b School of Medicine, Ji'nan University, Guangzhou 510632, China
Euphorbia fischeriana Steud., a traditional Chinese medicinal herb, is rich in diterpenoids [1-10]. Many of these compounds possess significant anticancer activities. For example, 17-hydroxyjolkinolide B (Fig. S1 in Supporting information), one of the major diterpenoid constituents of E. fischeriana, can induce apoptosis of human cancer cells by activating STAT3-mediated signaling pathway [11]. Recently, fischeriana A (Fig. S1), a meroditerpenoid, was found in E. fischeriana and also showed inhibitory activity against HepG2 cell line [12]. Our chemical investigation on the roots of E. fischeriana led to isolation of another novel meroditerpenoid euphoractone (1) with an unusual ent-abietane-phloroglucinol skeleton (Fig. 1). Herein, we report its structural elucidation, plausible biosynthetic pathway and cytotoxic activity evaluation.
|
Download:
|
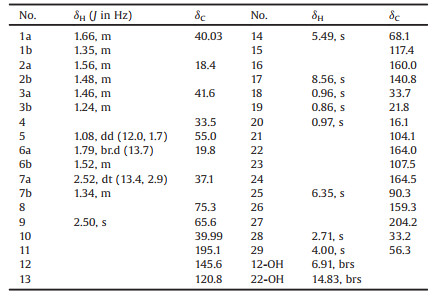
| Fig. 1. The structure of 1. | |
The ethanolic extracts of the dried roots of E. fischeriana were partitioned between water and ethyl acetate. The ethyl acetate fraction was chromatographed successively over silica gel and revered-phase preparative HPLC to yield 1 (15.2 mg).
Compound 1 was obtained as a yellow needle crystal with [α]D25 –84.5 (c 0.10, MeOH). Its molecular formula was determined to be C29H34O9 according to its quasi-molecular ion peak at m/z 527.2291 [M + H]+ (calcd. 527.2276) in the HR-ESI-MS, indicating 13 indices of hydrogen deficiency. The characteristic peaks attributed to hydroxyl (3414 cm-1), carbonyl (1725 and 1672 cm-1) and benzyl groups (1618 cm-1) were observed in the IR spectrum. Its 1H, 13C and HSQC NMR spectra indicated the presence of four methyl groups [δH 0.86 (s), δC 21.8; δH 0.96 (s), δC 33.7; δH 0.97 (s), δC 16.1; δH 2.71 (s), δC 33.2], one methoxyl [δH 4.00 (s), δC 56.3], two keto groups (δC 204.2 and 195.1), the other nine sp2 quaternary carbons (δC 104.1, 107.5, 117.4, 120.8, 145.6, 159.3, 160.0, 164.0 and 164.5), two sp2 methines [δH 8.56 (s), δC 140.8; δH 6.35 (s), δC 90.3], one sp3 oxygen-bearing methine [δH 5.49 (br.s), δC 68.1], one sp3 oxygen-bearing quaternary carbon (δC 75.3), five methylenes [δH1.56 (m) and 1.48 (m), δC 18.4; δH 1.79 (br.d, J =13.7 Hz) and 1.52 (m), δC 19.8; δH 2.52 (dt, J = 13.4, 2.9 Hz) and 1.34 (m), δC 37.1; δH 1.66 (m) and 1.35 (m), δC 40.03; δH 1.46 (m) and 1.24 (m), δC 41.6], two sp3 methines [δH 1.08 (m), δC 55.0; δH 2.50 (s), δC 65.6] and two sp3 quaternary carbons (δC 33.5 and 39.99) (Table 1). In addition, two active proton signals at δH 6.91 and 14.83 were observed in the 1H NMR data (Table 1).
