, alargeamount of effort has been devoted to improve the stability and intestinal penetration of delivered drugs for high oral bioavailability [22-25].
Biomacromolecules, including polysaccharides, peptides and microRNA etc., play key roles in regulating physiological activities, including improving gastrointestinal stability and enhancing intestinal permeability [26-28]. For example, chitosan and its derivative could bind with Ca2+ on intestinal cytomembrane and open its tight junctions with enhanced permeability and bioavailability of delivered cargo. Compared to other widely studied material applied for oral delivery (e.g., silica particles), biomacromolecules are more biocompatible, easy to modify, and pharmaceutical active, which could not only improve the delivery efficiency but also enhance the bioavailability [29-31]. Currently, a series of biomacromolecules have been applied for oral administration to promote absorption and bioavailability of drugs [32-40]. However, there is no systematic review and summary in the field of increasing intestinal penetration by biomacromolecules. Here, we summarized biomacromolecules and the related mechanisms of increasing oral bioavailability. Meanwhile, recent advances in biomacromolecules based oral delivery and related future directions were also included and predicted.
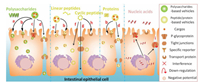
2. The strategies to increase intestinal permeabilityTo achieve efficient oral delivery and bioavailability, drug first need to remain stable within the stomach before reaching small intestine [39, 41-43] and overcome complex gastric environment, such as low pH (below 3.5), high levels of proteolytic enzymes and high mucosal viscosity [44-46]. Through optimizing the nanoarchitecture, solubility and isoelectric point, nanosized carrier could maintain its structure within stomach and intestine [47-49]. Besides, there are still numerous absorption barriers that lead to poor bioavailability [50, 51]. For example, the tight junction between small intestinal epithelial cells is one of the key barriers for intestinal absorption [52-54]. In addition, the short-term retention and high level of efflux in intestinal cells induced by P-glycoprotein (P-gp) and the low level of cellular uptake are the causes of low bioavailability [55-57]. Due to the diverse bioactivities, certain biomacromolecules could enhance the intestinal permeability via various mechanisms [58-60] (Scheme 1), such as directly penetration (Schemes 1e–g), promoting intestinal adhesion (Schemes 1a, b and h), slowing efflux (Scheme 1c), and even physically (Scheme 1d) or biologically (Scheme 1i) regulating tight junctions.
|
Download:
|
| Scheme 1. The illustration of various biomacromolecules to enhance intestinal permeability could be classified as promoting the ability to bind with cell membranes by (a) chemical or physical bonding and (e) autonomous penetration; increasing cellular uptake by attaching to (b) specific receptors or (h) transport proteins; (c) inhibiting the efflux capacity of P-glycoprotein; (d) opening tight junctions by interferences; directly crossing (f) cell membranes or (g) tight junctions; (i) down-regulating the protein expression related to tight junctions. | |
3. Polysaccharide-based vehicle for improving intestinal penetration
Polysaccharides are composed of monosaccharides bonded together by glycosidic linkages [61, 62], which have excellent biocompatibility and unique biological activities [61, 63-66]. Currently, naturally-occurring or artificially synthesized polysaccharides have been widely applied as vehicles for oral drug delivery [67, 68].
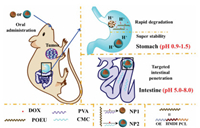
3.1. Chitosan and its derivativesChitosan (CS) and its derivatives are a class of aminopolysaccharide, which have shown diverse biological activities, such as improving the mucoadhesive [69-72] and opening tight junctions between intestinal epithelial cell [54, 73, 74]. It has been reported that CS could be complexed with plasmid DNA by electrostatic interactions to form CS-based DNA particles for efficient adhesion and penetration in gut [75]. Moreover, carboxymethyl chitosan (CMC) were also studied and demonstrated with enhanced capacity in opening tight junctions due to plentiful carboxyl group (negative potential) to anchor cadherin [54, 76-78]. Wang et al. [54] studied the enhanced epithelial permeation of the insulin:CMC/CS-NPs(-) and insulin:CMC/CS-NPs(+). In this work, the tight junctions between intestinal epithelial cells were almost completely detached, which were induced by the down-regulation and dephosphorylation of claudin-4, and Ca2+ deprivation by electronegative material [79, 80]. Meanwhile, cargo release characteristics in response to stomach and intestine environment revealed that the nanoparticles could withstand gastric environment and promote insulin release in small intestine. Based on this mechanism, a series of chitosan-based oral delivery systems with desired efficiency and oral bioavailability were developed [81-83]. For example, CMC-coated acid-sensitive nanoparticles via ultrasonic emulsification were developed to deliver doxorubicin into the hepatoma H22-bearing mice model by oral administration (Fig. 1) [84]. Single-layered acid sensitive nanoparticles prepared by polyvinyl alcohol were set as control groups to evaluate the performance for oral delivery. Double-layered nanoparticles demonstrated with robust stability under gastric tract, while the control group got degraded within 6 h. Afterwards, the outer layer would be peeled off to release free CMC due to the cleavage of Schiff base under pre-intestinal microenvironment (pH 5.0–8.0). Then, free CMC and the incompact CMC-coating would help to open tight junctions and improve the adhesion ratio of nanoparticles for enhanced intestinal permeability. In addition, relative experiments showed that the CMC-coated nanoparticles could remain stable and directly penetrate the intestine into blood circulation. Meanwhile, loaded DOX was released in the small intestine under mildly acidic conditions (pH 5.0).
|
Download:
|
| Fig. 1. Illustration of the delivery process after oral administration of intestinepenetrating and double-layered nanoparticles, which remained stable through stomach and improve the intestinal penetration by opening tight junctions. Copied with permission [84]. Copyright 2019, The Royal Society of Chemistry. | |
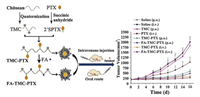
To further enhance the oral availability, various approaches have been applied to optimize the chemophysiological properties of CMC-based vehicles. One method is chemical modification of chitosan. For example, He et al. synthesized trimethyl embedded chitosan (TMC) [85] to improve the solubility in strong-acid environment. This TMC based vesicle demonstrated excellent capacity in opening tight junction and its transport rate is 15.5 folds higher than free paclitaxel (PTX). Meanwhile, pH sensitive release profile could prevent the rapid drug loss in the stomach and oral bioavailability during oral delivery. In addition, following the internalization into tumor cells through endocytosis, the conjugates might be held in intracellular compartments such as endosomes and lysosomes (pH 4–6), where the release of PTX from the conjugates would be further protracted [85]. Compared to intravenously injected free PTX [85, 86], TMC-based oral PTX delivery showed significant enhanced anti-cancer effects (Fig. 2). PTX loaded TMC nanoparticles presented a sustained release of PTX through the degradation of the ester bond without burst release, and the sustained drug release was benefit to prolong its half-life in gastrointestinal tract. In addition, arginine was also applied to chitosan modification by grafting onto carboxyl (CS-N-Arg), which can remain positively charged under neutral and basic pH to further improve the oral availability by interfering with P-gp and opening tight junctions [73]. In vitro Caco-2 cell paracellular transport measurement showed that CS-N-Arg based nanoparticles possess an enhanced transport ability with FITCgrafted macromolecule and hydrophobic drug [87]. Besides, thiomer chitosan 4-thiobutylamidine (TBA) [88] was prepared for high level absorption and inhibition in P-gp activity of intestinal epithelial cells. TBA based oral delivery system have shown that the apparent permeability coefficients (Papp) was increased to 21.9×10-6 cm/s and the efflux ratios was decreased to 0.5 [89]. For physical modification, Krajicek synthesized chitosan–EDTA complex by positive and negative electroadsorption, demonstrating greater effect on adhesion compared to free chitosan [90]. Meanwhile, chitosan could be also applied to incorporate with other materials as oral delivery platform [91-93]. For example, chitosan has been used to strengthen gastrointestinal stability and improve the absorption in intestine of porous silicon (PSI). In vitro studies using Caco-2/HT-29 cells indicated that the Papp of CS-conjugated PSI was increased to 7-fold compared to free insulin [94, 95].
