b School of Chemical Sciences, University of Chinese Academy of Sciences, Beijing 100049, China;
c Department of Chemical Engineering, Beijing Institute of Petro-chemical Technology, Beijing 102617, China;
d School of Pharmacy, Minzu University of China, Beijing 100081, China
Raltitrexed (RTX) is a specific quinazoline folate analog and can selectively inhibit thymidylate synthase [1]. It usually acts as a substitute of fluorouracil for radiotherapy in treatment of colonic and rectal cancer [2]. However, the bioavailability of RTX appeared to be low after oral dosing [3]. As a result, patients could mainly receive RTX administered intravenously undergoing in clinics [4]. Nevertheless, injection of excessive anticancer drugs RTX would not only lead to rapid and extensive metabolism, but also result in side effects, experiencing diarrhea or vomit. Therefore, it is very important and urgently needed to establish a method for detection of RTX in body fluids with high sensitivity and selectivity.
The common analytical technique on qualitative analysis of RTX [5] is high performance liquid chromatography [6, 7], radioimmunoassay [8] and fluorometry [2]. However, some of the methods are time consuming and some of the assays need organic solvents [6]. As the rapid growth of the drugs research field, fluorescence methods based on green gold nanoclusters (AuNCs) probes [9] have attracted great interests due to its high sensitivity and selectivity. Especially, comparing with organic dyes and quantum dots based probes, the advantages of fluorescent AuNCs possessing high quantum yield, good biocompatibility, facile synthesis and low toxicity make them to be excellent probes for construction of chemical/biological assay and for sensing drugs.
Various small molecules and biomacromolecules are commonly utilized as the stabilizers and reductants in the preparationof AuNCs due to the strong interaction between Au atom and amino groups, carboxyl groups and sulfhydryl groups[10].Forexample, glutathione reduced AuNCs have been made for monitoring urine mitoxantrone [9]. Meanwhile, DNA [11], bovine serum albumin [12], cysteine [13], poly(adenine) [14], L-arginine [15], dopamine binding aptamer [16] and peptide [17], also have been explored as the cappers to synthesize the fluorescent AuNCs for detection of drugs. Although the previous works have demonstrated that numerous small molecules and biomacromolecules indeed own the properties of stabilizers and reductants, there is no report for synthesis of AuNCs using D-proline (D-Pro) as the protector and reducing agent till now. Importantly, none of the AuNCs combing with gold nanopartilces (AuNPs) was utilized for testing fluids RTX. Therefore, exploration of facile synthetic protocol for making D-Pro capped AuNCs (D-Pro@AuNCs) to monitor RTX is purposeful and valuable.
Herein, a strategy for simple and rapid synthesis of D-Pro stabilized AuNCs was specifically designed. The fluorescence of D-Pro@AuNCs could be quenched by AuNPs via the electrostatic interaction and aurophilicity, which could be recovered in addition of RTX due to the stronger electrostatic interaction between RTX and AuNPs. Based on the fluorescence "turn-on" principle, an assay for highly sensitive and selective detection of RTX was fabricated for the first time. The proposed method was further applied for measurement of anticancer drug RTX in rat serums after an intravenous injection.
D-Pro@AuNCs was synthesized with a facile "one pot" protocol. Typically, aqueous HAuCl4 solution (8.0 mL, 6.0 μmol/L) was added to D-Pro solution (2.0 mL, 2.0 mol/L), the mixture was stirred at room temperature for 1.0 min. Then, it was vigorously stirred and the reaction process proceeded at 100 ℃ for 10.0 min. The mixture solution changed its color from canary yellow to light brown (Fig. 1A-a) and then cooled down to room temperature. The final product, D-Pro@AuNCs, was produced and stored at 4.0 ℃.
|
Download:
|
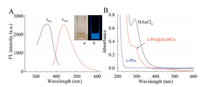
| Fig. 1. (A) Fluorescence spectra of the resultant D-Pro@AuNCs; (B) UV–vis absorption spectra of aqueous HAuCl4, D-Pro and D-Pro@AuNCs; Inset: photograph of the synthesized D-Pro@AuNCs under the visible light (a) and UV light (b). | |
Fig. S1 (Supporting information) illustrates the process for preparation of D-Pro@AuNCs based on the charges transfer between the donor (D-Pro) and the acceptor (gold atoms) [18], which exhibited its maximal emission at 435 nm (Fig. 1A). As described in reference [19], kiwi juice-coated AuNPs was obtained by a simple method. The maximum UV–vis absorption of the resultant AuNPs was at 540 nm (Fig. S2a in Supporting information). Figs. S2b and S2c (Supporting information) demonstrate that the obtained AuNPs is spherical particles in shape and 15.7 ± 0.5 nm in size.
