b State Key Laboratory of Chemical Engineering, Center for Chemistry of High Performance & Novel Materials, Department of Chemistry, Zhejiang University, Hangzhou 310027, China;
c Key Laboratory of Mesoscopic Chemistry of MOE, Jiangsu Key Laboratory of Advanced Organic Materials, School of Chemistry and Chemical Engineering, Nanjing University, Nanjing 210023, China
Supramolecular polymers (SPs), which combine the advantages of traditional polymer science and supramolecular chemistry, are advanced materials constructed by reversible non-covalent interactions [1-6]. In recent years, SPs have been developed rapidly due to their fascinating properties and wide applications. Compared with SPs constructed only by one type of non-covalent interaction, such as hydrogen bonding, host–guest interactions, metal–ligand coordination, or π-π interactions, functional SPs self-assembled by the combination of multiple non-covalent interactions in an orthogonal way can lead to some excellent properties [7-10]. The fabrication strategy can also be learned from nature. For example, for DNA, multiple non-covalent interactions including hydrogen bonding, π-π interaction, and hydrophobic interaction contribute to its double helix structure and properties [11-13]. In artificial systems, the orthogonal self-assembly means the fact that "different kinds of non-covalent interactions within one supramolecular complex do not interfere with each other" or "two or more non-covalent interactions occur in system with no crosstalk" [14, 15]. In 1997, Reinhoudt reported the first discrete nanostructure assembled by hydrogen bonding and metal–ligand coordination in an orthogonal way [16]. Multiple non-covalent interactions endow the complexes with strong ability to undergo reorganization and dynamic exchange processes by means of the reversible assembly procedures.
Among different types of non-covalent interactions employed in the construction of functional SPs, hydrogen bonding and host–guest interactions are two main types of supramolecular interactions and have been researched extensively. Hydrogen bonding plays an important role in biological systems and is also a powerful tool to construct dynamic supramolecular architectures. Hydrogen bonding is very attractive for their high directionality and fidelity. Although individual hydrogen bonding is weak, multiple hydrogen bonding array may offer high stability and show much greater binding strength [17]. The most well-known multiple hydrogen bonding ureidopyrimidinone (UPy) motif, first reported by Meijer et al., showed extremely strong dimerization constant (Kass > 107 L/mol in CHCl3) via quadruple hydrogen bonding [18, 19]. In recent years, a large number of functional SPs have been constructed from UPy motif. For example, Xu, Zhang et al. developed supramolecular interfacial polymerization by using UPy groups [20]. Tang et al. reported an aggregation-induced emission SP from a ditopic UPy monomer [21]. Moreover, Yang et al. constructed photores-ponsive SPs and fluorescent nanoparticles based on the UPy motif [22, 23].
In addition, macrocyclic host–guest interactions have played a critical role in supramolecular chemistry and developed rapidly due to the diversity of the macrocycles. Recently, Liu et al. reviewed water soluble calix[n]arenes and its biological applications [24]. Tian et al. systematically summarized photoresponsive host–guest functional systems [25]. Wang et al. reviewed the applications of cucurbit[n]urils in pharmaceutical sciences [26]. T. Xiao, L. Wang et al. carefully summarized dynamic hydrogels and artificial lightharvesting systems constructed by host–guest interactions [27, 28]. In recent years, macrocyclic host–guest interactions have also been extensively employed to construct SPs, especially by crown ethers or pillar[n]arene macrocycles. For example, Huang et al. summarized cryptand-based host–guest system and crown ether-based SPs [29, 30]. Li, Tian et al. reviewed SPs fabricated from pillar[n] arenes [31]. Yin et al. fabricated a series of fluorescent SPs driven by orthogonal self-assembly based on host–guest interactions and metal–ligand coordination [32-37]. In short, host–guest interactions endow SPs with inherent adaptability, leading their responsiveness to multiple external stimuli and therefore gaining more interesting functions [38, 39].
In this review, we aim to describe the recent developments in functional SPs driven by orthogonal multiple hydrogen bonding and host–guest interactions, with an emphasis on those having applications in the fields of dynamic materials, molecular machines, and soft matter. Herein, we describe the SPs depending on the types of the macrocycles that were employed. Therefore, the content of this review is organized as follows: (a) SPs constructed by multiple hydrogen bonding and crown etherbased host–guest interactions, (b) SPs constructed by multiple hydrogen bonding and pillararene-based host–guest interactions, (c) SPs constructed by multiple non-covalent interactions more than hydrogen bonding and host–guest recognition. We will discuss those SPs constructed from orthogonal self-assembly in detail, covering their fabrication, properties, and potential applications. Finally, current challenges and future perspectives for SPs constructed by orthogonal self-assembly are also illustrated.
2. SPs constructed by multiple hydrogen bonding and crown ether-based host–guest interactionsIn 2002, Tokunaga et al. reported a pseudorotaxane dimer driven by crown ether-based host–guest interactions and quadruple hydrogen bonding [40]. Next year, Li et al. constructed welldefined supramolecular heterodimers with unique pseudo[2]rotaxane structures which were also stabilized by the host–guest interactions and quadruple hydrogen bonding [41]. Following the above studies, Wang et al. have also employed these two supramolecular orthogonal interactions to construct various supramolecular complexes that possessed dynamic behaviors with responses to external stimuli. The electron deficient macrocycles, such as cyclobis(paraquat-p-phenylene)cyclophane (known as "blue box" or CBPQT4+), have attracted much attention due to their interesting properties [42, 43]. Wang et al. prepared a novel "blue box"-based dynamic [2]catenane, in which the CBPQT4+ was interlocked by the quadruple hydrogen bonding UPy motif [44]. In this work, a ditopic UPy derivative involving 1, 5-dioxynaphthalene (DNP) unit was firstly synthesized, which could undergo a concentration-dependent ring-opening polymerization to form linear SPs. However, in the presence of CBPQT4+, a dynamic donor– acceptor [2]catenane interlocked by dimerized UPy could be obtained selectively. Interestingly, the dynamic properties of the [2]catenane were further characterized via a ring-exchange experiment by employing a similar [2]catenane which was formed by CBPQT4+ and a 1, 4-dioxyphenylene bridged ditopic UPy compound. In the ring-exchange experiment, the color of the solution changed gradually from magenta to purple, due to the different strengths of host–guest interactions. This is a new type of dynamic catenane interlocked by hydrogen bonding and it represents a new kind of functional supramolecular complex constructed by quadruple hydrogen bonding and host–guest interactions via orthogonal self-assembly.
In 2011, Wang et al. first constructed a polymeric supramolecular complex based on two small-molecular-weight heteroditopic monomers by using UPy-based quadruple hydrogen bonding and crown ether-based host–guest recognition [45]. Dynamic light scattering (DLS) experiments showed that the equimolar solution of two monomers (40 mmol/L for each monomer) had a hydrodynamic radius of 31 nm in a mixed solvent of chloroform and acetonitrile (1/1, v/v). When the concentration increased to 433 mmol/L, polymerization degree of 38.5 was determined according to 1H NMR by using the Carothers equation, which corresponds to a molecular weight of 65.9 kDa. The polymeric supramolecular complex constructed by orthogonal self-assembly was further confirmed by viscosity measurements, scanning electronic microscopy (SEM) and concentration- dependent 1H NMR experiments. In a follow-up study, they further fabricated a novel kind of crown ether–paraquat supramolecular polypseudorotaxanes with the hydrogen bonded SP as the main chain (Fig. 1) [46]. This polypseudorotaxane network was based on two homoditopic monomers: One is the bifunctional UPys with a bis(p-phenylene)-34-crown-10 (BPP34C10) located at the center of the monomer (1) and the other is the ditopic paraquat molecule (2). Monomer 1 can self-assemble into linear SPs via quadruple hydrogen bonding. Upon addition of monomer 2, the paraquat group can thread into the crown ether cavity to achieve polypseudorotaxane networks. It is worth noting that the molar ratios of the bisparaquat are critical for the solution viscosity due to the different cross-linking degrees. The DOSY NMR also demonstrated that larger aggregates were formed after the addition of 0.5 equiv. of 2.

