b University of Chinese Academy of Sciences, Beijing 100049, China;
c Sino-Danish College, University of Chinese Academy of Sciences, Beijing 100190, China
In recent years, organic solar cells (OSCs) have attracted much attention both in academicians and industrialists, because of their advantages of low cost, light weight, solution processing, easy preparation and preferable flexibility [1-6]. Additionally, with the efforts of researchers, the power conversion efficiencies (PCEs) of organic solar cells have been remarkably improved, and the highest PCEs of over 14% has been achieved [7,8]. Recently, both small molecular electron donors and small molecular electron acceptors exhibit excellent performances [9-14]. A high PCE exceeding 13% was obtained for all-small-molecule non-fullerene systems was reported by Zhu and co-workers [15]. Compared with polymeric molecules, small molecules have lots of advantages for applications in OSCs [16], such as their easily modified molecular structures, excellent batch-to-batch reproducibility and highly modifiable physical properties, including absorption profiles, molecular orbital energy levels, and charge carrier mobility [17-19]. Therefore, the design and synthesis of new photovoltaic small molecules are crucial for improving the performance of solar cells.
Up to now, A-π-D-π-A (A represents the acceptor unit and D represents the donor unit) backbone structure small molecules are easily synthesized and show good performance because of their limit controlling over molecular orientation and intermolecular aggregation which have attracted much attention [20-22]. This combination has advantages in providing broad absorption spectra and appropriate molecular orbital energy levels, but the PCEs obtained by this material combination are still low [23-25]. Therefore, how to improve the PCE through material design is a meaningful work. Among the A-π-D-π-A backbone small molecules, rhodamine [19] is often selected as the electron withdrawing group when choosing building blocks to construct non-fullerene donors. Rhodamine has strong electron pulling ability which can be used to obtain red-shifted absorption spectra and lower molecular orbital energy levels [26]. Meanwhile, the length, isomerization and position of the end alkyl chain can effectively influence the solubility of molecules [27]. Also the end alkyl chain can affect the crystallization of the molecules and the π–π interactions [28]. Generally the position of the end alkyl chain affects the planarity of molecules [29]. Recently, modulating the molecular orientations through changing the length of the end alkyl chain have reported by Hou and co-workers [14]. Therefore, choosing end alkyl chains with appropriate length and isomerization is of great significance to the stacking structures in the solid states and hence to the photovoltaic performances. Our group have designed and synthesized a series of small molecular donors based on the naphtho[1, 2-b:5, 6-b']dithiophene (NDT) elements with high crystallization [30,31]. NDT can be a good donor moiety for small molecules to study the influences induced by the end and alkyl side chains.
Herein, we designed and synthesized two novel A-π-D-π-A small-molecule electron donors, based on two dimensional conjugated NDT as the donor unit and 3-octylrhodamine or 3- (2-ethylhexyl)-rhodamine as the acceptor unit, triple thiophene as the π-bridge to increase the conjugation of the molecules, named as NDT-3T-EH and NDT-3T-O, respectively (Fig. 1). The two NDTbased small-molecule compounds were synthesized [32-35] by end-capping with rhodamine featuring different end alkyl groups, through choosing branched and straight chains with the same number of carbon atoms. This design provides a model of compounds with the same photoelectric properties and allows us to explore the influence of the end-capping alkyl chains on intermolecular aggregation behavior in solid-state films. Branched chains are good for material solubility, and straight chains are good for increasing crystallinity. Next, we have blended the model compounds NDT-3T-R with IDIC-4F [30] (a nonfullerene acceptor, Fig. 1) which could match well with the two small molecular donor materials both in absorption profile and molecular orbital energy level. And the nonfullerene all-small-molecule organic photovoltaic devices have been fabricated. Compared with the NDT-3T-EH (6.11%), the PCE of NDT-3T-O-based solar cells reached 7.06% with improved short circuit current (Jsc) and filling factor (FF). In addition, we have found that the end alkyl chains can affect intermolecular aggregation behavior in the solid-state films to some extent. The results not only prove that NDT-3T-R is a proper donor material, but also give us a guideline for the design of high efficiency nonfullerene all-small-molecule systems.

