b Key Laboratory of Material Chemistry for Energy Conversion and Storage (Huazhong University of Science and Technology), Ministry of Education, Hubei Key Laboratory of Material Chemistry and Service Failure, School of Chemistry and Chemical Engineering, Huazhong University of Science and Technology, Wuhan 430074, China
Direct methanol fuel cells (DMFCs) are high-effective energy conversion devices that can directly obtain electrical energy from methanol fuel [1-5]. Proton exchange membrane (PEM) is one of the key components of DMFCs, which plays a vital role in facilitating proton transport and preventing methanol shuttling [6-9]. Unfortunately, the power density of the current DMFCs is largely hindered by the severe methanol permeability of the commercial Nafion membranes [10-15]. Therefore, many current researches have been focused on PEMs with low methanol permeability as alternatives to Nafion membranes to overcome the severe energy reduction issue [16-21].
Sulfonated poly(ether ether ketone) (SPEEK) has been considered as one of the most promising alternative PEMs in DMFCs due to its inherently low methanol permeability resulted from the poorly interconnected hydrophilic domains (methanol transport channels) [22, 23]. In addition, the high mechanical strength, good thermal and electrochemical stabilities and low cost of SPEEK are also important attributes to its practical use [24-26]. However, it still suffers from the low proton conductivity problem caused by the poorly interconnected hydrophilic domains. An efficient way to improve proton conductivity of the neat SPEEK membrane is to increase the degree of sulfonation (DS). However, the high DS always results in serious dimensional swelling, which gives rise to poor overall cell performance [27, 28]. Great efforts have been made to solve the serious swelling of SPEEK by several strategies, such as polymer blends [29-31], self-crosslinking networks [32, 33], inorganic/organic blends [34-36] and semi-interpenetrating polymer networks (semi-IPNs) [37, 38]. For the semi-IPNs, as the SPEEK polymeric chains are entrapped into the crosslinking networks of another polymer, the swelling behavior of the SPEEK membrane can be effectively addressed. In addition, the molecular scale dispersion of the sulfonated polymer chains in the semi-IPNs can guarantee the uniform distribution of proton carriers [39].
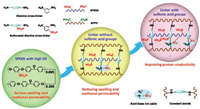
Bromomethylated poly(phenylene oxide) (BPPO), as a kind of industrial product, has been widely used in polymer electrolyte membranes due to its high reactivity of bromobenzyl groups, high mechanical strength and hydrolytic stability, good film-forming ability and low cost [40-42]. Fig. 1 displays the schematic illustration of molecular design for the construction of SPEEK based semi-IPNs through the formed crosslinking networks of BPPO and diamine cross-linkers. One hydrophobic linker with two amine groups, 1, 3-diaminobenzene, was firstly proposed to construct the crosslinking networks of the semi-IPN (CSB-NS) via the alkylation reaction between amine groups and bromobenzyl groups of BPPO. Besides the covalent C—N bonds, the ionic bonds were generated from the amine species of the cross-linking networks and the sulfonic acid groups of SPEEK to further reduce the dimensional swelling of SPEEK. To overcome the drawbacks of reduced proton conductivity of CSB-NS caused by the hydrophobic BPPO, another semi-IPN (CSB-S) was therefore prepared by making use of the sulfonated linker, 2, 4-diaminobenzenesulfonic acid. The results indicated that the high swelling and methanol permeability of the SPEEK membrane were indeed minimized by the fabricated semi-IPNs. Consequently, excellent DMFC performance in high concentration methanol solutions was successfully achieved for the semi-IPN membranes.
|
Download:
|
| Fig. 1. Schematic illustration of molecular design of diamine cross-linkers for constructing the semi-IPN membranes based on SPEEK and BPPO. | |
The chemical structures of BPPO and SPEEK were confirmed by the 1H NMR spectra. The degree of bromomethylation (DB) of BPPO is calculated according to the following Eq. (1):
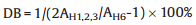
|
(1) |
where AH1, 2, 3 stands for the peak area of the phenyl ring hydrogen atoms (H1, 2, 3) and AH6 is assigned to the methylene peak area (H6) (Fig. S1a in Supporting information). The content of the bromobenzyl groups in the BPPO is calculated to be 57.8%. The bromobenzyl groups are favorable to form the crosslinking networks by reacting with the amine groups of diamine linkers. Otherwise, the degree of sulfonation of the SPEEK is obtained by the Eq. (2) [43]:
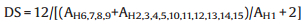
|
(2) |
where AH1 belongs to the H1 peak area (Fig. S1b in Supporting information). The DS of SPEEK is calculated to be 69.5%.
