b Key Laboratory of Bioorganic Chemistry and Molecular Engineering of Ministry of Education and Beijing National Laboratory for Molecular Science (BNLMS), and Peking-Tsinghua Center for Life Sciences, Peking University, Beijing 100871, China
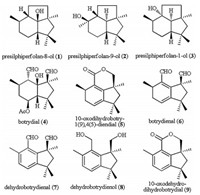
Presilphiperfolanols (1-3 in Fig. 1) are a family of structurally diverse sesquiterpenoids, which were first isolated in 1981 from Eriophyllum staechadifolium, a flowering plant commonly known as seaside woolly sunflower [1]. Presilphiperfolanols have a compact, high-energy tricyclic presilphiperfolane skeleton bearing five contiguous stereocenters, two all-carbon quaternary centers, and a tertiary hydroxyl group. These structural features enable presilphiperfolanols to undergo thermodynamically favorable cationic skeletal rearrangements to yield diverse sesquiterpenoid skeletons, which exist in numerous biologically important natural products [2]. Botryanes (4-9 in Fig. 1), sesquiterpenoids with excellent structural diversity, are derived from the phytopathogenic fungus Botrytis cinerea [3]. The botryane skeleton is a bicyclic sesquiterpenoid, a nonisoprenoid system, which has been the subject of many investigations, especially with regard to its biosynthetic pathway [4]. Recent experiments have shown that presilphiperfolanols and botryanes are biosynthetically related. Botryane metabolites [5] differ in substitution pattern, degree of saturation, and altered sites of oxidation, alkylation, unsaturation, etc., and they show a wide range of biological activities, including phytotoxic, antibiotic and cytotoxic activities [3a, 4].
|
Download:
|
| Fig. 1. Presilphiperfolanols and botryanes. | |
Given the importance of presilphiperfolanol and botryane natural products in sesquiterpene biosynthesis and their potential biological activities, much effort has been undertaken for their synthesis [6]. In 2015, we reported the total syntheses of 7-epi-presilphiperfolan-1-ol and 4-epi-presilphiperfolan-8-ol by employing Co-tetramethylthiourea (TMTU)-catalyzed tandem Pauson–Khand and 6π-electrocyclization reactions as the key step [7]. Herein, we describe the application of this reaction to the total syntheses of dehydrobotrydienal (7), dehydrobotrydienol (8), and 10-oxodehydrodihydrobotrydial (9).
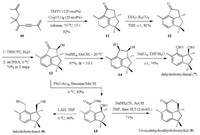
We commenced the synthesis with the Co-TMTU-catalyzed tandem Pauson–Khand and 6π-electrocyclization reactions of trienyne 10 to furnish tricyclic compound 11 in 92% yield [7] (Scheme 1). Compound 11 was converted to the corresponding aromatic ketone 12 by 2, 3-dichloro-5, 6-dicyano-1, 4-benzoquinone (DDQ)-mediated oxidative aromatization [8]. α-Hydroxylation of ketone 12 was realized using a two-step sequence: conversion of 12 to the corresponding silyl enol ether by treatment with trifluoromethanesulfonic acid tert-butyldimethylsilylester (TBSOTf) in the presence of Et3N, and oxidation by meta-chloroperoxybenzoic acid (mCPBA) to afford compound 13 in 70% overall yield. Reduction of 13 with NaBH4 in methanol provided a cis-diol 14 in 97% yield (dr = 10:1), which was then treated with NaIO4 [9] to give botryane 7 in 76% yield. Oxidation of 13 with Pb(OAc)4 [10] afforded aldehyde 15 in 83% yield. With 15 in hand, we proceeded to investigate the synthesis of botryanes 8 and 9. Treatment of 14 with LiAlH4 in THF produced 8 in 96% yield. The details were deposited in Supporting information. Selective reduction of 15 with NaBH3CN [11], followed by lactonization under acidic conditions, gave 9 in 71% yield. The NMR spectra of compounds 7–9 (Supporting information) were in good agreement with the data reported for the natural sample [3].
|
Download:
|
| Scheme 1. Total syntheses of botryanes 7-9. | |
In summary, we have achieved the concise total syntheses of three botryanes: dehydrobotrydienal, dehydrobotrydienol, and 10-oxodehydrodihydrobotrydial. The key to the success of this protocol is the application of our recently developed Co-TMTU-catalyzed tandem Pauson–Khand and 6π-electrocyclization reactions to construct the core structure of the botryanes. Further investigations on the application of this chemistry in natural product synthesis are in progress, and the results will be reported in due course.
