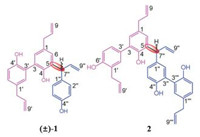
Magnolia officinalis Rehd. et Wils var. biloba (M. officinalis var. biloba), one kind of plant of Magnolia genus, distributed in southern China [1, 2]. It has been reported that the main secondary metabolites of M. officinalis var. biloba [3-5] are neolignanes, phenylethanoid glycosides, alkaloids and meroterpenoids, those compounds showed different biological activities [4-9] including antitumor, PTP1B inhibitory activity, free radical scavenging activity, and neuroprotective activity. The bark of M. officinalis var. biloba, known as 'Hou Pu' in Chinese, has been used as an important traditional Chinese medicine for the treatment and prevent diseases for centuries in China. This is a part of our continuous endeavor to search for more bioactive metabolites from traditional Chinese medicine. Our group had already reported that novel meroterpenoids with remarkable bioactivity were isolated from the bark of M. officinalis var. biloba [5]. In our continuous work, three novel compounds (±)-1 and 2 (Fig. 1) were isolated from the 95% ethanolic extract of the bark of M. officinalis var. biloba, (±)-1 possessed three C6-C3 subunits with new parallel approach, 2 was an unprecedented biphenyl derivative with new connection mode between magnolol and honokiol. Hence, the detailed information including isolation, absolute configuration elucidation and bioactivity evaluation of (±)-1 and 2 was reported.
|
Download:
|
| Fig. 1. Chemical structures of compounds (±)-1 and 2. | |
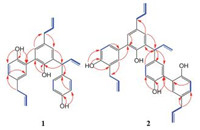
compound 1 was obtained as colorless oil, HRESI-MS of 1 showed pseudomolecule ion peak at m/z 421.1764 [M + Na]+ revealing a molecular formula of C27H26O3 (calcd. for C27H26O3Na, 421.1774), which suggested 15 degrees of unsaturation. The 1H NMR (Table 1) showed a 1, 3, 4-trisubstituted benzene ring [δH 6.93 (brs, 1H, H-20), 6.84 (d, 1H, J = 8.4 Hz, H-50) and 6.97 (dd, 1H, J = 1.8, 8.4 Hz, H-60)], a set of 1, 4-disubstitued phenyl proton signals at dH 6.63 (d, 2H, J = 8.4 Hz, H-3'0, H-500), 6.92 (d, 2H, J = 8.4 Hz, H-200, H-600), and a 1, 3, 4, 5-tetrasubstituted benzene ring proton signals at δH 6.88 (d, 1H, J = 1.8 Hz, H-2), 6.81 (d, 1H, J = 1.8 Hz, H-6), two methylene δH 3.26 (d, 2H, J = 6.6 Hz, H-7), 3.25 (d, 2H, J = 6.0 Hz, H-7') and 5.03 (d, 1H, J = 7.8 Hz, H-7"). With the help of 1H, HSQC, HMBC, and 1H-1H COSY NMR spectra (Figs. S6, S8 - S10 in Supporting information), the 13C NMR spectrum gave 18 aromatic carbon signals, six olefin carbons and three sp3 carbon signals.
|
|
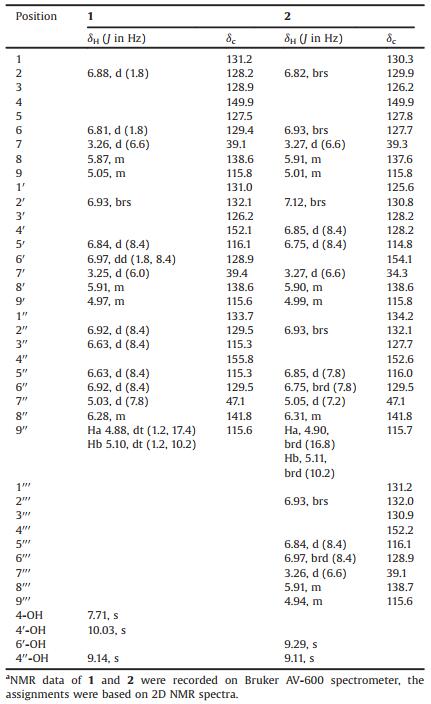
Table 1 1H (600 MHz) and 13C NMR (150 MHz) data of compounds 1 and 2 in DMSO-d6.a |
Along with the 1H-1H COSY cross-peaks (Fig. 2) of H-7/H-8/H-9, H-7'/H-8'/H-9' and H-7"/H-8"/H-9", the key HMBC correlations from H-7 to C-6, H-7' to C-6', from H-7" to C-1" suggested the presence of three C6-C3 subunits (C-1-C-9, C-1'-C-9' and C-1"-C-9") [10, 11]. The HMBC correlation of H-3'/C-2 indicated two allylbenzene moieties (C-1-C-9 and C-1'-C-9') conjugated through a carbon-carbon bond between C-3 and C-3', the HMBC correlations of H-7"/C-4, H-6/C-7" revealed fragment of C-1"-C-9" attached at C-5 by a carbon-carbon bond between C-7" and C-5. Thus, the structure of 1 was established.