|
Download:
|
| Fig. 2. Illustration of the synthesis of TMC-chitosan nanoparticles and the comparison of therapeutic effects of different formulations. Compared to intravenous administrated PTX group, oral PTX-loaded TMC-chitosan nanoparticles achieved similar therapeutic effects with lower toxicity. Copied with permission [85]. Copyright 2017, Elsevier. | |
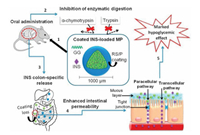
Besides chitosan and its derivatives, other polysaccharides could also affect the intestinal permeability. For instance, gellan gum, a linear anionic polysaccharide with good water solubility, was self-assembled into microparticles and coated with colonspecific films for oral delivery of insulin [96-99]. A pH-dependent release behavior was discovered from gellan gum-based drug carriers under gastrointestinal environments, which indicated that this vehicle could reduce the risk of transient high drug concentration in gastrointestinal tract. Moreover, both in vitro and in vivo permeation tests indicated that the enhancement of insulin permeation was due to the influence of gellan gum on tight junctions opening (Fig. 3), which further extended the effective activity of insulin to 7 h [100]. In addition, hydroxypropyl methylcellulose acetate succinate and celecoxib were also applied synergistically for saturable amorphous drug-loading, which could improve the drug stability and efficiency by 1.5-fold [101]. Alibolandi et al. used the poly(lactic-co-glycolic acid)-grafted dextran to synthesize polymersomes for oral delivery of insulin. In this work, experimental data showed that the transport rate could reach to 16.89%, while free insulin was 3.5%, which could be attributed to the enhanced intestinal penetration capability of dextran-based polymersomes [102-104]. This polymersome could retain the insulin against the proteolytic degradation in the stomach at a lower pH and release the insulin sustainedly in the intestine. Likewise, various small molecules with P-gp inhibition capacity have been introduced onto polysaccharides for enhanced intestinal permeability [55, 56, 105, 106]. Nakashima et al. discovered that the dextran sodium sulfate could weaken the function of P-gp and even down-regulate its expression in intestinal epithelial cells [107]. Besides, lipopolysaccharide could specially target toll-like receptor 4/MyD88 signal-transduction pathway to down-regulate the expression of tight junctions-relative protein for enhanced intestinal permeability [108].
|
Download:
|
| Fig. 3. The oral delivery process of gellan gum (GG) based microparticles, which could enhance the intestinal permeability by interfering with tight junctions and then accelerate paracellular pathway. Copied with permission [100]. Copyright 2018, Elsevier. | |
In general, a number of polysaccharides could improve the intestinal permeability with various strategies and could be applied for efficient oral drug delivery via combining different mechanisms by modification or synergies.
4. Peptide/protein-based intestinal permeability improverPeptide and protein are a kind of biological macromolecules composed of amino acids [109, 110], which perform a wide range of functions in the body [111, 112]. Therefore, peptides and proteins have been widely studied and applied to enhance bioavailability for oral drug delivery [109, 113-116].
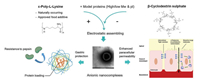
4.1. Linear peptideChristina [117] et al. synthesized a series of amphiphilic peptides, such as ac-A6K-CONH2, KA6-CONH2, ac-A6D-COOH, and DA6-COOH lipid-like linear peptides. These peptides could enhance the electrostatic adhesion to small epithelial cells and therefore increase the transport ratio of FITC-dextran by 7.6-fold compare to free FITC-dextran. Meanwhile, ε-polylysine (εPL, polyL-lysine and poly-D-lysine) produced by Streptomyces albulus is a cationic antimicrobial peptide, which could efficiently sterilize and directly affect intestinal tight junctions by electrostatic interference [118]. Niu [119] et al. utilized the electrostatic interactions of ePL and cyclodextrin (CD) to form nano-complexes as drug carriers (Fig. 4), which could prevent premature release and degradation by digestive enzyme in stomach and release its payload to the small intestine. The peptide based nano-complex could be optimized with appropriate zeta potential (-39±1.5 mV) to avoid clearance by gastric mucosa and overcome the trans-intestinal epithelial electrical resistance for enhanced oral absorption. Besides, certain small molecules were also applied to interact with peptides to achieve desired biological functions. For instance, deuterohemin [120-122], a liposoluble biological iron supplement, was modified with AHTVEK-NH2, which could effectively remove reactive oxygen species and penetrate Caco-2 cell monolayers with enhanced permeability [123]. Likewise, certain bioactive peptides can also be embedded into drug carriers for enhanced oral delivery [124, 125]. For instance, R8 was a typical cell-penetrating peptide to penetrate across intestinal mucosa. Yang et al. [126] grafted R8 onto the carboxymenthyl-β-cyclodextrin (R8-CM-β-CD) to assemble with insulin by pore size recombination, demonstrating desired insulin release kinetics and high level of permeability through interfering the efflux effect of P-gp in streptozotocin-induced diabetic mouse model. Besides, the chemotherapeutic drugs that cannot be absorbed orally could also be modified by these peptides to increase intestinal penetration. Yan et al. [127] used Gly-Sar, a typical substrate of intestinal oligopeptide transporter, to modify didanosine by esterification. In vivo experiment showed that the oral absolute availability of peptide modified prodrug was 47.2%, compared to 7.9% of free drug. Besides, the active parent drug could not be released from the prodrug in gastrointestinal tract, but could be released in intestine. Moreover, there are also a series of peptides with specific bioactivity modified with polysaccharide, chitosan and its derivative for better oral delivery efficiency. For example, chitosan–histidine–cysteine (CHC) was synthesized via specific sequence grating, which could integrate the advantages of peptide and polysaccharide to conquer various cellular and systemic barriers [128]. Then, the shRNA with tumor inhibiting effect was loaded into CHC-based nano-complex, whose oral delivery efficiency was 4 times higher than free shRNA. Similarly, a series of cell penetrating peptide, such as SAR6EW [129], CSKSSDYQC (CSK) [124] and FQSIYPpIK (FQS) [130] were also grafted onto chitosan or chitosan derivative for enhanced intestinal permeability.
