b Key Laboratory of Advanced Carbon Materials and Wearable Energy Technologies of Jiangsu Province, Soochow University, Suzhou 215006, China
Metal-organic frameworks (MOFs), a class of porous crystal materials with high surface and ordered channel structure [1], have been widely studied in many fields, such as gas separation and storage [2], catalysis [3, 4], sensors [5, 6], drug delivery [7], wastewater treatment [8]. Typically the structure of most MOFs are three-dimensional, and the precise control the low-dimensional microstructure of MOFs usually requires additional additives as structure guide or template agent [9]. So in recent years, the research about morphological control of low-dimensional MOFs is common occurrence.
Among these colorful MOFs materials, 1D structures such as rods, wires, tubes and belts have always been a research hotspot due to their excellent utilization potentiality in electronic or optoelectronic devices [10-12]. So far, the 1D MOFs materials is mainly focused on two types: one is MOFs-based composite structure, in which the MOFs particles are combined with an onedimensional materials (such as metal and semiconductor) to form the package or core-shell structure [13]. For example, Liu et al. first reported a Si NWs/MOF hybrid structure of MOF-199 wrapped on the -COOH modified silicon nanowires through step-by-step growth, they found that the interaction of the MOFs coordinating groups with the Si NWs surface functional group is crucial for the growth of the hybrid materials [14]. Zhan et al. used the ZnO array as a pecursor material to in-situ growth ZIF-8. After adjusted the corresponding parameters of reaction temperature, ligand concentration and reaction time, they successfully prepared a core-shell structure of ZnO@ZIF-8 nanorods arrays, which displayed distinct photoelectrochemical response to H2O2 and ascorbic acid [15]. Furthermore, Zhang et al. prepared a 1D package structure of CuNWs@ZIF-8 by microwave heating method, and the composite material exhibited an excellent performance in hydrogen conversion with the hydrolysis of ammonia borane [16]. From the above, it can be seen that reasonable packaging of onedimensional materials by MOFs can effectively improve their properties. The other method for the preparation of 1D MOFs materials is using end-capping reagent or template. For example, Luz et al. reported a one-step synthetic strategy for Cu@Cu-MOF-74 rods, they discussed in detail the effects of various factors in the reaction on the structure, including reaction time, the ligand-to-Cu molar ratio, solvent and reaction temperature [17]. While Zou et al. reported the fabrication of single-crystal MOF nanotubes via an amorphous MOF-mediated recrystallization approach. The obtained MOF nanotubes can be used to separate large molecules such as rhodamine B [18]. Zhang et al. used ultrathin tellurium nanowires as templates to direct growing ZIF-8 nanofibers, the derived porous doped carbon nanofibers exhibited excellent electrocatalytic performance for oxygen reduction reaction [19]. Furthermore, Pachfule et al. synthesized MOF-74 rods in the presence of salicylic acid as a modulator. After heat treatment of the MOFs rods, the obtained carbon nanorods exhibited excellent performance in supercapacitor electrodes [20]. Although both of these two strategies can construct a good one-dimensional structure of MOFs, it is still a significant challenge to conduct dynamic control of one-dimensional MOFs without any assistance.
Herein, we demonstrate a strategy for the synthesis of 1D MOFs (HKUST-1 nanobelts) from Cu NWs (ligand-free) by a traditional hydrothermal method. The Cu NWs act as a copper source and template during the transformation process of HKUST-1 nanobelts, and the molar ratio of Cu NWs/H3BTC and the reaction time play important roles in the morphology of the final products. Finally, a possible reaction mechanism is proposed based on the kinetic process. This work provides a strategy to future design and synthesize 1D structure of MOFs and metals@MOFs hybrids.