The infrared spectra of the SPEEK, BPPO and CSB-NS membranes are illustrated in Fig. S2a in Supporting information. The peak at 1646 cm-1 is assigned to the stretching vibration of C=O of SPEEK. The peaks at 1252 cm-1 and 1080 cm-1 correspond to the asymmetric and symmetric stretching of O=S=O of SPEEK, respectively. The peak at 598 cm-1 belongs to the stretching vibration of CH-Br [30, 44], which disappears in the CSB-NS membrane due to the alkylation reaction between bromobenzyl groups of BPPO and amine groups of the linkers. It suggests that the construction of crosslinking networks in semi-IPNs is successfully achieved.
The crosslinking networks of the semi-IPNs can be further proved by determining the existence of secondary amine, —NH—, formed from the alkylation reaction between bromobenzyl groups and amine groups. Here, the secondary amine groups are confirmed by determining the valence states of N elements through making use of the XPS spectra. As depicted in Figs. S2b and c (Supporting information), both CSB-NS and CSB-S membranes display a strong N 1s peak with binding energy of 399.7 eV, which is assigned to the N elements in the secondary amine species. It indicates that the covalent crosslinking networks of semi-IPNs are successfully obtained. It is further noted that another weak N 1s peak with the binding energy of 401.5 eV is observed for both membranes. This peak disappears after the membranes are treated with 2 mol/L KOH solution for two days, indicating that the peak is attributed to the protonated amine groups (Figs. S2b' and c' in Supporting information) [45]. It can be seen from the molecular structures of the semi-IPNs that the protonated amine groups can be formed from the interaction between amine species and sulfonic acid groups, which are favorable to the construction of the ionic crosslinking networks. As a consequence, the two types of crosslinking networks (covalent bonds and ionic bonds) can be formed in the semi-IPNs, which are beneficial for the enhanced performance of SPEEK.
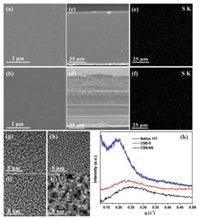
Uniform and compact micromorphology is dispensable for high methanol-permeation resistant PEMs. As seen from the SEM and elemental mapping images, the CSB-NS and CSB-S membranes display dense and homogeneous morphologies at both surface direction (Figs. 2a and b) and cross-section direction (Figs. 2c and d), indicating the good compatibility between BPPO and SPEEK. In addition, the uniform dispersion of S element in the cross-section direction suggests that the SPEEK chains distributes homogeneously in the semi-IPN membranes (Figs. 2e and f). Besides of the similar fully aromatic polymeric backbones of SPEEK and BPPO [46], the formed ionic bonds are also beneficial for improving the compatibility. For the CSB-S membrane, the ionic bonds can be generated not only between sulfonated groups of the linker and amine species, but also between sulfonated groups of SPEEK and amine species (Fig. S3 in Supporting information). Importantly, the acid-base ion pairs can provide facile proton transport pathways for further enhancing proton conductivity [47, 48].
|
Download:
|
| Fig. 2. SEM images of the CSB-NS and CSB-S membranes: surface: (a) CSB-NS and (b) CSB-S; Cross-section: (c) CSB-NS and (d) CSB-S; EDS images: (e) CSB-NS and (f) CSB-S. TEM images of membranes in the dry state: (g) SPEEK, (h) CSB-NS, (i) CSB-S and (j) Nafion 117. (k) The SAXS curves of the hydrated membranes. | |
The construction of high-efficient proton transport channels derived from the aggregation of hydrophilic sulfonic acid groups is responsible for high proton conductivity of a PEM. In general, the efficiency of proton transference can be promoted by improving the size and connection of proton transport channels [49]. In order to directly investigate the effect of non-sulfonated and sulfonated cross-linkers on proton transport channels, the micro-morphologies were determined by the TEM technique as depicted in Figs. 2g–j. The dark regions correspond to the lead acetate stained hydrophilic regions, while the bright regions represent the unstained hydrophobic domains. It is found that the hydrophilic regions of the SPEEK membrane (Fig. 2g) are much smaller than those of Nafion 117 (Fig. 2j), which is consistent with the reported works [22]. Clearly, the hydrophilic regions of the CSB-NS membrane (Fig. 2h) are reduced compared to the neat SPEEK membrane due to the formed covalent and ionic crosslinking networks. However, the hydrophilic regions of the CSB-S membrane (Fig. 2i) are slightly increased after introducing the hydrophilic sulfonated cross-linker. In addition, the distances between hydrophilic regions of the semi-IPN membranes are smaller than SPEEK, which is beneficial for improving the connection of conduction channels and reducing the barriers of proton transfer.
