Although many traditional technologies that have been successfully applied for organic wastewater treatment, mainly include physical [1, 2], biological [3, 4], and chemical methods [5, 6], there still has a much big promotion space for the process efficiency and economy. Since 70's of last century, semiconductor photocatalytic technology is booming and considered to be the most prospective method to resolve enhancing environmental and power source problems [7, 8]. It can transform sunlight to chemical energy to degrade organic pollutants with environment-friendly and no secondary pollution [9, 10]. However, the photogenerated carriers recombination rate of single-component photocatalyst is too fast, which would decrease the light energy utilization and photocatalytic efficiency [11, 12]. Therefore, how to enhance the separation of photogenerated carriers is the key to improve the photocatalyst performance.
There are several main strategies for improving the photocatalytic performance of semiconductors: Noble metal deposition [13], ion doping [14], and nanocomposites fabrication [15, 16]. In contrast to the former two ways, the preparation of composite semiconductor is to change the transfer path of photogenerated carriers, which would not influence the intrinsic properties of material, and the synthetic method is easy with minimal cost [17, 18]. At present, the transfer mode of photoinduced carriers in composite semiconductors are mainly divided into heterojunctions and Z-scheme [19]. Z-scheme simulates the natural photosynthesis mechanism, usually consisting of a reducing end (PS Ⅰ), an oxidation end (PS Ⅱ), and an electron transporter (PS Ⅰ-R-PS Ⅱ) [20, 21]. Compared with the heterojunction, Z-scheme is more conducive to improve the redox capability of the two semiconductors while suppressing the carrier recombination, but it is usually limited by the band position of semiconductor [22]. Therefore, selecting the band matched semiconductors for hybrid has become an important issue for the Z-scheme method.
BiVO4 is a typical ternary metal oxide, which has a narrower band gap, higher response to visible light, and photochemical stability than conventional TiO2 [23]. The BiVO4 possesses a higher valence band (VB) position, which is appropriate for PS Ⅱ material [24]. On the other hand, ternary indium-based sulfide AInxSy (A = Cu, Ag, Zn, Cd, and Sn) has been extensively studied in the fields of photocatalysis [25]. Among them, the narrow bandgap of ZnIn2S4 stands out among many ternary sulfides because of its good response to visible light, green material composition, and relatively strong photo-corrosion resistance [26], which are suitable as the PS Ⅰ material. Additionally, the LED light source has low cost and low energy consumption, and its operation is simple and safe, which has attracted much attention recently [27].
Based on above, the coupling of BiVO4 with ZnIn2S4 would significantly improve charge separation efficiency under visible light irradiation, but the relative report is seldom. Therefore, in this work, a Z-scheme composite was constructed using ZnIn2S4 as PS Ⅰ and BiVO4 as PS Ⅱ to promote the photocatalytic degradation performance under the LED illumination. The Z-scheme composite was fabricated and characterized, and its photodegradation capability was investigated through the methyl orange (MO) decomposition under visible light irradiation, and then the function mechanism was further studied and proposed.
The reagents used in this experiment were introduced in Text S1 (Supporting information). The BiVO4 nanosheets, ZnIn2S4, and BiVO4/ZnIn2S4 (BZ) composite were all prepared by the respective specific hydrothermal methods. Those detailed preparation procedures were described in Text S2 to Text S4 (Supporting information), respectively. Based on the mass ratio of BiVO4 to ZnIn2S4, the prepared composites were recorded as 1% BZ, 3% BZ, 5% BZ, 10% BZ, and 20% BZ, respectively.
The prepared catalysts were characterized by X-ray diffraction (XRD, D-max-2500), scanning electron microscopy (SEM, SUPRA55), energy-dispersive X-ray spectroscopy (EDS), X-ray photoelectron spectroscopy (XPS, Thermo Scientific ESCA Lab 250), UV– vis diffuse reflectance spectroscopy spectrophotometer (DRS, UV-3100), nitrogen adsorption (Nova 40000e), Fourier transform infrared spectra spectrometer (FTIR, Necolet IS 10), Raman spectrometer (HORIBA), zeta potential tester (Zetasizer NanoZS), and photoluminescence (PL) spectra fluorescence spectrophotometer (Hitachi F-7000). The photoelectric test of this study was tested in the electrochemical workstation CHI660E. The photocatalytic elimination of MO was conducted in a photocatalytic reaction system. All the details of the characterizations and measurements were revealed in Text S5 (Supporting information).
