b Wuya College of Innovation, Shenyang Pharmaceutical University, Shenyang 110016, China
Mitochondria, as important subcellular organelles in eukaryotic cells, play a critical role in many cellular processes, including the generation of metabolic energy, cellular differentiation and cell death [1]. Mitochondrial damage is well known to promote the development and progression of various diseases, such as cancer, diabetes, neurodegenerative diseases, heart failure and ischemiareperfusion injury. Therefore, mitochondria have long been used as therapeutic targets to improve the treatment outcome of therapeutics [2, 3].
In recent years, some research groups, including ours, have revealed that mitochondria-targeted delivery could enhance the protective potential of therapeutics against ototoxic agents, such as cisplatin and aminoglycosides [4-7]. The most common targeted delivery is predominantly based on the modification of lipophilic cations, which direct the molecules to mitochondria via mitochondrial inner membrane potential. Despite improving efficacy, some lipophilic cations were recognized to exhibit potential toxicity due to their accumulation in mitochondria which causes membrane depolarization [8, 9]. Recently, extensive research has focused on identifying new motifs or sequences that confer mitochondria-specific delivery properties.
Notably, Szeto and Schiller accidentally discovered a line of tetrapeptides with highly selective mitochondrial targeting properties when working on synthetic opioid molecules with superior affinity and selectivity for the μ-opioid receptor [10]. SS-02 and SS-31 are two typical peptides of this class, which are able to penetrate a variety of cell types without the need for specific transporters or receptors. Later studies have indicated that their uptake is independent of the mitochondrial potential, yielding less side effects on mitochondria [11]. Compared with SS-02, SS-31 with Dmt residue in the second position displays mitochondrial uptake ability but negligible affinity for opioid receptors. More importantly, the promising antioxidant activity owing to the Dmt residue strenghtens the therapeutic potential of SS-31 in the treatment of mitochondrial dysfunction-associated diseases [10]. We previously successfully utilized SS-31 as a targeting sequence to induce PLGA nanoparticles for mitochondrial uptake, resulting in improved protective efficacy against aminoglycosides in common formulations. Nevertheless, absolute protection was absent for the designed delivery system, probably due to the moderate targeting efficacy [12].
In the present study, we designed and synthesized a series of SS-31 analogues with the aim to improve the antioxidant efficacy and mitochondrial targeting ability of inducing drug delivery system of the lead compound, which were known to be important for the treatment of mitochondrial dysfunctional diseases. To gain a systematic insight into mitochondrial-targeting peptide-mediated delivery, we firstly synthesized a small library of SS-31 analogs. The aim of optimization was to provide controlled toxicity, favorable antioxidant activity and suitable mitochondrial delivery. We concluded that the RF-2 peptide indeed facilitates the mitochondrial delivery of PLGA NPs in a non-toxic manner.
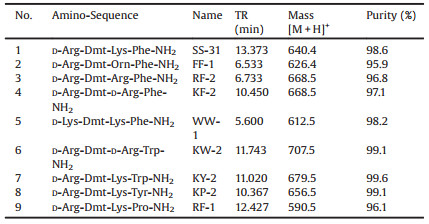
The study was conducted in accordance with the Basic & Clinical Pharmacology & Toxicology policy for experimental and clinical studies [13]. Peptides in this study were synthesized by the previously described manual solid-phase procedure, using techniques for Nα-Fmoc-protected or Boc(t-butyloxycarbonyl)-protected amino acids on Rink amide resin (0.03 μmol scale) and p-methylbenzhydrylamine (MBHA) resin (1% cross-linked, 100~200 mesh, 0.3 μmol/g, Aladdin Industrial Corporation). The Nε-amino groups of Lys and D-Lys were protected by Fmoc (9-fluorenylmethyloxycarbonyl), and the hydroxy group of Tyr by 2-Br-Z (2- bromobenzyloxycarbonyl). Peptides were assembled using Nα- Fmoc-protected amino acids and 1-hydroxybenzotriazole (HOBt)/ 1, 3-diisopropylcarbodiimide (DIC)/N, N-diisopropylethylamine (DIEA) or 1-hydroxybenzotriazole (HOBt)/1-(3-dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride (EDC·HCl)/4-dimethylaminopyridine (DMAP) as coupling agents. The side chains of Dmt were also Fmoc-protected. The phenolic hydroxyl group of Dmt was unprotected. Other side chain protection was as follows: tBu for Tyr and Cys, Boc for D-Lys, Trp, L-Phe and Orn, Pbf for Arg and DArg. A 4-fold excess of building blocks (Fmoc-L-Pro-OH, Fmoc-Orn (Boc)-OH, Fmoc-D-Lys(Boc)-OH, Fmoc-Arg(Pbf)-OH, Fmoc-D-Arg (Pbf)-OH, Fmoc-Lys(Boc)-OH, Fmoc-L-Phe(Boc)-OH, Fmoc-Trp (Boc)-OH, Fmoc-Tyr(tBu)-OH, Fmoc-Cys(Trt)-OH, Fmoc-Cys (tBu)-OH, Fmoc-Tyr-OH and Fmoc-Dmt-OH) and activating agents was applied, and dry DMF was used as a solvent. Fmoc deprotections were carried out by treatment of the resin with 95% piperidine in DMF, twice, for 30 min each. Fmoc-Dmt-OH was coupled with 1, 3-diisopropylcarbodiimide (DIC) and 1-hydroxybenzotriazole (HOBt) to avoid side reactions when uroniumbased coupling reagents were used. Final cleavage of the peptide, as well as the 2, 2, 4, 6, 7-pentamethyldihydrobenzofuran-5-sulfonyl (Pbf) side chain protection group removal were accomplished by treatment with 95% (v/v) TFA/H2O cleavage conditions for 2 h. Crude peptides were obtained after evaporation of the cleavage mixture, precipitated in cold Et2O, and purified by reversed-phase high-performance liquid chromatography (HPLC). The purified peptides (>95% purity) were then lyophilized, and the molecular mass of peptides was analyzed by agreement with those obtained. Molecular weights were subsequently confirmed by ESI–MS. Analytical data of the peptides are presented in Table 1.
|
|
Table 1 Structure of designed peptides used in the present study. |
Larval zebrafish (Danio rerio) were produced through paired mating of the AB wild-type strains adult fish from the China Zebrafish Resource Center (Wuhan, China). Animals were tested at 5–7 days post-fertilization (dpf) and held in an incubator in E3 embryo medium (EM, consisting of 5 mmol/L NaCl, 0.17 mmol/L KCl, 0.33 mmol/L CaCl2 and 0.33 mmol/L MgSO4; pH 7.2) at 28.5 ℃, during treatments unless otherwise noted. Embryos/larvae were raised at 28.5 ℃, at a density of 50 larvae per 100-mm2 petri dish. All experimental protocols were performed on animals in accordance with the guidelines of the Animal Care Ethics Committee of Shenyang Pharmaceutical University Medical Center and the National Institutes of Health.
To examine the potential toxicity of designed peptides, zebrafish larvae were incubated with each peptide at 100 or 50 μg/mL for 6 h (n = 12 for each group). After exposure, the larvae were observed under a stereomicroscope, and lethal endpoints were recorded.
The protective effect of peptides against gentamicin was screened as we previously reported [12]. The evaluated peptide (~75 μmol/L) was imposed on zebrafish for 1 h before acute exposure to gentamicin (200 μmol/L, 1 h), followed by another 1 h recovery. For rapid assessment of hair cell survival, the larvae were immersed in 1 μmol/L of DiOC2(3) for 30 min, then rinsed 4 × with EM and anesthetized with 0.02% MS-222. DiOC2(3) labelling was then evaluated on a Leica epifluorescent microscope (Chroma Technologies, Brattleboro, VT) for 4 neuromasts (SO2, SO3, O1 and MI1). The area of labelled neuromasts was quantified per animal, summed to calculate one value per animal, and averaged for each group (Image-Pro Plus). Results were presented as the mean area of neuromast, as a percentage of the group treated only with EM. The treatment of gentamicin-only was used as negative control. To further highlight the protective effects, we also used SYTOX Green (5 μmol/L, 1 min) for hair cell counting. The applied protocol was conducted as descibed above.