The hydrophilic/hydrophobic phase separation of the membranes under water-saturated level was also investigated by determining the sizes of hydrophilic regions using the SAXS technique. The SAXS curves are depicted in Fig. 2k and the sizes (d, nm) of hydrophilic regions are calculated according to the equation: d = 2π/q, where q (Å-1) is the peak position [50, 51]. The size of hydrophilic regions is calculated to be 2.79 nm for the CBS-S membrane, which is much higher than that of 2.45 nm for the CBS-NS membrane. The enlarged size of hydrophilic regions is attributed to the introduction of the sulfonated cross-linker (2, 4- diaminobenzenesulfonic acid), which gives rise to the increased IEC value and subsequently the high water uptake. In particular, the Nafion 117 membrane displays a much larger hydrophilic region size (3.20 nm) than the CSB-NS and CSB-S membranes due to the superior hydrophilic/hydrophobic phase separation of Nafion 117. The large hydrophilic region size is disadvantageous to prevent methanol molecular shuttling through the Nafion 117 membrane.
Adequate water uptake is essential for a PEM to construct proton conductivity channels as water molecules can serve as medium for proton transport [52]. However, excessive water uptake always leads to high dimensional swelling and insufficient mechanical strength, which give rise to reduced DMFC performance [53, 54]. The water uptake and dimensional swelling of the membranes at both 30 ℃ and 60 ℃ are depicted in Figs. 3a and b, respectively. It clearly indicates that the neat SPEEK membrane with a high IEC value displays an over swelling behavior at 60 ℃. Therefore, the neat SPEEK membrane is not suitable to be used in DMFCs. It is noted that the dimensional swelling of the SPEEK membrane is significantly decreased by blending with BPPO (the SP/BP membrane). This is due to the reduced IEC value and water uptake caused by hydrophobic BPPO polymeric chains. However, the high area swelling of 64.16% for SP/BP at 60 ℃ is still not suitable to the practical application in DMFCs. Fortunately, the dimensional swelling of SPEEK can be further decreased by constructing the crosslinking networks. The SPEEK polymer chains are well interpenetrated into the in-situ fabricated crosslinking networks to form the semi-IPNs (CBS-NS and CSB-S). In addition, the produced ionic bonds between amine species of the crosslinking networks and sulfonic acid groups of SPEEK give another contribution to enhance dimensional stability.
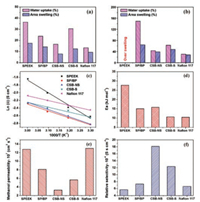
|
Download:
|
| Fig. 3. Water uptake and area swelling of the membranes at (a) 30 ℃ and (b) 60 ℃. (c) Proton conductivity, (d) activation energy, (e) methanol permeability and (f) relative selectivity of the membranes. | |
Proton conductivity is one of the most critical parameters for a PEM [55]. The temperature dependence of proton conductivity of the membranes is shown in Fig. 3c. It is observed that the proton conductivity of the SP/BP membrane is decreased compared to the neat SPEEK membrane due to the reduced density of sulfonated groups. However, the proton conductivity of the CSB-NS and CSB-S membranes is inversely increased compared to the SP/BP membrane even though they possess the similar IEC values (Table S1 in Supporting information). The enhanced proton conductivity is aroused from the formed acid-base interaction between amine species and sulfonic groups as the protophilic nature of amine species promotes the dissociation of protons from sulfonated groups [36]. In particular, the CSB-S membrane shows much higher proton conductivity than the CSB-NS membrane, which is due to the enlarged size of hydrophilic regions generated from the sulfonated cross-linker (Fig. 2).