|
Download:
|
| Fig. 1. Molecular structures of the donor NDT-3T-R and nonfullerene acceptor IDIC-4F. | |
The NDT-3T-R was synthesized as the routes shown in Scheme 1. Compounds 1 [34] and 2 were both prepared according to our previous work and reported literatures [34,36,37]. And through a Stille coupling reaction, we can get compound 3. The target molecules NDT-3T-R were synthesized by a Knoevenagel reaction between compound 3 and 3-octylrhodamine or 3-(2-ethylhexyl)- rhodamine with a high yield of 74%. NDT-3T-R show good solubility and can be dissolved in common solvents such as tetrahydrofuran and chloroform, which is beneficial for the device fabrication. The detailed synthesis processes, purification methods, and the nuclear magnetic resonance spectroscopy data are provided in the Supporting information.

|
Download:
|
| Scheme 1. Synthetic route of NDT-3T-R. | |
The absorption spectra of NDT-3T-R in thin film and solution are displayed in Fig. 2a. In solution, NDT-3T-EH and NDT-3T-O exhibited similar absorption in the range of 300–600 nm. For the film, a significant red-shift of about 100 nm appears compared with that in solution. Furthermore, NDT-3T-O shows a stronger shoulder peak at 606 nm in the film, indicating a stronger π–π intermolecular interaction [38]. Branched and straight end alkyl groups with the same number of carbon atoms have little effect on the absorption of the materials in solution, which is almost identical. Branched chains just increase molecules solubility. However, in the film, the shoulder peak enhancement indicates that the straight chain end group is conducive to the improvement of molecular aggregation ability. The optical bandgaps (Egopt) of NDT-3T-EH and NDT-3T-O were calculated to be 1.82 eV [39]. The absorption spectrum of non-fullerene acceptor IDIC-4F is mainly in the long wavelength band which could match well with the two small molecular donor materials [30]. Thus, the two all-smallmolecule systems could be expected to reach high Jsc.

|
Download:
|
| Fig. 2. (a) Absorption spectra of NDT-3T-R in solution (S) and film; (b) energy levels of NDT-3T-R and IDIC-4F. | |
The electrochemical properties of NDT-3T-R were measured by electrochemical cyclic voltammetry (CV) measurements with Ag/Ag+ as reference electrode, and the results are shown in Fig. S1 in Supporting information. Calculated by using the formula EHOMO/LUMO = -e(Eox/red - E1/2(Fc/Fc+) + 4.8) (eV) [40,41], the highest occupied molecular orbital (HOMO) energy levels of NDT-3T-EH and NDT-3T-O are determined to be –5.27 eV and –5.34 eV, respectively. The corresponding lowest unoccupied molecular orbital (LUMO) energy levels are –3.41 eV and –3.46 eV, respectively. Compare with NDT-3T-O, the molecular orbital energy level of NDT-3T-EH is little higher, because of the branched end alkyl groups is likely to reduce the aggregation ability of materials [35]. So, using branched end alkyl groups not change the band gap, but only increases the molecular orbital energy level. Moreover, the electrochemical bandgaps of NDT-3T-EH and NDT-3T-O are consistent with their corresponding optical bandgaps. The energy levels of the two donors are well matched with those of IDIC-4F. The HOMO and LUMO levels of IDIC-4 F can be obtained from related literatures [30], as shown in figure in Fig. 2b, which could be well matched with those of the two small molecule donors. Besides, these two kinds of small molecules have almost similar HOMO and LUMO level, which means the two small molecules used as the donor have almost similar open circuit voltage (Voc) in the devices [42].
