b The State Key Laboratory of Refractories and Metallurgy, School of Chemistry and Chemical Engineering, Wuhan University of Science and Technology, Wuhan, China;
c State Key Laboratory of Supramolecular Structure and Materials, Institute of Theoretical Chemistry, Jilin University, Changchun, China;
d California NanoSystems Institute and Department of Chemistry, Biochemistry, University of California, Los Angeles, CA, United States
Supramolecular polymers constructed by hydrogen bonding [1, 2], host–guest complexation [3, 4], hydrophobic interaction [5], and metal– ligand coordination [6, 7] have gained wide attention in the past decade due to their excellent properties, in particular stimuli-responsiveness and self-healing capability [8-10]. Orthogonal self-assembly as a promising strategy to construct smart supramolecular polymers attracts much interest because it can integrate multiple noncovalent inter-actions in one system, i.e., host–guest inclusion complexation and metal–ligand coordination, and endow the resulting materials with unique properties and great potentials [11-13].
Synthetic cyclic oligomers with arenes as basic building blocks, namely macrocyclic arenes [14-22], as a promising type of supramolecular macrocycles, have advanced rapidly during the past several decades and played a major role in the construction of functional supramolecular polymers [23-28]. By inducing desym-metrization to traditional pillar[6]arenes, we discovered a new class of macrocyclic arenes named leaning tower[6]arenes (LT6 for short), which adopt a much favorable leaning tower conformation instead of traditional pillar structure, thus rendering them remarkable performance in supramolecular self-assembly and host-guest chemistry [29, 30]. Further selective modification of LT6 to extend the applications has yet to be explored. Here we report the selective mono-functionalization on one rim of LT6 for application in the construction of functional supramolecular polymer network (SPN) with tunable luminescent property and multi-stimuli responsiveness, which will expand the library of functional building blocks for macrocyclic arenes-based supramo-lecular gel and may provide special functions different from traditional pillararenes [12, 24, 25].
On the other hand, fluorescent supramolecular polymers constructed from a special AIEgen tetraphenylethene (TPE) [31, 32] and its derivatives usually possess attractive aggregation-induced emission (AIE) properties [33-37]. Tunable host-guest interactions between synthetic macrocycles and TPE-based guest molecules will endow the supramolecular system with desirable performance. Meanwhile, terpyridine becomes a versa-tile functional motif due to its good binding affinity to various metal ions. Therefore, we come up with an idea of constructing a multi-responsive fluorescent SPN via orthogonal self-assembly, taking advantages of both the tunable macrocyclic host-guest interactions and the terpyridine-based metal–ligand coordination.
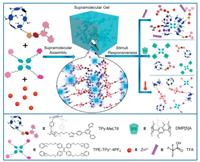
Herein, we synthesized a new monofunctionalized LT6 bearing one terpyridine arm, i.e., TPy-MeLT6, for further construction of fluorescent SPN upon host–guest complexation with a TPE tetratopic guest possessing four pyridinium arms, i.e., TPE-TPy+·4PF6-, and metal–ligand coordination of its terpyridine arm with Zn2+ ions (Scheme 1).
|
Download:
|
| Scheme 1. Graphical representation of the construction of SPN from TPy-MeLT6, TPE-TPy+·4PF6-, and Zn(OTf)2 through supramolecular self-assembly and its stimuli-responsiveness triggered by trifluoroacetic acid (TFA), dimethoxypillar[5]arene (DMP[5]A) and thermal heating. | |
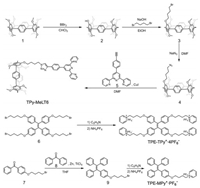
TPy-MeLT6 was synthesized by a facile four-step synthetic strategy involving fragmentation reaction, substitution reaction, and copper(I)-catalyzed azide–alkyne cycloaddition (CuAAC) reaction (Scheme 2). Mono-hydroxylated LT6 (2) as a basic starting reagent was facilely obtained by the reaction of MeLT6 (1) with a proper ratio of BBr3 in CHCl3. Compound 2 was converted to a hydrazoate derivative (4) via a two-step sequence of nucleophilic substitution by 1, 4-dibromobutane and azide. Finally, the inter-molecular CuAAC reaction between 5 and 4 gave the target product (TPy-MeLT6) in a yield of 65.9%. The syntheses of all the LT6 derivatives (see the Supporting information for details) are coherent, straightforward, and convenient, proving the good controllability and diversification of substituent groups in the modification of the synthetic macrocyclic arenes.