To further compare the protective effects of optimal peptides, the larvae were pretreated with optimal peptides for chronic exposure to gentamicin (6 h). Various concentrations of gentamicin (2, 5, 10, 15 μmol/L) were individually administrated to larvae for chronic exposure after peptide pretreating for 1 h and cotreatment for 6 h. TMRE (1 nmol/L, 20 min) was applied to stain remaining neuromasts for dose-response function models and pharmacological evaluation. Images were analyzed by Image-Pro Plus by drawing an area of interest (AOI) around individual neuromasts with background substracting, after which the mean fluorescent intensity within the AOI region was exported for analyses. The remaining area of stained neuromasts was then calculated by Image-Pro Plus 6.0.
To assess the targeting efficiency of RF-2, PLGA NPs were prepared through the emulsification solvent evaporation method, according to the procedures reported previously. In brief, PLGAmPEG and PLGA-PEG-Mal at various mass ratios (total 20 mg) were dissolved in dichloromethane (1 mL) to prepare MPP-NPs bearing different ratios of peptides, and the oil phase was added to 2 mL of 1% CHA under probe sonication (75% amplitude), for 3 min, in an ice bath. The preliminary emulsion was added dropwise to another 20 mL of 0.5% sodium cholate solution under magnetic stirring. Organic solvents were removed by reduced pressure distillation, and NPs were then filtrated through a 0.8-μm membrane, and subsequently collected by centrifugation at 12, 000 rpm for 30 min at 4 ℃, followed by careful washing 3 times with deionized water. To prepare the NPs for fluorescence imaging, PLGA13000-PEG3400- rhodamine (1% of total polymer) was mixed with unlabeled polymers prior to emulsification in the dark.
For the fabrication of peptide-modified NPs, the sulfhydryl group of RF-2 or SS-31 with a cysteine on the N-terminal was linked to the maleimide function located at the distal end of PEG on the NP surface, at a molar ratio of 1:1. The reaction was conducted with gentle shaking, for 1 h, at 37 ℃. The free peptide was subsequently removed by centrifugation at 10, 000 rpm for 30 min. The size, polydispersity index (PDI), and ζ-potential of the NPs were determined by dynamic light scattering (DLS, Nano-ZS, Malvern Instruments, UK). The morphology of NPs was observed by transmission electron microscopy (TEM, H7600 Hitachi, Japan).
The human breast adenocarcinoma cell line A549 was grown in DMEM medium supplemented with 10% fetal bovine serum (FBS) and antibiotics (1% (v/v) penicillin-streptomycin). The cells were cultured in a humidified incubator, at 37 ℃ and 5% CO2. The medium was replaced every other day.
A549 cells were seeded in 12-well plates (1.5 × 105 cells per well) and cultured in DMEM for 24 h. For co-localization of the tested formulations and mitochondria, the cells were treated with labelled PLGA NPs or peptide-modified NPs for 2 h or 6 h. Then, the cells were washed with PBS and incubated with Mitotracker Green (35 nmol/L) for 30 min. After staining, the cells were washed with PBS three times, then fixed with 4% paraformaldehyde for 15 min at 37 ℃. Lastly, the cells were observed using a confocal C2-si laser scanning microscope (CLSM, Nikon C2 Plus, Japan).
All data obtained in the present study were expressed as the mean ± standard deviation (SD). Statistical analysis was conducted using the two-tailed unpaired t-test for comparison between two groups. One-way ANOVA was performed for three or more experimental groups, followed by the Tukey multiple comparison test, using GraphPad Prism. P values < 0.05 were considered statistically significant.
To gain a systematic understanding of the impact of peptide sequence on the mitochondria targeting ability of peptides, we designed and synthesized a small library of SS-31 analogues by replacing aromatic and cationic residues (Table 1) [14]. Previous reports indicated that the alternation of aromatic and cationic residues plays key roles in the mitochondrial targeting ability of peptides. D-amino acid in either the first or third position was used to minimize degradation by peptidase and increase stability in serum. Since the peptide-modulated modification of NPs is based on the sulfhydryl group of CPP with a cysteine on the N-terminal and the maleimide function located at the distal end of PEG on the NP surface, a peptide containing 5 residues was also synthesized by only adding a cysteine on the N-terminal of SS-31.
