b College of Chemistry and Molecular Engineering, Zhengzhou University, Zhengzhou 450001, China;
c Henan Joint International Research Laboratory of Green Construction of Functional Molecules and Their Bioanalytical Applications, Zhengzhou 450001, China
Epigenetic regulation of tumor is the advancing spot in the present international biochemistry research [1]. Recent studies have indicated that epigenetic alterations play an essential role in this type of cancer [2]. DNA methyltransferases (DNMTs) are the important representations of epigenetic alterations. There are numerous of reports have shown that the aberrant expressions of DNMTs in breast cancer [3-5], liver cancer [6], lung cancer [7] and gastric cancer [8]. DNA methylation is mainly formed by the addition of 1-methyl at the 5′-position of cytosine to 5-methylcytosine catalyzed by methyltransferase [9]. One of 5-methylcytosine synthesis pathways is DNA methyltransferase 1 (DNMT1) catalyzes complete methylation of the unmethylated chain in the semi-methylated DNA and maintains methylation consistent with the protocell. In other words, the activity and concentration of DNMT1 directly affect the degree of DNA methylation. Overexpression of DNMT1 may cause aberrant methylation of tumor suppressor gene or proto-oncogene promoter, further leading to tumorigenesis.
Many methods for detecting DNMT1 activity were well established which include radioactive labeling strategy [10], high performance liquid chromatography [11], gel electrophoresis [12], and immune-based assay [13-15]. However, they are usually used for the DNMT1 activity detection, hardly applied for the quantitative analysis. The detection of enzyme activity must be based on the measurement of enzyme content, so the determination of enzyme concentration is of great concern.
Common enzyme quantitative methods include enzyme-linked immunosorbent assay [16, 17] (ELISA), chemiluminescence (CLIA) [18-20] and immunochromatography [21]. The advantages of ELISA are cost reduction and automation, but non-specific reaction caused by HOOK effect, false positive caused by cross-contamination, low sensitivity, low detection rate of window phase is its inherent defect. As a new detection method in recent years, CLIA method [22, 23] has good specificity for immunological reaction and high sensitivity for the detection of luminescence method.Compared with ELISA, CLIA is a heterogeneous immunoassay, so that antigen and antibody can bind efficiently without the shortcomings of traditional one-step method. At the same time, CLIA results are more stable, and the detection time is shorter. However, few reports have been found on the clinical application of DNMT1 using the CLIA without the participation of enzyme.
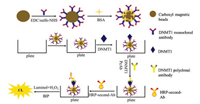
In this study, we developed a novel magnetic enhanced chemiluminescence enzyme immunoassay (MCLEIA) to achieve the simple, economical and easily commercialized DNMT1 detection in serum samples, and this method was compared with the purchasable ELISA kits. The experimental conditions, including the reaction time of DNMT1 and MBs@PcAbDNMT1, the dilution ratio of PcAbDNMT1, and the dilution ratio of HRP-second-Ab were examined and optimized. The methodology parameters including precision, accuracy and specificity were also evaluated. By comparing the established two methods, the results indicated that MCLEIA has shorter total reaction time, fewer capture antibodies, and lower detection limit. Therefore, the proposed MCLEIA could be used for the high-throughput detection of DNMT1 in clinical (Scheme 1).
|
Download:
|
| Scheme 1. The principle of MCLEIA. | |
The experimental section, including materials and instruments, buffers and solutions, immobilization of antibody on magnetic beads, characterization of the MBs@McAbDNMT1, theory and program of MCLEIA immunoassay and theory and program of ELISA immunoassay can be found in Supporting information.
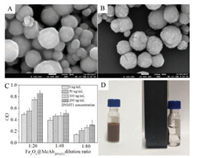
We first conducted characterization of MBs@McAbDNMT1. The coupling rate determined by coomassie brilliant blue staining was more than 75%. As shown in Figs. 1 A and B, the MBs@McAbDNMT1 surface morphology has changed; the surface is rough and covered with a layer of white substance, presumed to be McAbDNMT1. Therefore, the results show that the coupling is successful. The Fig. 1C showed that the 20 times dilution of immunomagnetic beads has the highest absorbance compared with the other two dilutions and there is a gradient between the three groups. It was shown that the carboxylate magnetic particle surface was successfully modified with McAbDNMT1 and maintained the immune activity of the antibody, which can be used in subsequent experiments. The magnetism characterization of MBs@McAbDNMT1 were presented in Fig. 1D, from which we could see in the left figure, the magnetic bead solution was brown, the solution was well dispersed, and no precipitation occurred within a few hours without the effect of external magnetic field. In the right figure, the immune magnetic beads were rapidly enriched with good magnetic response, and the solution was partially clarified in magnetic field. Therefore, the prepared MBs@McAbDNMT1 still had good magnetic response.