The activation energy Ea (kJ/mol) of proton conductivity can be calculated according to the following Arrhenius-type Equation: lnσ =-Ea/RT + lnσ0, where R is the pure gas constant (8.314 J mol-1 K-1), T is the temperature (K) and σ0 is the pre-exponential factor (S/cm). Fig. 3d shows the calculated Ea values of the membranes. The neat SPEEK membrane possesses the highest activation energy (27.7 kJ/mol) due to the poorly connected proton transport channels. The SP/BP membrane demonstrates much lower activation energy than the neat SPEEK membrane, which is attributed to the enhanced hydrophilic/hydrophobic phase separation by the hydrophobic BPPO. The same result is also observed for the SPEEK/PVDF composite membranes [56]. The activation energy CSB-NS membrane is slightly increased due to the obstruction of the hydrophobic cross-linker on proton transport. However, the activation energy of the CSB-S is largely reduced to 10.64 kJ/mol, which is even comparable to the value of 10.48 kJ/mol for Nafion 117. The result suggests that introducing sulfonated cross-linker in the semi-IPN is favorable for constructing highefficient proton channels.
High methanol-permeation resistance is desired for a PEM to approach excellent DMFC performance in high concentration methanol solutions. The methanol permeability of membranes is shown in Fig. 3e. It is found that the neat SPEEK membrane displays high methanol permeability of 12.79×10-7 cm2/s, which is close to the value of 13.01×10-7 cm2/s offered by Nafion 117. However, the SP/BP membrane displays reduced methanol permeability of 8.10×10-7 cm2/s due to the hydrophobic feature of BPPO. The value is further decreased to 3.26×10-7 cm2/s for the CSB-NS membrane, which is almost 4 times lower than the value of Nafion 117. The improved methanol-permeability resistance is attributed to the reduced size of hydrophilic regions. In addition, the formed ionic "barriers" from the acid-base interactions between sulfonated groups and amine species also play important roles in reducing methanol crossover through the CSB-NS membrane [57]. Unfortunately, the methanol permeability of CSB-S is increased up to 5.67×10-7 cm2/s due to the enlarged hydrophilic region size. Relative selectivity, which is defined as the ratio of proton conductivity to methanol permeability, has been commonly used to evaluate the comprehensive performance of a PEM. The CSB-NS membrane exhibits the highest relative selectivity of 18.12×104 S s cm-3, which is almost three times higher than that of Nafion 117 (6.64×104 S s cm-3) (Fig. 3f). The results imply that the as-prepared CSB-NS membrane is more suitable than Nafion 117 for the DMFC application.
Fig. S4a shows the mechanical strength of the hydrated membranes. The mechanical strength of the SPEEK membrane is recorded to be 26.1 MPa, which is much lower than the reported value of 52.3 MPa for SPEEK with a low DS (61%) in our previous work [58]. This is attributed to the weakened interaction between the SPEEK polymeric backbones derived from the high water uptake and dimensional swelling. However, the SP/BP membrane displays an enhanced mechanical strength due to the inherently excellent mechanical property of BPPO and the reduced dimensional swelling. The value is further increased for the CSB-NS membrane due to the formed covalent and ionic crosslinking networks. It is noted that the CSB-S membrane shows a slightly reduced mechanical strength of 44.9 MPa compared to the CSB-NS membrane because of the higher water uptake and swelling. In addition, the CSB-NS membrane with robust mechanical strength is shown in Fig. S4b in Supporting information. The results suggest that the semi-IPN technique is beneficial for improving mechanical strength of PEMs. The oxidative stability of the membranes is determined in Fenton reagent at 60 ℃ and the remaining weight is applied to reflect the oxidative stability. As seen from the results in Table S1 (Supporting information), the oxidative stability of the semi-IPN membranes is significantly improved by the formed covalent and ionic crosslinking networks, which is comparable to that of the Nafion 117 membrane.