|
Download:
|
| Fig. 1. Graphical representation of the construction of polypseudorotaxane networks from monomers 1 and 2. Reproduced with permission [46]. Copyright 2011, the Royal Society of Chemistry. | |
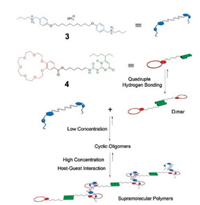
To develop responsive SPs, Wang et al. further explored the construction of metal-ion-responsive dynamic SPs driven by orthogonal quadruple hydrogen bonding and benzo-21-crown-7 (B21C7)-based host–guest interactions [47]. They first synthesized two types of monomers: the homoditopic building block 3, which bears two dialkylammonium units, and the heteroditopic building block 4, which is comprised of one B21C7 motif and one UPy group (Fig. 2). As we can see, a supramolecular dimer from 4 was initially obtained, followed by polymerization upon addition of 3. The disassembly/assembly of this SP could be reversibly switched by addition/removal of K+ without interfering with UPy dimerization due to the stronger binding of K+ with B21C7 moiety than dialkylammonium moiety.
|
Download:
|
| Fig. 2. Graphical representation of the SPs constructed from monomers 3 and 4 by orthogonal self-assembly. Reproduced with permission [47]. Copyright 2013, the Royal Society of Chemistry. | |
In 2015, Zimmerman, Huang et al. reported a double supramolecular crosslinked polymer gel which showed macroscale expansion and contraction behavior and multistimuli responsiveness [48]. As we know, most of the supramolecular polymeric gels contain only one type of non-covalent interaction. In this example, they constructed a novel supramolecular polymeric gel possessing two types of supramolecular crosslinks. Two types of non-covalent interactions are orthogonal: diamido- 1, 8-naphthyridine (DAN)-based quadruple hydrogen bonding and B21C7-based host–guest interactions. As shown from Fig. 3, polymer 5 is polystyrene with pendant DAN groups and dialkylammonium salt units, and polymer 6 is poly(butyl methacrylate) bearing deazaguanosine (DeUG) groups and B21C7 units. Notably, DAN can bind DeUG to form stable quadruple hydrogen bonding complexes, in which the hydrogen bonding interaction is highly orthogonal to B21C7-based host–guest interactions. Thus polymers 5 and 6 could be crosslinked by these two orthogonal non-covalent interactions, resulting in the formation of a supramolecular polymeric gel. This double crosslinked gel could undergo macroscale expansion and contraction behavior via the introduction of competitive compounds or ions, such as the DAN monomer (eDAN), K+ or Cl-. This example demonstrated that the macroscopic property of SPs can be induced by controlling the orthogonal self-assembly on a molecular scale.

|
Download:
|
| Fig. 3. Cartoon representations of polymers 5 and 6 and the illustration of the advantage of doubly vs. singly supramolecular crosslinked polymer gels. Reproduced with permission [48]. Copyright 2015, the Royal Society of Chemistry. | |
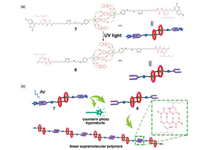
In 2016, Qu et al. reported a quadruple hydrogen-bonded SP with a [c2]daisy chain structure in its backbone [49]. This SP was constructed by phototriggered supramolecular polymerization of the [c2]daisy chain monomer (Fig. 4). The monomer 7 is a daisy chain rotaxane containing two coumarin protected UPy terminals at two ends. The supramolecular polymerization was driven by light irradiation that can remove the coumarin substituents and yield 8 with two terminal UPy groups. Then, linear SPs can be self-assembled by 8 through quadruple hydrogen bonding. This strategy of phototriggered supramolecular polymerization represents a good supplement for the fabrication of stimuli-responsive SPs.
|
Download:
|
| Fig. 4. (a) Chemical structures of 7 and 8; (b) Graphical representation for the phototriggered supramolecular polymerization of the [c2]daisy chain rotaxane 7. Reproduced with permission [49]. Copyright 2016, the Royal Society of Chemistry. | |
Molecular machines have attracted much research interest in recent years due to their ability to produce mechanical work at the nanoscale and may create new functions by energy transduction. When running in an integrated system, this kind of molecular machines can amplify their motion to a macro scale. In 2012, Giuseppone et al. synthesized muscle-like SPs by using bistable [c2]daisy chain and terpyridine-based metal–ligand coordination [50]. In the next work, they constructed an acid–base switchable [c2]daisy chain rotaxane terminated with two 2, 6-diacetylamino pyridine units, which could self-assemble with a bis(uracil) linker through complementary hydrogen bonding [51]. As a result, unidimensional SPs associated in bundles of muscle-like fibers could be obtained. In 2017, they further reported the integration of bistable [c2]daisy chain rotaxanes by employing UPy-based quadruple hydrogen bonding SP [52]. The monomer exhibits a chemical structure similar to the previous example reported by Qu et al. However, the big difference in this case is that the [c2]daisy chain is a bistable rotaxane. Therefore, the monomer can form "extended" and "contracted" SPs by controlling the pH of the system. In toluene, the "extended" SP forms a gel while the "contracted" one remains as a solution. The sol–gel transition could be changed in situ by adjusting the bistable [c2]daisy chains. Therefore, the macroscopic physical state of the material could be controlled by changing the mechanical bond at nanoscale. This example demonstrates the possibility of integrating molecular machines into novel smart materials.
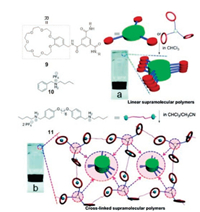
In recent years, Dong et al. prepared many novel supramolecular polymers based on host–guest interactions [53-58]. Very recently, they constructed a cross-linked SP network based on the B21C7/secondary ammonium salt host–guest recognition and 1, 3, 5-benzenetricarboxamide (BTA) hydrogen bonding motif [59]. The BTA motif is a well-known building block for supramolecular polymerization, which could be stabilized by threefold H-bonding [60]. They firstly designed and synthesized the BTA derivative with three B21C7 units (9) (Fig. 5). Monomer 9 can self-assemble into one-dimensional linear SPs through the aggregation of BTA cores via hydrogen bonding. Then, cross-linked SPs could be formed by adding guest molecule 11, which possesses two secondary ammonium salts and plays the role of a linker to connect the linear polymers formed by 9 together. The formation of the crosslinked polymers was evidenced by 1H NMR, DOSY, and viscosity measurements. Interestingly, soft and long viscous fibers could be pulled out from a concentrated solution of the cross-linked polymers. Notably, the cross-linked supramolecular (9 at 160 mmol/L and 11 at 240 mmol/L) existed in the gel-like state below 45 ℃ and converted into a viscous solution above 50 ℃. The SPs constructed in this work provide new understanding for supramolecular polymerization.