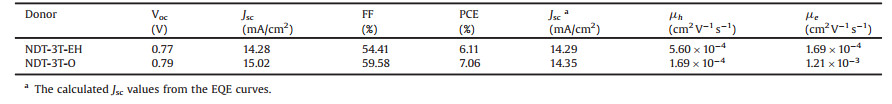
In order to explore the photovoltaic performance of NDT-3T-EH and NDT-3T-O, conventional devices with a structure ITO/ PEDOT:PSS/active layer/Ca/Al were fabricated and the detailed photovoltaic performance parameters are listed in Table 1. When blended with IDIC-4F without any treatment, NDT-3T-EH and NDT-3T-O exhibited low PCEs of 2.56% and 2.95%, respectively. The processing conditions were further optimized to improve the performance, and after being thermally annealed at 100 ℃ for 10 min, the PCE of NDT-3T-EH and NDT-3T-O increased to 6.11% and 7.06%, respectively. The detailed photovoltaic performance parameters can be seen in Table S1. The PCE is higher when using NDT-3T-O as donor, and IDIC-4F as acceptor. NDT-3T-O-based solar cells exhibited higher Voc, Jsc and FF compared with those of NDT-3T-EH. The higher Voc is attributed to the molecular orbital energy level of NDT-3T-O is little lower. The higher Jsc and FF is attributed to the improvement of molecular aggregation ability, which should be future investigated by the morphological characterization. The current–voltage (J–V) curves and the corresponding external quantum efficiencies (EQEs) are shown in Fig. 3. NDT-3T-O showed higher EQEs in the range of 600–800 nm which results in the improved Jsc compared with that of NDT-3T-EH. In addition, the Jsc values calculated from the EQE spectra are strongly in consistent with those acquired from J–V tests, the Jsc values calculated from the EQE are shown in Table 1, as well. The results reveal that the PCEs of two systems are reliable.
|
|
Table 1 Photovoltaic performance of NDT-3T-EH and NDT-3T-O. |
|
Download:
|
| Fig. 3. (a) J–V curves; (b) EQEs of the best performance for NDT-3T-EH:IDIC-4F and NDT-3T-O:IDIC-4F blends. | |
Simultaneously, the hole mobility and electron mobility of the two systems were measured via the space charge limited current (SCLC) method [43]. And the hole mobility was investigated by preparing the device structure of ITO/PEDOT:PSS/Active layer/ MoOx/Ag, while the electron mobility was measured with the structure of ITO/ZnO/Active layer/Ca/Al. From the J–V curves of these devices and the thickness of the active layer, the hole mobility of NDT-3T-EH: IDIC-4 F and NDT-3T-O:IDIC-4F were calculated to be 5.60 ×10–4 cm2 V–1 s–1 and 1.69 × 10–4 cm2 V–1 s–1, while the electron mobility was 1.69 × 10–4 cm2 V–1 s–1 and 1.21 ×10–3 cm2 V–1 s–1, respectively.
Atomic force microscopy (AFM), transmission electron microscopy (TEM) and the grazing-incidence wide-angle X-ray scattering (GIWAXS) were used to study the morphology of the two systems to further understand the reason for the different photovoltaic performance of the two systems.
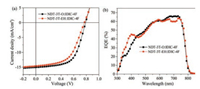
The AFM images are shown in Fig. 4. The surface of NDT-3T-O: IDIC-4F film is uniform and smooth with a roughness of 3.11 nm, while in NDT-3T-EH:IDIC-4F film the roughness increased to 8.78 nm, which might affect the contact between the active layer and the electrode. As can be seen from the phase pattern, NDT-3T-EH:IDIC-4F system exhibited larger domain size. For NDT-3T-O: IDIC-4F system, it showed relatively appropriate phase separation and bicontinuous interpenetrating network, which was conducive to exciton diffusion and charge transportation to some extent. Moreover, As can be seen from the TEM images shown in Fig. 4, NDT-3T-O:IDIC-4F system showed more appropriate phase separation and fibrillar structures within the active layer, which promotes charge transfer [44]. Phase separation is an crucial factor that influence excitons diffusion and charges transportation, from the AFM and TEM result, NDT-3T-O:IDIC-4F blend film has appropriate phase separation and smooth surface, which lead to improvements in Jsc and FF.