Fig. 4 displays the polarization curves of the DMFCs assembled with the SPEEK, CSB-NS, CSB-S, and Nafion 117 membranes. As depicted in Fig. 4a, the CSB-NS and CSB-S DMFCs display higher power output than the Nafion 117 and SPEEK DMFCs in 1 mol/L methanol solution. The highest power output of 24.1 mW is obtained for the CSB-NS DMFC, which is ca. 28% higher than that of the Nafion 117 DMFC. When the methanol concentration is increased to 5 mol/L, the value is largely increased up to ca. 281% (Fig. 4b). The excellent cell performance of the CSB-NS DMFC is attributed to the high methanol-permeability resistance of the CSB-NS membrane. In addition, the cell performance of the SPEEK DMFC with the methanol concentrations from 1 mol/L to 4 mol/L was also determined for comparison in Fig. 4c. It is observed that the polarization curves of the SPEEK DMFC become irregular when the methanol concentration is increased to 4 mol/L, which is caused by the poor dimensional stability of the SPEEK membrane with a high DS of 69.5%.
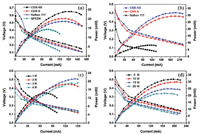
|
Download:
|
| Fig. 4. (a) DMFC performance comparison of the CSB-NS, CSB-S, Nafion 117 and SPEEK membranes with 1 mol/L methanol solution, (b) DMFC performance comparison of the CSB-NS, CSB-S and Nafion 117 membranes with 5 mol/L methanol solution, (c) DMFC performance of the SPEEK DMFC with methanol concentrations from 1 mol/L to 4 mol/L; (d) DMFC performance of the CSB-NS DMFC with methanol concentrations from 5 mol/L to 20 mol/L at 25 ℃. | |
Fig. 4d exhibits the polarization curves of the CSB-NS DMFC with the methanol solution from 5 mol/L to 20 mol/L. The power output of the CSB-NS DMFC decreases as the methanol concentrations increase. However, the CSB-NS DMFC displays only a 6.3% decline of the maximum power in 10 mol/L methanol solution compared to the value offered by the 5 mol/L methanol solution, which is due to the excellent methanol-permeation resistance of the CSB-NS membrane. The results suggest that the CSB-NS membrane is much more suitable than the Nafion 117 membrane to be applied in high concentration DMFCs.
In conclusion, the semi-IPNs were constructed by forming the crosslinking networks via the reaction between BPPO and diamine cross-linkers. The constructed semi-IPNs are beneficial for improving the dimensional stability, mechanical strength, oxidative stability and methanol-permeation resistance of the SPEEK membrane. In particular, the CSB-NS membrane exhibits remarkably reduced methanol permeability compared to the Nafion 117 membrane. Thus, the CSB-NS membrane possesses almost three times higher relative selectivity than Nafion 117. As a consequence, the CSB-NS DMFC exhibits much higher maximum power output than the Nafion 117 DMFC in high concentration methanol solution. The results suggest that preparing semi-IPN membranes toward SPEEK is a facile way to achieve high-performance PEMs in DMFCs.
AcknowledgmentsThe authors gratefully acknowledge support of the National Natural Science Foundation of China (Nos. 21603197, 21703212,21233006 and 21473164), Natural Science Foundation of Hube Province of China (No. 2016CFB181), Fundamental Research Funds for the Central University, China University of Geosciences (Wuhan) (No. CUGL180403) and China University of Geosciences (Wuhan) for the program of Center for Advanced Energy Research and Technologies.
Appendix A. Supplementary dataSupplementary material related to this article can be found, in the online version, at doi: https://doi.org/10.1016/j.cclet.2018.09.021.