AcknowledgmentsThis work was supported by the National Natural Science Foundation of China (Nos. 21772008, 21632002 and U1606403), Natural Science Foundation of Guangdong Province (No. 2016A030306011), Shenzhen Basic Research Program (Nos. JCYJ20170818090044432 and JCYJ20160226105337556) and Qingdao National Laboratory for Marine Science and Technology (No. LMDBKF201703).
Appendix A. Supplementary dataSupplementary material related to this article can be found, in the online version, at doi:https://doi.org/10.1016/j.cclet.2019.03.033.
| [1] |
(a) A.Y. Hong, B.M. Stoltz, Angew. Chem. Int. Ed. 53 (2014) 5248-5260; (b) F. Bohlmann, C. Zdero, J. Jakupovic, H. Robinson, R.M. King, Phytochemistry 20 (1981) 2239-2244. |
| [2] |
(a) J.R. Hanson, Pure Appl. Chem. 53 (1981) 1155-1162; (b) C.M. Wang, R. Hopson, X. Lin, D.E. Cane, J. Am. Chem. Soc.131 (2009) 8360-8361; (c) R.M. Coates, Z. Ho, M. Klobus, S.R. Wilson, J. Am. Chem. Soc. 118 (1996) 9249-9254; (d) P. Weyerstahl, H. Marschall, I. Seelmann, J. Jakupovic, Eur. J. Org. Chem. (1998) 1205-1212; (e) C.E. Davis, B.C. Duffy, R.M. Coates, J. Org. Chem. 68 (2003) 6935-6943. |
| [3] |
(a) I.G. Collado, A.J. Sanchez, J.R. Hanson, Nat. Prod. Rep. 24 (2007) 674-686; (b) I.G. Collado, J. Aleu, R. Hernández-Galán, R. Durcn-Patrón, Curr. Org. Chem. 4 (2000) 1261; (c) H.W. Fehlhaber, R. Geipel, H.J. Mercker, R. Tschesche, K. Welmar, F. Schönbeck, Chem. Ber. 107 (1974) 1720-1730; (d) I.G. Collado, R. Hernández-Gálan, R. Durán-Patrón, J.M. Cantoral, Photochemistry 38 (1995) 647-650; (e) F. Bohlmann, J. Ziesche, R.K. Gupta, Photochemistry 21 (1982) 1331-1334; (f) F. Bohlmann, C. Zdero, Photochemistry 21 (1982) 2537-2541; (g) A.H. Mericli, J. Jakupovic, F. Bohlmann, X.A. Dominguez, H.S. Vega, Photochemistry 28 (1989) 1149-1153. |
| [4] |
J.L. Reino, Rosa Durán-Patrón, I. Segura, et al., J. Nat. Prod. 66 (2003) 344-349.
|
| [5] |
K. Krohn, J. Dai, U. Flörke, et al., J. Nat. Prod. 68 (2005) 400-405. DOI:10.1021/np0498206 |
| [6] |
(a) P. Weyerstahl, H. Marschall, M. Schulze, I. Schwope, Liebigs Ann. (1996) 799-807; (b) A.Y. Hong, B.M. Stoltz, Angew. Chem. Int. Ed. 51 (2012) 9674-9678; (c) C. Qiao, W. Zhang, J.C. Han, C.C. Li, Org. Lett. 18 (2016) 4932-4935; (d) C. Qiao, W. Zhang, J.C. Han, W.M. Dai, C.C. Li, Tetrahedron 75 (2019) 1739-1745. |
| [7] |
Z. Zhang, Y. Li, D. Zhao, et al., Chem. Eur. J. 23 (2017) 1258-1262. DOI:10.1002/chem.201605438 |
| [8] |
P. Tharra, B. Baire, Chem. Commun. 52 (2016) 12147-12150. DOI:10.1039/C6CC06639A |
| [9] |
E.E. Maciver, P.C. Cridland, A.L. Thompson, M.D. Smith, Chem. Sci. 3 (2012) 537-540. DOI:10.1039/C1SC00697E |
| [10] |
F.A. Khan, J. Dash, Sudhee Ch., Chem. Commun. 10 (2004) 2507-2519. |
| [11] |
S. Basu, V. Gupta, J. Nickel, C. Schneider, Org. Lett. 16 (2014) 274-277. DOI:10.1021/ol403275k |
 2019, Vol. 30
2019, Vol. 30 

