b College of Animal Science and Veterinary Medicine, Shandong Agricultural University, Taian 271018, China;
c School of Chemistry and Chemical Engineering, Shandong University, Ji'nan 250100, China;
d State Key Laboratory for Chemistry and Molecular Engineering of Medicinal Resources, School of Chemistry and Pharmaceutical Sciences of Guangxi Normal University, Guilin 541004, China
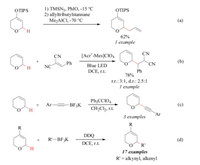
α-Substituted 3, 6-dihydro-2H-pyrans (DHPs) are common structural motifs in a number of biologically active natural products and synthetic pharmaceuticals [1]. Their current synthesis predominantly relies on four types of oxygen-heterocycle construction approaches: (1) Prins-type cyclization of homopropargylic alcohols and aldehydes [2]; (2) [4 + 2] cycloaddition of dienes and aldehydes [3]; (3) ring-closing metathesis [4]; (4) intramolecular alkene alkoxylation [5]. Despite wide synthetic applications, these methods rely heavily on the functional group transformations. On the other hand, a great number of methods have been established for the facile access to the DHP skeletons with diverse substituent patterns. Accordingly, direct manipulation of the DHP skeletons with a multitude of readily available coupling components through the structural-core diversification strategy represents an attractive alternative to traditional oxygenheterocycle construction strategy. Pallidium-catalyzed Heck reaction of 3, 4-dihydro-2H-pyrans represents a practical protocol for α- substituted DHP synthesis. However, the method is only suitable for the access to α-aryl substituted DHPs [6]. In this context, the Ferrier reaction involving the substitution of C-O bonds with C-C bonds at C1 position of DHP-based acetals is the most widely adopted approach [6, 7]. However, the method suffers from extra steps for pre-installation of the acetal functionality.
Direct C-H functionalization of DHPs represents an ideal approach to access the target with a minimal amount of intermediary refunctionalizations and with high atom economy [8-10]. Magnus has reported an isolated example of one-pot allylation of DHP-based triisopropylsilyl enol ethers with allyl tri-n-butylstannane and Me2AlCl (Scheme 1a) [11]. Wu disclosed one elegant example of photoredox induced C-H alkylationof DHP with electron-deficient benzylidenemalononitrile (Scheme 1b) [12]. Our group documented a Ph3CClO4 mediated C-H functionalization of DHP with potassium (phenylethynyl)boronate (Scheme 1c) [13]. Albeit great innovation, these studies still suffer from the employment of toxic organotin, limited DHP scope and α-substituent pattern, or expensive oxidative reagent. On the other hand, alkynes are common structural elements pervading the realms of biology, chemistry, material science, and medicine and serve as valuable building blocks due to their versatile chemical reactivities [14]. Given the importance of 2, 4-disubstituted DHPs in pharmaceutical science [1 e-g], a systematic study on the C-H alkynylation of 4-substituted DHPs using readily available oxidant would be a highly attractive project to pursue (Scheme 1d).
|
Download:
|
| Scheme 1. Overview of C-H functionalization of DHPs. | |
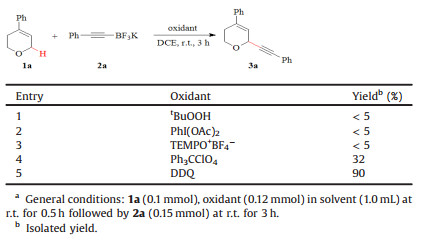
Initially, the C-H alkynylation of 4-phenyl substituted DHP 1a with boronate 2a was selected for the optimization of suitable oxidant (Table 1). Common oxidants, including tBuOOH, PhI(OAc)2, and TEMPO+BF4- (TEMPO = 2, 2, 6, 6-tetramethylpiperidin-1-oxyl) failed to effect the coupling (entries 1–3). Ph3CClO4 promoted the reaction, and expected 3a was isolated in 32% yield (entry 4). When DDQ (2, 3-dichloro-5, 6-dicyano-1, 4-benzoquinone) was used, the reaction proceeded smoothly, affording 3a in 90% yield.
