b Institute of Polymers and Energy Chemistry(IPEC), Nanchang University, Nanchang 330031, China
In recent years, organic polymer solar cells, with advantages of low-cost, light weight and flexibility, have achieved rapid development and attracted great attention in organic electronics [1-11]. They have large commercial value for the generation of inexpensive, clean, and sustainable energy [12-17]. An organic photovoltaic device adopts a donor and an acceptor (fullerene derivatives [3, 18-20], conjugated polymers [21-23], or organic small molecules [24, 25]) through solution-processing method for highly efficient PSCs. To enhance the photovoltaic performance of PSCs, a series of novel polymer donors have been developed by donor (D) unit and acceptor (A) segment copolymerization [26-29]. The highly efficient conjugated polymer donor can not only adjust micro-structured phase separation morphology to increase Jsc, but also improve charge transport ability to raise FF, as well as formation of an interpenetrating network structure with acceptor derivatives, e.g., ITIC [30], perylene diimides (PDIs) [31-35] and naphthalene diimides (NDIs) homologues [36-39], etc. However, the development of random conjugated polymers (donors and acceptors) [40-47] lags behind that of D-A copolymers, including the types of molecules and the efficiency of photovoltaic devices. One of the main reasons is due to relatively difficult to adjust and control morphology, resulting in the unsatisfactory efficiency of organic solar cells (OSCs). Thus, attempting novel molecule design is necessary, which plays an important role on the optimized topography to improve the efficiency of organic photovoltaics (OPVs).
In devices, the optimization of active layer morphology is an important process. In order to shape ideal micro-phase morphology of bi-continuous interpenetrating networks, several approaches of optimized active layer morphology have been adopted such as small amount of solvent additives [48, 49], thermal annealing (TA) [50], and solvent vapor annealing (SVA) [51, 52]. In recent literature, representative high-boiling-point liquid additives 1, 8-octanedithiol (ODT) [53, 54], 1, 8-diiodooctane (DIO) [55-57], nitrobenzene (NB) [58], N-methylpyrrolidone (NMP) [11, 59], 1-chloronapthalene (CN) [60, 61], 1-phenylnaphthalene (PN) [17], 1, 2, 3, 4-tetrahydronaphthalene (THN) and 1-methyl-naphthalene (MN) [62], etc. have been used in the D/A photovoltaic active layer. The micro-nanomorphology of the active layer have been successfully operated by these additives, and the high efficiency of OSCs have been remarkably enhanced. With the assist of these additives, PCEs of exceeding 14% have been obtained in single-junction OSCs [63].
However, the different content of additive is added to the parent solvent to increase PCE value, which is a cockamamie procedure, and the reproducibility of the photovoltaic efficiency is also destroyed by these additives, because efficiency of the device is very sensitive to additive ratio and structure, and the residual high-boiling-point additive in the photovoltaic active layer displayed the tardiness drying process, generating unsatisfactory topographic variety [64]. More noteworthy is that residual additive also immensely expedites the photo-oxidative degradation and observably debases the lifetime of the photovoltaic device [65]. In order to remove these additives, several experimental procedures have been carried out using high-vacuum, thermal annealing at high temperature and rinsing the active layer film with methanol. However, these methods cannot eradicate some of the problems mentioned above caused by these additives.
In this study, we attempt the random copolymerization method, with an electron-rich planar conjugated unit benzo [1, 2-b:4, 5-b']dithiophene, with two-dimensional conjugated thienyl side chains (BDT) and different ratios of two electrondeficient segments benzo[1, 2-c:4, 5-c']dithiophene-4, 8-dione (BDD) and difluorobenzothiadizole (BT) linked alkylthiophene, to construct a series of PBDB-TBTn random terpolymer. The aromatic conjugated BT unit with alkylthiophene has stronger electron pull-push effect and intermolecular π-π interaction, leading to red-shifted absorption, which is favor for photoresponse and Jsc value. Thus, we added different proportion of BT into the polymer PBDB-T system to modulate morphology of active layers, ultimately to further increase photovoltaic performance of non-fullerene PSCs, with ITIC as acceptor. In addition to characterization of random copolymer absorption and electrochemical properties, we fabricate the PBDB-TBTn:ITIC-based photovoltaic devices without and with 1% DPE and 1% DPE + 100 ℃ to compare their performance, exciton separation probability, light-intensity dependence, charge transport and morphology. We found that PBDB-TBT1 with 10% BT unit displays the optimal photovoltaic performance, achieving a PCE of 9.09% with Voc of 0.86 V, Jsc of 16.84 mA/cm2, and FF of 63%. Through a series of studies, we demonstrated the PBDB-TBTn-based devices are insensitive to the process with 1% DPE and 1% DPE + 100 ℃ as well as that without solvent additive. And a simple and practiced synthesized method are provided to obtain random copolymers in high efficiency non-fullerene PSCs.