To evaluate the potential toxicity of the designed peptides, the zebrafish larvae were incubated with the peptides for 6 h, and then the lethality was recorded (Table 2). The percentage of dead larvae was calculated for two peptide concentrations (100 or 50 μg/mL), since SS-31 at 100 μg/mL was found to provide favorable protective effect against gentamicin. As shown in Table 2, there were no dead fish counted after exposure to SS-31, CSS-31 or RF-2, at either of the two investigated levels. In contrast, FF-1, KP-2 and RF-1 exhibited toxicity as indicated by the dead larvae observed during the incubation time. Additionally, lethality reached 100% when the larvae were exposed to 100 μg/mL KF-2, WW-1, KW-2 or KY-2, and dead larvae were still observed at 50 μg/mL, indicating considerable toxicity. Notably, RF-2 and KF-2 share nearly the same sequence of amino acids, but with one difference in the L or D of the third position, which led to significant variations in their toxicities. Similarly, KW-2, which contains D-amino acid in the third position, also induced severe toxic effects upon the larvae. The possible explanation for the different toxic effects observed could stem from the oxidation of D-amino acids by D-amino acid oxidase, which produces α-keto acids with hydrogen peroxide as a byproduct [15]. The latter is reactive oxygen with high permeability to cellular and nuclear membranes, interrupting DNA, proteins and lipids via the produced hydroxyl radical. Chen et al. found that the activities of D-amino acid oxidase were expressed in a dynamic and stage-dependent manner, reaching a maximum amount in zebrafish at 5–6 dpf [16]. This may explain the toxic sensitivity observed in the present study. In mammals, D-amino acid oxidase is expressed in low levels, but still exists in some organs and tissues, such as the central nervous system [17]. Hence, the peptides containing more than one D-amino acid should be avoided when designing novel peptides.
|
|
Table 2 The toxicity of designed peptides in zebrafish. |
Hydrophobic peptides with similar cationic structures might severely permeabilize the cell membrane, thus affecting cell viability and inducing toxicity. To address this possibility, the HPLC retention times of designed peptides were measured (Table 1), however, no obvious relationship between lipophilicity and toxicity was observed. Compared with KP-2, the peptide KY-2 showed rather high toxicity to larvae, indicating the potential effect of Trp. This effect and its corresponding mechanism were further investigated in the following experiments.
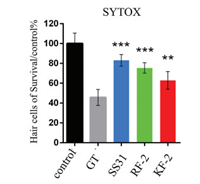
The zebrafish lateral line is emerging as an excellent in vivo model for the physiological analysis of hair cells and neurons [18]. Particularly, the hair cells in the zebrafish lateral line exhibited similar sensitivity to ototoxic agents to that observed in mammal. Hence, the protective potency of designed antioxidant peptides against acute exposure to gentamicin was then evaluated in hair cells, and the results are shown in Fig. 1. Except for WW-1, all the test peptides exhibited remarkable protective effect at the concentration of 50 μg/mL. However, SS-31 exhibited superior efficacy over the other peptides at the test level, followed by RF-2, CSS-31, FF-1, KF-2, RF-1, KP-2, KY-2 and KW-2. The pharmacological potency of RF-2 was also confirmed by direct hair cells counting via SYTOX labeling (Fig. 2). The results clearly demonstrated that RF-2 protected the hair cells against 200 μmol/L acute gentamicin exposure, with gentamicin alone killing approximately 50% of the hair cells and RF-2 conferring over 70% protection. The potency of RF-2 was still weaker than that of SS-31, but stronger than that of KF-2.