|
|
Table 1 Reaction condition optimization.a |
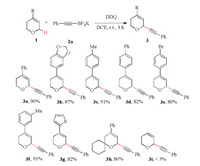
The substituent effect on DHPs 1 was explored (Scheme 2). DHPs bearing either electron-donating (1b-1d) or -withdrawing aryl moieties (1e) at the C4-position were found to be well compatible with the oxidation condition, providing corresponding 3b-3e in high efficiency. DHP 1f with a meta-methyl substituted aryl group at the C4-position proved to be a suitable substrate for the C-H alkynylation process, and 3f was obtained in 91% yield. DHP 1g having a heteroaryl moiety at the C4-position was well tolerated, and 2-thiophenyl substituted 3g was isolated in 82% yield. DHPs bearing a spirocycle at α positionwere also competent components, as demonstrated by the formation ofmultiple-substituted 3h in 80% yield. No reactionwas observed for DHP 1i, which might be ascribed to the increased oxidation potential of the substrate.
|
Download:
|
| Scheme 2. Scope of DHP components. Reaction conditions: 1a (0.1 mmol), 2a (0.12 mmol), and DDQ (0.11 mmol) in DCE (1.0 mL) at r.t. for 3 h. The yield refers to isolated yield. | |
The scope of alkynyl boronates 2 was next investigated (Scheme 3). Electronically varied potassium (arylethynyl) boronates 2b-2f participated in the oxidative coupling with DHP 1a smoothly, affording corresponding 4b-4f in good yields. Heteroarylacetylenes were competent components for the process, as demonstrated by the efficient generation of 4g in 83% yield. Alkylacetylene 2h also proved to be a suitable component, and expected 4h was obtained in 80% yield. Alkylacetylene 2i bearing benzyl ether was well tolerated for further manipulation. A gramscale C-H alkynylation of 1a proceeded in 83% yield, thus indicating the practicability of the method.

|
Download:
|
| Scheme3. Scope of alkynyl boronates. aReaction in a 1.0 g scale. | |
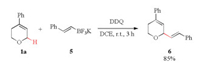
Besides the alkynylation process, C-H alkenylation of DHP 1a with 5 was well tolerated under the standard oxidation condition, furnishing the expected 6 in 85% yield (Scheme 4). No expected C-H arylation or allylation product was observed when respective aryl or allyl potassium trifluoroborate was used as the coupling component.
|
Download:
|
| Scheme 4. Oxidative C-H alkenylation of DHP 1a. | |
The synthetic utilities of the protocol were next examined (Scheme 5). The alkene moiety in DHPs can act as a reactive handle for diverse functionalization, as exemplified by the efficient hydrogenation of 4c and diastereoselective epoxidation of 3a, giving respective tetrahydropyrans 7 and 8 with diverse substituent patterns.

|
Download:
|
| Scheme5. Synthetic utilities. | |
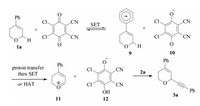
According to the mechanistic studies on DDQ-mediated benzylic ether oxidation [15], a mechanism for C–H alkynylation of DHPs was suggested (Scheme 6). DHPs 1a proceeded through a single electron transfer (SET) to DDQ provided radical cation 9 together with DDQ radical anion 10. The radical cation 9 might either undergo a proton abstraction by 10 followed by another SET or a hydrogen atom transfer (HAT) to 10, furnishing α, β-unsaturated oxocarbenium ion 11. Subsequent nucleophilic attack of potassium trifluoroborates onto 11 afforded the expected product 12.
|
Download:
|
| Scheme6. A proposed mechanism. | |
In conclusion, an efficient and practicalmethodfor the preparation of α-substituted DHPs is described. Under DDQ-mediated mild metal-free conditions, diverse DHPs undergo oxidative C–H alkynylation and alkenylation with a range of potassium trifluoroborates smoothly, rapidly providing a library of 2, 4-disubstituted DHPs with diverse patterns of α-functionalities for further diversification and bioactive small molecule identification.
AcknowledgmentsThis work was financial supported by the National Natural Science Foundation of China (No. 21722204), Fok Ying Tung Education Foundation (No. 151035), the Key Laboratory for Chemistry and Molecular Engineering of Medicinal Resources (Guangxi Normal University) (No. CHEMR2016-B09), and Guangxi Funds for Distinguished expert.