The XRD of HKUST-1 nanobelts was shown in Fig. 1a. Comparing with the particles, the HKUST-1 nanobelts exhibit a same XRD patterns, confirming both of the HKUST-1 nanobelts and particles have the same structure. Although the (222) plane is still a dominant crystal surface (Fig. S1 in Supporting information), the peak at around 2θ value of 9°, which can be assigned to (220) planes, shows a stronger peak intensity in HKUST-1 nanobelts, indicating the exposure of the crystal surface is enhanced [21, 22]. From the FESEM images of Cu NWs, HKUST-1 nanobelts (Figs. 1b and c), it can be seen that the Cu NWs exhibited extremely smooth surface but with some Cu nanoparticles, which is inevitable in our preparation [23]. But after the Cu NWs react with H3BTC under 80 ℃ for 20 h, there is no Cu NWs but uniform HKUST-1 nanobelts can be obtained from the products, and the surface of nanobelts remain smooth with a width of about 300 nm.

|
Download:
|
| Fig. 1. (a) XRD patterns of HKUST-1 particles and HKUST-1 nanobelts; SEM images of (b) CuNWs and (c) HKUST-1 nanobelts; AFM image of (d) CuNWs and (e) HKUST-1 nanobelts. | |
To further study the microscopic property of the HKUST-1 nanobelts, the atomic force microscope (AFM) was used to characterize the surface property of the nanobelts. From the AFM images, it can be seen that the width of Cu NWs is 100 nm with a thickness of 50 nm (Fig. 1d), and there is no obvious defect on the surface. However, although the nanobelts are inherited the smooth surface property of the Cu NWs, the width is up to 300 nm and the thickness reached 170 nm (Fig. 1e), which are both larger than the Cu NWs. In addition, the average length of the belts could reach up to about 60 μm, which is twice longer than the Cu NW with an average length of 30 μm, indicating the HKUST-1 nanobelts may be grown by a process of Cu NWs surface dissolving and MOFs self-assembling growth. Due to the high aspect ratio (1:100) with the nano-sized diameter, the HKUST-1 nanobelts exhibited a lower thermal stability than the HKUST-1 polyhedrons from the thermogravimetric analysis (Fig. S2 in Supporting information), and it can be calculated out that the molar ratio of Cu/H3BTC of the nanobelts is about 3:2 from the mass loss of TGA, which is consistent with the theoretical value of HKUST-1. Furthermore, it should be worth noting that the mass loss of the nanobelts before 250 ℃ is much less than HKUST-1 nanoparticles, indicating that the guest molecules adsorbed in nanobelts is lower than particles, which is in agreement with the lower BET of nanobelts (161 m2/g) than the particles (1217 m2/g) (Fig. S3 in Supporting information).
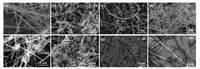
To better understanding the structural transformation mechanism of HKUST-1 nanobelts from Cu NWs, we first investigated the influence of the concentration of H3BTC. The SEM images of different mole ratios of Cu/H3BTC are shown in Fig. 2 and Fig. S4 (Supporting information) corresponding to different size of scales. It can been seen that the surface of Cu NWs are rugged after hydrothermal reaction without the organic ligands, and the characteristic peak of Cu2O can be found at 2θ = 36.6°, 42.5° and 61.6° from the XRD characterization (Fig. S5 in Supporting information), indicating the surface of the Cu NWs has been oxidized to Cu2O. When the H3BTC is added, a lot of particles of HKUST-1 are present in the products, and at the same time, a layer of HKUST-1 particles appeared on the surface of the nanowires (Fig. 2b). With the increase of ligand addition, the more particles were produced. However, when the molar ratio of Cu/H3BTC reached 1:40, only nanobelts can be found in the final products (Fig. 2g), and its structure was verified by XRD (Fig. S6). More organic ligands do not affect the formation of nanobelts (Fig. 2h).
|
Download:
|
| Fig. 2. SEM images of the products at different ligand concentration with small scale, the mass ratio of Cu NWs and H3BTC are (a) 1:0, (b) 1:0.4, (c) 1:1, (d) 1:4, (e) 1:10, (f) 1:20, (g) 1:40, (h) 1:80. | |
Next, we investigated the influence of the reaction time with a fixed ratio of Cu to H3BTC at 1:40. The SEM images of different reaction time are shown in Fig. 3. It can been seen that when the hydrothermal reaction is 2 h, the thin layer of HKUST-1 appeared on the copper nanowire, which can be confirmed by XRD (Fig. 3a and Fig. S7 in Supporting information). With the prolonging of reaction time, the HKUST-1 layer on the surface of copper nanowires becomes thicker, but the crystallinity is getting better and better. Finally, all of the Cu NWs are transformed into nanobelts at 20 h. However, when use Cu2+ as the Cu source, there is only HKUST-1 particles in the final products (Fig. S8 in Supporting information).