|
Download:
|
| Scheme 2. Synthetic routes to TPy-MeLT6, TPE-TPy+·4PF6- and TPE-MPy+·PF6-. | |
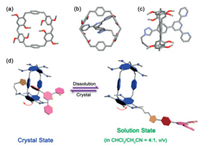
Single crystal of TPy-MeLT6 suitable for X-ray diffraction was grown by slow diffusion of CH3OH into its acetic ether solution. Crystal structural analyses revealed a conformation of lariat complex, where the substituted phenylene units of TPy-MeLT6 possess a nearly vertical dihedral angle with the plane of its six methylene bridges and the two unsubstituted phenylene units. Compared with the full tilt rings in MeLT6 (Fig. 1a) and other LT6 derivatives [30], this prop-opened structure ensures the self-inclusion of the side chain within the LT6 cavity.
|
Download:
|
| Fig. 1. (a) Top view of single crystal structure of MeLT6. (b) Top view and (c) side view of single crystal structure of TPy-MeLT6. (d) Schematic representation of different confirmations of TPy-MeLT6 in the crystalloid state and in solution. Note: in the single crystal structures, C gray, O red, N blue. | |
To confirm the conformation of TPy-MeLT6 in solution, nuclear Overhauser effect spectroscopy (NOESY) was measured (Fig. S25 in Supporting information), where no obvious cross-peaks between the protons of side chain (H1, H2, H5, H6, H7) and the parent macrocyclic ring was observed, suggesting that the side chain does not easily occupy the cavity of MeLT6 in solution and enabling its application in the construction of supramolecular polymer with the aid of host-guest complexation. Therefore, we conclude that two conformations can be captured in one TPy-MeLT6 compound, i.e., a self-inclusion state in the crystalloid state and an arm-stretching state in solution (Figs. 1b-d). To understand the conformational extensibility of TPy-MeLT6, we carried out variable temperature NMR (VT-NMR, from -50 ℃ to +40 ℃) experiments in CDCl3 and CDCl3/CD3CN (4:1, v/v). As in Fig. S26 (Supporting information), proton signals (Ha and Hb) of TPy-MeLT6 were broadened at above 20 ℃ (in CDCl3) and 30 ℃ (in CDCl3/CD3CN), respectively, and were all split at below 0 ℃ consistently, suggesting the slow conformation interconversion between the self-inclusion and arm-stretching states. Such conformational flexibility is the key property desired for the construction of SPN in solution.
Meanwhile, two new TPE-based cationic guests, i.e., TPE-TPy+·4PF6- and TPE-MPy+·PF6-, were synthesized by substitution reaction in conjunction with counterions exchange for the subsequent self-assembly and function studies. As in Scheme 1, TPy-MeLT6, TPE-TPy+·4PF6-, and Zn(OTf)2 formed a cross-linked SPN through the cooperation of the 1:2 metal–ligand coordination [38, 39] of Zn2+ and the terpyridine entities of TPy-MeLT6 and the 4:1 host–guest inclusion complexation between TPy-MeLT6 and the four pyridine arms of TPE-TPy+·4PF6- in CHCl3/CH3CN (4:1, v/v).
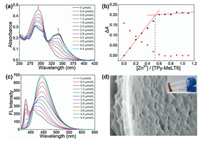
To study the coordination effect of TPy-MeLT6 toward Zn2+, UV–vis absorption spectroscopy of TPy-MeLT6 in CHCl3/CH3CN (4:1, v/v) was measured upon titration with different concentrations of Zn(OTf)2 (Fig. 2a). With an increasing concentration of Zn2+, the UV–vis absorption peak of TPy-MeLT6 at 293 nm (attributed to π-π* transition of terpyridine groups) was gradually decreased in accompany with a continuous blue shift. Accordingly, a new absorption peak at 327 nm appeared as a result of the coordination of TPy-MeLT6 and Zn2+ [40]. In addition, the binding stoichiometry between TPy-MeLT6 and Zn2+ was determined to be 2:1 by a plot of absorption changes versus Zn2+ concentration (Fig. 2b). According to fluorescence emission experiments (Fig. 2c), characteristic emission peak of TPy-MeLT6 at 385 nm disappeared, accompanied by the appearance of a new peak at 427 nm, whose intensity was increased with an increasing amount of Zn2+. This further indicated the effective coordination of TPy-MeLT6 and Zn2+. The fluorescence enhancement can be attributed to two factors: (a) Zn2+ possesses d10 electron configuration that is beneficial for the emission enhancement attributed to the intra-ligand charge transfer processes [41]; (b) the emission intensity of the complexes is obviously larger than that of free linker, further suggesting that the enhancement of complex rigidity upon Zn2+ coordination contrib-utes greatly to the fluorescence enhancement [42]. Compared with the coordinated complex of the corresponding monomer (TPy-Mono) and Zn2+ (Fig. S27 in Supporting information), the coordination complex of TPy-MeLT6 and Zn2+ exhibited stronger fluorescence emission because the high electron density of TPy-MeLT6 is favorable for charge transfer.
