b University of Chinese Academy of Sciences, Chinese Academy of Sciences, Beijing 10039, China;
c Department of Chemistry, Waterloo Institute for Nanotechnology, University of Waterloo, Waterloo, Ontario N2L 3G1, Canada;
d State Key Laboratory of Applied Organic Chemistry, Lanzhou University, Lanzhou 730000, China
Fluorescence polarization (FP)/anisotropy (FA) is a powerful and reliable signaling transduction approach that provides quantitative information on molecular mobility, rotational motion, interaction process [1, 2]. In principal, FP/FA value can be easily obtained by detecting the fluorescence intensities from two polarization planes. The FP (P) value can be defined according to the equation P = (I// - I⊥)/(I// + I⊥). Similarly, the FA (r) value is given by the equation r = (I// - I⊥)/(I// + 2I⊥), where I// stands for the fluorescence intensities with parallel polarization to the excitation plane, I⊥ represents the fluorescence intensity with perpendicular polarization to the excitation plane [2-6]. The FP (P) and FA (r) values can be interchangeable by means of r = 2 P/(3-P) [2, 3]. The polarization and anisotropy are independent of the total intensity of the sample and the fluorophore concentration, which mainly depends on intrinsic properties of the fluorescently labeled molecules, such as molecular weight (molecular volume), shape as well as the strength of the binding interaction, and by environmental factors, such as the solution viscosity and temperature [7-10]. Namely, if fluorescent molecule is free in solution, it will rotate fast and thus will exhibit a small P/r value. On the contrary, when the fluorescent molecule is bound with another substance to form a large molecule, its rotational rate will decrease and will display a high P/r value [11, 12]. Furthermore, the anisotropy and polarization are less sensitive to the fluorescence fluctuation and the photobleaching than other fluorescent assay method [13, 14]. Up to now, FP/FA has been widely introduced for the detecting various targets in a variety of application fields including environmental monitoring, food safety, and clinical diagnosis due to its automated high-throughput capability, homogeneous-phase, accuracy, and speed [15-21]. But just as a coin has two sides, conventional FP/FA inevitably has its disadvantage, poor detection sensitivity and substantial background owing to the lack of an efficient amplification mechanism. To solve the above limitations, different paths have been established for FP/FA signal amplification determination [22-24]. Of these paths, mass-augmented amplification strategies have been usually used for increasing the sensitivity of FP/FA measurement in a series of recent studies. For example, large biomolecules (e.g., oligonucleotides [25] and proteins [16]), and a variety of nanomaterials [26-31]. Check out the number of scientific publications in the field of FP/FA (Fig. 1), we can see the number of articles published in the field of FP/FA was basically stable in recent five years. In order to further promote the development of FP/FA technology, the careful summary of the essentials of the FP/ FA assay based on nanomaterial for is highly desired.

|
Download:
|
| Fig. 1. The number of scientific publications about FP/FA was sorted by year. Data were obtained from the "Web of Science". The words "fluorescence polarization" or "fluorescence anisotropy" were keyed into the "topic" search box. | |
In this present review, we highlight recent progress about nanomaterial-based FP/FA technology, and their applications in immunoassay, and highly selective, excellently sensitive detection of protein, nucleic acid, small molecule and metal ion.
2. Nanomaterial-based FP/FAThe emergence and significant advance of nanotechnology and nanoscience provide new opportunities for the use of nanomaterials in analytical chemistry [32-39]. Recently, a number of nanomaterial, such as carbon nanoparticles (CNPs), gold nanoparticals (AuNPs), metal–organic frameworks (MOFs), SiO2 nanoparticals (SiO2 NPs), graphene oxide (GO), quantum dots (QDs), carbon nanotubes (CNTs), 2D transition metal dichalcogenide nanosheets (TMD NSs), MnO2 nanosheets were introduced into FP/FA sensor system, has brought revolutionary effect for greatly improving detection sensitivity and application range of FP/FA, due to its unique structure and extraordinary properties such as simple/controllable synthesis and modification, different sizes, large volumes and masses, chemical compositions and morphologies, excellent optical and chemical properties [26-31, 40-44]. Table 1 lists various targets of interest, FP/FA enhancer and signal amplification methods, analytical performance, and so on [40-43, 45-60].
|
|
Table 1 Summary of nanomaterial-based FP/FA assay for biomolecule with respect to target, FP enhancer and amplification technique, dynamic range, limit of detection (LOD) and application. |
3. Sensor application of nanomaterial-based FP/FA
At present, nanomaterial-based FP/FA is widely applied for immunoassay, protein analysis, nucleic acid analysis, small molecule and metal ion analysis, and so on.