|
Download:
|
| Fig. 4. The preparation of β-CD-based nano-complex and its mechanism in enhancing intestinal permeability. Copied with permission [119]. Copyright 2019, Elsevier. | |
Cyclic peptides contain a circular sequence of amino acids through a connection between the amino and carboxyl ends of peptides [131]. Many efforts have been devoted to the study of cyclic peptides, demonstrating a series of bioactivities, such as anticancer, antibacterial, antifungal, and intestine permeability [132, 133]. In this section, recent progress on cyclic peptide based oral delivery has been summarized. Cyclosporine A (CyA) was commonly used as immunosuppressant and enhancer of oral bioavailability, which could be contributed to the high lipophilicity and inhibition in transport function of P-gp [133, 134]. For example, poly(methacrylic acid-co-methacrylate) copolymer was co-assembled with CyA through quasi-emulsion solvent diffusion technique. In this work, the oral availability of CyA was increased by 32.5% due to the efflux capacity of intestinal epithelial cells [135]. Likewise, cyclosporine-like cyclic peptides such as KI-306 [136], NIM811 [137, 138], SCY-635 [138] and Alisporivir [139], were also demonstrated with great intestinal permeability. Qian et al. [140] prepared a series of amphiphilic cell penetrating cyclopeptide for oral delivery. Briefly, the cytosolic delivery efficiency had increased up to 120% than that was 2% in HIV Tat peptide (YGRKKRRQRRR), a typical mammalian cell membrane translocate peptide. More importantly, the maximal plasma concentration reached 3.2 μmol/L after oral administration of cyclo(FΦRRRRQ, Φ: L-2-naphthylalanine), which was induced by its proteolytic stability and high membrane permeability. Besides, the poly alanine cyclic hexapeptide with N-methylation library was prepared for evaluation of intestinal permeability [141]. In vitro membrane permeability assay showed that N-methylation (2-5 N-Me groups) of cyclic peptides possess great permeability (Papp > 2×10-5 cm/s). This work could be beneficial for design and improving the intestinal permeability of cyclic peptide-based drug and delivery [142]. Nielsen et al. [143] discovered that the cyclization makes peptides more stable under enzyme or acid environment. And the further modification of cyclic peptides by N-methyl and N-heterocycle could result in an enhanced gastrointestinal stability and cell adhesion for enhancing intestinal permeability. For instance, cyclic N-methylated hexapeptide containing Arg-Gly-Asp (RGD) was modified by lipophilic prodrug charge masking (LPCM) (Fig. 5). Afterwards, the intestinal absorption pathway was change from paracellular to transcellular, and the Papp of the modified cyclopeptide was significantly increased [144]. In addition, Hess et al. [145] studied the impact of peptide modification on stability and intestinal permeability. For hexapeptide Phe-Gly-Gly-Gly-Gly-Phe, modification of C-methylation (Gly to Ala), N-methylation and cyclization revealed increased enzymatic and acidic stability and cell permeability.

|
Download:
|
| Fig. 5. The absorption strategy of cyclic peptides was changed from paracellular to transcellular by lipophilic prodrug charge masking (LPCM) approach. Copied with permission [144]. Copyright 2018, American Chemical Society. | |
Compared with peptides, proteins have larger molecular weight and more complex spatial structure with more biological functions, which were also used in the field of oral delivery [146-148]. For example, fish sarcoplasmic protein (FSP) has been studied for oral delivery due to its excellent acidic tolerance in gastrointestinal tract [146]. Stephansen et al. [149] used FSP to prepare nanofibers and load insulin by electrospinning, which could maintain a sustained release of insulin for 8 h. More importantly, the permeation efficiency of loaded insulin from FSP-based nanofibers into intestine was increased to 12%, which could be contributed to FSP induced tight junction modulations by disturbing relative protein (ZO-1 and CL-4). Likewise, Whey protein, a common food ingredient, was discovered for oral drug delivery [150, 151], demonstrating a controllable release of encapsulated cargo without any crosslinking in gastrointestinal tract. What is more, Chen et al. [152] discovered that the carboxyl groups in Whey protein could specially bind to Ca2+, and the Whey protein-based delivery could also target the intestinal epithelial cells for sustained drug release in intestinal tract for enhanced intestinal absorption. Meanwhile, this Whey protein based microsphere allows delayed cargo release in stomach and completely release in small intestine by pancreatic degradation. In addition, Lammers et al. [153] observed that Gliadin, the mixture of alcohol soluble protein, could increase intestinal permeability via immune mediation. The mechanism is that Gliadin could induce epithelial zonulin release and then disassembly of tight junctions. Besides, dietary soya saponins have also been developed to increase gut permeability by interfering cell membrane [154], which could reduce enteritis causing by oral administration.
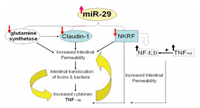
5. MicroRNA-based intestinal penetration promoterMicroRNA is a kind of genetic material, which could regulate protein's expression in intestinal epithelial cells to improve the permeability of small intestine [155]. Meanwhile, different from microRNA, polysaccharides and linear peptide based vehicles could increase intestinal absorption of cargos through physical absorption, bio-specific recognition, and interfering protein activity [67, 68, 118, 119, 131, 147]. Meanwhile, miRNA itself can increase the permeability of small intestine via regulating the expression of intracellular proteins. Similarly, cyclic peptide itself also enhances intestinal permeability via targeted absorption or regulating absorption strategy from paracellular to transcellular. A lot of efforts have been devoted to the study of microRNA and its effects of improving intestinal permeability. For example, Zhou et al. [156] evaluated the performance of microRNA-29a (MIR29A) in 19 diarrhea-predominant IBS patients, indicating increased intestinal membrane permeability with upregulation of miRNA expression. Besides, it was also discovered that the tight junction between intestinal epithelial cells was related to the level of tumor necrosis factor (TNF)-α [157, 158]. In addition, the mechanism and process of microRNA to increase intestinal permeability were further studied (Fig. 6) [159]. The high level of MIR29A and B could decrease the amount of glutamine synthetase to down-regulate claudin-1 (CLDN1) signaling for increased intestinal permeability, which was proved to be associated with various proteins between small intestinal epithelial cells. In addition, it was demonstrated that both CLDN1 and nuclear factor-kB–repressing factor (NKRF) were both the key regulators for intestinal permeability [160]. In general, miR-29a/b can obviously improve intestinal permeability by the un-regulated CLDN1 and NKRF. Moreover, bioactive microRNA could also facilitate intestinal absorption by opening the tight junction. For instance, due to the binding between miR- 122A and 30-untranslated region of occludin mRNA for induced degradation, the expression of occludin was significantly reduced and the intestinal permeability was increased [161]. In this work, Papp of insulin increased to ~10-fold than that in the control group. Zhang et al. [162] delved into the performance of TNF-α caused by miR-21 in Caco-2 monolayers. The results showed that the overexpressed TNF-α could decrease the level of transepithelial electrical resistance by 20% compared with the control group. The downstream proteins such as IL-6, IL-8 and PGE2 level was downregulated, and then the amount of occludin and CLDN1 were also decreased to unlock the tight junctions. In addition, many other RNAs, such as miR-122a [163], miR-223 [164], miR-155 [165], also have similar biological activities to increase the intestinal permeability [166].
|
Download:
|
| Fig. 6. The influence of intestinal permeability by miR-29 regulation. Briefly, miR- 29 directly down-regulated glutamine synthetase, CLDN1 and NKRF, then expression of NF-κB obviously increase. Finally, relative combined action devoted to increase intestinal permeability. Copied with permission [159]. Copyright 2015, Elsevier. | |
In recent years, an increasing interest of biomacromolecule based oral delivery has been developed due to its excellent biocompatibility, tunable degradability, and tunable biofunctions. In addition, the diverse bioactivities of biomacromolecules could be applied to increase intestinal permeability, which is the key factor for efficient drug delivery and bioavailability. In this review, we summarized the mechanism and applications of relevant biomacromolecules in enhancing intestinal permeability.