|
Download:
|
| Fig. 5. Cartoon representation for the construction of one dimensional SPs and cross-linked SPs. Reproduced with permission [59]. Copyright 2018, the Royal Society of Chemistry. | |
3. SPs constructed by multiple hydrogen bonding and pillar[n] arene-based host–guest interactions
Pillar[n]arenes are a relatively new type of macrocyclic host in supramolecular chemistry and have displayed versatile host–guest behavior with various kinds of guests [61-66]. The synthetic route of pillar[n]arenes is easy and pillar[n]arenes have unique shape, versatile solubility, and functionality. The symmetrical pillar architecture and easy derivatization endow pillar[n]arenes with a good ability to bind different kinds of guests, such as cationic guests, anionic guests, neutral guests, flexible guests, and rigid guests. In recent years, pillar[n]arenes have been employed to construct various kinds of functional supramolecular materials, such as gels [67-77], hydrogels [78-83], mechanically interlocked systems [84-86], supramolecular vesicles/nanocarriers [87-113], functional nanoparticles [114, 115], sensors [116-120], crystal materials [121-130], stimuli-responsive materials [131-138], and SPs [139-148]. In this section, we focus on SPs constructed orthogonally by hydrogen bonding and pillar[n]arene-based host– guest interactions.
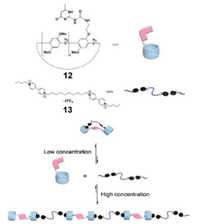
In recent years, Wang et al. fabricated a series of SPs orthogonally by employing pillar[n]arene-based host–guest interactions and UPy-based quadruple hydrogen bonding. For example, they synthesized a UPy functionalized pillar[5]arene (12) and a bisparaquat derivative (13), which can orthogonally self-assemble into polymeric architectures (Fig. 6) [149]. The binding constant was calculated to be 4.46 × 103 L/mol via fluorescent titration by using a model compound. The complexation of 12 with 13 was confirmed by 1H NMR titration and electrospray ionization mass spectrometry (ESI-MS). The formation of SPs at high concentration driven by two orthogonal interactions, quadruple hydrogen bonding and host–guest interactions, was proven by concentration-dependent 1H NMR, 2D NOESY NMR, DOSY, viscosity measurements, and cyclic voltammetry. This study opened a new window to construct orthogonal SPs by employing pillar[n] arenes.
|
Download:
|
| Fig. 6. Illustration of the formation of the SP from 12 and 13. Reproduced with permission [149]. Copyright 2012, the Royal Society of Chemistry. | |
As we can see from the above examples, the combination of different supramolecular interactions is a very easy and efficient way for the construction of complicated supramolecular structures. In another examples, Wang et al. fabricated a pillar[5]arene-based supramolecular polypseudorotaxane [150]. In this study, they first synthesized a ditopic UPy-functionalized pillar[5]arene 14 (Fig. 7). Monomer 14 can form linear SPs at high concentration. After that, when the guest molecule 1, 4-butanediamine (15) was added, a polypseudorotaxane could be obtained in the concentrated solution. This study affords the first example of a linear supramolecular polypseudorotaxane driven by efficient quadruple hydrogen bonding interactions and pillar[n]arene based host–guest recognition, and it also enriches the applications of the host–guest chemistry of pillar[n]arene. In the follow-up study, they further constructed polypseudorotaxane-based polymer networks by using 14 and a bisparaquat derivative, in which the backbones were constructed from quadruple hydrogen bonding [151].
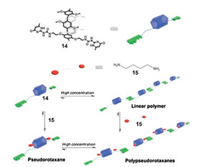
|
Download:
|
| Fig. 7. Graphical representation of the construction of polypseudorotaxane polymers from 14 and 15. Reproduced with permission [150]. Copyright 2012, the Royal Society of Chemistry. | |
Immediately after the above work, they further constructed polyrotaxanes by employing the method of "diamine threading followed by end-capping with UPy units" to access the solventresponsive SPs [152]. The obtained dynamic polyrotaxanes were fabricated from pillar[5]arene derivatives 16 and diamines 17 or 17' (Fig. 8). The polyrotaxanes were interlocked by orthogonal quadruple hydrogen bonding and host–guest interactions. The UPy groups played a dual role, acting not only as an end-capping stopper but also as a hydrogen bonded linking group to regulate the conformation and dynamic properties of the supramolecular polyrotaxanes. Notably, the reversibility of main-chain backbones and the sliding process of the axle could be achieved by changing the polarity of the solvent.
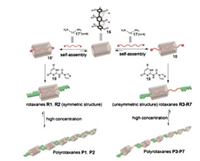
|
Download:
|
| Fig. 8. Graphical representation of the construction of dynamic polyrotaxanes. Reproduced with permission [152]. Copyright 2012, American Chemical Society. | |
Very recently, a dual-responsive hyperbranched SP was constructed by Zhang, Guo et al. [153]. This hyperbranched SP was driven by cooperative hydrogen bonding interaction and host–guest recognition (Fig. 9). They first prepared a homotritopic pillar[5]arene containing adenine groups (21). Then they synthesized a uracil derivative (6-(2, 4-dioxo-3, 4-dihydropyrimidin-1 (2H)-yl)hexanenitrile, 22). 1H NMR suggested that 22 could thread into the cavity of the model compound 20 to form a [2]pseudorotaxane and the binding stoichiometry was determined to be 1:1. The hyperbranched SP could be formed from 21 and 22 at a low concentration due to the strong cooperative interactions. The supramolecular polymerization was proved by the combination of experiments including 1H NMR, viscosity measurements, and DOSY experiments at various concentrations. This material displayed dual-responsiveness to heating and cooling, or the addition of acid/base.
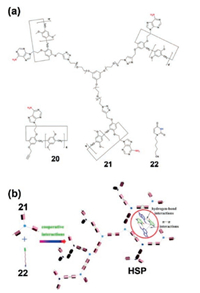
|
Download:
|
| Fig. 9. (a) Chemical structures of 20, 21 and 22. (b) The illustration of the construction of hyperbranched SP HSP from 21 and 22 through cooperative host– guest recognition and hydrogen-bond interactions. Reproduced with permission [153]. Copyright 2018, the Royal Society of Chemistry. | |
4. SPs constructed by multiple non-covalent interactions involving hydrogen bonding and host–guest recognition
Nature has provided lots of examples which display the ability to construct exquisite and complex biological systems from relatively simple building blocks via multiple types of noncovalent interactions. It is also noteworthy that hierarchical selfassembly strategy is an effective way to fabricate well-organized structures. Inspired by these facts, chemists began to adopt multiple orthogonal non-covalent interactions to fabricate functional SPs hierarchically. In an integrated system, multiple orthogonal non-covalent interactions will endow the SPs with more responsivenesses upon external stimuli and improve the dynamic properties of the material. For example, Li et al. first constructed a three-component dynamic [2]catenane by employing hydrogen bonding, metal–ligand coordination, and host–guest interaction in 2007 [154]. In this section, we will give some typical examples to illustrate the diversity of the construction of SPs by employing multiple orthogonal non-covalent interactions involving multiple hydrogen bonding and host–guest interactions.