|
Download:
|
| Fig. 3. SEM images of the products at different time of (a) 2 h, (b) 4 h, (c) 8 h, (d) 12 h, (e) 16 h and (f) 20 h. | |
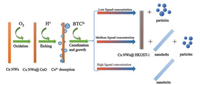
On the basis of the above observations, a formation mechanism can be proposed (Scheme 1). In the whole process, the Cu NWs play roles of both Cu source and structure-directing agent. First, the Cu NWs are oxidized by the dissolved O2 to form Cu2O (4Cu + O2 → 2Cu2O). However, the Cu2O is not stable in the acid solution with disproportionation (Cu2O + 2H+ → Cu2+ + Cu +H2O). Once the Cu2+ formed and dissociated into the solution, the H3BTC will immediately coordinate with it to form coordination polymer of HKUST-1. So the balance between the release rate of Cu2+ and heterogeneous nucleate rate of HKUST-1 is the key for the formation of HKUST-1 nanobelts. When the concentration of the ligand is low, the excess Cu2+ will diffuse into the solution and coordinate with the BTC anion to form HKUST-1 with anisotropic growth. However, when the concentration of ligand is high enough, the ligand would coordinate with Cu2+ as soon as the Cu2+ diffused out from the Cu NWs, and then nucleated and grown along the MOFs that is wrapped on the Cu NWs. In this case, Cu NWs play the roles of Cu source and structure-directing agent. Due to the porous structure of the MOFs, the O2 and H+ could still etching Cu NWs though the channels of the MOFs, and the Cu2+ could diffused out though the pores. The pore size of different crystal planes will affect the mass transfer of Cu2+ and organic ligand, as well as the O2, which will affect the growth rate of the crystal direction, resulting in the formation of the nanobelts with high aspect ratio.
|
Download:
|
| Scheme 1. reaction mechanism of CuNWs @ HKUST-1 and HKUST-1 nanobelts. | |
In summary, we successfully synthesized the HKUST-1 nanobelts by one-step hydrothermal method from Cu NWs. The novel structural transformation process of Cu NWs to HKUST-1 nanobelts is attributed to the template dissolving and self-assembling process with coordination reaction. This study is crucial to develop the nanotechnology of design for MOFs-based one-dimensional materials, which represent an attractive synthetic strategy of 1D MOFs-based material for applications.
AcknowledgmentsThis work was supported by the National Natural Science Foundation of China (No. 21701118), the Natural Science Foundation of Jiangsu Province (Nos. BK20161209 and BK20160323), Natural Science Research Project of Jiangsu Higher Education Institutions (No. 18KJA480004) and Priority Academic Program Development (PAPD) of Jiangsu Higher Education Institutions. We also extend our sincere appreciation to the support by Suzhou Key Laboratory for Advanced Carbon Materials and Wearable Energy Technologies, Suzhou 215006, China.
Appendix A. Supplementary dataSupplementary material related to this article canbefound, in the online version, at doi:https://doi.org/10.1016/j.cclet.2019.05.005.