Briefly, biomacromolecules could enhance the intestinal permeability via various mechanisms, such as directly penetration, promoting intestinal adhesion, accelerating intestinal cell uptake, slowing efflux, and even physically or biologically regulating tight junctions. For example, some biomacromolecule-based vehicles could penetrate intestine via naturally absorption (Schemes 1e–g) or physical bonding (Scheme 1a), which could be contributed to the absorbability and electropositivity of biomacromolecules, such as chitosan and ε-polylysine. Moreover, the specific reporters and transport protein on the cell member could also be used to accelerate capturing and transporting drug carriers (Schemes 1b and h), such as increased intestinal permeability by lipopolysaccharides in targeting toll-like receptor 4. In addition, the high efflux active of P-glycoprotein on small intestinal epithelial cell membrane is one of the major biological delivery barriers, which could be interfered by certain biomacromolecules, such as arginine-grafted chitosan, dextran sodium sulfate and cyclosporine A (Scheme 1c). Besides, the tight junctions could be opened by the negative chitosan (such as carboxymethyl chitosan) via coordination with Ca2+ on the surface of intestinal epithelial cells. Meanwhile, microRNA could biologically down-regulate the proteins associated with tight junctions to improve intestinal penetrability (Scheme 1d).
The approach for increasing intestinal permeability could be applied for a series of clinical oral administration, such as improving insulin absorption, increasing the bioavailability of oral cargos, facilitating direct intestinal penetration of large-sized drug carriers and even changing non-oral drugs to oral dosage forms. These biomacromolecules for oral drug delivery could reduce administered dose, and increase bioavailability and versatility to accommodate various types of drugs. Although tremendous progress has been made in oral drug administration, there are still certain directions worthy of exploration: (1) The combined effects of different biomacromolecules in oral administration need to be studied. For example, the unsatisfied stability of microRNA could be improved by the introducing chitosan and its derivatives. Meanwhile the polysaccharide could also be implemented with microRNA for additional protein regulation function; (2) The intersecting sections of the mechanism of biomacromolecules and drug should be further studied. For example, the therapeutic biomacromolecules (peptide or miRNA) and chemotherapeutic drug might share similar action pathway, which could help to design multi-component delivery platform; (3) Current targeting efficiency of oral delivered drug is still low, which could be improved with the help of external fields. For instance, magnetic, electrical, and gravitational fields could actively guide the drug delivery and release. Overall, biomacromolecules capable of enhancing intestinal permeability could advance the oral drug administration and will be of great value to scientists and clinician in a wide range of sectors.
Declaration of competing interestThe authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
AcknowledgmentsThis research was supported by the National Natural Science Foundation of China (No. 51803152) and Natural Science Foundation of Shanghai (No. 19ZR1478800).
| [1] |
S. Isanaka, O. Guindo, C. Langendorf, et al., N. Engl. J. Med. 376 (2017) 1121-1130. DOI:10.1056/NEJMoa1609462 |
| [2] |
A. Freifeld, D. Marchigiani, T. Walsh, et al., N. Engl. J. Med. 341 (1999) 305-311. DOI:10.1056/NEJM199907293410501 |
| [3] |
C.W. Pouton, C.J. Porter, Adv. Drug Delivery Rev. 60 (2008) 625-637. DOI:10.1016/j.addr.2007.10.010 |
| [4] |
K. Maisel, L. Ensign, M. Reddy, R. Cone, J. Hanes, J. Control. Release 197 (2015) 48-57. DOI:10.1016/j.jconrel.2014.10.026 |
| [5] |
M.W. Tibbitt, J.E. Dahlman, R. Langer, J. Am. Chem. Soc. 138 (2016) 704-717. DOI:10.1021/jacs.5b09974 |
| [6] |
M. Koziolek, M. Grimm, F. Schneider, et al., Adv. Drug Delivery Rev. 101 (2016) 75-88. DOI:10.1016/j.addr.2016.03.009 |
| [7] |
J. Florek, R. Caillard, F. Kleitz, Nanoscale 9 (2017) 15252-15277. DOI:10.1039/C7NR05762H |
| [8] |
Q. Hu, M. Wu, C. Fang, C. Cheng, et al., Nano Lett. 15 (2015) 2732-2739. DOI:10.1021/acs.nanolett.5b00570 |
| [9] |
T.X. Nguyen, L. Huang, M. Gauthier, et al., Nanomedicine 11 (2016) 1169-1185. DOI:10.2217/nnm.16.9 |
| [10] |
D.C. Forbes, N.A. Peppas, J. Control, Release 162 (2012) 438-445. DOI:10.1016/j.jconrel.2012.06.037 |
| [11] |
E. Mathiowitz, J.S. Jacob, Y.S. Jong, et al., Nature 386 (1997) 410-414. DOI:10.1038/386410a0 |
| [12] |
B. Devriendt, B.G. de Geest, B.M. Goddeeris, E. Cox, J. Control, Release 160 (2012) 431-439. DOI:10.1016/j.jconrel.2012.02.006 |
| [13] |
L.M. Ensign, R. Cone, J. Hanes, Adv. Drug Delivery Rev. 64 (2012) 557-570. DOI:10.1016/j.addr.2011.12.009 |
| [14] |
M. Goldberg, I. Gomez-Orellana, Nat. Rev. Drug Discovery 2 (2003) 289-295. DOI:10.1038/nrd1067 |
| [15] |
S. Rojas, T. Baati, L. Njim, et al., J. Am. Chem. Soc. 140 (2018) 9581-9586. DOI:10.1021/jacs.8b04435 |
| [16] |
T. Ren, Q. Wang, Y. Xu, et al., J. Control. Release 269 (2018) 423-438. DOI:10.1016/j.jconrel.2017.11.015 |
| [17] |
H.A. Gordon, E. Bruckner-Kardoss, Am. J. Physiol. 201 (1961) 175-178. DOI:10.1152/ajplegacy.1961.201.1.175 |
| [18] |
P. Kotanko, M. Carter, N.W. Levin, Dial. Nephrol, Transplant. 21 (2006) 2057-2060. |
| [19] |
X. Zhu, J. Wu, W. Shan, et al., Adv. Funct. Mater. 26 (2016) 2728-2738. DOI:10.1002/adfm.201505000 |
| [20] |
J. Huang, Q. Shu, L. Wang, et al., Biomaterials 39 (2015) 105-113. DOI:10.1016/j.biomaterials.2014.10.059 |
| [21] |
F. Xia, W. Fan, S. Jiang, et al., ACS Appl. Mater. Interfaces 9 (2017) 21660-21672. DOI:10.1021/acsami.7b04916 |
| [22] |
A. Banerjee, J. Qi, R. Gogoi, J. Wong, S. Mitragotri, J. Control, Release 238 (2016) 176-185. DOI:10.1016/j.jconrel.2016.07.051 |
| [23] |
A. Banerjee, K. Ibsen, T. Brown, et al., Proc. Natl. Acad. Sci. U. S. A. 115 (2018) 7296-7301. DOI:10.1073/pnas.1722338115 |
| [24] |
A.A. Date, J. Hanes, L.M. Ensign, J. Control, Release 240 (2016) 504-526. DOI:10.1016/j.jconrel.2016.06.016 |