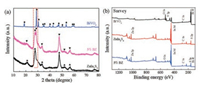
The crystal structure properties for the prepared catalysts were evaluated by XRD. As shown in Fig. 1a, all the diffraction peaks of BiVO4 and ZnIn2S4 were consistent well with the standard patterns for the pure monoclinic phase of BiVO4 (JCPDS No. 14-0688) and hexagonal phase of ZnIn2S4 (JCPDS No. 65-2023), respectively [28-30]. The characteristic peak at 28.7º for the 5% BZ proved that the composite was fabricated successfully. Besides, Fig. S1 (Supporting information) displays that most of the diffraction peaks in the composites with different BiVO4 mass ratios were most similar to the diffraction peaks of ZnIn2S4, which could be due to the decreasing content of BiVO4 in the composite.
|
Download:
|
| Fig. 1. (a) XRD patterns and (b) XPS spectrum of BiVO4, ZnIn2S4, and 5% BZ catalysts. | |
To prove the existence and state of each element in ZnIn2S4, BiVO4, and 5% BZ, the XPS characterization was performed on the three materials (Fig. 1b). It can be seen from the full spectrum that the elements on the surface of these materials had almost the same results as the following EDS analysis (Fig. 2). The C 1s peak was caused by the adsorption of carbonaceous compounds in the atmosphere on the sample surface [31-33]. The other elements in ZnIn2S4, BiVO4, and 5% BZ were analyzed and discussed in Text S6 and Fig. S2 (Supporting information). Moreover, FTIR and Raman were applied to further characterize and prove the chemical structures of the three materials in Text S7 and Fig. S3 (Supporting information). In addition, the surface pore structure of those materials were determined through nitrogen adsorption-desorption isotherms (Table S1 and Fig. S4 in Supporting information), and the results showed that the combination of BiVO4 and ZnIn2S4 decreased the surface area of hybrid material, which would affect the adsorption capability of 5% BZ.

|
Download:
|
| Fig. 2. SEM images and EDS of (a) ZnIn2S4, (b) BiVO4, and (c, d) 5% BZ. | |
The surface morphologies of the three materials were investigated by SEM. As shown in Fig. 2a, the pure ZnIn2S4 presented a uniform flower-like microspheres. Fig. 2b shows that the typical sheet-like morphologies were observed on the prepared BiVO4. Fig. 2c displays that the sample of 5% BZ was in the same microsphere shape as ZnIn2S4. On closer inspection of Fig. 2d, the surface of hybrid material had a more prominent petallike structure than ZnIn2S4, which was formed by the intercalation of sheet-like BiVO4 on the microspheres of ZnIn2S4. The produced intimate combination between ZnIn2S4 and BiVO4 could be conducive to the electrons transfer from ZnIn2S4 to BiVO4, so the recombination of photogenerated electron-holes would be suppressed in ZnIn2S4 [33-35]. The energy dispersive X-ray spectroscopy (EDS) spectrum for the three as-prepared samples indicated that the typical signals of all relevant elements were observed.