|
Download:
|
Fig. 2. Key 1H-1H COSY  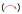 |
|
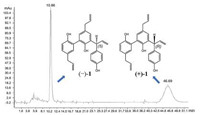
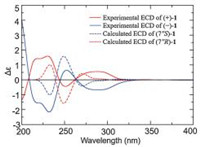
The lack of optical activity and no Cotton effects in the ECD spectrum of 1 indicated that 1 was a racemic mixture. Consequently, an AD-H column yielded (+)-1 ([α]D20 = + 35.8 (c 0.10, CHCl3)) and (-)-1 ([α]D20 = - 36.9 (c 0.10, CHCl3)), in an approximate ratio of 1:1 by chiral-phase HPLC (Fig. 3). The experimental ECD (Fig. 4) of (+)-1 and (-)-1 well match with the theoretically calculated ECD of 7"R-1 and 7"S-1, respectively [12, 13]. Thus, the absolute structures of (+)-1 and (-)-1 were defined as 7"R-1 and 7"S-1, respectively.
|
Download:
|
| Fig. 3. HPLC separation chromatogram of (±)-1 on the chiral AD-H column. | |
|
Download:
|
| Fig. 4. Experimental ECD spectra of (+)-1 and (-)-1 and calculated ECD spectra of (7"R)-1 and (7"S)-1. | |
compound 2, colorless oil, [α]D20 = - 45.2 (c 0.10, CHCl3), the molecular formula, C36H34O4, was deduced by the ion peak (-)-HRESI-MS m/z 529.2385 [M-H]-, (calcd. for C36H33O4, 529.2384), revealing 20 degrees of unsaturation. In combination with the 1D and 2D NMR data (Figs. S14-S18 in Supporting information), the 13C NMR (Table 1) gave four sp3 carbon signals at 39.3 (C-7), 34.3 (C-7'), 47.1 (C-7"), and 39.1 (C-7"'), three double carbon signals and 24 aromatic carbon signals [including four oxygen-bearing carbons δC 149.9 (C-4), 154.1 (C-6'), 152.6 (C-4"), and 152.2 (C-4"')]. Analysis of its 1H and 13C NMR (Table 1) data and comparison with 1H and 13C NMR data of 1 suggested 2 containing four C6-C3 subunits.
The planer structure of 2 was constructed basis on 2D NMR data of 2 (Fig. 2), the HMBC correlations from H-6 to C-7 and C-4, H-20 to C-7', from OH-60 to C-1', C-50, and C-6', the 1H-1H COSY peaks of H-7/H-8/H-9 and H-7'/H-80/H-90 defined two C6-C3 fragments (C-1-C-9 and C-1'-C-9'). The key HMBC correlations from H-20 to C-3 revealed a bond-bond between C-3 and C-3' connected the two subunits (C-1-C-9 and C-1'-C-9') which was described as honokiol [10]. Likewise, along with the 1H-1H COSY cross-peaks of H-7"/H-8"/H-9", H-7"'/H-8"'/H-9"', the HMBC correlations from H-7" to C-1", from OH-4" to C-3'0, C-4", and C-5", from H-8000 to C-1"', from H-2000 to C-3'00 gave a magnolol fragment (C-1"-C-9" and C-1"'-C-9"') [10], the HMBC correlations from H-7" to C-5, from H-6 to C-7" confirmed C-5 of honokiol linked to C-7" of magnolol fragment. Hence, the structure of 2, an adduct containing magnolol and honokiol, was established.