| [25] |
E. Moroz, S. Matoori, J.C. Leroux, Adv. Drug Delivery Rev. 101 (2016) 108-121. DOI:10.1016/j.addr.2016.01.010 |
| [26] |
K. Liang, R. Ricco, C.M. Doherty, et al., Nat. Commun. 6 (2015) 7240.
|
| [27] |
J. Wang, X.C. Chen, Y.F. Xue, et al., Biomaterials 192 (2019) 15-25. DOI:10.1016/j.biomaterials.2018.10.038 |
| [28] |
Y. Zhang, T. Sun, C. Jiang, Acta Pharm. Sin. B 8 (2018) 34-50. DOI:10.1016/j.apsb.2017.11.005 |
| [29] |
S.P. Rigby, M. Fairhead, C.F. van der Walle, Curr. Pharm. Design 14 (2008) 1821-1831. DOI:10.2174/138161208784746671 |
| [30] |
Y. Luo, Z. Teng, Y. Li, Q. Wang, Carbohydr. Polym. 122 (2015) 221-229. DOI:10.1016/j.carbpol.2014.12.084 |
| [31] |
A.O. Elzoghby, W.M. Samy, N.A. Elgindy, J. Control. Release 161 (2012) 38-49. DOI:10.1016/j.jconrel.2012.04.036 |
| [32] |
Y. Liu, F.Q. Wang, Z. Shah, et al., Colloids Surf. B 145 (2016) 492-501. DOI:10.1016/j.colsurfb.2016.05.035 |
| [33] |
T.X. Nguyen, L. Huang, L. Liu, et al., J. Mater. Chem. B 2 (2014) 7149-7159. DOI:10.1039/C4TB00876F |
| [34] |
K.Y. Win, S.S. Feng, Biomaterials 26 (2005) 2713-2722. DOI:10.1016/j.biomaterials.2004.07.050 |
| [35] |
J. Bhandari, H. Mishra, P.K. Mishra, et al., Int. J. Nanomed. 12 (2017) 2021-2031. DOI:10.2147/IJN.S124318 |
| [36] |
D.C. Aduba Jr., J.A. Hammer, Q. Yuan, et al., Acta Biomater. 9 (2013) 6576-6584. DOI:10.1016/j.actbio.2013.02.006 |
| [37] |
A.H. Krauland, D. Guggi, A. Bernkop-Schnürch, J. Control, Release 95 (2004) 547-555. DOI:10.1016/j.jconrel.2003.12.017 |
| [38] |
S. Mansuri, P. Kesharwani, R.K. Tekade, N.K. Jain, Eur. J. Pharm. Biopharm. 102 (2016) 202-213. DOI:10.1016/j.ejpb.2015.10.015 |
| [39] |
Y.H. Lin, H.F. Liang, C.K. Chung, M.C. Chen, H.W. Sung, Biomaterials 26 (2005) 2105-2113. DOI:10.1016/j.biomaterials.2004.06.011 |
| [40] |
M. Yoshida, N. Kamei, K. Muto, et al., Eur. J. Pharm. Biopharm. 112 (2017) 138-142. DOI:10.1016/j.ejpb.2016.11.029 |
| [41] |
K. Iwanaga, S. Ono, K. Narioka, et al., Int. J. Pharm. 157 (1997) 73-80. DOI:10.1016/S0378-5173(97)00237-8 |
| [42] |
J. Salonen, L. Laitinen, A. Kaukonen, et al., J. Control. Release 108 (2005) 362-374. DOI:10.1016/j.jconrel.2005.08.017 |
| [43] |
Y. Zheng, J. Wu, W. Shan, et al., ACS Appl. Mater. Interfaces 10 (2018) 34039-34049. DOI:10.1021/acsami.8b13707 |
| [44] |
Y.W. Wang, L.Y. Chen, F.P. An, M.Q. Chang, H.B. Song, Food Hydrocolloid 84 (2018) 68-74. DOI:10.1016/j.foodhyd.2018.05.039 |
| [45] |
W. Fan, D. Xia, Q. Zhu, et al., Biomaterials 151 (2018) 13-23. DOI:10.1016/j.biomaterials.2017.10.022 |
| [46] |
S.V. Lale, H.S. Gill, Int. J. Pharm. 552 (2018) 352-359. DOI:10.1016/j.ijpharm.2018.10.016 |
| [47] |
B. Ghalandari, A. Divsalar, A.A. Saboury, K. Parivar, J. Iran. Chem. Soc. 12 (2015) 613-619. DOI:10.1007/s13738-014-0519-2 |
| [48] |
O. Zupanci 9 c, A. Partenhauser, H.T. Lam, J. Rohrer, A.B. Schnürch, Eur. J. Pharm. 9 Sci. 81 (2016) 129-136. DOI:10.1016/j.ejps.2015.10.005 |
| [49] |
S. Jana, N. Maji, A.K. Nayak, K.K. Sen, S.K. Basu, Carbohydr. Polym. 98 (2013) 870-876. DOI:10.1016/j.carbpol.2013.06.064 |
| [50] |
Y. Zhang, L.Z. Benet, Clin. Pharmacokinet. 40 (2001) 159-168. DOI:10.2165/00003088-200140030-00002 |
| [51] |
M.J. Alonso, Biomed. Pharmacother. 58 (2004) 168-172. DOI:10.1016/j.biopha.2004.01.007 |
| [52] |
Y.H. Ma, M.Y. Wei, Y.Y. Liu, et al., Chin. Chem. Lett. 27 (2016) 215-220. DOI:10.1016/j.cclet.2015.10.003 |
| [53] |
M. Liu, J. Zhang, X. Zhu, et al., J. Control. Release 222 (2016) 67-77. DOI:10.1016/j.jconrel.2015.12.008 |
| [54] |
J. Wang, M. Kong, Z. Zhou, et al., Carbohydr. Polym. 157 (2017) 596-602. DOI:10.1016/j.carbpol.2016.10.021 |
| [55] |
L.Z. Benet, T. Izumi, Y. Zhang, J.A. Silverman, V.J. Wacher, J. Control. Release 62 (1999) 25-31. DOI:10.1016/S0168-3659(99)00034-6 |
| [56] |
D. Mandracchia, A. Trapani, G. Tripodo, et al., Carbohydr. Polym. 166 (2017) 73-82. DOI:10.1016/j.carbpol.2017.02.096 |
| [57] |
A.F. Räder, F. Reichart, M. Weinmüller, H. Kessler, Bioorg. Med. Chem. 26 (2018) 2766-2773. DOI:10.1016/j.bmc.2017.08.031 |