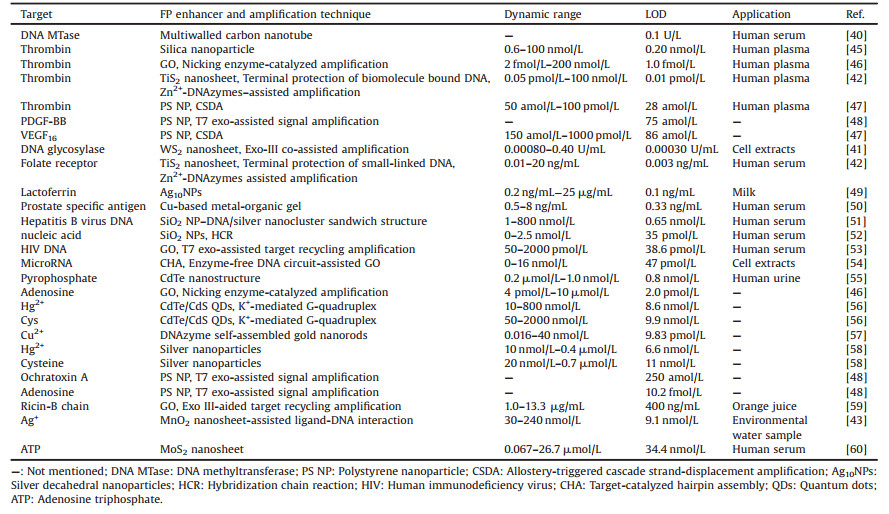
3.1. ImmunoassayFP immunoassay (FPIA) that combines FP and competitive immunoassay is a widespread homogeneous technique, with no washing or separation steps, which meets the requirements of a reliable, simple, cost-effective and fast analysis [61]. The analytical signal of FPIA is also proportional only to molecular size and does not depend on intensity changes. Up to now, the use of this approach for the determination of various compounds, including pesticides [62], therapeutic drug [63-66], toxins or antibiotics [67-73], etc. [74-76], have been reported in literatures. Zhang et al. [70] developed a dual-wavelength FPIA for simultaneous determination of total aflatoxins and family zearalenones in maize. This strategy provided higher efficiency, higher throughput, more information in one test and less assay time derived from the design of dual-wavelength fluorescence signals. Meanwhile, it was successfully used for screening naturally contaminated maize flour samples, and the results of the analysis were similar to those reported by HPLC-MS/MS. It is worth mentioning that Tian et al. [77] combined the high stability and sensitivity of CdTe/CdS QDs and the advantage of the FPIA resulting in a rapid, and portable, competitive immunoassay tool for selective and sensitive multiplexed determination of α-fetoprotein (α-AFP) and carcino embryonic antigen (CEA). The sensitivity of the detection was 0.28 ng/mL for α-AFP and 0.36 ng/mL for CEA. Furthermore, the proposed approach could also be extended to detect other antigen-antibody immune complexes by using the respective antigens and corresponding antibodies. Subsequently, the same group demonstrated another ultrasensitive CdSe/CdS QDs-based FPIA system [78] for adenosine triphosphate (ATP) detection by means of anti-digoxin-Au NPs amplification. As demonstrated in Fig. 2, DNA hybridization was formed when ATP presented in the system containing the DNA conjugated CdSe/CdS QDs and antidigoxin-Au NPs, leading to the volume of the fluorescence molecule get bigger. Meanwhile, the fluorescence molecule rotated slower and thus produced a larger FP value. Under the optimal conditions, the limit of detection could reach picomolar level (1.8 pmol/L).
|
Download:
|
| Fig. 2. Schematic illustration of the anti-digoxin-Au NPs-based FP platform and its use in detecting ATP. Reproduced with permission [78]. Copyright 2013, Elsevier Publishing Group. | |
3.2. Protein analysis
Protein plays a critical role in regulating various physiological functions. Extremely sensitive and accurate detection of protein at ultra-trace level is essential to biomedical diagnostics and clinical therapy [79].
The group of Zhao reported a series of sensing strategies using nanomaterials as FP enhancers for protein detection [40, 46, 47]. In 2015, Zhao et al. [46] introduced a new kind of amplified FP aptasensors for the determination of thrombin (Tb) based on GO enhancement effect as well as nicking enzyme-assisted signal amplification (NESA). As presented in Fig. 3, in the absence of model analyte Tb, FAM-labeled DNA and two aptamer subunits (aptamer subunit-1 and aptamer subunit-2) could be adsorbed onto the GO surface, making FAM-labeled DNA exhibited a high FP value because of the extraordinarily large volume of GO. However, after the introduction of Tb into the system, two aptamer subunits would assemble into Tb/aptamer complex, and formed a duplex region. This resulted duplex region containing the recognition sequence and cleavage site of enzyme Nb.BbvCI. This triggered selective cleavage of FAM-labeled DNA by means of Nb.BbvCI, leading to the release of a short DNA carrying dye FAM and the Tb/ aptamer complex from the surface of GO, the decrease of the FP value. The released complex of Tb/aptamer then hybridized with another FAM-labeled DNA associated with GO, and the cleavage cycle started anew, generating substantial decrease of FP value. By monitoring the changed FP value, the detection of Tb could be achieved successfully. This approach exhibited high specificity, low sensitivity (1.0 fmol/L) and wide dynamic ranges over 6 orders of magnitude.