| [1] |
M.J. Rosseinsky, Microporous Mesoporous Mater. 73 (2004) 15-30. DOI:10.1016/j.micromeso.2003.05.001 |
| [2] |
Y. Lin, C. Kong, Q. Zhang, L. Chen, Adv. Energy Mater. 7 (2017) 1601296. DOI:10.1002/aenm.201601296 |
| [3] |
A. Dhakshinamoorthy, A.M. Asiri, H. Garcia, Angew. Chem. Int. Ed. 55 (2016) 5414-5445. DOI:10.1002/anie.201505581 |
| [4] |
C.C. Wang, Y.Q. Zhang, J. Li, P. Wang, J. Mol. Struct. 1083 (2015) 127-136. DOI:10.1016/j.molstruc.2014.11.036 |
| [5] |
I. Stassen, N. Burtch, A. Talin, et al., Chem. Soc. Rev. 46 (2017) 3185-3241. DOI:10.1039/C7CS00122C |
| [6] |
F.Y. Yi, D. Chen, M.K. Wu, L. Han, H.L. Jiang, ChemPlusChem 81 (2016) 675-690. DOI:10.1002/cplu.201600137 |
| [7] |
M.X. Wu, Y.W. Yang, Adv. Mater. 29 (2017) 1606134. DOI:10.1002/adma.201606134 |
| [8] |
Q. Fu, L. Wen, L. Zhang, Y. Yang, H. Zhang, ACS Appl. Mater. Inter. 9 (2017) 33979-33988. DOI:10.1021/acsami.7b10872 |
| [9] |
K. Okada, R. Ricco, Y. Tokudome, et al., Adv. Funct. Mater. 24 (2014) 1969-1977. DOI:10.1002/adfm.201303303 |
| [10] |
J.W. Jiang, T. Rabczuk, Appl. Phys. Lett. 102 (2013) 123104. DOI:10.1063/1.4799029 |
| [11] |
J.W. Jiang, N. Yang, B.S. Wang, T. Rabczuk, Nano Lett. 13 (2013) 1670-1674. DOI:10.1021/nl400127q |
| [12] |
N.P. Dasgupta, J. Sun, C. Liu, et al., Adv. Mater. 26 (2014) 2137-2184. DOI:10.1002/adma.201305929 |
| [13] |
V. André, F. Galego, M. Martins, Cryst. Growth Des. 18 (2018) 2067-2081. DOI:10.1021/acs.cgd.7b01523 |
| [14] |
N. Liu, Y. Yao, J.J. Cha, et al., Nano Res. 5 (2011) 109-116. DOI:10.1007/s12274-011-0190-1 |
| [15] |
W.W. Zhan, Q. Kuang, J.Z. Zhou, et al., J. Am. Chem. Soc. 135 (2013) 1926-1933. DOI:10.1021/ja311085e |
| [16] |
D. Zhang, P. Liu, S. Xiao, et al., Nanoscale 8 (2016) 7749-7754. DOI:10.1039/C5NR07505J |
| [17] |
I. Luz, A. Loiudice, D.T. Sun, W.L. Queen, R. Buonsanti, Chem. Mater. 28 (2016) 3839-3849. DOI:10.1021/acs.chemmater.6b00880 |
| [18] |
L. Zou, C.C. Hou, Z. Liu, H. Pang, Q. Xu, J. Am. Chem. Soc. 140 (2018) 15393-15401. DOI:10.1021/jacs.8b09092 |
| [19] |
W. Zhang, Z.Y. Wu, H.L. Jiang, S.H. Yu, J. Am. Chem. Soc. 136 (2014) 14385-14388. DOI:10.1021/ja5084128 |
| [20] |
P. Pachfule, D. Shinde, M. Majumder, Q. Xu, Nat. Chem. 8 (2016) 718-724. DOI:10.1038/nchem.2515 |
| [21] |
B. Mortazavi, M. Shahrokhi, M. Makaremi, G. Cuniberti, T. Rabczuk, Mater. Today Energy 10 (2018) 336-342. DOI:10.1016/j.mtener.2018.10.007 |
| [22] |
B. Mortazavi, M. Shahrokhi, T. Hussain, X. Zhuang, T. Rabczuk, Appl. Mater. Today 15 (2019) 405-415. DOI:10.1016/j.apmt.2019.03.002 |
| [23] |
S. Ye, I.E. Stewart, Z. Chen, et al., Accounts Chem. Res. 49 (2016) 442-451. DOI:10.1021/acs.accounts.5b00506 |
 2020, Vol. 31
2020, Vol. 31 