|
Download:
|
| Fig. 4. The AFM phase images and TEM images of NDT-3T-R:IDIC-4F blend films. | |
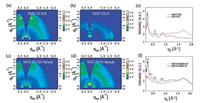
GIWAXS measurements were conducted to investigate the crystallinity and molecular orientation of the films. It is can be found in Fig. 5, the neat NDT-3T-EH film exhibits a preferred faceon orientation, and the π–π stacking peaks of neat NDT-3T-EH locates at 1.69 Å–1 along the out-of-plane direction, the corresponding π–π stacking distance is 3.72 Å. For NDT-3T-O, the neat NDT-3T-O film shows distinct diffraction peaks at 0.30Å–1, 0.59 Å-1 and 0.89 Å-1 in the out-of-plane direction, which are assigned to the (100), (200) and (300) lamellar stacking scattering respectively, indicating a high degree of molecular ordering. The π–π stacking peaks of neat NDT-3T-O located at 1.69 Å-1 in the in-plane direction, and shows the same π–π stacking distance with NDT-3T-EH. However, for the neat NDT-3T-EH film, the 2D GIWAXS pattern only exhibits strong (100) lamellar stacking scattering in the inplane direction, indicating a weaker crystallinity than NDT-3T-O. This indicates that the alkyl chain of the end groups can regulate the stacking of molecules. The 2D GIWAXS pattern of IDIC-4F is shown in Fig. S2 (Supporting information). When the two donors were blended with IDIC-4F, the π–π stacking of the two donors exhibit no difference with the neat film, and the peaks located at 0.38 Å-1 and 1.74 Å-1 in the out-of-plane direction are attributed to the (100) and (010) lamellar stacking scattering of IDIC-4F, corresponding to the lamellar stacking distance of 16.53 Å and 3.61 Å, respectively. For the NDT-3T-O:IDIC-4F blend film, the 2D GIWAXS pattern shows sharp lamellar (100) diffraction peak, and strong (200) and (300) diffraction peaks in the out-of-plane direction, indicating that NDT-3T-O keeps the good crystallinity after blend with IDIC-4F. Though NDT-3T-O tends to keep a preferred edge-on orientation stacking in the blend film, the good crystallinity can get the similar hole mobility with NDT-3T-EH. Adjustment of the end alkyl chain is an effective way to modulate the molecular stacking orientation, and the increased crystallization performance indicates the high degree of molecular ordering is beneficial to the charge transport and result in less charge recombination, as evidenced by the improved FF. It is supposed that the better crystallinity and stacking orientation should be the main reason for the high PCE of the two all-small-molecule systems.
|
Download:
|
| Fig. 5. (a, b) Two-dimensional GIWAXS patterns of the pristine films; (c, d) Two-dimensional GIWAXS patterns of the blend films; (e, f) the out-of-plane cuts of the blend film GIWAXS patterns. | |
Two novel A-π-D-π-A small molecular donors with different end alkyl chains were designed and synthesized. The performances of devices based on NDT-3T-O were obviously better than that of NDT-3T-EH. Compared with NDT-3T-EH, the molecular length and planar properties of NDT-3T-O have been increased. It can be found that by changing the shape of the alkyl chain, the stacking state between the molecules and the crystallinity of the molecule can be well changed when blended with IDIC-4F. This phenomenon verifies appropriate alkyl chains with appropriate length and isomerization is of great significance to the solubility of organic solar cell materials and the performance of devices.
AcknowledgmentsThe authors acknowledge the financial support from the National Natural Science Foundation of China (Nos. 21822503, 21534003, 21125420, 21603044 and 91427302), the Ministry of Science and Technology of China (Nos. 2016YFA0200704 and 2016YFF0203803), the Beijing Nova Program (No. Z17110001117062), the Youth Innovation Promotion Association CAS and the Chinese Academy of Sciences.