|
Download:
|
| Fig. 2. (a) UV–vis spectra (CHCl3/CH3CN = 4:1, v/v, 298 K) of TPy-MeLT6 (4.0 μmol/L) upon titration with Zn2+ (0–4.4 μmol/L). (b) Absorption changes at 293 nm (red-circle) and 327 nm (black-square). (c) Emission spectra (CHCl3/CH3CN =4:1, v/ v, 298 K, λex = 349 nm, slit widths: ex. 5 nm, em. 5 nm) of TPy-MeLT6 (4.0 μmol/L) upon titration with Zn2+ (0–4.4 μmol/L); (d) SEM image and photograph of the supramolecular polymer gel. | |
As aforementioned, terpyridine group decorated on MeLT6 via flexible alkyl chain is at a state of swing in solution, facilitating its application in the construction of SPN through host-guest and coordination interactions. TPy-MeLT6 and TPE-MPy+·PF6- were used as model molecules to study host-guest interactions. Upon addition of TPy-MeLT6 (Fig. S28a in Supporting information). In addition, the changes in the UV–vis absorption spectra and emission spectra (Fig. S29 in Supporting information) after adding TPE-TPy+·4PF6- into TPy-MeLT6 further confirmed the existence of host-guest interactions. As calculated from NMR titrations (Fig. S28b in Supporting information), the binding constant (Ka) between TPy-MeLT6 and TPE-MPy+·PF6- was 19.5 L/mol (stoichiometry is 1:1). This relatively weak host-guest interaction was caused by the polar solvent of acetonitrile [43, 44].
On the basis of the above investigations, we fabricated the fluorescent SPN via orthogonal self-assembly by combining host molecule TPy-MeLT6, guest molecule TPE-TPy+·4PF6-, and Zn2+ in one system, taking advantage of both host–guest interactions and metal-ligand coordination. Briefly, TPy-MeLT6 (100 μmol/L), Zn(OTf)2 (50 μmol/L), and TPE-TPy+·4PF6- (25 μmol/L) were dissolved in CHCl3/CH3CN (200 mL, 4:1, v/v, 298 K). Then, after shaking, the above solution gradually changed from pale yellow to purple. Finally, supramolecular polymer gel was obtained after the system was standing for 24 h. SEM image and digital photograph of the material (Fig. 2d) also support the morphology in the gel state, that is, a typical glutinous surface morphology. Compared with TPy-MeLT6, TPy-MeLT6@Zn2+, and TPE-TPy+·4PF6-⊂TPy-MeLT6, an apparently small weight-average diffusion coefficient (D = 2.67×10-10 m2/s) of TPE-TPy+·4PF6-⊂TPy-MeLT6@Zn2+ in DOSY experiments strongly supported the formation and practi-cability of supramolecular polymeric aggregates (Fig. 3).