|
Download:
|
| Fig. 3. Schematic illustration of the FP atasensor based on GO and nicking enzyme-catalyzed amplification for Tb assay. Reproduced with permission [46]. Copyright 2015, Elsevier Publishing Group. | |
In order to further improve the sensitivity of detection, the same group demonstrated [47] another new kind of amplified FP sensor based on polystyrene nanoparticle (PS NP) enhancement and allostery-triggered cascade strand-displacement amplification (CSDA) for attomolar determination of Tb. As exhibited in Fig. 4, compared with the homogeneous aptasensors in the previous work [45], the determination sensitivity (28 amol/L) could be improved at least 2 orders of magnitude.

|
Download:
|
| Fig. 4. Schematic illustration of the CSDA and PS NP enhancement-based amplified FP aptasensor for Tb determination. Reproduced with permission [47]. Copyright 2015, American Chemical Society. | |
Furthermore, Zhao et al. [41] also described layered tungsten disulfide nanosheet (WS2) and exonuclease Ⅲ (Exo Ⅲ) co-assisted cyclic amplification strategy for excellently sensitive and desirable selective FP detection of the activity of DNA glycosylase. Furthermore, this approach was successfully applied to measure uracil-DNA glycosylase (UDG) activity in cell extracts and the study of the inhibition of UDG.
Similar to other two-dimensional (2D) graphene analogues, TiS2 nanosheet also exhibited the capability of discriminating ssDNA versus dSDNA [80]. Inspired by this facts, Li et al. [42] developed a general FP strategy by using TiS2 nanosheet as FP enhancer, combined with Zn2+-dependent DNAzyme-assisted signal amplification and terminal protection of single moleculelinked DNA for highly efficient determination folate receptor (FR) and Tb. The analysis results were in good agreement with those obtained by enzyme-linked immunosorbent assay.
Chen et al. [49] designed a novel bivalent FP aptasensor for rapid determination of lactoferrin (Lac) in milk based on silver decahedral nanoparticals (Ag10NPs) enhancement, metal-enhanced fluorescence (MEF) effect and split aptamers enlargement. In this system, two split aptamers bound to different sites of Lac and assembled into a split aptamers-target complex, thus narrowing the distance between Ag10NPs and dye FITC, resulting in a high FP valve. This strategy substantially broadened the perspective for FP amplified strategies.
Recently, Zhao et al. [50] firstly described that Cu-based metalorganic gel (Cu-MOG) was capable of acting as mass amplifier in FA for quantitative prostate specific antigen assay. This FA mode was based on π-π stacking interaction and electrostatic incorporations between the FAM-labeled aptamer sequence and Cu-MOG, resulting in the rotation of fluorophore was restricted apparently, and generating a high FA value. However, in the presence of target PSA, FAM-labeled aptamer sequence specific combined with PSA, which made the detachment of FAM-labeled aptamer sequence from Cu-MOG and FA value was reduced.
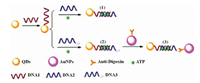
3.3. Nucleic acid analysisThe specific sequence determination of nucleic acid has attracted numerous research interests because of its widespread applications in environmental monitoring, medical diagnostics, genetics therapy, drug discovery, and early screening of cancers. It is highly establish to develop highly sensitive, selective, costeffective determination approach for specific nucleic acid sequences, especially for low-abundant nucleic acid. Chen et al. [81-84] designed various strategies for the dermination of nucleic acid at ultra-low levels. FP sensor based on nanomaterial has also been reported for the determination of nucleic acid target. Our group [51] described a new type of FP sensor based on the FP enhancement effect of the SiO2 NP–DNA/silver nanocluster sandwich structure by using hepatitis B virus (HBV) DNA as a model analyte. As shown in Fig. 5, Ag-DNA and Biotin-En-DNA were used in this study. Firstly, SiO2 nanoparticles modified with streptavidin (streptavidin-SiO2 NPs) was chosen as a model of nanoparticle amplifier. In the absence of HBV DNA, a relatively weak FP value could be obtained owing to the small molecular volume of DNA/silver nanoclusters (DNA/Ag NCs). When HBV DNA was introduced, the aforementioned sandwich structure would be formed at SiO2 NPs surface, leading to a notable increase of the FP value owing to the enlargement of the molecular volume of the formed SiO2 NP-functionalized DNA/Ag NCs sandwich structure with slow rotation. This assay protocol was successfully used to detect HBV levels in human serum, and the approach has significant advantage of excellent specificity with the capability of discriminating differentiate mismatched DNA. In addition, this strategy could be easily extended to other DNA or RNA assay by simply switching the corresponding sequences of nucleic acids substrates.