It has been reported that the surface ligands specifically could affect the fluorescence intensity of AuNCs [20]. More electron-rich groups [21-25] (such as imidazole groups, amino groups, heterocycle amine groups, carboxyl groups, pyrrole groups) are introduced in the ligands, the stronger the fluorescence intensity of the AuNCs is. There are abundant heterocycle amine groups in DPro, which have appropriate reducing ability [21]. Therefore, in this work, it is believed that D-Pro plays a key role as both capping agent and reductant for synthesis of fluorescent AuNCs. Based on the good reducibility of capping ligand, the Au3+ ions can be reduced by D-Pro to form Au+ ions and Au0 atoms. Fig. S3 (Supporting information) exhibits the binding energy for 4f7/2 (85.6 eV) and 4f5/2 (89.3 eV) of AuNCs, which defined with X-ray photoelectron spectroscopy. The data indicates the AuNCs consists of Au+ ions and Au0 atoms [21]. Moreover, Fig. 1A depicts the fluorescence spectra of D-Pro@AuNCs, demonstrating that the D-Pro modified AuNCs was successfully prepared.
To better understand the generation of D-Pro@AuNCs, the fluorescence was measured under different synthesis conditions by changing the concentration of reactants when the mixture solution incubated at 100 ℃ for 10 min [26]. Fig. S4a (Supporting information) shows that the fluorescence intensity of D-Pro@AuNCs climbed up gradually with the concentration of DPro increasing from 0.1 mol/L to 2.0 mol/L and further increase led decrease of the fluorescence intensity. Fig. S4b (Supporting information) displays that the fluorescence intensity of D-Pro@AuNCs obviously enhanced with the HAuCl4 concentration increased from 0.4 μmol/L to 4.8 μmol/L and no dramatically enhancement was observed with further increase. The results clearly demonstrated that the reactant concentration is essential in the formation of D-Pro@AuNCs. In the green synthesis process, there are no other catalysts or templates involved, providing the final nontoxic product as a pale yellow solution (Fig. 1A-a), which emits strong blue fluorescence under UV light (365 nm) irradiation (Fig. 1A-b). In addition, the strong fluorescence could be maintained after storage for four weeks (Fig. S5B in Supporting information), exhibiting a long-term stability of the aqueous D-Pro@AuNCs.
As shown in Fig. 1A, the fluorescence of the prepared D-Pro@AuNCs steadily displayed an emission peak at 435 nm with excitation at 355 nm. Meanwhile, there is no characteristic surface plasmon absorption resonance peak found in the UV–vis spectra of D-Pro@AuNCs solution ranging from 500 nm to 600 nm (Fig. 1B). Instead, it moves upwards at around 280 nm due to its molecularlike properties [21] such as electronic transition and size-tunable fluorescence, which indicated that the D-Pro@AuNCs was constructed successfully.
The D-Pro and D-Pro@AuNCs have been characterized by FT-IR measurements to confirm the interaction between D-Pro and the D-Pro@AuNCs. Fig. S5A (Supporting information) shows that the strong N-H stretching vibration appeared at 2397.4 cm-1. While, the stretching vibration of C–N group in nitrogen heterocycle could be observed at 1620.4 cm-1 and 1564.0 cm-1. Furthermore, the bands presented at 1388.7 cm-1 and 1174.6 cm-1 are attributed to C–H stretching vibration [26]. It could be assumed that the D-Pro bound to the Au0 atoms through the heterocycle amine groups for preparation and stabilization of AuNCs. The D-Pro coated on the surface of the AuNCs and played the roles of stabilizer and reductant to avoid the formed AuNCs aggregation. Thus, the D-Pro capped AuNCs could be observed, underpinning with UV–vis spectra in Fig. 1B.
It has been reported that AuNCs could be applied in sensing of drugs [9-16], whether the proposed D-Pro@AuNCs integrating with gold nanoparticles (AuNPs) for selective and sensitive detection of RTX is still unclear. Due to the aurophilicity and the electrostatic interactions in gold species [27], the fluorescence of D-Pro@AuNCs was remarkably quenched in the present of AuNPs. It should be noted that using fruit juices as the liagnds for synthesis of AuNPs is meaningful. Therefore, kiwi juice protected AuNPs was constructed [28] in this study. The fluorescence intensity of D-Pro@AuNCs decreased continuously with the increasing of AuNPs. Fig. S6 (Supporting information) depicts that once 50.0 mL AuNPs was added into the D-Pro@AuNCs solution, about 50.0% of the fluorescence intensity of D-Pro@AuNCs was quenched. Then followed in addition of RTX, the fluorescence of AuNCs could be recoveried effectively. Therefore, 50.0 mL AuNPs was selected in the further investigation.