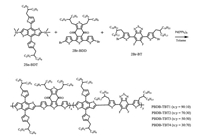
The synthetic routes and chemical structures of the PBDB-TBTn random copolymers are shown in Scheme 1. Various progressive proportions of acceptor monomers (BDB:BT = 90:10, 70:30, 50:50, and 30:70) were carried out during palladium-catalyzed Stille copolymerization, which generate random copolymers PBDB-TBT1, PBDB-TBT2, PBDB-TBT3 and PBDB-TBT4, respectively. The number average molecular weight (Mn) and polydispersity index (PDI) of a series of random copolymers PBDB-TBT1, PBDB-TBT2, PBDB-TBT3, and PBDB-TBT4 are 17.8/2.73 kDa, 13.4/2.86 kDa, 17.0/1.95 kDa, and 16.3/ 2.02 kDa, respectively, which were measured by high temperature gel permeation chromatography at 160 ℃ using 1, 2, 4-trichlorobenzene as the flowing solvent and polystyrene with narrow molecular weight distribution as a standard (Fig. S1 in Supporting information). Details of synthesis and properties are summarized in the experimental section of supporting information. Note that PBDB-TBT2-PBDB-TBT4 showed limited solubility due to their higher content of the rigid coplanar fused-ring BT unit with strong aggregation in this conjugated backbone, and thus four random copolymer donors were selected as compared objects in this study.
|
Download:
|
| Scheme 1. Synthetic routes of four PBDB-TBTn random copolymers. | |
Fig. 1 shows the absorption spectra of all random polymer donors in chloroform and films with ITIC as comparation. The corresponding optical data, including the absorption peaks (λabs), absorption edge wavelength (λonset) and their optical band gaps (Eg) are outlined in Table 1. Each random polymer shows similar absorption tendency in its solution and solid-state spectra. All polymers have three or four absorption peaks in the range of 300 nm to 700 nm. Absorption spectroscopy of PBDB-TBT1, PBDB-TBT2 and PBDB-TBT3 display four main wave profiles, composed of a π-π* transition peak of electron-rich group and a π-π* transition peak of electron-deficient units in the short wavelength region of 300– 500 nm, a strong intramolecular charge-transfer transition (ICT) peak and a π-π stacking peak, which is the shoulder and intense shape in the visible range of 500–700 nm.With increasing the ratios of BT segment, the π-π stacking peak weaken, showing only the ICT peak from 500 nm to 700 nm in solution and films, which indicates the destruction of π-π aggregation in the random copolymer films. However, the absorption spectra display the similar process for PBDB-TBT4 in solution and the neat films. Absorption of PBDB-TBT1, PBDB-TBT2 and PBDB-TBT3 film displays fourobvious peaks at 300– 700 nm, respectively, with absorption edge around 696 nm, and Eg of 1.78 eV, while absorption edge of PBDB-TBT4 shows at 708 nm, corresponding to Eg of 1.75 eV. Obviously, all the four random copolymers displayed better complementary absorption with that of ITIC acceptor in the vis-NIR range, covering a broader range from 300 nm to 780 nm. The external quantum efficiency (EQE) further confirms the tuned and matched complementary absorption of the polymer donor and the ITIC acceptor for subsequent discussion section.