| [58] |
A. Kotze, H. Luessen, A. de Boer, J. Verhoef, H. Junginger, Eur. J. Pharm. Sci. 7 (1999) 145-151. DOI:10.1016/S0928-0987(98)00016-5 |
| [59] |
B.J. Aungst, J. Pharm. Sci. 89 (2000) 429-442. DOI:10.1002/(SICI)1520-6017(200004)89:4<429::AID-JPS1>3.0.CO;2-J |
| [60] |
Z. Li, H. Jiang, C.M. Xu, L.W. Gu, Food Hydrocolloid 43 (2015) 153-164. DOI:10.1016/j.foodhyd.2014.05.010 |
| [61] |
Y. Yu, M. Shen, Q. Song, J. Xie, Carbohydr. Polym. 183 (2018) 91-101. DOI:10.1016/j.carbpol.2017.12.009 |
| [62] |
P. Cazón, G. Velazquez, J.A. Ramírez, M. Vázquez, Food Hydrocolloid 68 (2017) 136-148. DOI:10.1016/j.foodhyd.2016.09.009 |
| [63] |
A. Kumar, K.M. Rao, S.S. Han, Carbohydr. Polym. 180 (2018) 128-144. DOI:10.1016/j.carbpol.2017.10.009 |
| [64] |
Y. Dong, J.B. Matson, K.J. Edgar, Biomacromolecules 18 (2017) 1661-1676. DOI:10.1021/acs.biomac.7b00364 |
| [65] |
G.Y. Zhang, J. Wu, Y.R. Liu, et al., Chin. Chem. Lett. 29 (2018) 1861-1864. DOI:10.1016/j.cclet.2018.12.004 |
| [66] |
Y. Zhang, T. Sun, C. Jiang, Acta Pharm. Sin. B 8 (2018) 34-50. DOI:10.1016/j.apsb.2017.11.005 |
| [67] |
M.N. Sithole, Y.E. Choonara, L.C. du Toit, P. Kumar, V. Pillay, Pharm. Dev. Technol. 22 (2017) 283-295. DOI:10.1080/10837450.2016.1212882 |
| [68] |
L. Zhang, Y. Sang, J. Feng, Z. Li, A. Zhao, J. Drug Target. 24 (2016) 579-589. DOI:10.3109/1061186X.2015.1128941 |
| [69] |
I. Hamed, F. Özogul, J.M. Regenstein, Trends Food Sci. Tech. 48 (2016) 40-50. DOI:10.1016/j.tifs.2015.11.007 |
| [70] |
D.J. Fu, Y. Jin, M.Q. Xie, et al., Chin. Chem. Lett. 25 (2014) 1435-1440. DOI:10.1016/j.cclet.2014.06.027 |
| [71] |
R. LogithKumar, A. KeshavNarayan, S. Dhivya, et al., Carbohyd. Polym. 151 (2016) 172-188. DOI:10.1016/j.carbpol.2016.05.049 |
| [72] |
M. Sun, L. He, X. Wang, R.P. Tang, J. Mater. Chem. B 7 (2019) 240-250. DOI:10.1039/C8TB02742K |
| [73] |
C.H. Chen, Y.S. Lin, S.J. Wu, F.L. Mi, Carbohyd. Polym. 193 (2018) 163-172. DOI:10.1016/j.carbpol.2018.03.080 |
| [74] |
J. Sheng, L. Han, J. Qin, et al., ACS Appl. Mater. Interfaces 7 (2015) 15430-15441. DOI:10.1021/acsami.5b03555 |
| [75] |
K. Roy, H.Q. Mao, S.K. Huang, K.W. Leong, Nat. Med. 5 (1999) 387-391. DOI:10.1038/7385 |
| [76] |
C. Feng, Z. Wang, C. Jiang, et al., Int. J. Pharm. 457 (2013) 158-167. DOI:10.1016/j.ijpharm.2013.07.079 |
| [77] |
K. Sonaje, E.Y. Chuang, K.J. Lin, et al., Mol. Pharm. 9 (2012) 1271-1279. DOI:10.1021/mp200572t |
| [78] |
R. Rosenthal, D. Günzel, C. Finger, et al., Biomaterials 33 (2012) 2791-2800. DOI:10.1016/j.biomaterials.2011.12.034 |
| [79] |
T.H. Yeh, L.W. Hsu, M.T. Tseng, et al., Biomaterials 32 (2011) 6164-6173. DOI:10.1016/j.biomaterials.2011.03.056 |
| [80] |
L.W. Hsu, P.L. Lee, C.T. Chen, et al., Biomaterials 33 (2012) 6254-6263. DOI:10.1016/j.biomaterials.2012.05.013 |
| [81] |
M. Thanou, J. Verhoef, H. Junginger, Drug Delivery Rev. 52 (2001) 117-126. DOI:10.1016/S0169-409X(01)00231-9 |
| [82] |
F. Föger, T. Schmitz, A. Bernkop-Schnürch, Biomaterials 27 (2006) 4250-4255. DOI:10.1016/j.biomaterials.2006.03.033 |
| [83] |
A. Bernkop-Schnürch, S. Dünnhaupt, Eur. J. Pharm. Biopharm. 81 (2012) 463-469. DOI:10.1016/j.ejpb.2012.04.007 |
| [84] |
M. Sun, D. Li, X. Wang, et al., J. Mater. Chem. B 7 (2019) 3692-3703. DOI:10.1039/C9TB00212J |
| [85] |
R. He, C. Yin, Acta Biomater. 53 (2017) 355-366. DOI:10.1016/j.actbio.2017.02.012 |
| [86] |
F. Chen, Z.R. Zhang, Y. Huang, Int. J. Pharm. 336 (2007) 166-173. DOI:10.1016/j.ijpharm.2006.11.027 |
| [87] |
P.C. Pinheiro, C.T. Sousa, J.P. Araújo, A.J. Guiomar, T. Trindade, J. Colloid Interface Sci. 410 (2013) 21-26. DOI:10.1016/j.jcis.2013.07.065 |
| [88] |
M. Werle, M. Hoffer, J. Control. Release 111 (2006) 41-46. DOI:10.1016/j.jconrel.2005.11.011 |
| [89] |
S. Dünnhaupt, J. Barthelmes, D. Rahmat, et al., Mol. Pharmaceutics 9 (2012) 1331-1341. DOI:10.1021/mp200598j |
| [90] |
A. Bernkop-Schnürch, M.E. Krajicek, J. Control, Release 50 (1998) 215-223. DOI:10.1016/S0168-3659(97)00136-3 |