Appendix A. Supplementary dataSupplementary material related to this article can befound, in the online version, at doi:https://doi.org/10.1016/j.cclet.2019.03.027.
| [1] |
(a) M.M. Faul, B.E. Huff, Chem. Rev. 100 (2000) 2407-2474; (b) E.J. Kang, E. Lee, Chem. Rev. 105 (2005) 4348-4378; (c) T. Nakata, Chem. Rev. 105 (2005) 4314-4347; (d) S.D. Roughley, A.M. Jordan, J. Med. Chem. 54 (2011) 3451-3479; (e) L.G. León, P.O. Miranda, V.S. Martín, J.I. Padrón, J.M. Padrón, Bioorg. Med. Chem. Lett. 17 (2007) 3087-3090; (f) T.A. Johnson, K. Tenney, R.H. Cichewicz, et al., J. Med. Chem. 50 (2007) 3795-3803; (g) J. De Pascual Teresa, S. Vincente, M.S. Gonzalez, I.S. Bellido, Phytochemistry 22 (1983) 2235-2238. |
| [2] |
(a) P.O. Miranda, D.D. Díaz, J.I. Padrón, J. Bermejo, V.S. Martín, Org. Lett. 5 (2003) 1979-1982; (b) P.O. Miranda, R.M. Carballo, V.S. Martín, J.I. Padrón, Org. Lett. 11 (2009) 357-360; (c) Y. Lian, R.J. Hinkle, J. Org. Chem. 71 (2006) 7071-7074. |
| [3] |
(a) T.L. Gresham, T.R. Steadman, J. Am. Chem. Soc. 71 (1 949) 737-738; (b) K. Fujiwara, T. Kurahashi, S. Matsubara, J. Am. Chem. Soc. 134 (2012) 5512-5515; (c) L. Liu, H. Kim, Y. Xie, et al., J. Am. Chem. Soc. 139 (2017) 13656-13659. |
| [4] |
(a) B.M. Trost, B.S. Brown, E.J. McEachern, O. Kuhn, Chem.-Eur. J. 9 (2003) 4442-4451; (b) S. Basu, H. Waldmann, J. Org. Chem. 71 (2006) 3977-3979. |
| [5] |
(a) W. Kong, J. Cui, Y. Yu, et al., Org. Lett. 11 (2009) 1213-1216; (b) K.P. Kalbarczyk, S.T. Diver, J. Org. Chem. 74 (2009) 2193-2196; (c) K. Watanabe, J. Li, N. Veerasamy, A. Ghosh, R.G. Carter, Org. Lett. 18 (2016) 1744-1747; (d) B. Guo, G. Schwarzwalder, J.T. Njardarson, Angew. Chem. Int. Ed. 51 (2012) 5675-5678. |
| [6] |
(a) I.P. Beletskaya, A.V. Cheprakov, Chem. Rev. 100 (2000) 3009-3066; (b) G. Peh, P.E. Floreancig, Org. Lett. 17 (2015) 3750-3753. |
| [7] |
A.M. Gómez, F. Lobo, C. Uriel, J. Cristóbal López, Eur. J. Org. Chem. 2013 (2013) 7221-7262. |
| [8] |
(a) K. Godula, D. Sames, Science 312 (2006) 67-72; (b) W.R. Gutekunst, P.S. Baran, Chem. Soc. Rev. 40 (2011) 1976-1991; (c) R. Giri, B.F. Shi, K.M. Engle, N. Maugel, J.Q. Yu, Chem. Soc. Rev. 38 (2009) 3242-3272; (d) J. Robertson, J. Pillai, R.K. Lush, Chem. Soc. Rev. 30 (2001) 94-103; (e) H.M.L. Davies, Angew. Chem. Int. Ed. 45 (2006) 6422-6425; (f) S.Y. Zhang, F.M. Zhang, Y.Q. Tu, Chem. Soc. Rev. 40 (2011) 1937-1 949. |
| [9] |