Appendix A. Supplementary dataSupplementarymaterial related to this article canbefound, in the online version, at doi:https://doi.org/10.1016/j.cclet.2019.01.031.
| [1] |
J. Chen, Y. Cao, Acc. Chem. Res. 42 (2009) 1709-1718. DOI:10.1021/ar900061z |
| [2] |
A.J. Heeger, Adv. Mater. 26 (2014) 10-28. DOI:10.1002/adma.201304373 |
| [3] |
G. Li, V. Shrotriya, J.S. Huang, et al., Nat. Mater. 4 (2005) 864-868. DOI:10.1038/nmat1500 |
| [4] |
G. Li, R. Zhu, Y. Yang, Nat. Photonics 6 (2012) 153-161. DOI:10.1038/nphoton.2012.11 |
| [5] |
H. Li, Y. Zhao, J. Fang, et al., Adv. Energy Mater. 8 (2018) 1702377. DOI:10.1002/aenm.201702377 |
| [6] |
B. Qiu, L. Xue, Y. Yang, et al., Chem. Mater. 29 (2017) 7543-7553. DOI:10.1021/acs.chemmater.7b02536 |
| [7] |
S. Zhang, Y. Qin, J. Zhu, J. Hou, et al., Adv. Mater. 30 (2018) 1800868. DOI:10.1002/adma.v30.20 |
| [8] |
S. Li, L. Ye, W. Zhao, et al., J. Am. Chem. Soc. 140 (2018) 7159-7167. DOI:10.1021/jacs.8b02695 |
| [9] |
S. Badgujar, C.E. Song, S. Oh, et al., J. Mater. Chem. A 4 (2016) 16335-16340. DOI:10.1039/C6TA06367E |
| [10] |
H. Bin, Y. Yang, Z.G. Zhang, et al., J. Am. Chem. Soc. 139 (2017) 5085-5094. DOI:10.1021/jacs.6b12826 |
| [11] |
L. Yang, S. Zhang, C. He, J. Zhang, et al., J. Am. Chem. Soc. 139 (2017) 1958-1966. DOI:10.1021/jacs.6b11612 |
| [12] |
B. Qiu, L. Xue, Y. Yang, et al., Chem. Mater. 29 (2017) 7543-7553. DOI:10.1021/acs.chemmater.7b02536 |
| [13] |
H. Li, Y. Zhao, J. Fang, et al., Adv. Energy Mater. 8 (2018) 1702377. DOI:10.1002/aenm.201702377 |
| [14] |
H. Bin, J. Yao, Y. Yang, et al., Adv. Mater. 30 (2018) 1706361. DOI:10.1002/adma.201706361 |
| [15] |
Z.C. Zhou, S.J. Xu, J.N. Song, et al., Nat. Energy 3 (2018) 952-959. DOI:10.1038/s41560-018-0234-9 |
| [16] |
Y.Z. Lin, Y.F. Li, X.W. Zhan, Chem. Soc. Rev. 41 (2012) 4245-4272. DOI:10.1039/c2cs15313k |
| [17] |
S. Li, W. Liu, C.Z. Li, M. Shi, H. Chen, Small 13 (2017) 1701120. DOI:10.1002/smll.v13.37 |
| [18] |
Y. Lin, J. Wang, Z.G. Zhang, et al., Adv. Mater. 27 (2015) 1170-1174. DOI:10.1002/adma.201404317 |
| [19] |
M. Zhu, J. Miao, Z. Hu, et al., Dye. Pigment. 142 (2017) 39-50. DOI:10.1016/j.dyepig.2017.03.015 |
| [20] |
Q. Wu, D. Deng, K. Lu, Z.X. Wei, Chin. Chem. Lett. 28 (2017) 2065-2077. DOI:10.1016/j.cclet.2017.08.046 |
| [21] |
S. Holliday, R.S. Ashraf, A. Wadsworth, et al., Nat. Commun. 7 (2016) 11585. DOI:10.1038/ncomms11585 |
| [22] |
