b School of Biomedical Engineering, Sun Yat-sen University, Guangzhou 510006, China
Peptide vaccines, as the popular therapeutic strategy, are broadly applied in the fields of infection [1, 2], cancer [3-6], etc. due to the outstanding therapeutic effect as well as less side effects. Compared to the attenuated live vaccines and inactivated vaccines, peptide vaccines have the advantages of lower toxicity, convenient preparation and storage. Therefore, they have bright prospects and are expected to become the therapeutic strategy to overcome cancer. In the past five years, more than 50 peptide vaccines have been evaluated in clinical trials (data were obtained from www.Clinicaltrials.gov), however, the clinical evaluation of peptide vaccines is not satisfied as they are expected. A randomized, double-blind phase Ⅲ clinical trial of a personalized peptide vaccine for recurrent glioblastoma (rGBM) failed to reach to the primary endpoints (overall survival, OS) and secondary endpoints (progression-free survival, progression-free survival at 6 months, peptide-specific IgG response), which may be blamed for the peptide antigen [7]. Poor immunogenicity was observed in peptide antigens because they are T-cell independent, which results in the fast elimination and weak immune responses in vivo. At present, how to improve the immunogenicity of antigens and effectively transmit information of antigens to APCs (antigen presenting cells) are the main challenges of immunotherapy.
In order to improve immunogenicity of peptide antigens, a classic strategy is to develop nanovaccine carriers that conjugate with antigens [8, 9]. The investigations have shown that nano-materials have proved to be excellent carriers due to the characteristics of good biocompatibility, immunogenicity and safety; moreover, some adjuvants could be carried by nano-materials by chemical modification. The nanomaterials are not only able to protect antigens from rapid degradation and elimination, but promote antigens uptake by APCs and effectively prolong antigens in vivo [10]. Kuai et al. [11] loaded the antigen peptide to synthetic high-density lipoprotein (sHDL) and found that the nanodiscs could specifically transport Adpgk mutant peptide ASMTNMELM and OVA257-264SIINFEKL to dendritic cells, and promote presentation of peptide and maturation of dendritic cell and activation of T cell. It is reported that [12] peptide E749-57 was loaded onto the injectable scaffolds prepared of mesoporous silica rods (MSRs) and TC-1 tumours were completely eradicated. In the C57BL6/J melanoma model, the vaccine with B16F10 or CT26 as neoantigens displayed outstanding performance against melanoma.
Among a large number of nanomaterials, gold nanoparticles (GNPs) are the most common inorganic nanomaterials and are safe and biocompatible [13, 14]. Thanks to the large surface area, different types of ligands can be loaded onto GNPs [14]. The most popular ligand is glycoside, which is loaded onto the GNPs by covalent interaction together with the peptide antigens; moreover, GNPs can promote the ingestion of peptide antigens by APCs. According to the investigations [15-17], peptide vaccines based on glycosylated GNPs induce a stronger specific immune response in vivo and display inhibition of tumour; which demonstrated that gold nanoparticles have been proven to be the excellent vaccine carrier. Although outstanding performance was found in the GNPs nanovaccine based on the covalent interaction among peptide antigens, ligands and GNPs, however, the complicated route of chemical modification may results in the long cycle of preparation as well as the low yield [18]. Moreover, the nanoparticles size is closely related to the ability to elicit an immune response. However, the size of most glycosylated GNPs based on the covalent interaction are too small to meet the need for the nanovaccine carrier, thus it is necessary to develop a novel nanovaccine based on GNPs with the appropriate size [19-21].
With the continuous development of nano-self-assembly tech-nologies, the preparation of nanoparticles by non-covalent interac-tion has attracted wide attention due to the reversibility of valence interactions. Differing with the covalent interaction, non-covalent interaction realizes the controllable release for drugs. Therefore, we turn our attention to an innovative, effective nano-system based on the non-covalent interactions, the host-guest systems of β-cyclodex-trin (β-CD). β-CD was obtained from nature originally, it is the cyclic oligosaccharides composed of 7 glucose monomers linked by 1, 4-glycosidic bonds with the feature of low toxic, biodegradable, light-absorptive. What is more, there is hydrophilic rim but hydrophobic cavity of β-CD because the "rings" of β-CD are composed of OH of the oligosaccharides while the cavity is affected by shield of C–H bonds. Thus the β-CD often plays a role as a "host" to include some hydrophobic compounds (guest) of appropriate size that match to the cavity to form the host-guest complex [22]. These interactions could make β-CD based nanomaterials self-assembled from several nanometers to micrometers scale. Under the appropriate con-ditions, such as the stimulation of pH [23], light [24, 25], host-guest system will be dissociated, so that the drugs can be released. Studies have shown that the efficacy of drugs can be improved by β-CD host-guest self-assembling system; however, as we know, there has been no report about the application of β-CD host-guest self-assembly system on vaccine carrier.