The PL spectra of pure BiVO4, ZnIn2S4, and BZ composites with different BiVO4 content were measured (Fig. 3a). The pure BiVO4 and ZnIn2S4 presented the stronger PL spectra intensity than the BZ hybrid [36-38]. With the increasing mass percentage of BiVO4, the PL intensity of BZ composites obviously declined, indicating that the photogenerated electron-holes separation degree was augmented after the combination. Besides, as seen in Fig. 3b for the photocurrent measurement, 5% BZ displayed an obvious improved photocurrent, which produced a photocurrent of 1.4 μA/cm2, while the photocurrents were only 1.0 μA/cm2 and 0.7 μA/cm2 for BiVO4 and ZnIn2S4, respectively. Fig. 3c shows that the order of electrochemical impedance curvature of three curves from large to small was BiVO4 > ZnIn2S4 > 5% BZ, demonstrating that the photogenerated electron-holes separation of 5% BZ was the highest [39, 40]. Above results can be concluded that the combination of BiVO4 and ZnIn2S4 represented a synergistic effect, inhibiting the photo-generated electron-holes recombination and promoting the efficient separation of photogenerated carriers, and thus can improve the photocatalytic performance. Furthermore, the UV–vis DRS spectra testified that 5% BZ had a wider response range to visible light than the pure BiVO4 and ZnIn2S4 (Fig. S5 in Supporting information).

|
Download:
|
| Fig. 3. PL spectra (a), transient photocurrent density (b), and electrochemical impedance spectroscopy (c) of BiVO4 ZnIn2S4, and different BZ. (d) Time courses of MO decolorization for BiVO4, ZnIn2S4, and BiVO4/ZnIn2S4 with different BiVO4 content under visible light irradiation. (e) Consecutive adsorption and photocatalytic tests of 5% BZ for MO decolorization. (f) Free radicals scavenging test for 5% BZ. | |
The photocatalytic degradations of BiVO4, ZnIn2S4, and BZ composites with different BiVO4 mass ratio were investigated for the decolorization of MO (Fig. 3d). The experimental conditions were as follows: MO concentration 15 mg/L, initial pH 6.3, and catalyst dosage 0.20 g/L. Before the photocatalytic test begins, a 30 min dark reaction process was conducted to reach absorption equilibrium. The adsorption performances of the composites were slightly lower than that of pure ZnIn2S4. This is because that the specific surface area and pore volume of the hybrids both decreased after combination. After 240 min photocatalytic reaction, pure BiVO4 presented the lowest photocatalytic efficiency of 17% (corresponding rate constant 0.000963 min-1), meanwhile ZnIn2S4 achieved a decolorization of 62% (0.00515 min-1). For the series of BZ composites, the decolorization rate displayed an increase and decrease trend with the enhancing mass percentage of BiVO4 from 1% to 20%. Therein, 5% BZ reached the highest decolorization rate of 86% (0.00997 min-1), which was 1.9 and 10.3 times higher than pure ZnIn2S4 and BiVO4, respectively. But when the BiVO4 loading amount exceeded 5%, the decolorization efficiency gradually decreased, only 52% (0.00402 min-1) and 40% (0.00213 min-1) for 10% BZ and 20% BZ, respectively, which were even lower than that of ZnIn2S4. The optimal photocatalytic performance of 5% BZ could be ascribed to its better separation for photogenerated electron-holes. Besides, the photostability of 5% BZ was investigated by the consecutive adsorption and photocatalytic decomposition of MO (Fig. 3e). After four times repeated applications, the composite presented the analogical removal trend and rate, which could be ascribed to the well separation of photogenerated carriers for 5% BZ, proving it possessed a certain photostability.
The further degradation and mineralization of MO by 5% BZ in the photocatalytic process were assessed through the UV–vis spectra, chemical oxygen demand (COD) and total organic carbon (TOC) analyses (Figs. S6 and S7 in Supporting information). The COD and TOC removing efficiencies of 5% BZ were both higher than those of ZnIn2S4, indicating the better degradation and mineralization effects on the MO for 5% BZ.
A certain amount of methanol (MA), tert-butanol (TBA), and p-benzoquinone (BQ) were introduced as inhibitors of h+, ·OH, and ·O2- to investigate the photo-degradation mechanism, respectively (Fig. 3f) [41-43]. After 4 h irradiation, the decolorization ratios for the adding 30 mmol/L of MA and TBA were 86% and 78%, respectively. However, only 1 mmol/L BQ declined the MO removal to 29%. During the photocatalytic process, the photogenerated e- could react with O2 to form ·O2- and ·OH would be produced through h+ oxidative process on H2O or OH-. Besides, not only ·O2- plays a key role in the organics photodegradation, but also participate in the ·OH generation in the reactive species chain reactions [44-47].