| [1] |
C. Laberty-Robert, K. Valle, F. Pereira, C. Sanchez, Chem. Soc. Rev. 40 (2011) 961-1005. DOI:10.1039/c0cs00144a |
| [2] |
N. Li, M.D. Guiver, Macromolecules 47 (2014) 2175-2198. DOI:10.1021/ma402254h |
| [3] |
X. Liu, Y. Zhang, Y. Chen, et al., Energ. Technol.-Ger. 6 (2018) 2264-2272. DOI:10.1002/ente.201800274 |
| [4] |
J. Li, W. Cai, Y. Zhang, et al., Electrochim. Acta 151 (2015) 168-176. DOI:10.1016/j.electacta.2014.11.047 |
| [5] |
Y. Li, J. Song, J. Yang, Chin. Sci. Bull. 57 (2012) 4153-4159. DOI:10.1007/s11434-012-5448-9 |
| [6] |
Y. Choi, Y. Kim, K.Y. Kang, J.S. Lee, Carbon 49 (2011) 1367-1373. DOI:10.1016/j.carbon.2010.11.056 |
| [7] |
Y. Choi, Y. Kim, H.K. Kim, J.S. Lee, J. Membr. Sci. 357 (2010) 199-205. DOI:10.1016/j.memsci.2010.04.024 |
| [8] |
Y. Zhang, J. Li, L. Ma, W. Cai, H. Cheng, Energy Technol.-Ger. 3 (2015) 675-691. DOI:10.1002/ente.201500028 |
| [9] |
Y.F. Liang, H.Y. Pan, X.L. Zhu, Y.X. Zhang, X.G. Jian, Chin. Chem. Lett. 18 (2007) 609-612. DOI:10.1016/j.cclet.2007.03.031 |
| [10] |
V. Parthiban, S. Akula, S.G. Peera, N. Islam, A.K. Sahu, Energy Fuel. 30 (2016) 725-734. DOI:10.1021/acs.energyfuels.5b02194 |
| [11] |
A.K. Mohanty, E.A. Mistri, S. Banerjee, H. Komber, B. Voit, Ind. Eng. Chem. Res. 52 (2013) 2772-2783. DOI:10.1021/ie303380a |
| [12] |
H. Beydaghi, M. Javanbakht, E. Kowsari, Ind. Eng. Chem. Res. 53 (2014) 16621-16632. DOI:10.1021/ie502491d |
| [13] |
R. Guo, O. Lane, D. VanHouten, J.E. McGrath, Ind. Eng. Chem. Res. 49 (2010) 12125-12134. DOI:10.1021/ie100785t |
| [14] |
C. Felice, S. Ye, D. Qu, Ind. Eng. Chem. Res. 49 (2010) 1514-1519. DOI:10.1021/ie901600a |
| [15] |
T. Haolin, P. Mu, M. Shichun, Y. Runzhang, Chin. Sci. Bull. 50 (2005) 377-379. |
| [16] |
Y.M. Kim, K.W. Park, J.H. Choi, I.S. Park, Y.E. Sung, Electrochem. Commun. 5 (2003) 571-574. DOI:10.1016/S1388-2481(03)00130-9 |
| [17] |
H. Hou, G. Sun, Z. Wu, W. Jin, Q. Xin, Int. J. Hydrogen Energy 33 (2008) 3402-3409. DOI:10.1016/j.ijhydene.2008.03.060 |
| [18] |
S.E. Nam, S. Park, W.C. Choi, J.W. Lee, Y. Kang, Macromol. Res. 21 (2013) 1314-1321. DOI:10.1007/s13233-013-1183-6 |
| [19] |
J. Li, W. Cai, L. Ma, et al., Chem. Commun. 51 (2015) 6556-6559. DOI:10.1039/C4CC09420D |
| [20] |
C. Li, Y. Zhang, X. Liu, et al., J. Mater. Sci. 53 (2017) 5501-5510. |
| [21] |
H.Y. Pan, Y.F. Liang, X.L. Zhu, X.G. Jian, Chin. Chem. Lett. 18 (2007) 1148-1150. DOI:10.1016/j.cclet.2007.06.032 |
| [22] |
K.D. Kreuer, J. Membr. Sci. 185 (2001) 29-39. DOI:10.1016/S0376-7388(00)00632-3 |
| [23] |
C. Li, X. Liu, Y. Zhang, et al., Energy Technol.-Ger. (2018). DOI:10.1002/ente.201700754 |
| [24] |
T. Yang, Int. J. Hydrogen Energy 33 (2008) 6772-6779. DOI:10.1016/j.ijhydene.2008.08.022 |
| [25] |