As discussed above, glycoside-modified gold nanoparticles with covalent bonding are able to enhance the immunogenicity of the peptide antigens. Herein, we have developed a non-covalent glycosylated gold nanoparticles/peptides nanovaccine based on β-CD host-guest self-assembly system for the first time. 1-Adamantanecarboxylic acid (AD) was chosen as the guest because it is of appropriate polarity and matched size to the cavity of β-CD. β-CD was firstly attached to gold nanoparticles by chemical modification as a host group, peptides NY-ESO-1157-165 (SLLMWITQC) or OVA257-264 derived peptide (FFRKSIINFEKL) were conjugated with the AD to form the guest group. According to the investigation, the sequence FFRK of FFRKSIINFEKL is able to promote hydrolysis of the respective epitopes in APCs [26]. Then the novel nanovaccine was developed by host-guest interactions. When the vaccine was injected in vivo, the host and guest dissociated and the antigen was released.
As is shown in Fig. 1, Au-β-CD/AD-Acp-FFRKSIINFEKL (com-pound 4) was synthesized by the following steps. Briefly, the monomers containing host groups (sulfur-gold-β-cyclodextrin, Au-β-CD, compound 3) and guest groups (AD-Acp-FFRKSIINFEKL) were synthesized by classical coupling, respectively (Figs. S1, S2, S10 and S11 in Supporting information). Finally, 4 were prepared by stirring in double distilled water through host-guest inter-actions. Thermogravimetric analysis (TGA, Fig. S4 in Supporting information) was used for the analysis of β-CD content on gold nanoparticles and the ratio of graft is 36.55% (Table S1 in Supporting information).

|
Download:
|
| Fig. 1. Schematic diagram of non-covalent glycosylated gold nanoparticles/ peptides nanovaccine. Reagents and conditions: (a) NaBH4, DMF, HAuCl43H·2O, 24 h, 25.22%. (b) AD-Acp-FFRKSIINFEKL or AD-Acp-SLLMWITQC, r.t., 4 h. (c) C57BL/ 6 J mice were immunized with the vaccine on day 0, 7, 14, 21 and (d) antibody titers were analyzed by ELISA. | |
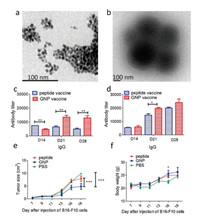
Then, the sizes of compounds 3 and 4 were confirmed by high resolution transmission electron microscopy (HTEM, Figs. 2a and b) and dynamic light scattering analysis (DLS, Figs. S5[26 and S6 in Supporting information). These results displayed that the size of 3 is around 10 nm while the size of 4 is around 200 nm after graft of AD-Acp-FFRKSIINFEKL.
|
Download:
|
| Fig. 2. HTEM analysis of compounds 3 (a) and 4 (b). IgG titers of mice received the vaccine containing 15.5 nmol 4 (c) and 31 nmol 4 (d). Independent t-test was used to analysis the differences in levels of antibody titers. *P < 0.05, **P < 0.01, Student's t-test. (e) Tumor size (e) and body weight (f) of mice bearing melanoma. *P < 0.05, **P < 0.01, ***P < 0.001. Two way ANOVA. | |
The robust antibody titers are the symbol of outstanding immunogenicity and the essential factor of vaccine, for further immunological evaluation of compound 4, immunological studies in vivo were performed. Each mouse of a group (n = 6) received the emulsion containing 15.5 nmol 4 with Freund's complete adjuvant(CFA) on day 0 and 15.5 nmol 4 with incomplete Freund's adjuvant (IFA) on day 7, 14, 21. For comparison, the group (n = 6) that each mouse of which received the same dose of AD-Acp-FFRKSIINFEKL mixed with CFA or IFA was set. On day 14, 21, 28, blood samples were collected then sera were used to analyze the antibody titers. The antibody titers including IgG (Fig. 2c), kappa, IgM (Figs. S8a and b in Supporting information) were analyzed by enzyme-linked immunosorbent assay (ELISA). From the results of ELISA, an obviously higher level of IgG titers of 4 than those of AD-Acp-FFRKSIINFEKL was observed, indicating that a strong immune response could be elicited by FFRKSIINFEKL with the nanovaccine based on β-CD host-guest self-assembly system.