|
Download:
|
| Fig. 1. (A) The SEM images of the carboxyl magnetic beads (MBs), Scale bar: 100 nm. (B) The SEM images of MBs@McAbDNMT1, Scale bar: 100 nm. (C) Biological activity verification of MBs@McAbDNMT1. (D) Magnetism constancy of MBs@McAbDNMT1. | |
Next, we optimized MCLEIA method. MBs@McAbDNMT1 with different dilution ratios was optimized (Fig. 2A).The concentration of DNMT1 was 100 ng/mL.When the dilution ratio of MBs@McAbDNMT1 was 1:40, the RLU intensity reached the maximum, and the difference between the experimental group and the control group was the largest. Therefore, 1:40 was chosen as the optimal dilution ratio of MBs@McAbDNMT1. Results as shown in Fig. 2B, the RLU value was relatively high when the reaction time between MBs@McAbDNMT1 and DNMT1 was 90 min and 120 min, but the RLU value did not change significantly after 90 min, indicating that the balance of antigen antibody complexes has reached stabilization. Thus, the optimal reaction time was 90 min for further experiments. After different dilutions of PcAbDNMT1, the chemiluminescence values of DNMT1 (100 ng/mL) were determined. RLU was determined with 0 ng/mL as blank control, and the difference value △RLU (RLU100 - RLU0) was used to dependent variable (Fig. 2C). When the dilution ratio of PcAbDNMT1 was 1: 2000, the △RLU intensitywas the strongest. The result indicated the best dilution ratio of PcAbDNMT1 was 1: 2000. After different dilutions HRP-second-Ab, the chemiluminescence values of DNMT1 (100 ng/mL) were determined. RLU was determined with 0 ng/mL as blank control, and the difference value △RLU (RLU100 - RLU0) was used to dependent variable (Fig. 2D). When the dilution ratio of HRP-second-Ab was 1: 4000, the △RLU intensity was the strongest. The result indicated the best dilution ratio of HRP-second-Ab was 1: 4000.

|
Download:
|
| Fig. 2. (A) The result of MBs@McAbDNMT1 dilution ratio on RLU intensity. (B) The best reaction time of MBs@McAbDNMT1 with DNMT1 in serum sample. (C) The effect of PcAbDNMT1 dilution ratio on △RLU intensity. (D) The effect of HRP-Second-Ab dilution ratio on △RLU intensity. | |
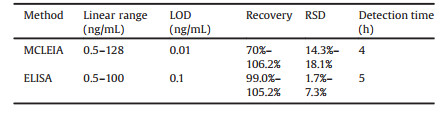
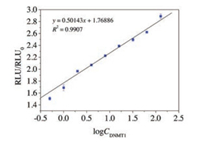
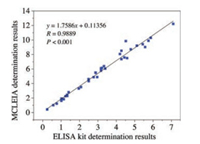
Then, we explored the MCLEIA methodology parameters including precision, accuracy and specificity. In the 0.5–128 ng/mL, the standard curve equation was y = 0.5014x + 1.769 (x was logCDNMT1, y was RLU/ RLU0 with a correlation coefficient of 0.9907) (Fig. 3). On the one hand, same sample (0.5, 4.0, 32.0 ng/mL) detected six times to value the inter-assay precision. To value the intra-assay precision, comparing data from two microplates. The RSD of intra- and inter-assays were 15.8%–16.9% and 14.3%–18.1%, respectively. The recoveries of the method were 70%–106.2% (Table 1). Because of the complexity of the components in serum sample, the established MCLEIA method is required to have high specificity for DNMT1. Therefore, DNMT3A and DNMT3B with similar structure were selected. Their cross-reactivity rates were 0.31% and 0.34%, respectively. The low cross-reaction rate indicated that McAbDNMT1 had no specific reaction to other DNMTs. Therefore, the established MCLEIA method had good specificity and could be used to detect DNMT1 in samples. Although there are several ways to detect DNMT1 concentration, the only commercialkits areELISA. Thus, the results obtained using the proposed MCLEIA in the quantity of DNMT1 in serum samples were compared with the commercial ELISA kit. The correlation between the two groups of data was analyzed. The results showed that there was a good correlation between the two groups (correlation coefficient R = 0.9889, P < 0.01) (Fig. 4). The result suggests that the MCLEIA displays stable performance for determination of DNMT1 and is appropriate for high-throughput detection in clinical diagnosis.
|
Download:
|
| Fig. 3. The relationship between RLU/RLU0 and logCDNMT1 (mL). | |
|
|
Table 1 Comparison of key of the analytical parameters. |
|
Download:
|
| Fig. 4. The correlation between the proposed MCLEIA and the commercial ELISA kits. | |
In summary, a simple, sensitive and selective enhanced chemiluminescence system was composed of HRP, luminal, H2O2 and BIP was developed for the detection of DNMT1. The proposed MCLEIA method was finally applied to determine DNMT1 in 36 samples of lung cancer serum. Results obtained using the method showed high correlation with the purchasable ELISA kit. Thus, the MCLEIA established in this study could be used for the sensitive quantitative detection of DNMT1 in serum and presented a promising application potential.