|
Download:
|
| Fig. 1. The protective effects(Acute Exposure)of peptide at concentration of 50 μg/mL on hair cell damage, induced by gentamicin, with menbrane potential dye DiOC2(3). (Data were analyzed by one way ANOVA using SPSS, **P < 0.01, ***P < 0.001). | |
|
Download:
|
| Fig. 2. The protective effects of peptide at concentration of 50 μg/mL on hair cell damage induced by acute exposure gentamicin. The hair cells were counted by SYTOX staining. (Data were analyzed by one way ANOVA using SPSS, **P < 0.01, ***P < 0.001). | |
RF-2 was then selected for further evaluation due to its low risk of toxicity and relatively greater otoprotection upon screening. To determine the effectiveness of otoprotection, RF-2 was compared with SS-31 in providing protection against 2–15 μmol/L continuous gentamicin exposure (Fig. 3). Overall, RF-2 offered better protection than SS-31 against chronic exposure to gentamicin in the range of 2–10 μmol/L, however, this was no longer observed when the level of gentamicin was increased to 15 μmol/L. It was recently discovered that SS-31 selectively binds to cardiolipin of mitochondrial inner membrane via both electrostatic and hydrophobic interactions. Specially, the interaction between peptide and cardiolipin lies in the electrostatic interaction between the two basic amino acids on SS-31 or RF-2 and the phosphate head group of cardiolipin aligns the aromatic residues within the hydrophobic acyl chain region. A couple of SS-31 analogues were designed and synthesized by introducing alternating aromatic and cationic residues in the molecules. The insertion of the aromatic groups into the lipid environment might be optimal for the antioxidant potency: a small aromatic group (Phe or Dmt) reserved the affinity with the hydrophobic acyl chain region of cardiolipin, whereas an increased size of aromatic group (Trp or Tyr in KW-2, KY-2, KP-2) led to the loss of the affinity. More importantly, basicity was prerequisite for high activity of peptides and several basic residues with different pKa values were assessed. It was demonstrated that at least one Arg was required for the electrostatic interaction between peptides and cardiolipin. In general, guanidinium heads are known to enhance the membrane interaction of cell-penetrating peptides via hydrogen bonding with phospholipids [19, 20]. The two guanidinium heads and aromatic hydrophobic residues in RF-2 could make the cationic, amphipathic constructs more permeable.

|
Download:
|
| Fig. 3. The protective effectsof peptide SS-31 and RF-2 on hair cell damage induced by chronic exposure of gentamicin, with mitochondrial hair cells membrane potential dye TMRE. (Data were analyzed by one way ANOVA using SPSS, *P < 0.05, **P < 0.01, ***P < 0.001). | |
To study the behavior of optimal peptide-carrying NPs, PLGA NPs conjugated with RF-2 or SS-31 were synthesized and their physicochemical characteristics were evaluated. The particle size of both peptide-modified NPs was comparable to that of the unmodified control. Besides, due to the cationic profiles of designed peptides, the zeta potential of RF-2- or SS-31-modified NPs was approximately -5~-7 mV, which was lower than that of unmodified NPs (~20 mV).
The co-localization of the peptide-modified NPs and MitoTracker Green in A549 cells was determined by confocal microscopy. Overall, both types of peptide-modified NPs showed greater cellular uptake than the unmodified NPs, suggesting that the two peptides promoted cellular penetration. The merged (yellow) red and green fluorescence indicated that the peptide-modified NPs can enter the mitochondria. The cellular uptake of NPs increased with time. In particular, RF-2- and SS-31-modified NPs exhibited strong red fluorescence that overlapped with the green fluorescence signals from Mitotracker after 6 h of incubation (Fig. 5, compared with 2 h of incubation as shown in Fig. 4), indicating that these peptides enabled the selective targeting of mitochondria. In contrast, the unmodified NPs exhibited low red fluorescence, likely due to their low membrane permeability.

|
Download:
|
| Fig. 4. Co-localization of rhodamine-labeled mPEG-PLGA NPs, RF-2 NPs and SS-31 NPs on A549 cells. Cells were treated with mPEG-PLGA NPs, RF-2 NPs and SS-31 NPs for 2 h respectively. Yellow area in the merged picture denoted the co-localization of the NPs (labeled with rhodamine, red fluorescence) with mitochondrial compartments (stained with Mitotracker Green, green fluorescence). Confocal microscopy was used to observe the co-localization of the modified NPs into the mitochondria and PLGA NPs as comparison. Scale bar =10 μm. | |

|
Download:
|
| Fig. 5. Co-localization of rhodamine-labeled mPEG-PLGA NPs, RF-2 NPs and SS-31 NPs on A549 cells. Cells were treated with mPEG-PLGA NPs, RF-2 NPs and SS-31 NPs for 6 h respectively. Yellow area in the merged picture denoted the co-localization of the NPs (labeled with rhodamine, red fluorescence) with mitochondrial compartments (stained with Mitotracker Green, green fluorescence). Confocal microscopy was used to observe the co-localization of the modified NPs into the mitochondria and PLGA NPs as comparison. Scale bar = 10 μm. | |
In summary, in the present study, we developed a small library of SS-31 analogues, and identified a cationic, amphipathic tetrapeptide (RF-2) that specifically accumulates in mitochondria. RF-2 exhibited controlled toxicity even at high concentrations, as well as favorable antioxidant activity. Thus, RF-2 was regarded as a potential mitochondrial inducer for the delivery of therapeutic cargoes to this organelle. The present findings might provide useful guidance in designing novel cellular or subcellular penetrating peptides for not only small molecular candidates but also the development of drug delivery systems in the treatment of mitochondrial dysfunctional diseases.