|
Download:
|
| Fig. 5. (A) The process for the preparation of the functionalized streptavidin–SiO2 NPs. (B) Scheme of the established amplified FP sensor for HBV DNA assay. Reproduced with permission [51]. Copyright 2015, Royal Society of Chemistry. | |
In addition, Zhao et al. developed a lot of amplified FP system for the determination of DNA or miRNA based on hybridization chain reaction (HCR) and SiO2 NPs [52], T7 exo-assisted target recycling amplification and GO [53], T7 exo-assisted quadratic recycling amplification and GO [85], dendrite-modified Au NPs [86], T7 exoassisted dual-cycle signal amplification and PS NP [87], respectively. These amplification strategies are not limited to the particular DNA but are perceived to provide universal platforms for highly sensitive determination of a wide spectrum of nucleic acids.
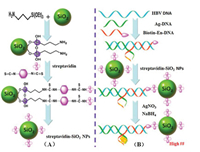
GO has been widely proven as an outstanding FP/FA enhancer due to its unique 2D nanosheet structure with a much slow rotation rate as well as a large surface area to interact with various probes [46]. Zhen et al. [54] reported a new FA strategy for the determination of microRNA-21 (miRNA-21) on the base of enzyme-free DNA circuit assisted GO amplification and targetcatalyzed hairpin assembly (CHA). As illustrated in Fig. 6, upon addition of miRNA-21, the CHA was initiated, continuously generating numerous H1–H2 duplexes. The resulted H1–H2 duplex could induce the formation of a dye-modified H1–H2-probe DNA complex by means of toehold-mediated strand exchange reaction, which led to the dye-modified H1–H2-probe DNA complex leave away from the surface of GO, producing a decreased FA value. By monitoring the change of FA value, miRNA-21 could be determined in the range from 0 to16 nmol/L. The experimental result indicated that the limit of detection (47 pmol/L) was about 200 times lower than that without CHA. Recently, Zhang's group [88] introduced a FP platform for ligand-RNA interaction detection using GO as a "nanoamplifer" to obtain the signal amplification. In this system, proflavine was used as FP signal readout. Before and after the introduction of Rev antagonist to the preformed proflavine-RRE complex, the release of proflavine only induced slight change of FP, while in the presence of GO, a dramatically increase of FP could be observed.
|
Download:
|
| Fig. 6. Principle of the proposed amplified enzyme-free FA sensor for the determination of miRNA-21. Reproduced with permission [54]. Copyright 2017, American Chemical Society. | |
3.4. Small molecule and metal ion analysis
The determination of small molecule and metal ion is associated with many diseases and biological process [89, 90]. A great deal of small molecules and metal ions, e.g., silver ions [43], adenosine [48], toxin [48, 59, 91], mercury (Ⅱ) ions or biothiols [56, 58], copper (Ⅱ) ions [57], ATP [60], organophosphorus pesticide [92], and so on [55], were highly sensitively measured by nanomaterial-based FP/FA. For example, Tian's group [56] constructed a simple, highly selective and sensitive FP assay based on CdTe/CdS QDs using a K+-mediate G-quadruplex as a FP enhancer for the determination of biothiols and Hg2+ in complex biological matrix. Another example, Qi and co-workers [43] demonstrated a MnO2 nanosheet-assisted ligand-DNA interaction-based FP sensor for the determination of Ag+ using proflavine as a FP indicator. As displayed in Fig. 7, the introduction of Ag+ to the preformed proflavine-DNA complex induced the leave away of proflavine, which led to weak change of FP value. The subsequent introduction of MnO2 nanosheet amplified the observed change, making it a viable approach for Ag+ determination. This amplified strategy exhibited a detection limit of 9.1 nmol/L with a linear range of 30–240 nmol/L. Furthermore, the proposed method could be potentially used for other types of target analytes by means of simply changing the sequence of nucleic acid to a specific target.
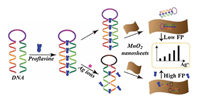
|
Download:
|
| Fig. 7. Schematic illustration of principle of MnO2 nanosheet-assisted ligand-DNA interaction-based FP sensing for Ag+ assay. Reproduced with permission [43]. Copyright 2017, Elsevier Publishing Group. | |
4. Conclusions and future perspectives
This review summarizes the recent research progress in the development of FP/FA sensor platform mainly based on various nanomaterials in the past five years. FP/FA is a fast homogeneous analysis technique, and less affected by environmental interferences. Nanomaterials have extraordinary physicochemical properties that can significantly improve detection sensitivity through enhancing the signals of FP/FA.