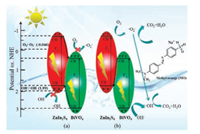
Based on above results and discussions, the assumption for the BZ composite was depicted in Fig. 4. According to heterojunctions function as in the Fig. 4a, there would be hard to generate ·O2- and ·OH [48, 49]. Nevertheless, previous results testified the existence of ·O2- and ·OH, so the heterojunctions assumption is untenable. As seen in the Z-scheme path (Fig. 4b), e- on conduction band (CB) of BiVO4 would migrate to the VB of Znln2S4 under illumination. Especially, e- produced by BiVO4 would be recombined with h+ form by ZnIn2S4, which could inhibit the recombination of e- and h+ for Znln2S4 and BiVO4, respectively. Hence, for Z-scheme composite, e- and h+ would be aggregated on the CB of ZnIn2S4 and the VB of BiVO4, respectively [50]. The CB position of ZnIn2S4 is -0.68 eV, which is much lower than the O2/·O2- redox (-0.33 eV), so it is more favorable for the reaction with O2 adsorbed on the composite surface to produce ·O2-. Meanwhile, the VB position of BiVO4 is at +2.98 eV, which is much higher than that of OH-/·OH (+2.4 eV), which is beneficial to ·OH production. In addition, h+ on BiVO4 can also directly participate in the reaction. Above Zscheme path was proved by previous characterizations and photocatalytic tests.
|
Download:
|
| Fig. 4. Photocatalysis mechanism for BiVO4/ZnIn2S4 composite. (a) Heterojunctions path; (b) Z-scheme path. | |
To sum up, the BiVO4/ZnIn2S4 all-solid-state Z-scheme composite with the excellent photocatalytic performance under the LED visible light illumination was constructed. The highest MO decolorization rate of 86% (corresponding rate constant 0.00997 min-1), was achieved over 5% BZ nanocomposite, which was higher than pure BiVO4 and ZnIn2S4. The Z-scheme composite demonstrated good photocatalytic degradation of MO because its enhanced separation for photogenerated electron-holes, thus promoting the photocatalytic performance and reactive species generation. This work proved that constructing an all-solid-stateZ-schemenanocomposite is a hopeful method to improve the photocatalytic degradation for organic wastewater of photocatalyst.
Declaration of competing interestsThe authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
AcknowledgementsWe are thankful to the financial supports from the National Natural Science Foundation of China (Nos. 51908485 and 51608468), the China Postdoctoral Science Foundation (No. 2019T120194), the University Science and Technology Program Project of Hebei Provincial Department of Education (No. QN2018258).
Appendix A. Supplementary dataSupplementary material related to this article can befound, in the online version, at doi:https://doi.org/10.1016/j.cclet.2019.09.051.
| [1] |