|
Download:
|
| Fig. 1. (a) Chemical structures of four PBDB-TBTn random terpolymer donors, and small molecule acceptor ITIC. (b) Energy level diagrams of four PBDB-TBTn random copolymers, and ITIC. (c) UV–vis spectra of PBDB-TBTn random copolymers in chloroform solution; (d) UV–vis absorption spectra of four neat polymer films and ITIC film. | |
|
|
Table 1 Optical and electrochemical properties of all random copolymers. |
The orbital energy levels of all four random conjugated copolymer films were carried out through electrochemical cyclicvoltammetry (CV). CV plots of the random copolymer films are displayed in Fig. S2 (Supporting information) and is outlined in Table 1, respectively, which is calculated from the onset reduction and onset oxidation potentials (Ered and Eox). The highest occupied molecular orbital (HOMO) and the lowest unoccupied molecular orbital (LUMO) electron energy levels of four random copolymers can be investigated from the Ered and Eox values according to the equations [66]: HOMO = -e(Eox + 4.71) (eV); LUMO = -e(Ered + 4.71) (eV). The tested LUMO and HOMO levels are -3.42/ -5.30 eV, -3.45/-5.33 eV, -3.47/-5.35 eV, and -3.45/-5.36 eV for random copolymers PBDB-TBT1-PBDB-TBT4, respectively, as outlined in Table 1. Obviously, The HOMO levels of all random conjugated copolymers are gradually down-shifted, which could be mainly due to the increase of BT content with the strong electron-deficient ability. Thus, from the viewpoint of electronic energy level, all four random copolymers are suitable donors with ITIC as small molecular acceptor.
The photovoltaic properties of random conjugated PSCs were investigated with the single-junction device structure ITO/ZnO/ random copolymer:ITIC/MoO3/Ag. The photovoltaic devices were fabricated by using chlorobenzene (CB) as solvent to dissolve the random copolymer:ITIC mixture. Through changing ratios of donor and aceeptor copolymer, we found that the optimized blend films random copolymer:ITIC (D/A) ratios is 1:1. To furtherenhance device performance, we also attemped to add solvent additive of 1 vol% diphenyl ether (DPE) and 100 ℃ thermal annealing (TA). To one's delight, the efficiencies of device have not been improved and are obtained the similar results as that without solvent additive treatment.
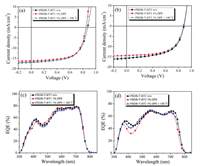
The photovoltaic performances of random conjugated tercopolymer solar cells were tested under the illumination of AM 1.5 G, 100 mW/cm2. Fig. 2 and Fig. S3 (Surporting information) show the current density-voltage (J-V) plots of the photovoltaic devices with D/A ratios (1:1), 1 vol% DPE solvent additive and 100 ℃ TA for PBDB-TBT1, PBDB-TBT2, PBDB-TBT3 and PBDB-TBT4, respectively, the corresponding photovoltaic parameters with Jsc, Voc, FF, and PCE are outlined in Table 2. The photovoltaic property of copolymer PBDB-TBT1:ITIC-based device without any treatment exbibits the optimized PCE value of 9.09% with an Voc of 0.86 V, a Jsc of 16.84, and an FF of 62.85%, as shown in Fig. 2a. This PCE value is similar to that of 8.82% and 9.01% with 1% DPE and 1% DPE + 100 ℃ TA process, respectively. The devices based on PBDB-TBT2 without, with 1% DPE and 1% DPE + 100 ℃ treatment display the PCE value of 7.95%, 7.53%, and 7.91%, respectively (Fig. 2b). With increasement of BT proportion, the efficiencies of PBDB-TBT3, PBDB-TBT4-based devices are gradully reduced to 5.93% and 2.09%, respectively (Table 2, Figs. S3a and b). The reducement of these PCE values are mainly ascribled to the lowed Jsc and FF. Obviously, these device results indicated that the efficiencies of copolymer is insensitive to solvent additive and thermal annealing for non-fullerene PSCs.
|
Download:
|
| Fig. 2. J-V (a, b) and EQE (c, d) plots of the PBDB-TBT1 and PBDB-TBT2-based photovoltaic devices without, with 1% DPE and 1% DPE + 100 ℃ treatment, respectively. | |
|
|
Table 2 Photovoltaic performance of random conjugated copolymer solar cells based on PBDB-TBTn:ITIC (1:1, w/w) under the illumination of AM1.5 G, 100 mW/cm2. |
The external quantum efficiency (EQE) spectra of non-fullerene random copolymer solar cell devices are shown in Fig. 2. PBDBTBT1 without other treatment displays the highest EQE values of 79%, compared with that of the other copolymers, convering the whole wavelength range from 300 nm to 800 nm, which is similar to these with 1% DPE additive (EQE value of 78%) and 1% DPE + 100 ℃ mixed treatment (EQE value of 77%), as shown in Fig. 2c. The EQE value of PBDB-TBT2-based device without, with 1% DPE additive and 1% DPE + 100 ℃ mixed treatment is 10% lower than that of device based on PBDB-TBT1:ITIC, as shown in Fig. 2d. However, random copolymers PBDB-TBT3 and PBDB-TBT4 exhibit Obviously, These results are consistent with the performance meatured that the PBDB-TBT1-based device with the highest Jsc value of all the photovoltaic devices. The Jsc values of EQE spectra are obtained by integrating area of the EQE curves, which are reasonable with the Jsc values determined from the J-V curves (Table 2), the obtained data indicated that property of the J-V measurements are highly trustworthy.