The effect of buffer pH and ionic strength on fluorescence recovery efficiency of D-Pro@AuNCs-AuNPs system in addition of RTX has been investigated. As exhibited in Fig. S7b (Supporting information), NaCl solution ranging from 20.0 μmol/L to 160.0 μmol/L appears to have no obvious impact. While, the fluorescence recovery of D-Pro@AuNCs displayed notable difference when pH value changed, ranging from 5.0 to 11.0 (Fig. S7a in Supporting information). At pH 8.0, the maximum recoveried fluorescence was achieved [5]. Moreover, it could be observed that the fluorescence intensity of D-Pro@AuNCs-AuNPs/RTX system enhanced to the maximum within 4 min (Fig. S8 in Supporting information). Finally, buffer pH at 8.0 and incubation time of 4 min were chosen for further study.
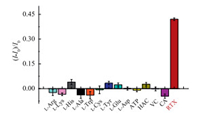
Especially, only trace D-amino acids can be tested in body fluids. Therefore, to evaluate the selectivity of the proposed assay for sensing RTX, the potential interferences, including normal L-amino acids (L-Arg, L-Lys, L-His, L-Ala, L-Trp, L-Cys, L-Tyr, L-Glu, L-Asp), adenosine triphosphate (ATP), acetic acid (HAC), ascorbic acid (VC), citric acid (CA), anticancer drug (doxorubicin, DOX) and common metal ions (Mg2+, Fe2+, Na+, Ni+, Ca2+, Cr3+, K+, Al3+, Zn2+, Fe3+) have been investigated. As exhibited in Fig. 2 and Fig. S9 (Supporting information), no notable change of fluorescence intensity induced by the potential interferences. Only RTX could lead to vary dramatically in the value of (I-I0)/I0, indicating this assay established based on fluorescence "turn on" strategy had high selectivity for sensing RTX and good resistance to interferences.
|
Download:
|
| Fig. 2. The relative fluorescence (I–I0)/I0 of D-Pro@AuNCs-AuNPs in the presence of L-Arg, L-Lys, L-His, L-Ala, L-Trp, L-Cys, L-Tyr, L-Glu, L-Asp, ATP, HAC, VC, CA and RTX, at pH 8.0. | |
As exposed to Fig. 3, the principle of D-Pro@AuNCs-AuNPs system fluorescence "turned on" by RTX was deduced. The asprepared D-Pro@AuNCs was measured by dynamic light scattering meter with a mean size of 5.6 ± 0.1 nm (Fig. S10a in Supporting information) and its zeta potential (ζ) was 3.5 mV. After adding of AuNPs (ζ = -14.1 mV). The average size of D-Pro@AuNCs-AuNPs was dramatically enlarged to 24.1 °0.1 nm (Fig. S10b in Supporting information). Its ζ changed to -10.9 mV, which indicated that the D-Pro@AuNCs indeed attached on the surface of AuNPs. Then, in the presence of RTX (ζ = 5.5 mV), the mean size of D-Pro@AuNCsAuNPs/RTX became 190.1 °0.4 nm (Fig. S10c in Supporting information) and its ζ was -5.7 mV. The increase in ζ [27, 29] of D-Pro@AuNCs-AuNPs/RTX should be attributed to the positively charged RTX, which further revealed that the RTX indeed induced the aggregation of the AuNPs. Fig. S10d displays (Supporting information) the TEM image of the D-Pro@AuNCs, which well dispersed in the solution with uniform spherical shapes. In addition of AuNPs, the size of the D-Pro@AuNCs-AuNPs changed greatly as depicted in Fig. S10e (Supporting information) and further quenched the fluorescence of D-Pro@AuNCs due to the electrostatic and gold-gold interactions [27, 29]. Fig. S10f (Supporting information) indicates that after adding RTX, the size of the D-Pro@AuNCs-AuNPs/RTX was enhanced obviously, which revealed that the RTX had stronger electrostatic interactions with AuNPs, led to its aggregation and further increased the distance between AuNPs and D-Pro@AuNCs. It should be mentioned that the surface electron density of AuNCs would affect its fluorescence intensity [30]. In this work, the formed D-Pro@AuNCs-AuNPs composites decreased the surface electron density because of the electrostatic interaction and aurophilicity, but addition of RTX could further "turned on" (Fig. 3) the fluorescence of the D-Pro@AuNCs (Fig. S6 in Supporting information).