(a) C.J. Li, Z. Li, Pure Appl. Chem. 78 (2006) 935-945; (b) S. Murahashi, D. Zhang, Chem. Soc. Rev. 37 (2008) 1490-1501; (c) C.J. Li, Acc. Chem. Res. 42 (2009) 335-344; (d) C.J. Scheuermann, Chem.-Asian J. 5 (2010) 436-451; (e) M. Klussmann, D. Sureshkumar, Synthesis (2011) 353-369; (f) C. Liu, H. Zhang, W. Shi, A. Lei, Chem. Rev. 111 (2011) 1780-1824; (g) C.L. Sun, B.J. Li, Z.J. Shi, Chem. Rev. 111 (2011) 1293-1314; (h) C. Zhang, C. Tang, N. Jiao, Chem. Soc. Rev. 41 (2012) 3464-3484; (i) S.H. Cho, J.Y. Kim, J. Kwak, S. Chang, Chem. Soc. Rev. 40 (2011) 5068-5083; (j) J. Le Bras, J. Muzart, Chem. Rev. 111 (2011) 1170-1214; (k) C.S. Yeung, V.M. Dong, Chem. Rev. 111 (2011) 1215-1292; (l) R. Rohlmann, O. García Mancheño, Synlett 24 (2013) 6-10; (m) S.A. Girard, T. Knauber, C.J. Li, Angew. Chem. Int. Ed. 53 (2014) 74-100. |
| [10] |
(a) Y. Zhang, C.J. Li, Angew. Chem. Int. Ed. 45 (2006) 1949-1952; (b) Y. Zhang, C.J. Li, J. Am. Chem. Soc. 128 (2006) 4242-4243; (c) M. Ghobrial, K. Harhammer, M.D. Mihovilovic, M. Schnürch, Chem. Commun. 46 (2010) 8836-8838; (d) H. Richter, R. Rohlmann, O. García Mancheño, Chem.-Eur. J. 17 (2011) 11622-11627; (e) S.J. Park, J.R. Price, M.H. Todd, J. Org. Chem. 77 (2012) 949-955; (f) D.J. Clausen, P.E. Floreancig, J. Org. Chem. 77 (2012) 6574-6582; (g) X. Liu, B. Sun, Z. Xie, X. Qin, L. Liu, H. Lou, J. Org. Chem. 78 (2013) 31 04-3112; (h) W. Chen, Z. Xie, H. Zheng, H. Lou, L. Liu, Org. Lett. 16 (2014) 5988-5991; (i) Z. Meng, S. Sun, H. Yuan, H. Lou, L. Liu, Angew. Chem. Int. Ed. 53 (2014) 543-547; (j) W. Muramatsu, K. Nakano, Org. Lett. 16 (2014) 2 042-2 045; (k) M. Xiang, Q.Y. Meng, X.W. Gao, et al., Org. Chem. Front. 3 (2016) 486-490; (l) Z. Peng, Y. Wang, Z. Yu, et al., J. Org. Chem. 83 (2018) 7900-7906; (m) L. Jin, J. Feng, G. Lu, C. Cai, Adv. Synth. Catal. 357 (2015) 2105-2110. |
| [11] |
P. Magnus, J. Lacour, P.A. Evans, P. Rigollier, H. Tobler, J. Am. Chem. Soc. 120 (1998) 12486-12499. DOI:10.1021/ja9829564 |
| [12] |
R. Zhou, H. Liu, H. Tao, X. Yu, J. Wu, Chem. Sci. 8 (2017) 4654-4659. DOI:10.1039/C7SC00953D |
| [13] |
M. Wan, Z. Meng, H. Lou, L. Liu, Angew. Chem. Int. Ed. 53 (2014) 13845-13849. DOI:10.1002/anie.201407083 |
| [14] |
F. Diederich, P.J. Stang, R.R. Tykwinski, Acetylene Chemistry:Chemistry, Biology and Material Science. Weinheim: Wiley-VCH, 2005.
|
| [15] |
H.H. Jung, P.E. Floreancig, Tetrahedron 65 (2009) 10830-10836. DOI:10.1016/j.tet.2009.10.088 |
 2019, Vol. 30
2019, Vol. 30