Besides the traditional covalent polymerization method, mechanically connected polyrotaxanes with unique topological structures can also be realized via the non-covalent methods. In recent years, Wang et al. constructed a series of supramolecular materials with various topological structures [155-160]. In 2015, they modified the [2]rotaxane backbone with two orthogonal recognition motifs and successfully realized the poly[2]rotaxanes by mechanical linkage through the hierarchical self-assembly method [161]. The authors firstly synthesized the [2]rotaxane 24, which was based on the B21C7/secondary ammonium salt recognition unit (Fig. 10). On one hand, terpyridine group was connected on the axle of the [2]rotxane, which could be coordinated with Zn2+ to form dimeric [3]rotaxane 26. On the other hand, a Hamilton receptor was connected to the ring of the [2]rotaxane, which can drive the [2]rotaxane to form hydrogen bonded complex 25 with cyanuric acid functionalized polycaprolactone (PCL) 23. Finally, when mixing all of three components together, the poly[2]rotaxanes 27 could be successfully obtained. Notably, self-standing films could be fabricated by solution casting of 1:2:1 mixture of monomers 23, 24 and Zn(OTf)2 in concentrated solution. This novel type of mechanically linked polyrotaxanes stand for a special SP driven by orthogonal self-assembly based on multiple non-covalent interactions.

|
Download:
|
| Fig. 10. Schematic representation for hierarchical construction of mechanically linked poly[2]rotaxanes 27. Reproduced with permission [161]. Copyright 2015, American Chemical Society. | |
As we can see from the above example, the employment of multiple orthogonal interactions to construct supramolecular complex provides advantages over traditional methods that use only one kind of non-covalent bond. In next example, we introduce a special SP constructed by triply orthogonal non-covalent interactions, which was reported by Yan, Stang and co-workers [162]. As shown from Fig. 11, supramolecular polymeric network 28 was fabricated from a 120° B21C7-tethered acceptor 29 and UPy-decorated 120° donor 30 through metal–ligand bond. The B21C7 moieties within the network offers a platform for further modification via host–guest interaction. For example, tetraphenylethylene (TPE)-functionalized dialkylammonium salt 31b were incorporated into network 28 to get supramolecular networks with aggregation induced emission property. The last step was highly modular and can only occur when the substrate does not interfere with the metal–ligand coordination or quadruple hydrogen bonds. The orthogonal strategy employed in this research based on non-covalent bonds afforded advantages over the covalent method for achieving complicated polymeric structures in a highly modular way.
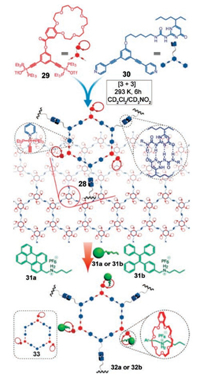
|
Download:
|
| Fig. 11. Cartoon representations of the formation of a supramolecular polymeric network from monomer 29, 30 and 31 via triply orthogonal non-covalent interactions. Reproduced with permission [162]. Copyright 2016, American Chemical Society. | |
The π-π stacking interaction is also prevalent in biological systems, such as the stacking of the base pairs in DNA. In recent years, an increasing number of supramolecular complexes have been constructed by employing π-π stacking interaction and other non-covalent interactions at the same time. For example, Wang et al. prepared a highly stable supramolecular cyclic monomer from a ditopic UPy molecule, which was stabilized by the π-π interaction between the aromatic group in the molecule and the dimeric UPy plane [163, 164]. In 2017, Q. Wang et al. prepared a UPy-based SP network by photo-cross-linking between the coumarin groups [165]. Later, they further constructed a neutral SP by three kinds of non-covalent interactions including UPy-based quadruple hydrogen bonding, pillar[5]arene-based molecular recognition, and π-π donor–acceptor interaction [166]. Firstly, they designed and synthesized an unsymmetrical guest 34 bearing a UPy motif and a carbamate group, which can dimerize through quadruple hydrogen bonding to form a homoditopic AA-type supramonomer (Fig. 12). Secondly, they prepared a pillar[5]arene derivative 35, which can bind with 36 to generate a 2:1 exo-wall complex via π-π interaction, leading to the formation of a homoditopic BB-type supramonomer. Finally, the neutral linear SPs could be successfully fabricated in concentrated solution. Interestingly, the degradation of the SP backbone could be effectively regulated by introducing the competitive adiponitrile guest. In follow-up study, they reported a pillar[5]arene-fused cryptand-based SP, in which three orthogonal supramolecular interactions, pillar[5]arene-based host–guest recognition, cryptand-based host–guest interaction, and quadruple hydrogen bonding were combined together [167]. Moreover, the obtained SPs could be effectively degraded with a change in fluorescence by the addition of a competitive cryptand host.
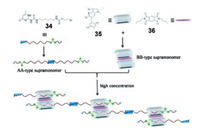
|
Download:
|
| Fig. 12. Graphical representation of SPs constructed by three orthogonal interactions. Reproduced with permission [166]. Copyright 2017, the Royal Society of Chemistry. | |
5. Conclusions and perspectives
In conclusion, recent developments in the construction of SPs based on orthogonal host–guest interactions and multiple hydrogen bonding are reviewed. The orthogonal construction strategy is versatile by employing multiple hydrogen bonding and host–guest interactions due to the versatility of macrocyclic hosts, such as crown ethers and pillar[n]arenes. In addition, SPs constructed by multiple non-covalent interactions involving hydrogen bonding and host–guest interactions are also concerned. The potential to introduce stimuli-responsive motifs into the architectures and the dynamic property of the supramolecular interactions endow these SPs with multiple stimuli-responsive properties. With these characters, many of these species can be made to be used as mechanically interlocked molecules or molecular machines, adaptive SPs, functional gels, nanofiber and self-standing film materials.
Compared with the complex biological systems, SPs constructed by orthogonal self-assembly are still in the initial stage of research. Therefore, the orthogonal self-assembly is still a powerful tool for scientists to understand and mimic the biological systems and also a necessary way to prepare dynamic materials. The current research in this field is still immature and still requires continued exploration. Before getting more practical applications, we are still facing some basic challenges. Herein, we prefer to describe some future directions that may have significant effects in this field. Firstly, the mechanism of orthogonal self-assembly process needs further in-depth study to gain clear insight into the relationship between building blocks and macroscopic functions. Secondly, more water-soluble supramolecular complexes fabricated by orthogonal self-assembly need to be developed. Particularly, the construction of biocompatible supramolecular complexes, which can be incorporated into biological systems, can provide a versatile platform to understand, study, and replicate biological processes. Thirdly, how to promote dynamic SPs to real functional materials is another challenge. For example, more attention needs to be placed on the synthetic simplicity, economic suitability, mechanical strength, and environmental friendliness of such materials. Nevertheless, we believe that the examples described in this review will motivate scientists to further develop multifunctional SPs, which will enrich nanoscience, materials science, and biological technology.