| [91] |
P. Mukhopadhyay, R. Mishra, D. Rana, P.P. Kundu, Prog. Polym. Sci. 37 (2012) 1457-1475. DOI:10.1016/j.progpolymsci.2012.04.004 |
| [92] |
H. Yamamoto, Y. Kuno, S. Sugimoto, H. Takeuchi, Y. Kawashima, J. Control. Release 102 (2005) 373-381. DOI:10.1016/j.jconrel.2004.10.010 |
| [93] |
C. Prego, D. Torres, E. Fernandez-Megia, et al., J. Control. Release 111 (2006) 299-308. DOI:10.1016/j.jconrel.2005.12.015 |
| [94] |
N. Shrestha, M.A. Shahbazi, F. Araújo, et al., Biomaterials 35 (2014) 7172-7179. DOI:10.1016/j.biomaterials.2014.04.104 |
| [95] |
N. Zhang, Q. Ping, G. Huang, et al., Int. J. Pharmaceut. 327 (2006) 153-159. DOI:10.1016/j.ijpharm.2006.07.026 |
| [96] |
T. Osmałek, A. Froelich, S. Tasarek, Int. J. Pharm. 466 (2014) 328-340. DOI:10.1016/j.ijpharm.2014.03.038 |
| [97] |
K.M. Zia, S. Tabasum, M.F. Khan, et al., Int. J. Biol. Macromol. 109 (2018) 1068-1087. DOI:10.1016/j.ijbiomac.2017.11.099 |
| [98] |
J. Li, K. Kamath, C. Dwivedi, J. Biomater. Appl. 15 (2001) 321-343. DOI:10.1106/R3TF-PT7W-DWN0-1RBL |
| [99] |
B. Jansson, H. Hägerström, N. Fransén, K. Edsman, E. Björk, Eur. J. Pharm. Biopharm. 59 (2005) 557-564. DOI:10.1016/j.ejpb.2004.10.001 |
| [100] |
A.B. Meneguin, E. Beyssac, G. Garrait, H. Hsein, B.S.F. Cury, Eur. J. Pharm. Biopharm. 123 (2018) 84-94. DOI:10.1016/j.ejpb.2017.11.012 |
| [101] |
A.L. Lainé, D. Price, J. Davis, et al., Int. J. Pharm. 512 (2016) 118-125. DOI:10.1016/j.ijpharm.2016.08.034 |
| [102] |
M. Alibolandi, F. Alabdollah, F. Sadeghi, et al., J. Control. Release 227 (2016) 58-70. DOI:10.1016/j.jconrel.2016.02.031 |
| [103] |
C. Houga, J. Giermanska, S. Lecommandoux, et al., Biomacromolecules 10 (2008) 32-40. |
| [104] |
W.H. Chiang, Y.J. Lan, Y.C. Huang, et al., Polymer 53 (2012) 2233-2244. DOI:10.1016/j.polymer.2012.03.030 |
| [105] |
M.J. Gomes, P.J. Kennedy, S. Martins, B. Sarmento, Nanomedicine 12 (2017) 1385-1399. DOI:10.2217/nnm-2017-0023 |
| [106] |
C. Chang, T.R. Wang, Q.B. Hu, et al., Food Hydrocolloid. 70 (2017) 143-151. DOI:10.1016/j.foodhyd.2017.03.033 |
| [107] |
H. Iizasa, N. Genda, T. Kitano, et al., J. Pharm. Sci. 92 (2003) 569-576. DOI:10.1002/jps.10326 |
| [108] |
M. Nighot, R. Al-Sadi, S. Guo, et al., Am. J. Pathol. 187 (2017) 2698-2710. DOI:10.1016/j.ajpath.2017.08.005 |
| [109] |
M. Abbas, Q. Zou, S. Li, X. Yan, Adv. Mater. 29 (2017) 1605021.
|
| [110] |
I. Pusterla, J.W. Bode, Nat. Chem. 7 (2015) 668-672. DOI:10.1038/nchem.2282 |
| [111] |
J. Rizo, L.M. Gierasch, Annu. Rev. Biochem. 61 (1992) 387-416. DOI:10.1146/annurev.bi.61.070192.002131 |
| [112] |
R. Neme, C. Amador, B. Yildirim, E. McConnell, D. Tautz, Nat. Ecol. Evol. 1 (2017) 0127.
|
| [113] |
N. Zhang, F. Zhao, Q. Zou, et al., Small 12 (2016) 5936-5943. DOI:10.1002/smll.201602339 |
| [114] |
N. Habibi, N. Kamaly, A. Memic, H. Shafiee, Nano Today 11 (2016) 41-60. DOI:10.1016/j.nantod.2016.02.004 |
| [115] |
M. Kristensen, D. Birch, H. Mørck Nielsen, Int. J. Mol. Sci. 17 (2016) 185.
|
| [116] |
S. Zhao, J. Li, F. Wang, et al., Chin. Chem. Lett. 31 (2020) 1147-1152. DOI:10.1016/j.cclet.2019.07.009 |
| [117] |
C. Karavasili, M. Spanakis, D. Papagiannopoulou, et al., J. Pharm. Sci. 104 (2015) 2304-2311. DOI:10.1002/jps.24484 |
| [118] |
Y. Liu, L. Shi, L. Su, H. C, et al., Chem. Soc. Rev. 48 (2019) 428-446. DOI:10.1039/C7CS00807D |
| [119] |
Z. Niu, I. Thielen, A. Barnett, S.M. Loveday, H. Singh, J. Colloid InterfaceSci. 546 (2019) 312-323. DOI:10.1016/j.jcis.2019.03.006 |
| [120] |
F. van de Manakker, K. Braeckmans, N. el Morabit, et al., Adv. Funct. Mater. 19 (2009) 2992-3001. DOI:10.1002/adfm.200900603 |
| [121] |
Y. Ping, Q. Hu, G. Tang, J. Li, Biomaterials 34 (2013) 6482-6494. DOI:10.1016/j.biomaterials.2013.03.071 |
| [122] |
W. Jiang, X. Wang, J. Chen, et al., ACS Appl. Mater. 9 (2017) 26948-26957. DOI:10.1021/acsami.7b09218 |
| [123] |
Q.G. Dong, Y. Zhang, M.S. Wang, et al., Amino Acids 43 (2012) 2431-2441. DOI:10.1007/s00726-012-1322-y |