|
|
Table 1 1H NMR (400 MHz) and 13C NMR (100 MHz) data of compound 1 (δ in ppm, in CDCl3). |
The structure of 1 was established by analysis of its 2D NMR spectra (Fig. 2). The HMBC correlations from H-25 to C-21, C-23, C-24 and C-26, from H3-28 to C-27 and C-23, and from the active proton (δH 14.83) to C-21, C-22 and C-23, together with the chemical shifts of C-22, C-24 and C-26, indicated the presence of a phloroglucinol moiety (2, 4, 6-trihydroxyacetophenone) (Fig. 2). And the HMBC correlation between H3-29 and C-24 implied that the hydroxyl group at C-24 was methylated (Fig. 2). The HMBC correlations from H3-18 and H3-19 to C-3, C-4 and C-5, from H-2 and H-3 to C-1, from H3-20 to C-1, C-5, C-9 and C-10, from H-5 to C-6, from H-6 to C-4, C-8 and C-10, and from H-9 to C-8, as well as the 1H-1H COSY correlation between H-6 and H-7, established the rings A and B (Fig. 2). The construction of the ring C was based on the correlations from H-9 to C-11, C-12 and C-14, from H-14 to C-12 and C-13 and from the active proton (δH 6.91) to C-11, C-12 and C-13 in the HMBC spectrum. The remaining three sp2 carbons at δC 117.4, 140.8 and 160.0 were readily assigned to one double bond and one carbonyl carbon (carboxyl or ester). The HMBC correlations from H-17 to C-13, C-16, C-22 and C-26 indicated the presence of an abietane or ent-abietane moiety which is tethered with a 2, 4, 6-trihydroxyacetophenone unit. Thus, the semi-finished structure accounted for 12 out of the total 13 indices of hydrogen deficiency, indicating the presence of an additional ring in 1. This made us readily infer that a lactone ring should form between C-16 and one of three oxygen-bearing carbons (C-8, C-14 and C-26). Based on the fact that the proton signals of two hydroxyl groups locating at the sp2 carbons (C-12 and C-22) were observed in the 1H NMR spectrum while the proton signal of the hydroxyl group at C-26 was obscure, we thus inferred that the lactone ring (ring D) was closed by the formation of an ester bond between C-16 and C-26 (Fig. 1). In the NOESY spectrum of 1, the correlations of H-9/H-5/H3-19 and H-14/H3-20 were observed, indicating that H-5, H-9 and CH3-19 are on the same side, and H-14 and CH3-20 have the same orientations (Fig. 2).

|
Download:
|
| Fig. 2. Selective 2D NMR correlations of 1. | |
Finally, the planar structure of 1 and its absolute configurations were determined by X-ray crystallography using anomalous scattering of Cu Kα radiation (the Flack parameter is 0.078) [13] (Fig. 3). Two molecules of 1 and one molecule of chloroform were found in each asymmetric unit cell (Fig. S2 in Supporting information). Due to the presence of chlorine, the absolute configurations of 1 were unambiguously determined. Moreover, these two molecules of 1 in each asymmetric unit cell adopt the opposite conformations with respect to the spatial arrangement of the A/B/C ring system and the D/E ring system about the bond between C-13 and C-15 (Fig. S2), indicating the free rotation about the bond between C-13 and C-15 is not hindered. This excludes the presence of axial chirality in 1.

|
Download:
|
| Fig. 3. ORTEP drawing of 1. | |
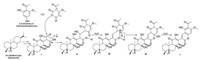
The plausible biosynthetic pathway for 1 was proposed (Scheme 1). C-5 of the diketo form of 4, 6-dihydroxy-2-methox-yacetophenone [14] attacks C-17 (aldehyde group) of the ent-abietane derivative Ⅰ to yield Ⅱ. Ⅱ is subsequently converted into Ⅲ by dehydration. Next, the carboxyl group (C-16) attacks C-26 to afford the hemiketal intermediate Ⅳ [15] which then eliminates one molecule of water to yield 1.
|
Download:
|
| Scheme 1. The plausible biosynthetic pathway of 1. | |
In the cytotoxicity assay [16], 1 exhibited inhibitory activities against H23 and H460 cells with IC50 values of 21.07 ± 3.54 and 20.91 ± 4.07 μmol/L. The IC50 values of the positive control cisplatin against H23 and H460 cells were 5.23 ± 1.13 and 3.81 ± 0.97 μmol/L, respectively.
Most of meroditerpenoids were isolated from fungi and marine organisms [17, 18], and only an extremely limited number of them were found in plants [12]. Many meroditerpenoids harbor diterpenoid moieties with 6/6/6 ring systems [17, 18]. And the diterpenoid units of these compounds so far found share a common skeleton [17, 18], as shown in the structure of epitaondiol (the first meroditerpenoid of this type) [19]. Several meroditerpenoids of this type possess potent anticancer activities, e.g., strongylophorine-26 which can inhibit cancer cell invasion [20]. 1 represents a novel meroditerpenoid harboring an ent-abietanetype diterpenoid moiety (with a 6/6/6 ring system, as well), and possesses cytotoxic activity against cancer cells. Accordingly, its discovery enriched the structural diversity of anticancer meroditerpenoids harboring diterpenoid moieties with 6/6/6 ring systems.