AcknowledgmentWe gratefully thank the financial support from the National Natural Science Foundation of China (Nos. 21702020, 21672102, 21572026).
| [1] |
L. Yang, X. Tan, Z. Wang, et al., Chem. Rev. 115 (2015) 7196-7239. DOI:10.1021/cr500633b |
| [2] |
F. Huang, O.A. Scherman, Chem. Soc. Rev. 41 (2012) 5879-5880. DOI:10.1039/c2cs90071h |
| [3] |
T. Aida, E.W. Meijer, S.I. Stupp, Science 335 (2012) 813-817. DOI:10.1126/science.1205962 |
| [4] |
T.F.A. de Greef, E.W. Meijer, Nature 453 (2008) 171-173. DOI:10.1038/453171a |
| [5] |
L. Brunsveld, B.J. Folmer, E.W. Meijer, et al., Chem. Rev. 101 (2001) 4071-4098. DOI:10.1021/cr990125q |
| [6] |
X. Zheng, Q. Miao, W. Wang, et al., Chin. Chem. Lett. 29 (2018) 1621-1624. DOI:10.1016/j.cclet.2018.04.002 |
| [7] |
S. Datta, M.L. Saha, P.J. Stang, Acc. Chem. Res. 51 (2018) 2047-2063. DOI:10.1021/acs.accounts.8b00233 |
| [8] |
P. Wei, X. Yan, F. Huang, Chem. Soc. Rev. 44 (2015) 815-832. DOI:10.1039/C4CS00327F |
| [9] |
X.Y. Hu, T. Xiao, C. Lin, et al., Acc. Chem. Res. 47 (2014) 2041-2051. DOI:10.1021/ar5000709 |
| [10] |
S.L. Li, T. Xiao, C. Lin, et al., Chem. Soc. Rev. 41 (2012) 5950-5968. DOI:10.1039/c2cs35099h |
| [11] |
J.D. Watson, F.H.C. Crick, Nature 171 (1953) 737-738. DOI:10.1038/171737a0 |
| [12] |
P. Yakovchuk, E. Protozanova, M.D. Frank-Kamenetskii, Nucleic Acids Res. 34 (2006) 564-574. DOI:10.1093/nar/gkj454 |
| [13] |
C.K. McLaughlin, G.D. Hamblin, H.F. Sleiman, Chem. Soc. Rev. 40 (2011) 5647-5656. DOI:10.1039/c1cs15253j |
| [14] |
C.H. Wong, S.C. Zimmerman, Chem. Comm. 49 (2013) 1679-1695. DOI:10.1039/c2cc37316e |
| [15] |
M.L. Saha, S. De, S. Pramanik, et al., Chem. Soc. Rev. 42 (2013) 6860-6909. DOI:10.1039/c3cs60098j |
| [16] |
W.T.S. Huck, R. Hulst, P. Timmerman, et al., Angew. Chem. Int. Ed. 36 (1997) 1006-1008. DOI:10.1002/anie.199710061 |
| [17] |
C.Z. Steven, S.C. Perry, Struct. Bond. 96 (2000) 64-94. |
| [18] |
R.P. Sijbesma, F.H. Beijer, L. Brunsveld, et al., Science 278 (1997) 1601-1604. DOI:10.1126/science.278.5343.1601 |
| [19] |
F.H. Beijer, R.P. Sijbesma, H. Kooijman, et al., J. Am. Chem. Soc. 120 (1998) 6761-6769. DOI:10.1021/ja974112a |
| [20] |
B. Qin, S. Zhang, Q. Song, et al., Angew. Chem. Int. Ed. 56 (2017) 7639-7643. DOI:10.1002/anie.201703572 |
| [21] |
H.Q. Peng, X. Zheng, T. Han, et al., J. Am. Chem. Soc. 139 (2017) 10150-10156. DOI:10.1021/jacs.7b05792 |
| [22] |
H.Q. Peng, C.L. Sun, L.-Y. Niu, et al., Adv. Funct. Mater. 26 (2016) 5483-5489. DOI:10.1002/adfm.201600593 |
| [23] |
J.F. Xu, Y.Z. Chen, D. Wu, et al., Angew. Chem. Int. Ed. 52 (2013) 9738-9742. DOI:10.1002/anie.201303496 |
| [24] |
D.S. Guo, Y. Liu, Acc. Chem. Res. 47 (2014) 1925-1934. DOI:10.1021/ar500009g |
| [25] |
D.H. Qu, Q.C. Wang, Q.W. Zhang, et al., Chem. Rev. 115 (2015) 7543-7588. DOI:10.1021/cr5006342 |
| [26] |
H. Yin, R. Wang, Isr. J. Chem. 58 (2018) 188-198. DOI:10.1002/ijch.201700092 |
| [27] |
T. Xiao, W. Zhong, L. Zhou, et al., Chin. Chem. Lett. 30 (2019) 31-36. DOI:10.1016/j.cclet.2018.05.034 |
| [28] |
T. Xiao, L. Xu, L. Zhou, et al., J. Mater. Chem. B 7 (2019) 1526-1540. DOI:10.1039/C8TB02339E |
| [29] |
M. Zhang, X. Yan, F. Huang, et al., Acc. Chem. Res. 47 (2014) 1995-2005. DOI:10.1021/ar500046r |
| [30] |
B. Zheng, F. Wang, S. Dong, et al., Chem. Soc. Rev. 41 (2012) 1621-1636. DOI:10.1039/C1CS15220C |
| [31] |
H. Li, Y. Yang, F. Xu, et al., Chem. Commun. 55 (2019) 271-285. DOI:10.1039/C8CC08085B |
| [32] |
B. Li, T. He, X. Shen, et al., Polym. Chem. 10 (2019) 796-818. DOI:10.1039/C8PY01396A |
| [33] |
L.N. Xu, D. Chen, Q. Zhang, et al., Polym. Chem. 9 (2018) 399-403. DOI:10.1039/C7PY01788J |
| [34] |
C. Lu, M. Zhang, D. Tang, et al., J. Am. Chem. Soc. 140 (2018) 7674-7680. DOI:10.1021/jacs.8b03781 |
| [35] |
J. Zhang, J. Zhu, C. Lu, et al., Polym. Chem. 7 (2016) 4317-4321. DOI:10.1039/C6PY00872K |
| [36] |
D. Chen, J.Y. Zhan, M.M. Zhang, et al., Polym. Chem. 6 (2015) 25-29. DOI:10.1039/C4PY01206B |
| [37] |
J. Zhan, Q. Li, Q. Hu, et al., Chem. Commun. 50 (2014) 722-724. DOI:10.1039/C3CC47468B |
| [38] |
Y. Chen, F. Huang, Z.T. Li, et al., Sci. China Chem. 61 (2018) 979-992. DOI:10.1007/s11426-018-9337-4 |
| [39] |
Y. Zhou, H. Li, Y.W. Yang, Chin. Chem. Lett. 26 (2015) 825-828. DOI:10.1016/j.cclet.2015.01.038 |
| [40] |
Y. Tokunaga, T. Seo, Chem. Commun. (2002) 970-971. |
| [41] |
X.Z. Wang, X.Q. Li, X.-B. Shao, et al., Chem. Eur. J. 9 (2003) 2904-2913. DOI:10.1002/chem.200204513 |
| [42] |