It is well known that immune effect is closely related to the dose administered of vaccine [27]. In order to explore the correlation between dose and immune effect of compound 4, a high dose group was performed. The emulsion containing 31 nmol 4 was injected into per mouse while the same dose of AD-Acp-FFRKSIINFEKL was performed at the same time. From the results of ELISA, the higher IgG antibody titers were found in mice received 31 nmol 4 (Fig. 2d) and higher antibody titers of kappa, IgM were observed at the same time (Figs. S8c and d in Supporting infor-mation), which suggested that the high dose vaccine containing 31 nmol 4 could induce a stronger immune response in vivo. Moreover, the high IgG1 antibody levels were observed among antibody levels of IgG1, IgG2b and IgG3 on day 28 (Fig. S9 in Supporting information).
The β-CD modified gold nanoparticle self-assembly system has proved to be an outstanding vaccine carrier. Next an anti-tumour peptide NY-ESO-1157-165 (SLLMWITQC) was chosen to investigate whether the carrier based on β-CD host-guest self-assembly system could be used in the field of cancer vaccine. The SLLMWITQC is an excellent antigen of the New York Esophageal Squamous Cell Carcinoma-1 (NY-ESO-1) and has excellent perfor-mance in clinical trials [28]. 1-Adamantanecarboxylic acid-Acp-SLLMWITQC (AD-Acp-SLLMWITQC, Figs. S14 and S15 in Supporting information) was used to prepare compound 5 by the host-guest interaction similar to compound 4. TEM analysis displayed that the size of 5 is around 200 nm similar to the size of 4 (Figs. S3 and S7 in Supporting information), which is the desired size for nanoparticle vaccines. The result of the tumour challenge suggested that the melanoma growth in mice receiving compound 5 was significantly inhibited compared to that received PBS or AD-Acp-SLLMWITQC, indicating nanovaccine is able to improve the therapeutic effect against melanoma (Fig. 2e). Moreover, there was no significant difference in body weight among the three groups of mice on day 18, which suggested that there is no obvious toxicity of vaccine based on β-CD modified gold nanoparticles (Fig. 2f) in vivo. The reason might be the host-guest interaction would combine β-CD with peptide antigens close to the glycosylation, which could improve the immunogenicity of peptide antigens as covalent glycosylated vaccines reported in previous studies [18, 29]. These results indicated that interactions between saccharides and antigens could enhance the immunogenicity of antigens when the forces increase to a certain value. Under in vivo microenviron-ment conditions, the enzyme would break the host-guest interaction and –SH-gold interaction so that the antigens would dissociate from the GNP after been injected in vivo to stimulate the body well.
In conclusion, a novel non-covalent glycosylated gold nano-particles/peptides nanovaccine based on β-CD host-guest self-assembly system was developed for the first time. The preparation of this nanovaccine is convenient and the particles are of appropriate sizes for vaccines. At present, most of vaccine carriers can be linked with a few antigens by covalent interaction, which hardly meet the need for the novel individualized vaccines. The nanovaccine reported here is based on non-covalent interac-tion between β-CD modified gold nanoparticles and AD linked with antigens; moreover, the immune studies in vivo show that the improved immune respond as well as therapeutic effect against melanoma were observed of the vaccine. Therefore, it is suitable for many types of antigens, especially for the T cell independent antigens, which provides a new choice for the design, application and promotion of individualized vaccines.
Declaration of competing interestThe authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
AcknowledgmentsIt is thankful that the Guangdong Key Laboratory for Translational Cancer Research of Chinese Medicine (No. 2018B030322011) and the National Natural Science Foundation of China (No. 81773580) offered financial support for this work.
Appendix A. Supplementary dataSupplementary material related to this article can be found, in the online version, at doi:https://doi.org/10.1016/j.cclet.2019.10.015.