|
Download:
|
| Fig. 3. DOSY spectra (600 MHz, DMSO-d6, 298 K) of (a) 10 μmol/L TPy-MeLT6, (b) 10 μmol/L TPy-MeLT6@Zn2+, (c) 10 μmol/L TPE-TPy+·4PF6-⊂TPy-MeLT6, and (d) 10 μmol/L TPE-TPy+·4PF6-⊂TPy-MeLT6@Zn2+. | |
The unique properties of host–guest interaction of TPy-MeLT6 and TPE-TPy+·4PF6- together with metal–ligand coordination of Zn2+ and terpyridine group provided a great possibility for disassembly of SPN in response to acid, competitive agent, and thermal heating. Based on the property that terpyridine groupt cannot bind with metal ions in acidic solution [45], we added trifluoroacetic acid (TFA) into the solution of SPN to investigate whether TFA could dissociate the SPN. The UV–vis spectroscopy results showed that absorption peak of TPy-MeLT6 Zn2+ at 292 nm was increased in intensity in accompany with a red shift upon addition of TFA, while the absorbance at 327 nm was decreased and the shape of the absorption peak turned gradually to resemble that of TPy-MeLT6, proving that the SPN was dissociated in response to TFA (Fig. 4a). In Fig. 4b, upon addition of TFA into the solution of SPN, the fluorescence intensity was decreased evidently and showed negligible change after adding 240 μmol/L TFA ([TPy-MeLT6] = 4μmol/L) because of the entire protonation of terpyr-idine group. Significantly, to further prove the TFA-responsive decomposition of SPN, we exposed the supramolecular polymer gel to TFA vapor and the gel gradually turned into solution, indicating that the cross-linked structure of gel was destroyed in the presence of TFA (Fig. 4d).
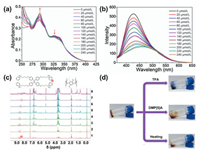
|
Download:
|
| Fig. 4. (a) UV–vis spectra of the SPN (1.0 μmol/L) upon addition of TFA (0–240 μmol/L, CHCl3/CH3CN = 4:1, v/v, 298 K); (b) Emission spectra (λex = 349 nm, slit widths: ex. 5 nm, em. 5 nm) of the SPN (1.0 μmol/L) upon addition of TFA (0–240 μmol/L, CHCl3/CH3CN = 4:1, v/v, 298 K); (c) 1H NMR spectra of the SPN (1.0 μmol/L) upon addition of DMP[5]A (0–16.0 μmol/L, 300 MHz, CDCl3/CD3CN = 4:1, v/v, 298 K); (d) Images of gel–sol transitions triggered by different stimuli (temperature, pH, and competitive reagent). | |
Then, the gel-sol transition in response to certain competitive agents was further studied by adding host competitive agent into the supramolecular polymer. There is no doubt that the weakened interactions between TPy-MeLT6 and TPE-TPy+·4PF6- is beneficial for competitive responsiveness, that is to say, when a supramo-lecular macrocycle with stronger binding capacity toward TPE-TPy+·4PF6- is added, the structure of polymer will be destroyed to achieve stimulus-responsiveness. To prove this concept, the construction mechanism of the as-synthesized SPN and its smart responsiveness were investigated in detail. DMP[5]A possesses better solubility in polar solvent (CDCl3:CD3CN, v/v = 4:1) with a higher binding constant (Ka =43.9 L/mol) toward TPE-MPy+·PF6- as compared with TPy-MeLT6 (Ka =19.5 L/mol), thus DMP[5]A would be a suitable competitive agent for the gel-sol transition (Figs. S28c and d in Supporting information). Firstly, the titration experiments of DMP[5]A and SPN were carried out by 1H NMR spectroscopy. Upon addition of DMP[5]A, the peak of pyridine salt motif at 8.64 ppm was gradually upshifted because the cavity of DMP[5]A interacted with the pyridine salt motif while the cavity of TPy-MeLT6 existed (Fig. 4c). More intuitively, when DMP[5]A was added into the supramolecular polymer gel, the original gel was transformed into solution and it could also be transformed into solution in response to an elevated temperature (Fig. 4d). Based on the above experimental results, the as-prepared SPN demonstrated excellent multi-stimuli responsiveness and fluorescence-respon-sive properties, showing great potential in fluorescence sensing.
In summary, we fabricated a multi-responsive fluorescent SPN from the newly synthesized terpyridine-monofunctionalized LT6 (TPy-MeLT6) host, TPE-based tetratopic guest linker, and Zn2+ ion through orthogonal self-assembly of host–guest interaction and metal–ligand coordination. The as-synthesized cross-linked SPN could be depolymerized by acid addition, competitive host, and thermal heating. This work not only presented a new function-alization method of synthetic macrocycles but also illustrated the synergistic construction of multi-stimuli responsive functional soft materials, providing a promising strategy for the development of fluorescent SPN materials.
Declaration of competing interestThe authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
AcknowledgmentWe thank the National Natural Science Foundation of China (No. 21871108) for financial support.