Z. Liu, S.K.M. Nalluri, J.F. Stoddart, Chem. Soc. Rev. 46 (2017) 2459-2478. DOI:10.1039/C7CS00185A |
| [43] |
O.Š. Miljanić, W.R. Dichtel, S. Mortezaei, et al., Org. Lett. 8 (2006) 4835-4838. DOI:10.1021/ol061864d |
| [44] |
T. Xiao, S.L. Li, Y. Zhang, et al., Chem. Sci. 3 (2012) 1417-1421. DOI:10.1039/c2sc01004f |
| [45] |
S.L. Li, T. Xiao, Y. Wu, et al., Chem. Commun. 47 (2011) 6903-6905. DOI:10.1039/c1cc12003d |
| [46] |
S.L. Li, T. Xiao, B. Hu, et al., Chem. Commun. 47 (2011) 10755. DOI:10.1039/c1cc14559b |
| [47] |
T. Xiao, X. Feng, Q. Wang, et al., Chem. Commun. 49 (2013) 8329-8331. DOI:10.1039/c3cc44525a |
| [48] |
X. Ji, K. Jie, S.C. Zimmerman, et al., Polym. Chem. 6 (2015) 1912-1917. DOI:10.1039/C4PY01715C |
| [49] |
X. Fu, R.R. Gu, Q. Zhang, et al., Polym. Chem. 7 (2016) 2166-2170. DOI:10.1039/C6PY00309E |
| [50] |
G. Du, E. Moulin, N. Jouault, et al., Angew. Chem. Int. Ed. 51 (2012) 12504-12508. DOI:10.1002/anie.201206571 |
| [51] |
A. Goujon, G. Du, E. Moulin, et al., Angew. Chem. Int. Ed. 55 (2016) 703-707. DOI:10.1002/anie.201509813 |
| [52] |
A. Goujon, G. Mariani, T. Lang, et al., J. Am. Chem. Soc. 139 (2017) 4923-4928. DOI:10.1021/jacs.7b00983 |
| [53] |
B. Zheng, Z. Luo, Y. Deng, et al., Chem. Commun. 55 (2019) 782-785. DOI:10.1039/C8CC09160A |
| [54] |
L. Wang, X. Li, Q. Zhang, et al., New J. Chem. 42 (2018) 8330-8333. DOI:10.1039/C8NJ01366G |
| [55] |
Z. Qi, L. Chiappisi, H. Gong, et al., Chem. -Eur. J. 24 (2018) 3854-3861. DOI:10.1002/chem.201705838 |
| [56] |
D. Huang, Q. Zhang, Y. Deng, et al., Polym. Chem. 9 (2018) 2574-2579. DOI:10.1039/C8PY00412A |
| [57] |
C. Han, D. Zhao, S. Dong, Chem. Commun. 54 (2018) 13099-13102. DOI:10.1039/C8CC07993E |
| [58] |
S. Dong, J. Leng, Y. Feng, et al., Sci. Adv. 3 (2017) eaao0900. DOI:10.1126/sciadv.aao0900 |
| [59] |
X. Li, L. Wang, Y. Deng, et al., Chem. Commun. 54 (2018) 12459-12462. DOI:10.1039/C8CC07657J |
| [60] |
S. Cantekin, T.F.A. de Greef, A.R.A. Palmans, Chem. Soc. Rev. 41 (2012) 6125-6137. DOI:10.1039/c2cs35156k |
| [61] |
Y. Yao, R. Zhao, Y. Shi, et al., Chem. Commun. 54 (2018) 8068-8071. DOI:10.1039/C8CC04423F |
| [62] |
T. Ogoshi, T.A. Yamagishi, Y. Nakamoto, Chem. Rev. 116 (2016) 7937-8002. DOI:10.1021/acs.chemrev.5b00765 |
| [63] |
C. Li, Chem. Commun. 50 (2014) 12420-12433. DOI:10.1039/C4CC03170A |
| [64] |
K. Yang, Y. Pei, J. Wen, et al., Chem. Commun. 52 (2016) 9316-9326. DOI:10.1039/C6CC03641D |
| [65] |
N. Song, T. Kakuta, T.-a. Yamagishi, et al., Chem. 4 (2018) 2029-2053. DOI:10.1016/j.chempr.2018.05.015 |
| [66] |
N. Song, Y.W. Yang, Sci. China Chem. 57 (2014) 1185-1198. DOI:10.1007/s11426-014-5190-z |
| [67] |
Q. Lin, X.W. Guan, S.S. Song, et al., Polym. Chem. 10 (2019) 253-259. DOI:10.1039/C8PY01299G |
| [68] |
B. Li, B. Wang, X. Huang, et al., Angew. Chem. Int. Ed. 58 (2019) 3885-3889. DOI:10.1002/anie.201813972 |
| [69] |
Y.M. Zhang, Y.F. Li, H. Fang, et al., Soft Matter 14 (2018) 8529-8536. DOI:10.1039/C8SM01838C |
| [70] |
Y.M. Zhang, W. Zhu, W.J. Qu, et al., Chem. Commun. 54 (2018) 4549-4552. DOI:10.1039/C8CC00814K |
| [71] |
C.W. Zhang, B. Ou, S.T. Jiang, et al., Polym. Chem. 9 (2018) 2021-2030. DOI:10.1039/C8PY00226F |
| [72] |
H. Yao, J. Wang, S.S. Song, et al., New J. Chem. 42 (2018) 18059-18065. DOI:10.1039/C8NJ04160A |
| [73] |
T. Xiao, L. Wang, Tetrahedron Lett. 59 (2018) 1172-1182. DOI:10.1016/j.tetlet.2018.02.028 |
| [74] |
X.M. Jiang, X.J. Huang, S.S. Song, et al., Polym. Chem. 9 (2018) 4625-4630. DOI:10.1039/C8PY01085D |
| [75] |
Y.Q. Fan, J. Liu, Y.Y. Chen, et al., J. Mater. Chem. C 6 (2018) 13331-13335. |
| [76] |
J.D. Ding, J.F. Chen, Q. Lin, et al., Polym. Chem. 9 (2018) 5370-5376. DOI:10.1039/C8PY01319E |
| [77] |
Q. Lin, K.P. Zhong, J.H. Zhu, et al., Macromolecules 50 (2017) 7863-7871. DOI:10.1021/acs.macromol.7b01835 |
| [78] |
S. Wang, Z. Xu, T. Wang, et al., Nat. Commun. 9 (2018) 1737. DOI:10.1038/s41467-018-03827-3 |
| [79] |
S. Wang, Z. Xu, T. Wang, et al., J. Photochem. Photobiol. A 355 (2018) 60-66. DOI:10.1016/j.jphotochem.2017.11.004 |
| [80] |
X. Zhang, J. Xu, C. Lang, et al., Biomacromolecules 18 (2017) 1885-1892. DOI:10.1021/acs.biomac.7b00321 |
| [81] |
H. Ju, F. Zhu, H. Xing, et al., Macro. Rapid Commun. 38 (2017) 1700232. DOI:10.1002/marc.201700232 |
| [82] |
M. Ni, N. Zhang, W. Xia, et al., J. Am. Chem. Soc. 138 (2016) 6643-6649. DOI:10.1021/jacs.6b03296 |
| [83] |
K. Jie, Y. Zhou, B. Shi, et al., Chem. Commun. 51 (2015) 8461-8464. DOI:10.1039/C5CC00976F |
| [84] |
Y.X. Wang, Q.F. Zhou, L.J. Chen, et al., Chem. Commun. 54 (2018) 2224-2227. DOI:10.1039/C7CC08729B |