In order to further investigate the effect of different proportion of BT content on the photovoltaic property of random copolymer solar cells based on PBDB-TBTn:ITIC, we adopted the spacecharge-limited current (SCLC) method to characterize the charge carrier mobilities for PBDB-TBTn:ITIC device without, with the solvent additive treatment of 1% DPE and 1% DPE + 100 ℃ process. The current-voltage (J1/2 -V) curves for the electron and hole mobility measurements were displayed in Figs. S4 and S5 (Supporting information), respectively, and the electron (μe) and hole (μh) mobility values were summarized in Table S1 (Supporting information). The μe and μh values of PBDB-TBT1:ITIC blend without any process are 4.12 × 10-4 cm2 V-1 s-1and 4.74 ×10-4 cm2 V-1 s-1, respectively. Through adding solvent additive and TA treatment, we found that this blend is in identical order of magnitude as that without any process. For PBDB-TBT2: ITIC blend, the electron mobilities without, with the solvent additive treatment of 1% DPE and 1% DPE + 100 ℃ mixed process are 4.23 ×10-4, 1.31 ×10-4 and 2.11 ×10-4 cm2 V-1 s-1, the corresponding hole mobilities are 4.58 × 10-4, 1.83 × 10-4 and 2.15 ×10-4 cm2 V-1 s-1, respectively. It is important to note that μe and μh values of PBDB-TBT1:ITIC blend are obviously superior to that of PBDB-TBT2:ITIC-based device. When BT content gradually increase to 50% and 70% in this system, the μe values of blend based on PBDB-TBT3:ITIC are more serious than the μh values, decreasing to 10-5 orders of magnitude. However, The μe and μh values of PBDB-TBT3:ITIC and PBDB-TBT4:ITIC blends are all reduced to 10-5 orders of magnitude. The above results display that the charge mobilities of PBDB-TBT1:ITIC blend are highest values in all device, and played an particular important role in the enhancement of photovoltaic performance, in especial for Jsc, while the discussed results of charge carrier mobilities are also consistent with the photovoltaic performance of PBDB-TBT1:ITIC blend without, with 1% DPE additive and 1% DPE + 100 ℃ treatment. More importantly, mobilities also reflect that it is insensitive to solvent additive and thermal annealing. And hole and electron mobilities show the higher and balanced values for PBDB-TBT1:ITIC-based device without any treatment, which is propitious to restraining charge recombination and improving photovoltaic property of the devices.
To further better understand the internal charge carriers recombination processes of random conjugated PSCs based on PBDB-TBTn:ITIC blends, light-intensity-dependent Jsc and Voc measurement were carried out [67]. By adopting plots of the photocurrent density (Jph) vs. effective voltage (Veff) at Jsc conditions, we reveal exciton generation rates and the charge collection probabilities (Pdiss, determined by the normalized Jph at the saturated current density), as shown in Figs. 3a and b and Fig. S6 (Supporting information). Jph has been confirmed by calculating JL - JD, where JL represents the current densities under illumination of AM 1.5 G, 100 mW/cm2, and JD is the current densities in the dark condition. And Veff = V0 - Vappl, where V0 is the voltage at which Jph = 0, and Vappl is the applied bias voltage. We observe the saturated photocurrent at Veff = 2~4 V and the resulting Pdiss was determined to be 78%/75%, 77%/73% and 73%/ 69% for PBDB-TBT1:ITIC and PBDB-TBT2:ITIC blend without and with the treatments of 1% DPE and 1% DPE + 100 ℃ under short-circuit conditions, respectively (Figs. 3a and b). PBDB-TBT3 and PBDB-TBT4 blend without any treatment display the Pdiss values of 74% and 65% (Fig. S6), respectively. Obviously, by comparison with all devices without and with 1% DPE solvent additive and 1% DPE + 100 ℃ mixed treatment, the highest Pdiss value of 78% for the PBDB-TBT1:ITIC blend without other treatment is consistent with the prominent photovoltaic property of the device. More importantly, these results demonstrate that all devices are insensitive to solvent additive and thermal annealing for random copolymer solar cells.