| [1] |
F. Zhang, Z. Li, B. Jia, J. Ding, et al., Sci. Rep. 9 (2019) 3512. DOI:10.1038/s41598-019-40084-w |
| [2] |
I.S. Ganda, Q. Zhong, M. Hali, et al., Int. J. Pharm. 527 (2017) 79-91. DOI:10.1016/j.ijpharm.2017.05.045 |
| [3] |
U. Sahin, E. Derhovanessian, M. Miller, et al., Nature 547 (2017) 222-226. DOI:10.1038/nature23003 |
| [4] |
P.A. Ott, Z. Hu, D.B. Keskin, et al., Nature 547 (2017) 217-221. DOI:10.1038/nature22991 |
| [5] |
A. Tsuboi, N. Hashimoto, F. Fujiki, et al., Cancer Immunol. Immunother. 68 (2019) 331-340. DOI:10.1007/s00262-018-2274-1 |
| [6] |
N.K. Botelho, B.O. Tschumi, J.A. Hubbell, et al., Front. Immunol. 10 (2019) 294. DOI:10.3389/fimmu.2019.00294 |
| [7] |
Y. Narita, Y. Arakawa, F. Yamasaki, et al., Neuro. Oncol. 21 (2018) 348-359. |
| [8] |
M. Luo, H. Wang, Z. Wang, et al., Nat. Nanotechnol. 12 (2017) 648-654. DOI:10.1038/nnano.2017.52 |
| [9] |
Y. Mi, C.C. Smith, F. Yang, et al., Adv. Mater. 30 (2018) e1706098. DOI:10.1002/adma.201706098 |
| [10] |
Z. Wang, W. Liu, J. Shi, N. Chen, C. Fan, Mater. Horiz. 5 (2018) 344-362. DOI:10.1039/C7MH00991G |
| [11] |
R. Kuai, L.J. Ochyl, K.S. Bahjat, A. Schwendeman, J.J. Moon, Nat. Mater. 16 (2017) 489-496. DOI:10.1038/nmat4822 |
| [12] |
A.W. Li, M.C. Sobral, S. Badrinath, et al., Nat. Mater. 17 (2018) 528-534. DOI:10.1038/s41563-018-0028-2 |
| [13] |
Y.P. Jia, B.Y. Ma, X.W. Wei, Z.Y. Qian, Chin. Chem. Lett. 28 (2017) 691-702. DOI:10.1016/j.cclet.2017.01.021 |
| [14] |
J. Chen, Y. Huang, X. Yang, et al., Anal. Chim. Acta 1023 (2018) 89-95. DOI:10.1016/j.aca.2018.02.082 |
| [15] |
Y. Liu, Y. Wang, F. Yu, et al., Chem. Commun. 53 (2017) 9486-9489. DOI:10.1039/C7CC04386D |
| [16] |
Y. Liu, W. Zhang, Q. He, et al., Chem. Commun. 52 (2016) 10886-10889. DOI:10.1039/C6CC04623A |
| [17] |
H. Wang, B. Yang, Y. Wang, F. Liu, S. Dong, Chem. Commun. 55 (2019) 253-256. DOI:10.1039/C8CC07691J |
| [18] |
M. Vetro, D. Safari, S. Fallarini, et al., Nanomedicine 12 (2017) 13-23. DOI:10.2217/nnm-2016-0306 |
| [19] |
S. Kang, S. Ahn, J. Lee, et al., J. Control. Release 256 (2017) 56-67. DOI:10.1016/j.jconrel.2017.04.024 |
| [20] |
W. Lin, C. Yang, Z. Xue, et al., J. Colloid. Interf. Sci. 528 (2018) 135-144. DOI:10.1016/j.jcis.2018.05.082 |
| [21] |
N. Benne, J. van Duijn, J. Kuiper, W. Jiskoot, B. Slütter, J. Control. Release 234 (2016) 124-134. DOI:10.1016/j.jconrel.2016.05.033 |
| [22] |
H. Shelley, R.J. Babu, J. Pharm. Sci. 107 (2018) 1741-1753. DOI:10.1016/j.xphs.2018.03.021 |
| [23] |
Y. Bai, C.P. Liu, X. Song, L. Zhuo, W. Tian, Chem.-Asian J. 13 (2018) 3903-3911. DOI:10.1002/asia.201801366 |
| [24] |
M. Yu, Z. Liu, Y. Du, et al., ACS. Appl. Mater. Inter. 13 (2019) 15103-15110. |
| [25] |
Z. Du, K. Ke, X. Chang, R. Dong, B. Ren, Langmuir 34 (2018) 5606-5614. DOI:10.1021/acs.langmuir.8b00470 |
| [26] |
R.J. Anderson, C.W. Tang, N.J. Daniels, et al., Nat. Chem. Biol. 10 (2014) 943-949. DOI:10.1038/nchembio.1640 |
| [27] |
G. Liao, Z. Zhou, S. Suryawanshi, M.A. Mondal, Z. Guo, ACS. Central. Sci. 2 (2016) 210-218. DOI:10.1021/acscentsci.5b00364 |
| [28] |
O. Gasser, K.J. Sharples, C. Barrow, et al., ACS. Central. Sci. 67 (2018) 285-298. |
| [29] |
S.Khamehchian, M.Nikkhah, R. Madani, S.Hosseinkhani, J. Biomed. Mater. Res. A 104 (2016) 2693 -2700.
|
 2020, Vol. 31
2020, Vol. 31 