| [85] |
Y. Wang, Y. Tian, Y.Z. Chen, et al., Chem. Commun. 54 (2018) 7991-7994. DOI:10.1039/C8CC04542A |
| [86] |
P. Liu, Z. Li, B. Shi, et al., Chem. -Eur. J. 24 (2018) 4264-4267. DOI:10.1002/chem.201800312 |
| [87] |
T. Xiao, W. Zhong, L. Xu, et al., Org. Biomol. Chem. 17 (2019) 1336-1350. DOI:10.1039/C8OB03095B |
| [88] |
H. Zhu, L. Shangguan, B. Shi, et al., Mater. Chem. Front. 2 (2018) 2152-2174. DOI:10.1039/C8QM00314A |
| [89] |
Y. Zhou, K. Jie, F. Huang, Chem. Commun. 54 (2018) 12856-12859. DOI:10.1039/C8CC06406G |
| [90] |
Q. Wang, P. Zhang, J. Xu, et al., ACS Appl. Bio Mater. 1 (2018) 70-78. DOI:10.1021/acsabm.8b00014 |
| [91] |
Q. Wang, L. Tian, J. Xu, et al., Chem. Commun. 54 (2018) 10328-10331. DOI:10.1039/C8CC05560B |
| [92] |
S. Sun, M. Geng, L. Huang, et al., Chem. Commun. 54 (2018) 13006-13009. DOI:10.1039/C8CC07658H |
| [93] |
Y. Zhou, K. Jie, F. Huang, Org. Chem. Front. 4 (2017) 2387-2391. DOI:10.1039/C7QO00736A |
| [94] |
D. Xia, P. Wang, B. Shi, Org. Lett. 19 (2017) 202-205. DOI:10.1021/acs.orglett.6b03486 |
| [95] |
M.X. Wu, Y.W. Yang, Adv. Mater. 29 (2017) 1606134. |
| [96] |
S. Wang, C.H. Yao, M.F. Ni, et al., Polym. Chem. 8 (2017) 682-688. DOI:10.1039/C6PY01961G |
| [97] |
L. Shangguan, Q. Chen, B. Shi, et al., Chem. Commun. 53 (2017) 9749-9752. DOI:10.1039/C7CC05305C |
| [98] |
X. Liu, K. Jia, Y. Wang, et al., ACS Appl. Mater. Interfaces 9 (2017) 4843-4850. DOI:10.1021/acsami.7b00643 |
| [99] |
L. Jiang, X. Huang, D. Chen, et al., Angew. Chem. Int. Ed. 56 (2017) 2655-2659. DOI:10.1002/anie.201611973 |
| [100] |
S. Guo, T. Liang, Y. Song, et al., Polym. Chem. 8 (2017) 5718-5725. DOI:10.1039/C7PY01259D |
| [101] |
L. Gao, T. Wang, K. Jia, et al., Chem. Eur. J. 23 (2017) 6605-6614. DOI:10.1002/chem.201700345 |
| [102] |
G. Yu, W. Yu, L. Shao, et al., Adv. Funct. Mater. 26 (2016) 8999-9008. DOI:10.1002/adfm.201601770 |
| [103] |
K. Yang, Y. Chang, J. Wen, et al., Chem. Mater. 28 (2016) 1990-1993. DOI:10.1021/acs.chemmater.6b00696 |
| [104] |
D. Xia, L. Shangguan, M. Xue, et al., New J. Chem. 40 (2016) 9890-9894. DOI:10.1039/C6NJ02269C |
| [105] |
S. Guo, X. Liu, C. Yao, et al., Chem. Commun. 52 (2016) 10751-10754. DOI:10.1039/C6CC05993G |
| [106] |
X. Chi, G. Yu, L. Shao, et al., J. Am. Chem. Soc. 138 (2016) 3168-3174. DOI:10.1021/jacs.5b13173 |
| [107] |
Y. Chang, C. Hou, J. Ren, et al., Chem. Commun. 52 (2016) 9578-9581. DOI:10.1039/C6CC03637F |
| [108] |
X. Wu, Y. Li, C. Lin, et al., Chem. Commun. 51 (2015) 6832-6835. DOI:10.1039/C5CC01393C |
| [109] |
L.B. Meng, W. Zhang, D. Li, et al., Chem. Commun. 51 (2015) 14381-14384. DOI:10.1039/C5CC05785J |
| [110] |
X.Y. Hu, K. Jia, Y. Cao, et al., Chem. -Eur. J. 21 (2015) 1208-1220. DOI:10.1002/chem.201405095 |
| [111] |
X. Chi, G. Yu, X. Ji, et al., ACS Macro Lett. 4 (2015) 996-999. DOI:10.1021/acsmacrolett.5b00525 |
| [112] |
X. Chi, X. Ji, D. Xia, et al., J. Am. Chem. Soc. 137 (2015) 1440-1443. DOI:10.1021/ja512978n |
| [113] |
D. Xia, G. Yu, J. Li, et al., Chem. Commun. 50 (2014) 3606-3608. DOI:10.1039/c3cc49686d |
| [114] |
M. Zuo, W. Qian, T. Li, et al., ACS Appl. Mater. Interfaces 10 (2018) 39214-39221. DOI:10.1021/acsami.8b14110 |
| [115] |
X. Wang, J. Yang, X. Sun, et al., Chem. Commun. 54 (2018) 12990-12993. DOI:10.1039/C8CC08168A |
| [116] |
N. Song, X.Y. Lou, W. Hou, et al., Macro. Rapid Commun. 39 (2018) 1800593. DOI:10.1002/marc.201800593 |
| [117] |
X. Li, Z. Li, Y.W. Yang, Adv. Mater. 30 (2018) 1800177. DOI:10.1002/adma.201800177 |
| [118] |
S. Guo, Y. Song, Y. He, et al., Angew. Chem. Int. Ed. 57 (2018) 3163-3167. DOI:10.1002/anie.201800175 |
| [119] |
J.F. Chen, X.B. Cheng, H. Li, et al., New J. Chem. 41 (2017) 2148-2153. DOI:10.1039/C6NJ03380F |
| [120] |
J.F. Chen, Q. Lin, Y.M. Zhang, et al., Chem. Commun. 53 (2017) 13296-13311. DOI:10.1039/C7CC08365C |
| [121] |
E. Li, Y. Zhou, R. Zhao, et al., Angew. Chem. Int. Ed. 58 (2019) 3981-3985. DOI:10.1002/anie.201900140 |
| [122] |
E. Li, K. Jie, Y. Zhou, et al., J. Am. Chem. Soc. 140 (2018) 15070-15079. DOI:10.1021/jacs.8b10192 |
| [123] |
K. Jie, Y. Zhou, E. Li, et al., J. Am. Chem. Soc. 140 (2018) 3190-3193. DOI:10.1021/jacs.7b13156 |
| [124] |
K. Jie, Y. Zhou, E. Li, et al., Angew. Chem. Int. Ed. 57 (2018) 12845-12849. DOI:10.1002/anie.201808998 |
| [125] |
K. Jie, Y. Zhou, E. Li, et al., Acc. Chem. Res. 51 (2018) 2064-2072. DOI:10.1021/acs.accounts.8b00255 |
| [126] |