Q. Zhang, S. Bolisetty, Y. Cao, et al., Angew. Chem. Int. Ed. 58 (2019) 6012-6016. DOI:10.1002/anie.201901596 |
| [2] |
Q. Zhang, Y. Han, L. Wu, Chem. Eng. J. 363 (2019) 278-284. DOI:10.1016/j.cej.2019.01.146 |
| [3] |
C. Grandclément, I. Seyssiecq, A. Piram, et al., Water Res. 111 (2017) 297-317. DOI:10.1016/j.watres.2017.01.005 |
| [4] |
J. Li, B. Li, H. Huang, et al., Sci. Total Environ. 687 (2019) 460-469. DOI:10.1016/j.scitotenv.2019.05.400 |
| [5] |
K. Ma, W. Chen, T. Jiao, et al., Chem. Sci. 10 (2019) 6821-6827. DOI:10.1039/C9SC01577A |
| [6] |
S. Yang, K. Yin, J. Wu, et al., Nanoscale 11 (2019) 17607-17614. DOI:10.1039/C9NR04381K |
| [7] |
A. Fujishima, K. Honda, Nature 238 (1972) 37-38. DOI:10.1038/238037a0 |
| [8] |
Y. Hou, M. Qiu, M.G. Kim, et al., Nat. Commun. 10 (2019) 1392. DOI:10.1038/s41467-019-09394-5 |
| [9] |
M. Gmurek, M. Olak-Kucharczyk, S. Ledakowicz, Chem. Eng. J. 310 (2017) 437-456. DOI:10.1016/j.cej.2016.05.014 |
| [10] |
G. Xia, C. Li, K. Wang, L. Li, Sci. Adv. Mater. 11 (2019) 1079-1086. DOI:10.1166/sam.2019.3487 |
| [11] |
J. Liu, J. Zhang, D. Wang, et al., ACS Sustain. Chem. Eng. 7 (2019) 12428-12438. |
| [12] |
J. Ke, Adnan Younis M., Y. Kong, et al., Nano-Micro Lett. 10 (2018). |
| [13] |
Y. Chen, Y. Wang, W. Li, et al., Appl. Catal. B.-Environ. 210 (2017) 352-367. DOI:10.1016/j.apcatb.2017.03.077 |
| [14] |
B. Huang, J. He, S. Bian, et al., Chin. Chem. Lett. 29 (2018) 1698-1701. DOI:10.1016/j.cclet.2018.01.004 |
| [15] |
J. Ke, J. Liu, H. Sun, et al., Appl. Catal. B.-Environ. 200 (2017) 47-55. DOI:10.1016/j.apcatb.2016.06.071 |
| [16] |
M. Ji, Z. Zhang, S. Zhang, et al., Chin. Chem. Lett. 29 (2018) 805-810. DOI:10.1016/j.cclet.2018.05.002 |
| [17] |
Y. Hong, Y. Jiang, C. Li, et al., Appl. Catal. B.-Environ. 180 (2016) 663-673. |
| [18] |
Y. Zhang, D. Zhang, X. Xu, B. Zhang, Chin. Chem. Lett. 29 (2018) 1350-1354. DOI:10.1016/j.cclet.2018.03.009 |
| [19] |
J. Chen, J. Zhan, Y. Zhang, Y. Tang, Chin. Chem. Lett. 30 (2019) 735-738. DOI:10.1016/j.cclet.2018.08.020 |
| [20] |
P. Zhou, J. Yu, M. Jaroniec, Adv. Mater. 26 (2014) 4920-4935. DOI:10.1002/adma.201400288 |
| [21] |
H. Zhou, Z. Wen, J. Liu, et al., Appl. Catal. B.-Environ. 242 (2019) 76-84. DOI:10.1016/j.apcatb.2018.09.090 |
| [22] |
L. Ye, J. Liu, C. Gong, et al., ACS Catal. 2 (2012) 1677-1683. DOI:10.1021/cs300213m |
| [23] |
F. Chen, Q. Yang, Y. Wang, et al., Chem. Eng. J. 348 (2018) 157-170. DOI:10.1016/j.cej.2018.04.170 |
| [24] |
F. Ye, H. Li, H. Yu, S. Chen, X. Quan, Appl. Catal. B.-Environ. 227 (2018) 258-265. DOI:10.1016/j.apcatb.2017.12.020 |
| [25] |
Z. Guan, Z. Xu, Q. Li, et al., Appl. Catal. B.-Environ. 227 (2018) 512-518. DOI:10.1016/j.apcatb.2018.01.068 |
| [26] |
S. Wan, M. Ou, Q. Zhong, S. Zhang, F. Song, Chem. Eng. J. 325 (2017) 690-699. DOI:10.1016/j.cej.2017.05.047 |