| [124] |
Y. Jin, Y. Song, X. Zhu, et al., Biomaterials 33 (2012) 1573-1582. DOI:10.1016/j.biomaterials.2011.10.075 |
| [125] |
M. Larche, D.C. Wraith, Nat. Med. 11 (2005) S69.
|
| [126] |
L. Yang, M. Li, Y. Sun, L. Zhang, Int. J. Biol. Macromol. 111 (2018) 685-695. DOI:10.1016/j.ijbiomac.2018.01.077 |
| [127] |
Z. Yan, J. Sun, Y. Chang, et al., Mol. Pharmaceutics 8 (2011) 319-329. DOI:10.1021/mp100376q |
| [128] |
H. Zheng, C. Tang, C. Yin, Biomaterials 70 (2015) 126-137. DOI:10.1016/j.biomaterials.2015.08.024 |
| [129] |
L. Li, L. Yang, M. Li, L. Zhang, Carbohydr. Polym. 174 (2017) 182-189. DOI:10.1016/j.carbpol.2017.06.061 |
| [130] |
Y. Xu, J. Xu, W. Shan, et al., Int. J. Pharm. 500 (2016) 42-53. DOI:10.1016/j.ijpharm.2016.01.028 |
| [131] |
A.K. Malde, T.A. Hill, A. Iyer, D.P. Fairlie, Chem. Rev. 119 (2019) 9861-9914. DOI:10.1021/acs.chemrev.8b00807 |
| [132] |
I.W. Hamley, Chem. Rev. 117 (2017) 14015-14041. DOI:10.1021/acs.chemrev.7b00522 |
| [133] |
Y. Jiang, H.Y. Long, Y.J. Zhu, Y. Zeng, Chin. Chem. Lett. 29 (2018) 1067-1073. DOI:10.1016/j.cclet.2018.05.028 |
| [134] |
X.Q. Wang, J.D. Dai, Z. Chen, et al., J. Control. Release 97 (2004) 421-429. DOI:10.1016/S0168-3659(04)00121-X |
| [135] |
A. Penttilä, P. Uusi-Kyyny, A. Salminen, J. Seppälä, V. Alopaeus, Int. J. Greenhouse Gas Control. 22 (2014) 313-324. DOI:10.1016/j.ijggc.2013.12.001 |
| [136] |
E.L. Lan, S.O. Ugwu, J. Blanchard, et al., J. Pharm. Sci. 83 (1994) 1081-1084. DOI:10.1002/jps.2600830805 |
| [137] |
J.E. Springer, N.P. Visavadiya, P.G. Sullivan, E.D. Hall, J. Neurotrauma 35 (2018) 492-499. DOI:10.1089/neu.2017.5167 |
| [138] |
J. Fu, C. Becker, L. Cao, et al., Bioorg. Med. Chem. 26 (2018) 957-969. DOI:10.1016/j.bmc.2017.09.008 |
| [139] |
C. Stanciu, A. Trifan, C. Muzica, C. Sfarti, Expert Opin. Pharmacother. 20 (2019) 379-384. DOI:10.1080/14656566.2018.1560424 |
| [140] |
Z. Qian, A. Martyna, R.L. Hard, et al., Biochemistry 55 (2016) 2601-2612. DOI:10.1021/acs.biochem.6b00226 |
| [141] |
O. Ovadia, S. Greenberg, J. Chatterjee, et al., Mol. Pharmaceutics 8 (2011) 479-487. DOI:10.1021/mp1003306 |
| [142] |
S.G. Albert, J.E. Morley, Clin. Endocrinol. (Oxf) 85 (2016) 436-443. DOI:10.1111/cen.13084 |
| [143] |
D.S. Nielsen, H.N. Hoang, R.J. Lohman, et al., Int. Ed. 53 (2014) 12059-12063. DOI:10.1002/anie.201405364 |
| [144] |
A. Schumacher-Klinger, J. Fanous, S. Merzbach, M. Weinmüller, et al., Mol. Pharmaceutics 15 (2018) 3468-3477. DOI:10.1021/acs.molpharmaceut.8b00466 |
| [145] |
S. Hess, O. Ovadia, D.E. Shalev, et al., J. Med. Chem. 50 (2007) 6201-6211. DOI:10.1021/jm070836d |
| [146] |
K. Stephansen, I.S. Chronakis, F. Jessen, Colloids Surf. B 122 (2014) 158-165. DOI:10.1016/j.colsurfb.2014.06.053 |
| [147] |
R.L. Merzel, J.J. Chen, E.N.G. Marsh, M.M.B. Holl, Chin. Chem. Lett. 26 (2015) 426-430. DOI:10.1016/j.cclet.2014.12.015 |
| [148] |
J. Xie, Y. Lu, B. Yu, J. Wu, J. Liu, Chin. Chem. Lett. 31 (2020) 1173-1177. DOI:10.1016/j.cclet.2019.10.030 |
| [149] |
K. Stephansen, M. García-Díaz, F. Jessen, I.S. Chronakis, H.M. Nielsen, Int. J. Pharm. 495 (2015) 58-66. DOI:10.1016/j.ijpharm.2015.08.076 |
| [150] |
M. Lehrke, N. Marx, Curr. Opin. Lipidol. 23 (2012) 569-575. DOI:10.1097/MOL.0b013e328359b19f |
| [151] |
K.B. Moore, C.D. Saudek, Am. J. Phytomed. Clin. Ther. 15 (2008) 484-491. DOI:10.1097/MJT.0b013e3180ed42dc |
| [152] |
L. Chen, M. Subirade, Biomaterials 27 (2006) 4646-4654. DOI:10.1016/j.biomaterials.2006.04.037 |
| [153] |
K.M. Lammers, R. Lu, J. Brownley, et al., Gastroenterology 135 (2008) 194-204. DOI:10.1053/j.gastro.2008.03.023 |
| [154] |
D. Knudsen, F. Jutfelt, H. Sundh, et al., Br. J. Nutr. 100 (2008) 120-129. DOI:10.1017/S0007114507886338 |
| [155] |
A. Grishok, A.E. Pasquinelli, D. Conte, et al., Cell 106 (2001) 23-34. DOI:10.1016/S0092-8674(01)00431-7 |
| [156] |
Q. Zhou, W.W. Souba, C.M. Croce, G.N. Verne, Gut 59 (2010) 775-784. DOI:10.1136/gut.2009.181834 |
| [157] |
R. Al-Sadi, S. Guo, D. Ye, M. Rawat, T.Y. Ma, Am. J.Pathol. 186 (2016) 1151-1165. DOI:10.1016/j.ajpath.2015.12.016 |
| [158] |
C. He, L. Yin, C. Tang, C. Yin, Biomaterials 34 (2013) 2843-2854. DOI:10.1016/j.biomaterials.2013.01.033 |
| [159] |
Q. Zhou, S. Costinean, C.M. Croce, et al., Gastroenterology 148 (2015) 158-169. DOI:10.1053/j.gastro.2014.09.037 |
| [160] |
M.B. Zeisel, P. Dhawan, T.F. Baumert, Gut 68 (2019) 547-561. DOI:10.1136/gutjnl-2018-316906 |
| [161] |
C. Cichon, H. Sabharwal, C. Rüter, M.A. Schmidt, Tissue Barriers 2 (2014) e944446.
|
| [162] |
L. Zhang, J. Shen, J. Cheng, X. Fan, Cell Biochem. Funct. 33 (2015) 235-240. DOI:10.1002/cbf.3109 |
| [163] |
D. Ye, S. Guo, R. Al-Sadi, T.Y. Ma, Gastroenterology 141 (2011) 1323-1333. DOI:10.1053/j.gastro.2011.07.005 |
| [164] |
V. Neudecker, M. Haneklaus, O. Jensen, et al., J. Exp. Med. 214 (2017) 1737-1752. DOI:10.1084/jem.20160462 |
| [165] |
Y. Li, Y. Tian, W. Zhu, et al., Biochem. Pharmacol. 116 (2016) 100-106. DOI:10.1016/j.bcp.2016.07.002 |
| [166] |
E. Tili, J.J. Michaille, V. Piurowski, B. Rigot, C.M. Croce, Curr. Opin. Pharmacol. 37 (2017) 142-150. DOI:10.1016/j.coph.2017.10.010 |
 2020, Vol. 31
2020, Vol. 31 