K. Jie, M. Liu, Y. Zhou, et al., J. Am. Chem. Soc. 140 (2018) 6921-6930. DOI:10.1021/jacs.8b02621 |
| [127] |
B. Hua, W. Zhou, Z. Yang, et al., J. Am. Chem. Soc. 140 (2018) 15651-15654. DOI:10.1021/jacs.8b11156 |
| [128] |
L.L. Tan, Y. Zhu, H. Long, et al., Chem. Commun. 53 (2017) 6409-6412. DOI:10.1039/C7CC03638H |
| [129] |
K. Jie, Y. Zhou, E. Li, et al., J. Am. Chem. Soc. 139 (2017) 15320-15323. DOI:10.1021/jacs.7b09850 |
| [130] |
K. Jie, M. Liu, Y. Zhou, et al., J. Am. Chem. Soc. 139 (2017) 2908-2911. DOI:10.1021/jacs.6b13300 |
| [131] |
T. Xiao, L. Zhou, L. Xu, et al., Chin. Chem. Lett. 30 (2019) 271-276. DOI:10.1016/j.cclet.2018.05.039 |
| [132] |
M. Zuo, W. Qian, Z. Xu, et al., Small 14 (2018) 1801942. DOI:10.1002/smll.201801942 |
| [133] |
T. Xiao, L. Xu, W. Zhong, et al., Isr. J. Chem. 58 (2018) 1183-1193. DOI:10.1002/ijch.201800026 |
| [134] |
J.R. Wu, C.Y. Wang, Y.C. Tao, et al., Eur. J. Org. Chem. 2018 (2018) 1321-1325. DOI:10.1002/ejoc.201800112 |
| [135] |
J.R. Wu, A.U. Mu, B. Li, et al., Angew. Chem. Int. Ed. 57 (2018) 9853-9858. DOI:10.1002/anie.201805980 |
| [136] |
Y. Sun, J. Wang, Y. Yao, Chem. Commun. 53 (2017) 165-167. DOI:10.1039/C6CC08452D |
| [137] |
G. Ping, Y. Wang, L. Shen, et al., Chem. Commun. 53 (2017) 7381-7384. DOI:10.1039/C7CC02799K |
| [138] |
B. Jiang, W. Wang, Y. Zhang, et al., Angew. Chem. Int. Ed. 56 (2017) 14438-14442. DOI:10.1002/anie.201707209 |
| [139] |
D. Dai, Z. Li, J. Yang, et al., J. Am. Chem. Soc. 141 (2019) 4756-4763. DOI:10.1021/jacs.9b01546 |
| [140] |
Y. Chen, S. Sun, D. Lu, et al., Chin. Chem. Lett. 30 (2019) 37-43. DOI:10.1016/j.cclet.2018.10.022 |
| [141] |
X. Zeng, H. Deng, X. Jia, et al., Chem. Commun. 54 (2018) 11634-11637. DOI:10.1039/C8CC07188H |
| [142] |
D.Y. Xia, L.Y. Wang, X.Q. Lv, et al., Macromolecules 51 (2018) 2716-2722. DOI:10.1021/acs.macromol.8b00354 |
| [143] |
C.L. Sun, H.Q. Peng, L.Y. Niu, et al., Chem. Commun. 54 (2018) 1117-1120. DOI:10.1039/C7CC09315B |
| [144] |
L. Shao, J. Yang, B. Hua, Polym. Chem. 9 (2018) 1293-1297. DOI:10.1039/C8PY00057C |
| [145] |
L. Shao, J. Sun, B. Hua, et al., Chem. Commun. 54 (2018) 4866-4869. DOI:10.1039/C8CC02077A |
| [146] |
H. Zhu, B. Shi, L. Gao, et al., Polym. Chem. 8 (2017) 7108-7112. DOI:10.1039/C7PY01669G |
| [147] |
B.B. Shi, H.X. Guan, L.Q. Shangguan, et al., J. Mater. Chem. A 5 (2017) 24217-24222. DOI:10.1039/C7TA08894A |
| [148] |
L. Shangguan, H. Xing, J.H. Mondal, et al., Chem. Commun. 53 (2017) 889-892. DOI:10.1039/C6CC08336F |
| [149] |
Y. Guan, M. Ni, X. Hu, et al., Chem. Commun. 48 (2012) 8529-8531. DOI:10.1039/c2cc33943a |
| [150] |
X.Y. Hu, P. Zhang, X. Wu, et al., Polym. Chem. 3 (2012) 3060-3063. DOI:10.1039/c2py20285a |
| [151] |
X.Y. Hu, X. Wu, S. Wang, et al., Polym. Chem. 4 (2013) 4292-4297. DOI:10.1039/c3py00575e |
| [152] |
X.Y. Hu, X. Wu, Q. Duan, et al., Org. Lett. 14 (2012) 4826-4829. DOI:10.1021/ol302149t |
| [153] |
Y.Q. Jiang, K. Wu, Q. Zhang, et al., Chem. Commun. 54 (2018) 13821-13824. DOI:10.1039/C8CC08226J |
| [154] |
J. Wu, W.Y. Fang, Lu, et al., J. Org. Chem. 72 (2007) 2897-2905. DOI:10.1021/jo062523g |
| [155] |
J. Chen, L. Ao, C. Wei, et al., Chem. Commun. 55 (2018) 229-232. |
| [156] |
Y. Han, Y. Tian, Z. Li, et al., Chem. Soc. Rev. 47 (2018) 5165-5176. DOI:10.1039/C7CS00802C |
| [157] |
Z. Gao, Y. Han, Z. Gao, et al., Acc. Chem. Res. 51 (2018) 2719-2729. DOI:10.1021/acs.accounts.8b00340 |
| [158] |
X. Wang, Y. Han, Y. Liu, et al., Angew. Chem. Int. Ed. 56 (2017) 12466-12470. DOI:10.1002/anie.201704294 |
| [159] |
Z. Gao, Y. Han, S. Chen, et al., ACS Macro Lett. 6 (2017) 541-545. DOI:10.1021/acsmacrolett.7b00241 |
| [160] |
T.F. Fu, Z.J. Li, Z.X. Zhang, et al., Macromolecules 50 (2017) 7517-7525. DOI:10.1021/acs.macromol.7b01149 |
| [161] |
Y. Shi, Z. Yang, H. Liu, et al., ACS Macro Lett. 4 (2015) 6-10. DOI:10.1021/mz500659f |
| [162] |
Z.X. Zhou, X.Z. Yan, T.R. Cook, et al., J. Am. Chem. Soc. 138 (2016) 806-809. DOI:10.1021/jacs.5b12986 |
| [163] |
T. Xiao, L. Xu, J. Wang, et al., Org. Chem. Front. 6 (2019) 936-941. DOI:10.1039/C9QO00089E |
| [164] |
T. Xiao, X. Feng, S. Ye, et al., Macromolecules 45 (2012) 9585-9594. DOI:10.1021/ma302459n |
| [165] |
Q. Wang, M. Cheng, J.L. Jiang, et al., Chin. Chem. Lett. 28 (2017) 793-797. DOI:10.1016/j.cclet.2017.02.008 |
| [166] |
Q. Wang, P. Zhang, Y. Li, et al., RSC Adv. 7 (2017) 29364-29367. DOI:10.1039/C7RA05351G |
| [167] |
Q. Wang, M. Cheng, L. Tian, et al., Polym. Chem. 8 (2017) 6058-6063. DOI:10.1039/C7PY01096F |
 2020, Vol. 31
2020, Vol. 31 