|
Download:
|
| Fig. 3. Schematic mechanism of D-Pro@AuNCs for fluorescence turn on sensing of RTX. | |
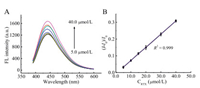
The "turn on" fluorescence of D-Pro@AuNCs-AuNPs strategy for sensitively sensing RTX could be achieved based on the stronger electrostatic interaction between AuNPs and RTX. Fig. 4A displays the fluorescence of D-Pro-AuNCs-AuNPs increased gradually with the increasing concentration of RTX. A good linear relationship between RTX and the fluorescence efficiency was gotten (Fig. 4B) in the range of 5.0–40.0 μmol/L (R2 = 0.999) with the detection limit as low as 1.9 μmol/L (3kb/k) [31]. The regression equation was:
|
Download:
|
| Fig. 4. (A) The fluorescence response of the D-Pro@AuNCs-AuNPs upon addition of various concentrations of RTX; (B) relative fluorescence intensity (I–I0)/I0 of D-Pro@AuNCs-AuNPs contrast to the RTX concentration. | |

|
where I0 and I are the fluorescence intensities in the absence and presence of RTX, respectively.
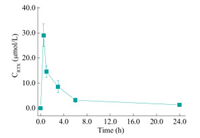
It demonstrates that the D-Pro@AuNCs-AuNPs/RTX system had a prosperous ability for monitoring RTX. Moreover, the system provided high selectivity towards RTX. Therefore, the proposed method was further applied in detection of RTX in metabolic process in real biological examples. The serum concentration of RTX in rat serums following intravenous injection versus time was exhibited in Fig. 5. At 0.5 h following abdominal injection, the amount of RTX content in the serums rose and reached the highest point, and then declined quickly for another 0.5 h, after that the decline became slow [4]. Moreover, the investigation on the recovery of RTXs was carried out. The proposed assay for testing serum RTX had good recovery rate (Table S1 in Supporting information) in the range of 95.2%-103.4% with satisfied relative standard deviation (RSD). The protocol has presented the advantage in rapid preparation of D-Pro@AuNCs and in "turn on" monitoring of RTX, boosting future practical application of D-Pro@AuNCs in pharmacokinetics study.
|
Download:
|
| Fig. 5. The drug concentration-time curve of serum RTX following intravenous injection in the abdominal cavity of rats. | |
In summary, a simple and rapid hydrothermal protocol was utilized for synthesis of D-Pro@AuNCs based fluorescent probe with D-Pro as the reductan and stabilizer. Consequently, a highly selective and sensitive method was established for detection of RTX using D-Pro@AuNCs-AuNP system. Due to the strong interaction between RTX and AuNPs, the presence of RTX could "turn on" fluorescence of D-Pro@AuNCs-AuNP. Moreover, the prepared probe was successfully applied to metabolic analysis of serum RTX following intravenous injection in rats. The proposed AuNCsAuNPs system has shown a great potential for pharmacokinetics study in real living bio-systems.
AcknowledgmentsThe authors are grateful for the financial support from the National Natural Science Foundation of China (Nos. 21575144, 91732103, 21874138, 21635008, 21621062). We also would like to thank Mr. Han Wu for his kind help.
Appendix A. Supplementary dataSupplementary material related to this article can be found, in the online version, at doi:https://doi.org/10.1016/j.cclet.2019.05.019.