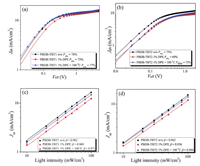
|
Download:
|
| Fig. 3. Jph-Veff (a, b) and Jsc-P (c, d) curves of the devices based on PBDB-TBT1:ITIC and PBDB-TBT2:ITIC, respectively. The solid lines are the fitted lines in the Jsc-P plots. | |
To more understand the effect of non-geminate recombination losses to photovoltaic performance, the light-intensity (P) dependences of Jsc and Voc were investigate to follow the relationship between Jsc and light intensity, Jsc∝Pβ [68]. The β values were estimated to be 0.982, 0.971, and 0.969, respectively from the Jsc-P plots in Fig. 3c and d for PBDB-TBT1:ITIC blend without and with the treatments of 1% DPE and 1% DPE + 100 ℃ respectively. And PBDB-TBT2:ITIC blend film also showed the declined β values around 0.962, 0.960, and 0.956 (Fig. 3d). However, the highest β values of PBDB-TBT3 and PBDB-TBT4 without any treatment from Figs. S7a and b (Supporting information) displayed 0.942 and 0.935, respectively. The β value of 0.982 (near to 1.00) for the PBDB-TBT1:ITIC device without any process foreboded that it was negligible to the bimolecular recombination in this device. These data also indicated that bimolecular recombination were availably suppressed at Jsc conditions for the PBDB-TBT1:ITIC devices without any treatment [2].
To further gain insight into the effect of morphology of random copolymer blend on photovoltaic performance by atomic force microscopy (AFM) and transmission electron microscopy (TEM) images to estimate surface and internal phase situation of blend films, as shown in Fig. 4. The surface roughness emerged legible changes for PBDB-TBTn:ITIC blends without, with solvent additive and thermal annealing, suggesting that the two phase aggregation of donor and acceptor was reduced to some extent, due to the compatibility difference of random copolymer donor with different proportions of BT units and acceptor in the CB solutionor solventadditive.The surface roughness of the PBDB-TBT1:ITIC and PBDB-TBT2:ITIC blend without, with the treatment of 1% DPE additive and 1% DPE + 100 ℃ process were 3.50 nm/2.58 nm, 2.12 nm/6.44 nm and 2.53 nm/4.24 nm, respectively, as shown in Figs. 4a–f. In addition, the PBDB-TBT3:ITIC and PBDB-TBT4:ITIC films also displayed the same tendency with PBDB-TBT1 and PBDB-TBT2 blends, the corresponding surface roughness were 1.95 nm/2.00 nm, 15.90 nm/9.49 nm and 8.85 nm/ 4.63 nm, respectively (Figs. S8a–f in Supporting information). The results of AFM images indicated that the suitable surface aggregation is beneficial to enhancing charge carrier transport and further improving photovoltaic performance of the device. From TEM images observed (Figs. 4g–l and Figs. S9a–f in Supporting information), it shows well-distributed microstructural morphologies, forming appropriate phase separation with a smaller domain size of around 10–20 nm for the PBDB-TBT1:ITIC blend. The result is consistent with the highest device performance obtained from PBDB-TBT1 blend film without any process.

|
Download:
|
| Fig. 4. AFM height (a–f) and TEM (g–l) images of the PBDB-TBT1:ITIC and PBDB-TBT2:ITIC blend films (1:1, w/w) without (a, d, g, j) and with the additive treatment of 1% DPE (b, e, h, k) and 1% DPE + 100 ℃ process (c, f, i, l), respectively. | |
In summary, an effective pattern was carried out in the additvie-free non-fullerene PSCs with a high PCE of 9.09% by adopting the PBDB-TBTn random copolymers with two simple electron-deficient segments BDD and BT as donors and ITIC as an acceptor. Elaborative tailoring of the content of BDD and BT units in the PBDB-TBT backbone generated an outstanding change in the molecular morphology, resulting in a significant influence on the optical bandgaps and energy levels. Careful regulation of the molecular topography lead to different photovoltaic performance of the device based on tercopolymers PBDB-TBTn as donor with ITIC as an acceptor. Furthermore, a uniform phase separation features with a micro-nanostructure was observed in the best-optimizing PBDB-TBT1:ITIC device, thereby enabling efficacious exciton dissociation, negligible bimolecular recombination and preferable charge carrier transport. These factors played an important role in realizing a remarkably increased Jsc of 16.84 mA/cm2 through this conjugated skeleton system. These results emphasize that the random copolymerization technique is a dexterous methodology for the abundant types of polymer donors match for the non-fullerene acceptors and emerge a bright future for the application and develop high efficiency non-fullerene PSCs based on random conjugated terpolymers.