The combination of FP/FA with nanomaterials has enabled the reliable and efficient determination of a large number of target analytes in medical diagnosis, food and drug safety, environmental monitoring. Notwithstanding so great progress in the area have been made, there are still challenges that need to be addressed. Firstly, the variety and quantity of nanomaterials are still quite limited. Hence, exploring and constructing more novel nanomaterials based on different family members is necessary. Secondly, it is still a rather difficult task to measure multiple target analytes in a single solution simultaneously. Up to now, there is few article focusing on the multiple determination of biomarkers based on multicolor QDs [77]. It is important to fabricate more advanced nanomaterials with multiplexed signaling reporters to detect the existence of different biomolecules. Thirdly, FP/FA sensors based on nanomaterials applicable for living cell analysis are still fairly few, although FP/FA is less sensitive to the fluorescence fluctuation and the photobleaching than other fluorescent assay method. It is highly desired to establish FP/FA sensors based on nanomaterials with low toxicities, desirable stability, etc. In addition, almost all the established FP sensors based on nanomaterials so far still stay at the stage of proof-ofprinciple. These works rarely reach commercial level. This is because that the real biological matrix is very complex, which makes the detection result susceptible to the influence of unexpected events. In response to these challenges and opportunities, it is highly demanded to prepare more new nanomaterials with superior physicochemical properties combined with other paths as FP/FA enhancers. Last but not the least, the analytical field only measures the overall FP/FA response of the sensors, and quantitative analysis of the origin of the values has yet to be demonstrated. This is especially important in systems with strong quenchers such as GO. The adsorbed molecules may not contribute much to the fluorescence signal. Theoretical advancement in this front will also further the development of the analytical field.
Along with the great progress in the field of nanoscience as well as sensing-technology, we believe that nanomaterial-based FP/FA sensor will bring about promising future and play a critical role in various applications in the coming years.
AcknowledgmentsThe authors gratefully acknowledge the financial support provided by the National Natural Science Foundation of China (Nos. 21822407, 21405163) and the top priority program of "OneThree-Five" Strategic Planning of Lanzhou Institute of Chemical Physics, CAS.
| [1] |
B.C. Ye, B.C. Yin, Chem. Angew., Int. Ed. 47 (2008) 8386-8389. DOI:10.1002/anie.200803069 |
| [2] |
D.M. Jameson, J.A. Ross, Chem. Rev. 110 (2010) 2685-2708. DOI:10.1021/cr900267p |
| [3] |
Q. Zhao, J. Tao, J.S. Uppal, et al., TrAC-Trend. Anal. Chem. 110 (2019) 401-409. DOI:10.1016/j.trac.2018.11.018 |
| [4] |
H. Zhang, S. Yang, K.D. Ruyck, et al., TrAC-Trend. Anal. Chem. 114 (2019) 293-313. DOI:10.1016/j.trac.2019.03.013 |
| [5] |
A.V. Samokhvalov, I.V. Safenkova, S.A. Eremin, et al., Anal. Chem. 90 (2018) 9189-9198. DOI:10.1021/acs.analchem.8b01699 |