| [1] |
J.M. Ferrero, E. Chamorey, N.M.F. Leccia, et al., Cancer Chemother. Phamacol. 50 (2002) 459-464. DOI:10.1007/s00280-002-0519-2 |
| [2] |
J.X. Zhang, Z.N. Yin, W. Wu, et al., Chem. Res. Chin. Univ. 28 (2012) 963-970. |
| [3] |
D.I. Jodrell, D.R. Newell, W. Gibson, et al., Cancer Chemother. Phamacol. 28 (1991) 331-338. DOI:10.1007/BF00685685 |
| [4] |
D.X. Wang, Y.L. Gao, L.H. Yun, Cancer Chemother. Phamacol. 57 (2006) 97-104. DOI:10.1007/s00280-005-0018-3 |
| [5] |
D.X. Wang, Y.L. Gao, L.H. Hui, Chin. J. Pharm. Anal. 25 (2005) 778-780. |
| [6] |
S. Kim, A. Bisch, D. Montange, et al., J. Chromatogr. B 1060 (2017) 240-246. DOI:10.1016/j.jchromb.2017.06.021 |
| [7] |
J. Hu, L. Ding, Y. Gao, et al., J. Chromatogr. B 853 (2007) 147-153. DOI:10.1016/j.jchromb.2007.03.028 |
| [8] |
G.W. Aherne, E. Ward, N. Lawrence, et al., Br. J. Cancer 77 (1998) 221-226. DOI:10.1038/bjc.1998.37 |
| [9] |
X.Y. Jiang, D.Q. Feng, G.L. Liu, et al., Sens. Actuators B-Chem. 232 (2016) 276-282. DOI:10.1016/j.snb.2016.03.100 |
| [10] |
J. Xu, L. Shang, Chin. Chem. Lett. 29 (2018) 1436-1444. DOI:10.1016/j.cclet.2017.12.020 |
| [11] |
M. Shamsipur, F. Molaabasi, S. Hosseinkhani, et al., Anal. Chem. 88 (2016) 2188-2197. DOI:10.1021/acs.analchem.5b03824 |
| [12] |
A.A.F. Nejad, F. Ghasemi, M.R. Hormozi-Nezhad, Anal. Chim. Acta 1039 (2018) 124-131. DOI:10.1016/j.aca.2018.07.043 |
| [13] |
Y.S. Borghei, M. Hosseini, M.R. Ganjali, Sens. Actuators B-Chem. 273 (2018) 1618-1626. DOI:10.1016/j.snb.2018.07.061 |
| [14] |
H.B. Wang, Y. Li, H.Y. Bai, et al., Food Anal. Method. 11 (2018) 3095-3102. DOI:10.1007/s12161-018-1289-8 |
| [15] |
Y.M. Wang, M.H. Lu, D.P. Tang, Biosens. Bioelectron. 109 (2018) 70-74. DOI:10.1016/j.bios.2018.03.007 |
| [16] |
T.Y. Lin, K.C. Wei, S.P. Ju, et al., J. Mater. Chem. B-Mater. Biol. Med. 6 (2018) 3387-3394. DOI:10.1039/C8TB00913A |
| [17] |
R.L. Zhang, S.S. Liang, M. Jin, et al., Sens. Actuators B-Chem. 253 (2017) 196-202. DOI:10.1016/j.snb.2017.06.136 |
| [18] |
S. Rai, H. Singh, J. Mol. Model. 19 (2013) 4099-4109. DOI:10.1007/s00894-012-1711-x |
| [19] |
R.S. Li, H. Liu, B.B. Chen, et al., Anal. Mehods 8 (2016) 2491-2498. |
| [20] |
Z.K. Wu, R.C. Jin, Nano Lett. 10 (2010) 2568-2573. DOI:10.1021/nl101225f |
| [21] |
X. Yang, M.M. Shi, R.J. Zhou, et al., Nanoscale 3 (2011) 2596-2601. DOI:10.1039/c1nr10287g |
| [22] |
K.L. Dong, J.S. Zhou, T.Q. Yang, et al., Appl. Spectrosc. 72 (2018) 1645-1652. DOI:10.1177/0003702818775704 |
| [23] |
F.R. Nie, L. Ga, J. Ai, et al., Micro Nano Lett. 13 (2018) 817-820. DOI:10.1049/mnl.2017.0871 |
| [24] |
Z.Q. Bai, X.L. Ren, Z. Gong, et al., Chin. Chem. Lett. 28 (2017) 1901-1904. DOI:10.1016/j.cclet.2017.05.005 |
| [25] |
J. Chen, Y.N. Huang, X.Y. Yang, et al., Anal. Chim. Acta 1023 (2018) 89-95. DOI:10.1016/j.aca.2018.02.082 |
| [26] |
X.Y. Mu, L. Qi, P. Dong, et al., Biosens. Bioelectron. 49 (2013) 249-255. DOI:10.1016/j.bios.2013.05.019 |
| [27] |
B.A. Rusell, B. Jachimska, I. Kralka, et al., J. Mater. Chem. B 4 (2016) 6876-6882. DOI:10.1039/C6TB01827K |
| [28] |
B.B. Chen, H. Liu, C.Z. Huang, et al., New J. Chem. 39 (2015) 1295-1300. DOI:10.1039/C4NJ01578A |
| [29] |
F. Scherbaum, A. Grohmann, C.B. Huber, et al., Angew. Chem. Int. Ed. 27 (1988) 1544-1546. DOI:10.1002/anie.198815441 |
| [30] |
Y.F. Chen, J. Qiao, Q.R. Liu, et al., Anal. Chim. Acta 1026 (2018) 133-139. DOI:10.1016/j.aca.2018.04.014 |
| [31] |
T. Yu, C.N. Xu, J. Qiao, et al., Chin. Chem. Lett. 30 (2019) 660-663. DOI:10.1016/j.cclet.2018.10.001 |
 2019, Vol. 30
2019, Vol. 30 