AcknowledgmentsThis work was supported by the National Natural Science Foundation of China (Nos. 21602150, 51763017, 51863012, 21861025, 51833004), Natural Science Foundation of Jiangxi Province in China (Nos. 20171ACB21012 and 2018ACB21022) and the Innovation Fund Designated for Graduate Students of Nanchang University (No. CX2018028).
Appendix A. Supplementary dataSupplementary material related to this article can befound, in the online version, at doi:https://doi.org/10.1016/j.cclet.2019.04.029.
| [1] |
G. Yu, J. Gao, J.C. Hummelen, F. Wudl, A.J. Heeger, Science 270 (1995) 1789. DOI:10.1126/science.270.5243.1789 |
| [2] |
Y. Zhong, M.T. Trinh, R. Chen, et al., Nat. Commun. 6 (2015) 8242. DOI:10.1038/ncomms9242 |
| [3] |
Y. Liu, J. Zhao, Z. Li, et al., Nat. Commun. 5 (2014) 5293. DOI:10.1038/ncomms6293 |
| [4] |
X. Ouyang, R. Peng, L. Ai, X. Zhang, Z. Ge, Nat. Photonics 9 (2015) 520. DOI:10.1038/nphoton.2015.126 |
| [5] |
C. Sun, F. Pan, H. Bin, et al., Nat. Commun. 9 (2018) 743. DOI:10.1038/s41467-018-03207-x |
| [6] |
C. Yan, S. Barlow, Z. Wang, et al., Nat. Rev. Mater. 3 (2018) 18003. DOI:10.1038/natrevmats.2018.3 |
| [7] |
P. Cheng, G. Li, X. Zhan, Y. Yang, Nat. Photonics 12 (2018) 131-142. DOI:10.1038/s41566-018-0104-9 |
| [8] |
W. Gao, M. Zhang, T. Liu, et al., Adv. Mater. 30 (2018) 1800052. DOI:10.1002/adma.v30.26 |
| [9] |
H. Yao, Y. Cui, R. Yu, et al., Angew. Chem. Int. Ed. 56 (2017) 3045-3049. DOI:10.1002/anie.201610944 |
| [10] |
A. Tang, B. Xiao, Y. Wang, et al., Adv. Funct. Mater. 28 (2018) 1704507. DOI:10.1002/adfm.v28.6 |
| [11] |
Y. Zhang, X. Guo, B. Guo, et al., Adv. Funct. Mater. 27 (2017) 1603892. DOI:10.1002/adfm.v27.10 |
| [12] |
W. Zhao, D. Qian, S. Zhang, et al., Adv. Mater. 28 (2016) 4734-4739. DOI:10.1002/adma.v28.23 |
| [13] |
Z. Zhang, L. Feng, S. Xu, et al., Adv. Sci. 4 (2017) 1700152. DOI:10.1002/advs.201700152 |
| [14] |
H. Yan, J. Chen, K. Zhou, et al., Adv. Energy Mater. 8 (2018) 1703672. DOI:10.1002/aenm.201703672 |
| [15] |
Z. Ding, X. Long, C. Dou, J. Liu, L. Wang, Chem. Sci. 7 (2016) 6197-6202. DOI:10.1039/C6SC01756H |
| [16] |
M. Li, Y. Liu, W. Ni, et al., J. Mater. Chem. A 4 (2016) 10409-10413. DOI:10.1039/C6TA04358E |
| [17] |
J. Zhao, Y. Li, G. Yang, et al., Nat. Energy 1 (2016) 15027. DOI:10.1038/nenergy.2015.27 |
| [18] |
Y. Zhang, D. Deng, Z. Wang, et al., Adv. Energy Mater. 7 (2017) 1701548. DOI:10.1002/aenm.201701548 |
| [19] |
G. Zhao, Y. He, Y. Li, Adv. Mater. 22 (2010) 4355-4358. DOI:10.1002/adma.v22:39 |
| [20] |
Y. He, H.Y. Chen, J. Hou, Y. Li, J. Am. Chem. Soc. 132 (2010) 1377-1382. DOI:10.1021/ja908602j |
| [21] |