| [27] |
Y. Gao, S. Li, Y. Li, L. Yao, H. Zhang, Appl. Catal. B.-Environ. 202 (2017) 165-174. DOI:10.1016/j.apcatb.2016.09.005 |
| [28] |
M. Ou, S. Wan, Q. Zhong, et al., Appl. Catal. B.-Environ. 221 (2018) 97-107. DOI:10.1016/j.apcatb.2017.09.005 |
| [29] |
C. Duan, F. Li, M. Yang, et al., Ind. Eng. Chem. Res. 57 (2018) 15385-15394. |
| [30] |
C. Duan, Y. Cao, L. Hu, et al., J. Hazard. Mater. 373 (2019) 141-151. DOI:10.1016/j.jhazmat.2019.03.064 |
| [31] |
M. Zhu, Z. Sun, M. Fujitsuka, T. Majima, Angew. Chem. Int. Ed. 130 (2018) 2182-2186. DOI:10.1002/ange.201711357 |
| [32] |
H. Guo, N. Jiang, H. Wang, et al., Appl. Catal. B.-Environ. 248 (2019) 552-566. DOI:10.1016/j.apcatb.2019.01.052 |
| [33] |
K. Wang, L. Li, Y. Lan, P. Dong, G. Xia, Math. Probl. Eng. 2019 (2019) 1-8. |
| [34] |
N. Jiang, C. Qiu, L. Guo, et al., J. Hazard. Mater. 369 (2019) 611-620. DOI:10.1016/j.jhazmat.2019.02.087 |
| [35] |
F. Chen, H. Huang, Y. Zhang, T. Zhang, Chin. Chem. Lett. 28 (2017) 2244-2250. DOI:10.1016/j.cclet.2017.09.017 |
| [36] |
X. Zhang, J. Xiao, M. Hou, Y. Xiang, H. Chen, Appl. Catal. B.-Environ. 224 (2018) 871-876. DOI:10.1016/j.apcatb.2017.11.038 |
| [37] |
P. Huo, J. Li, Z. Ye, et al., Chin. Chem. Lett. 28 (2017) 2259-2262. DOI:10.1016/j.cclet.2017.09.067 |
| [38] |
Y. Zhou, Y. Huang, J. Pang, K. Wang, J. Power Sources 440 (2019) 227149. DOI:10.1016/j.jpowsour.2019.227149 |
| [39] |
Z. Luo, L. Qu, J. Jia, et al., Chin. Chem. Lett. 29 (2018) 547-550. DOI:10.1016/j.cclet.2017.09.025 |
| [40] |
H. Li, S. Guo, K. Shin, M.S. Wong, G. Henkelman, ACS Catal. 9 (2019) 7957-7966. DOI:10.1021/acscatal.9b02182 |
| [41] |
N. Jiang, Y. Zhao, C. Qiu, et al., Appl. Catal. B.-Environ. 259 (2019) 118061. DOI:10.1016/j.apcatb.2019.118061 |
| [42] |
T. Zhang, X. Li, Q. Zhao, Y. Rao, Sustain. Cities Soc. 51 (2019) 101714. DOI:10.1016/j.scs.2019.101714 |
| [43] |
X. Nie, S. Feng, Z. Shudu, G. Quan, Adv. Civ. Eng. Mater. 2019 (2019) 1-12. |
| [44] |
S. Tang, N. Li, D. Yuan, et al., Chemosphere 234 (2019) 658-667. DOI:10.1016/j.chemosphere.2019.06.112 |
| [45] |
D. Yuan, C. Zhang, S. Tang, et al., Water Res. (2019) 114861. |
| [46] |
Y. He, X. Zhuang, C. Lei, et al., Nano Today 24 (2019) 103-119. DOI:10.1016/j.nantod.2018.12.004 |
| [47] |
H. Huang, S. Tu, C. Zeng, et al., Angew. Chem. Int. Ed. 56 (2017) 11860-11864. DOI:10.1002/anie.201706549 |
| [48] |
K. Wu, H. Yang, L. Jia, et al., Green Chem. 21 (2019) 1472-1483. DOI:10.1039/C8GC03702G |
| [49] |
D. Liu, Y. Cao, J. Liu, Y. Gao, Y. Wang, J. Eur. Ceram. Soc. 38 (2018) 817-820. DOI:10.1016/j.jeurceramsoc.2017.09.009 |
| [50] |
Y. He, R. Wang, T. Jiao, et al., ACS Sustain. Chem. Eng. 7 (2019) 10888-10899. DOI:10.1021/acssuschemeng.9b01780 |
 2020, Vol. 31
2020, Vol. 31 