| [6] |
S. Wang, Y. Zhang, L. Zhang, et al., Chin. Chem. Lett. 29 (2018) 1513-1516. DOI:10.1016/j.cclet.2018.08.002 |
| [7] |
J. Ruta, S. Perrier, C. Ravelet, et al., Anal. Chem. 81 (2009) 7468-7473. DOI:10.1021/ac9014512 |
| [8] |
M.E. McCarroll, F.H. Billiot, I.M. Warner, J. Am. Chem. Soc. 123 (2001) 3173-3174. DOI:10.1021/ja005604h |
| [9] |
M.U. Kumke, L.C. Shu, L.B. McGown, et al., Anal. Chem. 69 (1997) 500-506. DOI:10.1021/ac9608230 |
| [10] |
M.D. Barkley, B.H. Zimm, J. Chem. Phys. 70 (1979) 2991-3007. DOI:10.1063/1.437838 |
| [11] |
Z. Meng, R. Song, Y. Chen, et al., Nanoscale Res. Lett. 8 (2013) 118. DOI:10.1186/1556-276X-8-118 |
| [12] |
B.C. Yin, P. Zuo, H. Huo, et al., Anal. Biochem. 401 (2010) 47-52. DOI:10.1016/j.ab.2010.02.014 |
| [13] |
S.J. Zhen, Y. Yu, C.M. Li, et al., Analyst 140 (2015) 353-357. DOI:10.1039/C4AN01433B |
| [14] |
J. Liu, Z. Cao, Y. Lu, Chem. Rev. 109 (2009) 1948-1998. DOI:10.1021/cr030183i |
| [15] |
B. Yang, X.B. Zhang, L.P. Kang, et al., Anal. Chem. 85 (2013) 11518-11523. DOI:10.1021/ac402781g |
| [16] |
Z. Zhu, C. Ravelet, S. Perrier, et al., Anal. Chem. 84 (2012) 7203-7211. DOI:10.1021/ac301552e |
| [17] |
S. Perrier, V. Guieu, B. Chovelon, et al., Anal. Chem. 90 (2018) 4236-4248. DOI:10.1021/acs.analchem.7b04593 |
| [18] |
D. Qiao, J. Xu, P. Qin, et al., Anal. Chem. 90 (2018) 7171-7175. DOI:10.1021/acs.analchem.8b01374 |
| [19] |
L. Cui, Y. Zou, N. Lin, et al., Anal. Chem. 84 (2012) 5535-5541. DOI:10.1021/ac300182w |
| [20] |
M. Zhang, Y.M. Guan, B.C. Ye, Chem. Commun. (Camb.) 47 (2011) 3478-3480. DOI:10.1039/c0cc05703g |
| [21] |
T. Jia, C. Fu, C. Huang, et al., ACS Appl. Mater. Interfaces 7 (2015) 10013-10021. DOI:10.1021/acsami.5b02429 |
| [22] |
Z. Zhu, T. Schmidt, M. Mahrous, et al., Anal. Chim. Acta 707 (2011) 191-196. DOI:10.1016/j.aca.2011.09.022 |
| [23] |
X. Pei, H. Huang, Y. Chen, et al., Talanta 154 (2016) 567-573. DOI:10.1016/j.talanta.2016.01.016 |
| [24] |
B. Chovelon, E. Fiore, P. Faure, et al., Biosens. Bioelectron. 90 (2017) 140-145. DOI:10.1016/j.bios.2016.11.049 |
| [25] |
J.A. Cruz-Aguado, G. Penner, Anal. Chem. 80 (2008) 8853-8855. DOI:10.1021/ac8017058 |
| [26] |
J. Liu, J. Yu, J. Chen, et al., Mater. Sci. Eng. C 38 (2014) 206-211. DOI:10.1016/j.msec.2014.02.001 |
| [27] |
X. Wang, M. Zou, H. Huang, et al., Biosens. Bioelectron. 41 (2013) 569-575. DOI:10.1016/j.bios.2012.09.023 |
| [28] |
J.F. Guo, R.M. Fang, C.Z. Huang, et al., RSC Adv. 5 (2015) 46301-46306. DOI:10.1039/C5RA06654A |
| [29] |
Y. Huang, S. Zhao, Z.F. Chen, et al., Chem. Commun. 48 (2012) 7480-7482. DOI:10.1039/c2cc33021k |
| [30] |
X. Xiao, Y.F. Li, C.Z. Huang, et al., Chem. Commun. 51 (2015) 16080-16083. DOI:10.1039/C5CC05902J |
| [31] |
J. Tian, Y. Wang, S. Chen, et al., Microchim. Acta 180 (2013) 203-209. DOI:10.1007/s00604-012-0919-x |
| [32] |
J. Chen, L. Wang, Y. Huang, et al., Sens. Actuators B:Chem. 290 (2019) 15-22. DOI:10.1016/j.snb.2019.03.106 |
| [33] |
J. Fu, Z. Zhang, G. Li, Chin. Chem. Lett. 30 (2019) 285-291. DOI:10.1016/j.cclet.2018.10.031 |
| [34] |
J. Chen, Y. Li, Y. Huang, et al., Microchim. Acta 186 (2019) 58. DOI:10.1007/s00604-018-3143-5 |
| [35] |
J. Chen, Y. Huang, X. Yang, et al., Anal. Chim. Acta 1023 (2018) 89-95. DOI:10.1016/j.aca.2018.02.082 |