Z.G. Zhang, Y. Yang, J. Yao, et al., Angew. Chem. Int. Ed. 56 (2017) 13503-13507. DOI:10.1002/anie.201707678 |
| [22] |
A. Yin, D. Zhang, S.H. Cheung, et al., J. Mater. Chem. C 6 (2018) 7855-7863. DOI:10.1039/C8TC02689K |
| [23] |
T. Zhang, W. Feng, W. Wang, et al., J. Mater. Chem. C 6 (2018) 8418-8428. DOI:10.1039/C8TC02553C |
| [24] |
S. Li, W. Liu, M. Shi, et al., Energy Environ. Sci. 9 (2016) 604-610. DOI:10.1039/C5EE03481G |
| [25] |
Y. Liu, Z. Zhang, S. Feng, et al., J. Am. Chem. Soc. 139 (2017) 3356-3359. DOI:10.1021/jacs.7b00566 |
| [26] |
D. Liu, B. Yang, B. Jang, et al., Energy Environ. Sci. 10 (2017) 546-551. DOI:10.1039/C6EE03489F |
| [27] |
W. Zhao, S. Li, H. Yao, et al., J. Am. Chem. Soc. 139 (2017) 7148-7151. DOI:10.1021/jacs.7b02677 |
| [28] |
S. Li, L. Ye, W. Zhao, et al., J. Am. Chem. Soc. 140 (2018) 7159-7167. DOI:10.1021/jacs.8b02695 |
| [29] |
H. Chen, Z. Hu, H. Wang, et al., Joule 2 (2018) 1623-1634. DOI:10.1016/j.joule.2018.05.010 |
| [30] |
Y. Lin, Z.G. Zhang, H. Bai, et al., Energy Environ. Sci. 8 (2015) 610-616. DOI:10.1039/C4EE03424D |
| [31] |
A. Zhang, C. Li, F. Yang, et al., Angew. Chem. Int. Ed. 56 (2017) 2694-2698. DOI:10.1002/anie.201612090 |
| [32] |
Y. Duan, X. Xu, H. Yan, et al., Adv. Mater. 29 (2017) 1605115. DOI:10.1002/adma.201605115 |
| [33] |
D. Meng, H. Fu, C. Xiao, et al., J. Am. Chem. Soc. 138 (2016) 10184-10190. DOI:10.1021/jacs.6b04368 |
| [34] |
D. Meng, D. Sun, C. Zhong, et al., J. Am. Chem. Soc. 138 (2016) 375-380. DOI:10.1021/jacs.5b11149 |
| [35] |
J. Zhang, Y. Li, J. Huang, et al., J. Am. Chem. Soc. 139 (2017) 16092-16095. DOI:10.1021/jacs.7b09998 |
| [36] |
B. Fan, L. Ying, Z. Wang, et al., Energy Environ. Sci. 10 (2017) 1243-1251. DOI:10.1039/C7EE00619E |
| [37] |
H. You, D. Kim, H.H. Cho, et al., Adv. Funct. Mater. 3 (2018) 1803613. |
| [38] |
S. Chen, Y. An, G.K. Dutta, et al., Adv. Funct. Mater. 27 (2017) 1603564. DOI:10.1002/adfm.v27.2 |
| [39] |
N.B. Kolhe, H. Lee, D. Kuzuhara, et al., Chem. Mater. 30 (2018) 6540-6548. DOI:10.1021/acs.chemmater.8b03229 |
| [40] |
M.H. Hoang, G.E. Park, S. Choi, et al., J. Mater. Chem. C 7 (2019) 111-118. DOI:10.1039/C8TC05035J |
| [41] |
D. Chen, J. Yao, L. Chen, et al., Angew. Chem. Int. Ed. 57 (2018) 4580-4584. DOI:10.1002/anie.201800035 |
| [42] |
X. Liao, L. Zhang, L. Chen, et al., Nano Energy 37 (2017) 32-39. DOI:10.1016/j.nanoen.2017.05.008 |
| [43] |
X. Liu, C. Zhang, C. Duan, et al., J. Am. Chem. Soc. 140 (2018) 8934-8943. DOI:10.1021/jacs.8b05038 |
| [44] |
Y.J. Hwang, T. Earmme, B.A. Courtright, J. Am. Chem. Soc. 137 (2015) 4424-4434. DOI:10.1021/ja513260w |
| [45] |