AcknowledgmentsThis work was financially supported by the National Natural Science Foundation of China (No. 81673530) and Local Innovative and Research Teams Project of Guangdong Pearl River Talents Program (No. 2017BT01Y036).
Appendix A. Supplementary dataSupplementary material related to this article can be found, in the online version, at doi:https://doi.org/10.1016/j.cclet.2019.07.068.
| [1] |
B.Y. Jian, H. Zhang, C.C. Han, et al., Molecules 23 (2018) 387-397. DOI:10.3390/molecules23020387 |
| [2] |
M.H. Li, F. He, Y. Zhou, et al., Arch. Pharm. Res. 42 (2019) 1-7. DOI:10.1007/s12272-018-01102-z |
| [3] |
B.Y. Jian, H. Zhang, J.C. Liu, Molecules 23 (2018) 935-946>. DOI:10.3390/molecules23040935 |
| [4] |
H.B. Wang, W. Chen, Y.Y. Zhang, et al., Fitoterapia 91 (2013) 211-216. DOI:10.1016/j.fitote.2013.09.003 |
| [5] |
X.Z. Kuang, W. Li, Y. Kanno, et al., J. Nat. Med. 70 (2016) 412-422. DOI:10.1007/s11418-016-0987-z |
| [6] |
X.Z. Kuang, W. Li, Y. Kanno, et al., J. Nat. Med. 70 (2016) 120-126. DOI:10.1007/s11418-015-0940-6 |
| [7] |
J. Zhang, J. He, X.X. Wang, et al., RSC Adv. 7 (2017) 55859-55865. DOI:10.1039/C7RA11982H |
| [8] |
Y.L. Wei, Z.L. Yu, X.K. Huo, et al., J. Asian Nat. Prod. Res. 20 (2018) 977-984. DOI:10.1080/10286020.2017.1367923 |
| [9] |
J.W. Lee, C. Lee, Q.H. Jin, et al., J. Nat. Prod. 79 (2016) 126-131. DOI:10.1021/acs.jnatprod.5b00789 |
| [10] |
Y.B. Wang, R. Huang, H.B. Wang, et al., J. Nat. Prod. 69 (2006) 967-970. DOI:10.1021/np0600088 |
| [11] |
Y. Wang, X.Q. Ma, S.S. Yan, et al., Cancer Res. 69 (2009) 7302-7311. DOI:10.1158/0008-5472.CAN-09-0462 |
| [12] |
J. He, J.K. Xu, J. Zhang, et al., Org. Biomol. Chem. 17 (2019) 2721-2724. DOI:10.1039/C9OB00013E |
| [13] |
J. Chen, X.N. Fan, J.H. Zhu, et al., Acta Pharm. Sin. B 8 (2018) 639-644. DOI:10.1016/j.apsb.2017.12.009 |
| [14] |
K.O. Kim, D. Lee, N.T. Hiep, et al., Molecules 24 (2019) 195. DOI:10.3390/molecules24010195 |
| [15] |
W. Langschwager, H.M.R. Hoffmann, Liebigs Ann. 1995 (1995) 797-802. DOI:10.1002/jlac.1995199505117 |
| [16] |
Z.F. Wu, H. Bao, F.Y. Zhou, et al., Chin. Chem. Lett. 28 (2017) 1711-1715. DOI:10.1016/j.cclet.2017.04.023 |
| [17] |
R. Geris, T.J. Simpson, Nat. Prod. Rep. 26 (2009) 1063-1094. DOI:10.1039/b820413f |
| [18] |
Y. Matsuda, I. Abe, Nat. Prod. Rep. 33 (2016) 26-53. DOI:10.1039/C5NP00090D |
| [19] |
F. Sánchez-Ferrando, A. San-Martín, J. Org. Chem. 60 (1995) 1475-1478. DOI:10.1021/jo00110a062 |
| [20] |
L.M. McHardy, K. Warabi, R.J. Andersen, et al., Mol. Cancer Ther. 4 (2005) 772-778. DOI:10.1158/1535-7163.MCT-04-0310 |
 2020, Vol. 31
2020, Vol. 31