| [36] |
J. Chen, Q. Chen, J. Chen, et al., Microchim. Acta 183 (2016) 3191-3199. DOI:10.1007/s00604-016-1972-7 |
| [37] |
J. Chen, C. Gao, A.K. Malik, et al., J. Mater. Chem. B:Mater. Biol. Med. 4 (2016) 5161-5166. DOI:10.1039/C6TB00881J |
| [38] |
Z. Li, X. Zhang, H. Tan, et al., Adv. Funct. Mater. 28 (2018) 1805026. DOI:10.1002/adfm.201805026 |
| [39] |
Q. Ma, J. Wang, Z. Li, et al., Small 15 (2019) 1804969. DOI:10.1002/smll.201804969 |
| [40] |
Y. Huang, M. Shi, L. Zhao, et al., Biosens. Bioelectron. 54 (2014) 285-291. DOI:10.1016/j.bios.2013.10.065 |
| [41] |
J. Zhao, Y. Ma, R. Kong, et al., Anal. Chim. Acta 887 (2015) 216-223. DOI:10.1016/j.aca.2015.07.006 |
| [42] |
X. Li, X. Ding, Y. Li, et al., Nanoscale 8 (2016) 9852-9860. DOI:10.1039/C6NR00946H |
| [43] |
L. Qi, Z. Yan, Y. Huo, et al., Biosens. Bioelectron. 87 (2017) 566-571. DOI:10.1016/j.bios.2016.08.093 |
| [44] |
Y. Yu, Y. Liu, S.J. Zhen, et al., Chem. Commun. (Camb.) 49 (2013) 1942-1944. DOI:10.1039/c3cc38129c |
| [45] |
Q. Yue, T. Shen, L. Wang, et al., Biosens. Bioelectron. 56 (2014) 231-236. DOI:10.1016/j.bios.2014.01.021 |
| [46] |
Y. Huang, X. Liu, L. Zhang, et al., Biosens. Bioelectron. 63 (2015) 178-184. DOI:10.1016/j.bios.2014.07.036 |
| [47] |
Y. Huang, X. Liu, H. Huang, et al., Anal. Chem. 87 (2015) 8107-8114. DOI:10.1021/ac5041692 |
| [48] |
Y. Huang, X. Liu, M. Shi, et al., Chem.-Asian J. 9 (2014) 2755-2760. DOI:10.1002/asia.201402563 |
| [49] |
Z. Chen, H. Li, W. Jia, et al., Anal. Chem. 89 (2017) 5900-5908. DOI:10.1021/acs.analchem.7b00261 |
| [50] |
T.T. Zhao, Z.W. Peng, D. Yuan, et al., Spectrochim. Acta A. Mol. Biomol. Spectrosc. 192 (2018) 328-332. DOI:10.1016/j.saa.2017.10.072 |
| [51] |
J. Chen, Q. Chen, C. Gao, et al., J. Mater. Chem. B:Mater. Biol. Med. 3 (2015) 964-967. DOI:10.1039/C4TB01875C |
| [52] |
J. Zhao, Z. Chu, X. Jin, et al., Sens. Actuators B:Chem. 209 (2015) 116-121. DOI:10.1016/j.snb.2014.11.102 |
| [53] |
L. Wang, J. Tian, W. Yang, et al., Luminescence 31 (2016) 573-579. DOI:10.1002/bio.2997 |
| [54] |
S.J. Zhen, X. Xiao, C.H. Li, et al., Anal. Chem. 89 (2017) 8766-8771. DOI:10.1021/acs.analchem.7b00955 |
| [55] |
J. Du, L. Ye, M. Ding, et al., Analyst 139 (2014) 3541-3547. DOI:10.1039/C4AN00266K |
| [56] |
J. Zhang, J. Tian, Y. He, et al., Chem. Commun. (Camb.) 50 (2014) 2049-2051. DOI:10.1039/C3CC49424A |
| [57] |
Y. He, J. Tian, J. Zhang, et al., Biosens. Bioelectron. 55 (2014) 285-288. DOI:10.1016/j.bios.2013.12.032 |
| [58] |
Y. Jiang, J. Tian, K. Hu, et al., Microchim. Acta 181 (2014) 1423-1430. DOI:10.1007/s00604-014-1296-4 |
| [59] |
X. Xiao, J. Tao, H.Z. Zhang, et al., Biosens. Bioelectron. 85 (2016) 822-827. DOI:10.1016/j.bios.2016.05.091 |
| [60] |
Y.Y. Fan, Z.L. Mou, M. Wang, et al., Anal. Chem. 90 (2018) 13708-13713. DOI:10.1021/acs.analchem.8b04107 |
| [61] |
O. Wakao, Y. Fujii, M. Maeki, et al., Anal. Chem. 87 (2015) 9647-9652. DOI:10.1021/acs.analchem.5b01164 |
| [62] |
A.Y. Boroduleva, J.J. Manclús, Á. Montoya, et al., Anal. Bioanal. Chem. 410 (2018) 6923-6934. DOI:10.1007/s00216-018-1296-z |
| [63] |
L. Oberleitner, S.A. Eremin, A. Lehmann, et al., Anal. Methods 7 (2015) 5854-5861. DOI:10.1039/C5AY00617A |
| [64] |