Appendix A. Supplementary dataSupplementary material related to this article can be found, in the online version, at doi:https://doi.org/10.1016/j.cclet.2019.10.023.
| [1] |
Q. Wang, M. Cheng, J.L. Jiang, L.Y. Wang, Chin. Chem. Lett. 28 (2017) 793-797. DOI:10.1016/j.cclet.2017.02.008 |
| [2] |
W.H. Wang, Q. Gao, A.L. Li, et al., Chin. Chem. Lett. 29 (2018) 336-338. DOI:10.1016/j.cclet.2017.08.033 |
| [3] |
M.M. Zhang, D.H. Xu, X.Z. Yan, et al., Angew. Chem. Int. Ed. 51 (2012) 7011-7015. DOI:10.1002/anie.201203063 |
| [4] |
X. Zheng, Q. Miao, W. Wang, D.H. Qu, Chin. Chem. Lett. 29 (2018) 1621-1624. DOI:10.1016/j.cclet.2018.04.002 |
| [5] |
C. Zhou, M.A. Hillmyer, T.P. Lodge, J. Am. Chem. Soc. 134 (2012) 10365-10368. DOI:10.1021/ja303841f |
| [6] |
C. Xu, L. Xu, X. Ma, Chin. Chem. Lett. 29 (2018) 970-972. DOI:10.1016/j.cclet.2017.11.045 |
| [7] |
G.Q. Yin, H. Wang, X.Q. Wang, et al., Nat. Commun. 9 (2018) 567. DOI:10.1038/s41467-018-02959-w |
| [8] |
T. Aida, E.W. Meijer, S.I. Stupp, Science 335 (2012) 813-817. DOI:10.1126/science.1205962 |
| [9] |
T.F.A. de Greef, E.W. Meijer, Nature 453 (2008) 171-173. DOI:10.1038/453171a |
| [10] |
L.L. Yang, X.X. Tan, Z.Q. Wang, X. Zhang, Chem. Rev. 115 (2015) 7196-7239. DOI:10.1021/cr500633b |
| [11] |
P.F. Wei, X.Z. Yan, F.H. Huang, Chem. Soc. Rev. 44 (2015) 815-832. DOI:10.1039/C4CS00327F |
| [12] |
X.M. Qian, W.T. Gong, X.P. Li, et al., Chem. -Eur. J. 22 (2016) 6881-6890. DOI:10.1002/chem.201600561 |
| [13] |
Y. Zhou, H.Y. Zhang, Z.Y. Zhang, Y. Liu, J. Am. Chem. Soc. 139 (2017) 7168-7171. DOI:10.1021/jacs.7b03153 |
| [14] |
J.R. Wu, Y.W. Yang, Chem. Commun. 55 (2019) 1533-1543. DOI:10.1039/C8CC09374A |
| [15] |
M. Xue, Y. Yang, X.D. Chi, Z.B. Zhang, F.H. Huang, Acc. Chem. Res. 45 (2012) 1294-1308. DOI:10.1021/ar2003418 |
| [16] |
Y.L. Wang, K.D. Xu, B. Li, et al., Angew. Chem. Int. Ed. 58 (2019) 10281-10284. DOI:10.1002/anie.201905563 |
| [17] |
B. Gao, L.L. Tan, N. Song, K. Li, Y.W. Yang, Chem. Commun. 52 (2016) 5804-5807. DOI:10.1039/C6CC01892K |
| [18] |
T. Ogoshi, T.A. Yamagishi, Y. Nakamoto, Chem. Rev. 116 (2016) 7937-8002. DOI:10.1021/acs.chemrev.5b00765 |
| [19] |
P. Li, Y. Chen, Y. Liu, Chin. Chem. Lett. 30 (2019) 1190-1197. DOI:10.1016/j.cclet.2019.03.035 |
| [20] |
Z. Zhang, K. Sun, S. Li, G. Yu, Chin. Chem. Lett. 30 (2019) 957-960. DOI:10.1016/j.cclet.2019.01.018 |
| [21] |
J.C. Gui, Z.Q. Yan, Y. Peng, et al., Chin. Chem. Lett. 27 (2016) 1017-1021. DOI:10.1016/j.cclet.2016.04.021 |
| [22] |
Q.H. Guo, L. Zhao, M.X. Wang, Angew. Chem. Int. Ed. 54 (2015) 8386-8389. DOI:10.1002/anie.201503179 |
| [23] |
X. Yan, F. Wang, B. Zheng, F. Huang, Chem. Soc. Rev. 41 (2012) 6042-6065. DOI:10.1039/c2cs35091b |
| [24] |
M.F. Ni, N. Zhang, W. Xia, et al., J. Am. Chem. Soc. 138 (2016) 6643-6649. DOI:10.1021/jacs.6b03296 |
| [25] |
T.X. Xiao, L.Y. Wang, Tetrahedron Lett. 59 (2018) 1172-1182. DOI:10.1016/j.tetlet.2018.02.028 |
| [26] |
H.C. Zhang, Z.N. Liu, Y.L. Zhao, Chem. Soc. Rev. 47 (2018) 5491-5528. DOI:10.1039/C8CS00037A |
| [27] |
X.H. Wang, N. Song, W. Hou, et al., Adv. Mater. 31 (2019) 190362. |
| [28] |
D.H. Dai, Z. Li, J. Yang, et al., J. Am. Chem. Soc. 141 (2019) 4756-4763. DOI:10.1021/jacs.9b01546 |
| [29] |
D.H. Dai, J.R. Wu, Y.W. Yang, Emerging macrocyclic arenes related to calixarenes and pillararenes, in:Y. Liu, Y. Chen, H.Y. Zhang (Eds.), Handbook of Macrocyclic Supramolecular Assembly, Springer, Singapore, 2019, pp.14-16.