AcknowledgmentsThis study was financially supported by the National Natural Science Foundation of China (No. 81473162), the Program for Liaoning Excellent Talents in University and the Young and Middleaged Career Development Planning of Shenyang Pharmaceutical University.
| [1] |
J. Zielonka, J. Joseph, A. Sikora, et al., Chem Rev. 117 (2017) 10043-10120. DOI:10.1021/acs.chemrev.7b00042 |
| [2] |
R.K. Pathak, N. Kolishetti, S. Dhar, Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 7 (2015) 315-329. DOI:10.1002/wnan.1305 |
| [3] |
I. Pezzini, V. Mattoli, G. Ciofani, Expert. Opin. Drug Deliv. 14 (2017) 513-523. DOI:10.1080/17425247.2016.1218461 |
| [4] |
R. Esterberg, T. Linbo, S.B. Pickett, et al., J. Clin. Invest. 126 (2016) 3556-3566. DOI:10.1172/JCI84939 |
| [5] |
C.P. Ojano-Dirain, P.J. Antonelli, Laryngoscope 122 (2012) 2543-2548. |
| [6] |
C.P. Ojano-Dirain, P.J. Antonelli, C.G. Le Prell, Otol. Neurotol. 35 (2014) 533-539. DOI:10.1097/MAO.0000000000000192 |
| [7] |
Z. Wang, X. Kuang, J. Shi, et al., Biomater. Sci. 5 (2017) 1800-1809. DOI:10.1039/C7BM00224F |
| [8] |
S.E. Abu-Gosh, N. Kolvazon, B. Tirosh, et al., Mol. Pharm. 6 (2009) 1138-1144. DOI:10.1021/mp900032r |
| [9] |
M.P. Murphy, Trends Biotechnol. 15 (1997) 326-330. DOI:10.1016/S0167-7799(97)01068-8 |
| [10] |
H.H. Szeto, A.V. Birk, Clin. Pharmacol. Ther. 96 (2014) 672-683. DOI:10.1038/clpt.2014.174 |
| [11] |
H.H. Szeto, Br. J. Pharmacol. 171 (2014) 2029-2050. DOI:10.1111/bph.12461 |
| [12] |
X. Kuang, S. Zhou, W. Guo, et al., Drug Deliv. 24 (2017) 1750-1761. DOI:10.1080/10717544.2017.1402220 |
| [13] |
P. Tveden-Nyborg, T.K. Bergmann, J. Lykkesfeldt, Basic Clin. Pharmacol. Toxicol. 123 (2018) 233-235. |
| [14] |
C.P. Cerrato, M. Pirisinu, E.N. Vlachos, et al., FASEB J. 29 (2015) 4589-4599. DOI:10.1096/fj.14-269225 |
| [15] |
E. Rosini, L. Caldinelli, L. Piubelli, Front. Mol. Biosci. 4 (2017) 102. |
| [16] |
Y.H. Chen, W.L. Chen, Y.H. Wang, et al., Fish Physiol Biochem. 33 (2007) 73-80. DOI:10.1007/s10695-006-9118-1 |
| [17] |
J. Sasabe, Y. Miyoshi, S. Rakoff-Nahoum, et al., Nat. Microbiol. 1 (2016) 16125. DOI:10.1038/nmicrobiol.2016.125 |
| [18] |
H. Ou, J.A. Simon, E.W. Rubel, et al., Hear. Res. 288 (2012) 58-66. DOI:10.1016/j.heares.2012.01.009 |
| [19] |
C. Bechara, S. Sagan, FEBS Lett. 587 (2013) 1693-1702. DOI:10.1016/j.febslet.2013.04.031 |
| [20] |
E.I. Geihe, C.B. Cooley, J.R. Simon, et al., Proc. Natl. Acad. Sci. U. S. A. 109 (2012) 13171-13176. DOI:10.1073/pnas.1211361109 |
 2019, Vol. 30
2019, Vol. 30