AcknowledgmentsThis work was supported by the National Natural Science Foundation of China (Nos. 81402721, 81573203, 21605131) and Science and Technology Department of Henan Province (No. 22170004).
Appendix A. Supplementary dataSupplementary material related to this article canbefound, in the online version, at doi:https://doi.org/10.1016/j.cclet.2019.03.013.
| [1] |
P. Prasad, S.A. Mittal, J. Chongtham, S. Mohanty, T. Srivastava, Stem Cells 35 (2017) 1468-1478. DOI:10.1002/stem.2621 |
| [2] |
S.K. Botla, S. Savant, P. Jandaghi, et al., Cancer Res. 76 (2016) 4149-4159. DOI:10.1158/0008-5472.CAN-15-0390 |
| [3] |
J. Wu, Z. Shuang, J. Zhao, et al., Biomed. Pharmacother. 97 (2018) 1275-1281. DOI:10.1016/j.biopha.2017.11.055 |
| [4] |
M.Y. Sun, X.X. Yang, W.W. Xu, et al., Genet. Mol. Res. 11 (2012) 4330-4341. DOI:10.4238/2012.September.26.1 |
| [5] |
W. Zhang, Z. Chang, K.E. Shi, et al., Oncol. Lett. 11 (2016) 1995-2000. DOI:10.3892/ol.2016.4193 |
| [6] |
C. Zhang, Y. Peng, F. Yang, et al., J. Cancer 7 (2016) 446-452. DOI:10.7150/jca.13065 |
| [7] |
R.E. Husni, A. Shiba-Ishii, S. Iiyama, et al., Lung Cancer 97 (2016) 59-65. DOI:10.1016/j.lungcan.2016.04.018 |
| [8] |
H. Li, W. Li, S. Liu, et al., EBioMedicine 13 (2016) 125-131. DOI:10.1016/j.ebiom.2016.10.028 |
| [9] |
A. Meissner, T.S. Mikkelsen, H. Gu, et al., Nature 454 (2008) 766-770. DOI:10.1038/nature07107 |
| [10] |
B.Y. Kim, O.S. Kwon, S.A. Joo, et al., Anal. Biochem. 326 (2004) 21-24. DOI:10.1016/j.ab.2003.11.009 |
| [11] |
K. Chen, J. Liu, S. Liu, et al., Cell 170 (2017) 492-506. DOI:10.1016/j.cell.2017.06.042 |
| [12] |
C.D. Valdez, J.N. Davis, H.M. Odeh, et al., PLoS One 6 (2011) 251-256. |
| [13] |
H. Zhao, L. Wang, W. Li, et al., ACS Appl. Mater. Interfaces 9 (2017) 29554-29561. DOI:10.1021/acsami.7b09631 |
| [14] |
B. Zhu, J. Ge, S.Q. Yao, Bioorg. Med. Chem. 23 (2015) 2917-2927. DOI:10.1016/j.bmc.2015.03.006 |
| [15] |
J. Li, G. He, C. Mu, K. Wang, Y. Xiang, Anal. Chim. Acta 986 (2017) 131-137. DOI:10.1016/j.aca.2017.07.021 |
| [16] |
C. Hu, I. Zeimpekis, K. Sun, et al., Anal. Chem. 88 (2016) 4872-4878. DOI:10.1021/acs.analchem.6b00702 |
| [17] |
A.G. Pose, E.R. Rodriguez, M.J. Pineiro, et al., J. Immunol. Methods 459 (2018) 70-75. DOI:10.1016/j.jim.2018.05.008 |
| [18] |
H. Zhang, S. Qi, J. Rao, et al., Luminescence 28 (2013) 927-932. DOI:10.1002/bio.v28.6 |
| [19] |
Y. Li, Y.Z. Zheng, D.K. Zhang, et al., Chin. Chem. Lett. 28 (2017) 184-188. DOI:10.1016/j.cclet.2016.07.020 |
| [20] |
M.L. Yuan, T.Y. Jiang, L.P. Du, M.Y. Li, Chin. Chem. Lett. 27 (2016) 550-554. DOI:10.1016/j.cclet.2016.02.011 |
| [21] |
W.Y. Sun, W.Y. Liu, L.B. Qu, Chin. Chem. Lett. 18 (2007) 1107-1110. DOI:10.1016/j.cclet.2007.07.008 |
| [22] |
A.R. Rodríguez-Orozco, H. Ruiz-Reyes, N. Medina-Serriteno, Mini Rev. Med. Chem. 10 (2010) 113-116. |
| [23] |
H. Zhang, S. Qi, Clin. Chim. Acta 412 (2011) 1572-1577. DOI:10.1016/j.cca.2011.05.005 |
 2019, Vol. 30
2019, Vol. 30