Y. Lin, Q. He, F. Zhao, et al., J. Am. Chem. Soc. 138 (2016) 2973-2976. DOI:10.1021/jacs.6b00853 |
| [23] |
Y.Z. Lin, J.Y. Wang, Z.G. Zhang, et al., Adv. Mater. 27 (2015) 1170-1174. DOI:10.1002/adma.201404317 |
| [24] |
L.Y. Yang, S.Q. Zhang, C. He, et al., J. Am. Chem. Soc. 139 (2017) 1958-1966. DOI:10.1021/jacs.6b11612 |
| [25] |
C.B. Nielsen, S. Holliday, H.Y. Chen, S.J. Cryer, I. McCulloch, Acc. Chem. Res. 48 (2015) 2803-2812. DOI:10.1021/acs.accounts.5b00199 |
| [26] |
G.X. Jia, S.Q. Zhang, L.Y. Yang, et al., Acta Phys. Chim. Sin. 35 (2019) 76-83. |
| [27] |
H.X. Zhou, L.Q. Yang, W. You, Macromolecules 45 (2012) 607-632. DOI:10.1021/ma201648t |
| [28] |
D.A.M. Egbe, S. Turk, S. Rathgeber, et al., Macromolecules 43 (2010) 1261-1269. DOI:10.1021/ma902273s |
| [29] |
H.X. Zhou, L.Q. Yang, S.Q. Xiao, S.B. Liu, W. You, Macromolecules 43 (2010) 811-820. DOI:10.1021/ma902241b |
| [30] |
H. Li, Q. Wu, R. Zhou, et al., Adv. Energy Mater. (2018) 1803175. |
| [31] |
H. Li, Y.F. Zhao, J. Fang, et al., Adv. Energy Mater. 8 (2018) 8. |
| [32] |
X.W. Zhu, K. Lu, H. Li, R.M. Zhou, Z.X. Wei, Chin.Chem.Lett. 27 (2016) 1271-1276. DOI:10.1016/j.cclet.2016.06.015 |
| [33] |
S. Loser, H. Miyauchi, J.W. Hennek, et al., Chem. Commun. 48 (2012) 8511-8513. DOI:10.1039/c2cc32646a |
| [34] |
X. Zhu, B. Xia, K. Lu, et al., Chem. Mater. 28 (2016) 943-950. DOI:10.1021/acs.chemmater.5b04668 |
| [35] |
D. Deng, Y. Zhang, L. Yuan, et al., Adv. Energy Mater. 4 (2014) 1400538. DOI:10.1002/aenm.201400538 |
| [36] |
R.J. Kumar, J.M. MacDonald, T.B. Singh, L.J. Waddington, A.B. Holmes, J. Am. Chem. Soc. 133 (2011) 8564-8573. DOI:10.1021/ja110858k |
| [37] |
K. Sun, Z.Y. Xiao, S.R. Lu, et al., Nat. Commun. 6 (2015) 9. |
| [38] |
S. Badgujar, G.Y. Lee, T. Park, et al., Adv. Energy Mater. 6 (2016) 9. |
| [39] |
S.H. Liao, H.J. Jhuo, Y.S. Cheng, S.A. Chen, Adv. Mater. 25 (2013) 4766-4771. DOI:10.1002/adma.v25.34 |
| [40] |
Q.J. Sun, H.Q. Wang, C.H. Yang, Y.F. Li, J. Mater. Chem. 13 (2003) 800-806. DOI:10.1039/b209469j |
| [41] |
H.J. Bin, L. Gao, Z.G. Zhang, et al., Nat. Commun. 7 (2016) 11. |
| [42] |
M. Erray, M. Hanine, E. Boufounas, A. El Amrani, Eur. Phys. J. Appl. Phys. 82 (2018) 30201. DOI:10.1051/epjap/2018180070 |
| [43] |
P.W. Blom, M. De Jong, J. Vleggaar, Appl. Phys. Lett. 68 (1996) 3308-3310. DOI:10.1063/1.116583 |
| [44] |
M.H. Jao, H.C. Liao, W.F. Su, J. Mater. Chem. A 4 (2016) 5784-5801. DOI:10.1039/C6TA00126B |
 2019, Vol. 30
2019, Vol. 30