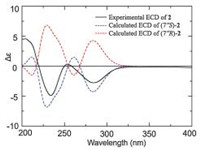
The ECD spectrum of 2 (Fig. 5) showed negative Cotton effects in 230 nm and 280 nm, which was similar to the ECD curve of (-)-1, indicating that 2 possessed the same absolute configuration as (-)-1, moreover, the experimental ECD of 2 well match with the theoretically calculated ECD of 7"S-2. Thus, the absolute structure of 2 was defined as 7"S-2.
|
Download:
|
| Fig. 5. Experimental ECD spectrum of 2 and calculated ECD spectra of (7"S)-2 and (7"R)-2. | |
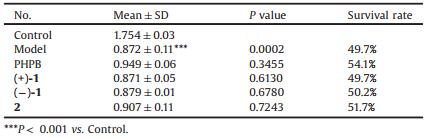
In this work, the isolated compounds (±)-1 and 2 were evaluated for their bioactivity. At a concentration of 10 mmol/L, the neuroprotective effects experiment of (±)-1 and 2 against oxygen glucose deprivation (OGD) and glutamic acid-induced SK-N-SH cell injury [14] indicated that (±)-1 and 2 exhibited weak neuroprotective effects (Tables 2 and 3). In particular 2 increased the OGD-induced SK-N-SH cell injury from 50.2% to 58.4% which more powerful than positive control drug potassium 2-(1-hydroxypentyl)-benzoate (PHPB, 53.8%).
|
|
Table 2 Neuroprotective effects of (±)-1 and 2 on ODG-induced injury of SK-N-SH cells (10 μmol/L). |
|
|
Table 3 Neuroprotective effects of (±)-1 and 2 on glutamic acid-induced injury of SK-N-SH cells (10 μmol/L). |
In summary, we reported (±)-magoilgomer A [(±)-1] and magoilgomer B (2) isolated from the 95% ethanolic extract of the bark of M. officinalis var. biloba, (±)-1 possessed three C6-C3 subunits with new parallel approach, 2 was an unprecedented biphenyl derivative containing magnolol fragment and honokiol fragment. The neuroprotective effects of (±)-1 and 2 were evaluated. 2 exhibited weak neuroprotective effect of ODGinduced SK-N-SH cell injury.
Declaration of competing interestThe authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
AcknowledgmentsThis work was supported by the National Natural Science Foundation of China (No. 81730093), the CAMS Innovation Fund for Medical Sciences (No. 2017-I2M-3-010), In Dependent Project of State Key Laboratory of Bioactive Substance and Function of Natural Medicines (No. GTZA201803).
Appendix A. Supplementary dataSupplementary material related to this article can befound, in the online version, at doi:https://doi.org/10.1016/j.cclet.2019.09.058.
| [1] |
National Pharmacopoeia Committee, Pharmacopoeia of Peoples Republic of China, Part 1, 2005, pp. 251
|
| [2] |
J.S. He, L. Chen, Y. Si, et al., Genetica 135 (2009) 233-243. DOI:10.1007/s10709-008-9272-8 |
| [3] |
J.Y. Wu, Y.B. Wu, Y. Jun, et al., Chin. Trad. Herb. Drugs 44 (2013) 2965-2968. |
| [4] |
L.L. Ge, W.H. Zhang, G. Zhou, et al., Sci. Rep. 7 (2017) 45342. DOI:10.1038/srep45342 |
| [5] |
C. Li, C.J. Li, J. Ma, et al., Org. Lett. 20 (2018) 3682-3686. DOI:10.1021/acs.orglett.8b01476 |
| [6] |
K.S. Suh, S. Chon, W.W. Jung, et al., Chem. Biol. Interact. 277 (2017) 101-109. DOI:10.1016/j.cbi.2017.09.014 |
| [7] |
J.H. Yang, W.S. Wu, J.L. Wen, et al., Biomaterials 141 (2017) 188-198. DOI:10.1016/j.biomaterials.2017.07.002 |
| [8] |
V.B. Pillai, S. Samant, N.R. Sundaresan, et al., Nat. Commun. 6 (2015) 6656. DOI:10.1038/ncomms7656 |
| [9] |
H.C. Ho, C.Y. Hong, J. Biomed. Sci. 19 (2012) 70-78. DOI:10.1186/1423-0127-19-70 |
| [10] |
W. Schuehly, W. Voith, H. Teppner, O. Kunert, J. Nat. Prod. 73 (2010) 1381-1384. |
| [11] |
H.C. Shih, T.L. Hwang, H.C. Chen, et al., PLoS One 8 (2013) e59502. |
| [12] |
L.B. Dong, X.D. Wu, X. Shi, et al., Org. Lett. 18 (2016) 4498-4501. DOI:10.1021/acs.orglett.6b02065 |
| [13] |
L.J. Meng, G.L. Guo, C.G. Zhu, et al., Chin. Chem. Lett. 29 (2018) 119-122. DOI:10.1016/j.cclet.2017.05.019 |
| [14] |
Y. Hu, Y. Peng, Y. Long, et al., Eur. J. Pharmacol. 680 (2012) 49-54. DOI:10.1016/j.ejphar.2012.01.031 |
 2020, Vol. 31
2020, Vol. 31