Z. Li, X. Xu, W. Zhang, et al., Energy Environ. Sci. 10 (2017) 2212-2221. DOI:10.1039/C7EE01858D |
| [46] |
C. Duan, K. Gao, van Franeker J.J., et al., J. Am. Chem. Soc. 138 (2016) 10782-10785. DOI:10.1021/jacs.6b06418 |
| [47] |
M. Jeong, S. Chen, S.M. Lee, et al., Adv. Energy Mater. 8 (2018) 170166. |
| [48] |
S. Albrecht, S. Janietz, W. Schindler, et al., J. Am. Chem. Soc. 134 (2012) 14932-14944. DOI:10.1021/ja305039j |
| [49] |
Y. Lin, Y. Jin, S. Dong, et al., Adv. Energy Mater. 8 (2018) 1701942. DOI:10.1002/aenm.201701942 |
| [50] |
L. Ye, W. Zhao, S. Li, et al., Adv. Energy Mater. 7 (2017) 1602000. DOI:10.1002/aenm.201602000 |
| [51] |
L. Zhang, B. Lin, Z. Ke, et al., Nano Energy 41 (2017) 609-617. DOI:10.1016/j.nanoen.2017.10.014 |
| [52] |
X. Zhang, C. Zhan, J. Yao, Chem. Mater. 27 (2015) 166-173. DOI:10.1021/cm504140c |
| [53] |
D.J.D. Moet, M. Lenes, M. Morana, et al., Appl. Phys. Lett. 96 (2010) 213506. DOI:10.1063/1.3435468 |
| [54] |
S. Zhang, L. Ye, W. Zhao, et al., Sci. China Chem. 58 (2015) 248-256. DOI:10.1007/s11426-014-5273-x |
| [55] |
A. Sharenko, D. Gehrig, F. Laquai, T.Q. Nguyen, Chem. Mater. 26 (2014) 4109-4118. DOI:10.1021/cm5010483 |
| [56] |
W. Zhao, S. Zhang, Y. Zhang, et al., Adv. Mater. 30 (2018) 1704837. DOI:10.1002/adma.v30.4 |
| [57] |
H.C. Liao, C.C. Ho, C.Y. Chang, et al., Mater. Today 16 (2013) 326-336. DOI:10.1016/j.mattod.2013.08.013 |
| [58] |
A.J. Moulé, K. Meerholz, Adv. Mater. 20 (2008) 240-245. DOI:10.1002/(ISSN)1521-4095 |
| [59] |
Y. Yao, J. Hou, Z. Xu, G. Li, Y. Yang, Adv. Funct. Mater. 18 (2008) 1783-1789. DOI:10.1002/adfm.200701459 |
| [60] |
C.V. Hoven, X.D. Dang, R.C. Coffin, et al., Adv. Mater. 22 (2010) E63-E66. DOI:10.1002/adma.v22:8 |
| [61] |
Y. Zhang, Q. Wan, X. Guo, et al., J. Mater. Chem. A 3 (2015) 18442-18449. DOI:10.1039/C5TA05014F |
| [62] |
Y. Zhou, K.L. Gu, X. Gu, et al., Chem. Mater. 28 (2016) 5037-5042. DOI:10.1021/acs.chemmater.6b01776 |
| [63] |
S. Li, L. Ye, W. Zhao, et al., J. Am. Chem. Soc. 140 (2018) 7159-7167. DOI:10.1021/jacs.8b02695 |
| [64] |
L. Ye, Y. Jing, X. Guo, et al., J. Phys. Chem. C 117 (2013) 14920-14928. DOI:10.1021/jp404395q |
| [65] |
Tremolet de Villers B.J., O'Hara K.A., D.P. Ostrowski, et al., Chem. Mater. 28 (2016) 876-884. DOI:10.1021/acs.chemmater.5b04346 |
| [66] |
Q. Sun, H. Wang, C. Yang, Y. Li, J. Mater. Chem. 13 (2003) 800-806. DOI:10.1039/b209469j |
| [67] |
S.R. Cowan, A. Roy, A.J. Heeger, Phys. Rev. B 82 (2010) 245207. DOI:10.1103/PhysRevB.82.245207 |
| [68] |
O.K. Kwon, M.A. Uddin, J.H. Park, et al., Adv. Mater. 28 (2016) 910-916. DOI:10.1002/adma.v28.5 |
 2019, Vol. 30
2019, Vol. 30