B. Glahn-Martínez, E. Benito-Peña, F. Salis, et al., Anal. Chem. 90 (2018) 5459-5465. DOI:10.1021/acs.analchem.8b00780 |
| [65] |
L. Oberleitner, U. Dahmen-Levison, L.A. Garbe, et al., J. Environ. Manage. 193 (2017) 92-97. DOI:10.1016/j.jenvman.2017.01.063 |
| [66] |
D. Shen, J. Zheng, X. Cui, et al., Sens. Actuators B:Chem. 256 (2018) 846-852. DOI:10.1016/j.snb.2017.10.013 |
| [67] |
C. Li, Y. Zhang, S.A. Eremin, et al., Food Chem. 227 (2017) 48-54. DOI:10.1016/j.foodchem.2017.01.058 |
| [68] |
X. Zhang, S.A. Eremin, K. Wen, et al., J. Agric. Food Chem. 65 (2017) 2240-2247. DOI:10.1021/acs.jafc.6b05614 |
| [69] |
F. Wang, J. Cai, S.A. Eremin, et al., Food Anal. Methods 11 (2018) 2455-2462. DOI:10.1007/s12161-018-1236-8 |
| [70] |
X. Zhang, Q. Tang, T. Mi, et al., Food Control 87 (2018) 100-108. DOI:10.1016/j.foodcont.2017.12.002 |
| [71] |
C. Li, K. Wen, T. Mi, et al., Biosens. Bioelectron. 79 (2016) 258-265. DOI:10.1016/j.bios.2015.12.033 |
| [72] |
N.V. Beloglazova, S.A. Eremin, Talanta 142 (2015) 170-175. DOI:10.1016/j.talanta.2015.04.027 |
| [73] |
C. Li, T. Mi, G.O. Conti, et al., J. Agric. Food Chem. 63 (2015) 4940-4946. DOI:10.1021/acs.jafc.5b01845 |
| [74] |
P. Huang, S. Zhao, S.A. Eremin, et al., Anal. Methods 7 (2015) 4246-4251. DOI:10.1039/C5AY00818B |
| [75] |
B. Dong, S. Zhao, H. Li, et al., Food Chem. 271 (2019) 9-17. DOI:10.1016/j.foodchem.2018.07.147 |
| [76] |
A. Fiene, Y. Baqi, J. Lecka, et al., Analyst 140 (2015) 140-148. DOI:10.1039/C4AN01694G |
| [77] |
J. Tian, L. Zhou, Y. Zhao, et al., Talanta 92 (2012) 72-77. DOI:10.1016/j.talanta.2012.01.051 |
| [78] |
Y. He, J. Tian, K. Hu, et al., Anal. Chim. Acta 802 (2013) 67-73. DOI:10.1016/j.aca.2013.10.011 |
| [79] |
Y. Huang, J. Chen, S. Zhao, et al., Anal. Chem. 85 (2013) 4423-4430. DOI:10.1021/ac3037443 |
| [80] |
Y. Zhang, B. Zheng, C.F. Zhu, et al., Adv. Mater 27 (2015) 935-939. DOI:10.1002/adma.201404568 |
| [81] |
J. Chen, H. Qiu, M. Zhang, et al., Biosens. Bioelectron. 68 (2015) 550-555. DOI:10.1016/j.bios.2015.01.054 |
| [82] |
J. Chen, Y. Huang, M. Shi, et al., Talanta 109 (2013) 160-166. DOI:10.1016/j.talanta.2013.02.003 |
| [83] |
J. Chen, Y. Huang, M. Vdovenko, et al., Talanta 138 (2015) 59-63. DOI:10.1016/j.talanta.2015.02.004 |
| [84] |
J. Chen, Y. Guo, Q. Chen, et al., Chinese J. Anal. Chem. 43 (2015) 1513-1519. DOI:10.1016/S1872-2040(15)60871-4 |
| [85] |
L. Wang, J. Tian, Y. Huang, et al., Microchim. Acta 183 (2016) 2147-2153. DOI:10.1007/s00604-016-1844-1 |
| [86] |
S. Liang, G. He, J. Tian, et al., Microchim. Acta 185 (2018) 119. DOI:10.1007/s00604-018-2673-1 |
| [87] |
X. Li, N. Huang, L. Zhang, et al., Talanta 202 (2019) 297-302. DOI:10.1016/j.talanta.2019.05.006 |
| [88] |
L. Qi, Y.Y. Fan, H. Wei, et al., Sens. Actuators B:Chem. 257 (2018) 666-671. DOI:10.1016/j.snb.2017.11.036 |
| [89] |
L. Mo, J. Li, Q. Liu, et al., Biosens. Bioelectron. 89 (2017) 201-211. DOI:10.1016/j.bios.2016.03.044 |
| [90] |
K.D. Bhatt, D.J. Vyas, B.A. Makwana, et al., Chin. Chem. Lett. 27 (2016) 731-737. DOI:10.1016/j.cclet.2016.01.012 |
| [91] |
H. Ye, N. Duan, H. Wang, et al., Microchim. Acta 186 (2019) 173. DOI:10.1007/s00604-019-3261-8 |
| [92] |
C. Zhang, L. Wang, Z. Tu, et al., Biosens. Bioelectron. 55 (2014) 216-219. DOI:10.1016/j.bios.2013.12.020 |
 2019, Vol. 30
2019, Vol. 30