|
| [30] |
J.R. Wu, A.U. Mu, B. Li, et al., Angew. Chem. Int. Ed. 57 (2018) 9853-9858. DOI:10.1002/anie.201805980 |
| [31] |
J. Mei, Y.N. Hong, J.W.Y. Lam, et al., Adv. Mater. 26 (2014) 5429-5479. DOI:10.1002/adma.201401356 |
| [32] |
Y.C. Chen, J.W.Y. Lam, R.T.K. Kwok, B. Liu, B.Z. Tang, Mater. Horiz. 6 (2019) 428-433. DOI:10.1039/C8MH01331D |
| [33] |
N. Song, D.X. Chen, Y.C. Qiu, et al., Chem. Commun. 50 (2014) 8231-8234. DOI:10.1039/c4cc03105a |
| [34] |
Y. Sun, J. Wang, Y. Yao, Chem. Commun. 53 (2017) 165-167. DOI:10.1039/C6CC08452D |
| [35] |
Q. Zhao, Y. Chen, Y. Liu, Chin. Chem. Lett. 29 (2018) 84-86. DOI:10.1016/j.cclet.2017.07.024 |
| [36] |
X.Y. Lou, Y.W. Yang, Adv. Opt. Mater. 6 (2018) 1800688. |
| [37] |
X. Li, Z. Li, Y.W. Yang, Adv. Mater. 30 (2018) 1800177. DOI:10.1002/adma.201800177 |
| [38] |
A. Fermi, G. Bergamini, M. Roy, M. Gingras, P. Ceroni, J. Am. Chem. Soc. 136 (2014) 6395-6400. DOI:10.1021/ja501458s |
| [39] |
Z.K. Zheng, L. Opilik, F. Schiffmann, et al., J. Am. Chem. Soc. 136 (2014) 6103-6110. DOI:10.1021/ja501849y |
| [40] |
A. Anthonysamy, S. Balasubramanian, V. Shanmugaiah, N. Mathivanan, Dalton Trans. (2008) 2136-2143. |
| [41] |
T. Tsukamoto, K. Takada, R. Sakamoto, et al., J. Am. Chem. Soc. 139 (2017) 5359-5366. DOI:10.1021/jacs.6b12810 |
| [42] |
Y.J. Cui, Y.F. Yue, G.D. Qian, B.L. Chen, Chem. Rev. 112 (2012) 1126-1162. DOI:10.1021/cr200101d |
| [43] |
P. Rios, T.S. Carter, T.J. Mooibroek, et al., Angew. Chem. Int. Ed. 55 (2016) 3387-3392. DOI:10.1002/anie.201510611 |
| [44] |
P. Rios, T.J. Mooibroek, T.S. Carter, et al., Chem. Sci. 8 (2017) 4056-4061. DOI:10.1039/C6SC05399H |
| [45] |
P.K. Chen, Q.C. Li, S. Grindy, N. Holten-Andersen, J. Am. Chem. Soc. 137 (2015) 11590-11593. DOI:10.1021/jacs.5b07394 |
 2019, Vol. 30
2019, Vol. 30 