Q. Che, B. Sun, R. He, Electrochim. Acta 53 (2008) 4428-4434. DOI:10.1016/j.electacta.2008.01.028 |
| [26] |
Q. Che, R. He, J. Yang, L. Feng, R.F. Savinell, Electrochem. Commun. 12 (2010) 647-649. DOI:10.1016/j.elecom.2010.02.021 |
| [27] |
T.Y. Inan, H. Doğan, E.E. Unveren, E. Eker, Int. J. Hydrogen Energy 35 (2010) 12038-12053. DOI:10.1016/j.ijhydene.2010.07.084 |
| [28] |
M. Li, G. Zhang, S. Xu, et al., J. Power Sources 255 (2014) 101-107. DOI:10.1016/j.jpowsour.2013.12.116 |
| [29] |
J. Tsai, H. Cheng, J.F. Kuo, Y. Huang, C. Chen, J. Power Sources 189 (2009) 958-965. DOI:10.1016/j.jpowsour.2008.12.071 |
| [30] |
Z. Li, L. Liu, L. Yu, et al., J. Power Sources 272 (2014) 427-435. DOI:10.1016/j.jpowsour.2014.08.101 |
| [31] |
C. Li, Z. Yang, X. Liu, et al., Int. J. Hydrogen Energy 42 (2017) 28567-28577. DOI:10.1016/j.ijhydene.2017.09.166 |
| [32] |
H. Li, G. Zhang, J. Wu, et al., J. Power Sources 195 (2010) 8061-8066. DOI:10.1016/j.jpowsour.2010.06.106 |
| [33] |
S.D. Mikhailenko, K. Wang, S. Kaliaguine, et al., J. Membr. Sci. 233 (2004) 93-99. DOI:10.1016/j.memsci.2004.01.004 |
| [34] |
A. Khabibullin, S.D. Minteer, I. Zharov, J. Mater. Chem. A 2 (2014) 12761-12769. DOI:10.1039/C4TA01202J |
| [35] |
E. Yan, J. Wang, Z. Jiang, et al., J. Mater. Chem. A 1 (2013) 11762-11777. DOI:10.1039/c3ta11620d |
| [36] |
Y. Yin, W. Deng, H. Wang, et al., J. Mater. Chem. A 3 (2015) 16079-16088. DOI:10.1039/C5TA03276H |
| [37] |
P. Deivanayagam, A.R. Ramamoorthy, J. Macromol. Sci. A 49 (2012) 191-200. DOI:10.1080/10601325.2012.649197 |
| [38] |
X. Wu, G. He, X. Li, J. Benziger, et al., J. Power Sources 246 (2014) 482-490. DOI:10.1016/j.jpowsour.2013.07.108 |
| [39] |
L. Chikh, V. Delhorbe, O. Fichet, J. Membr. Sci. 368 (2011) 1-17. DOI:10.1016/j.memsci.2010.11.020 |
| [40] |
T. Xu, D. Wu, L. Wu, Prog. Polym. Sci. 33 (2008) 894-915. DOI:10.1016/j.progpolymsci.2008.07.002 |
| [41] |
L. Wu, Q. Pan, J.R. Varcoe, et al., J. Membr. Sci. 490 (2015) 1-8. DOI:10.1016/j.memsci.2015.04.046 |
| [42] |
S.H. Yun, J.J. Woo, S.J. Seo, et al., J. Membr. Sci. 367 (2011) 296-305. DOI:10.1016/j.memsci.2010.11.017 |
| [43] |
X. Liu, Y. Zhang, Y. Chen, et al., J. Membr. Sci. 544 (2017) 58-67. DOI:10.1016/j.memsci.2017.09.013 |
| [44] |
L. Zeng, T.S. Zhao, J. Power Sources 303 (2016) 354-362. DOI:10.1016/j.jpowsour.2015.11.019 |
| [45] |
C. Fang, D. Julius, S.W. Tay, L. Hong, J.Y. Lee, J. Phys. Chem. B 116 (2012) 6416-6424. DOI:10.1021/jp2081353 |
| [46] |
Y. Zhang, R. Rohan, W. Cai, et al., ACS Appl. Mater. Interfaces 6 (2014) 17534-17542. DOI:10.1021/am503152m |
| [47] |
H. Wu, X. Shen, T. Xu, W. Hou, Z. Jiang, J. Power Sources 213 (2012) 83-92. DOI:10.1016/j.jpowsour.2012.04.003 |
| [48] |
J. Wang, H. Bai, H. Zhang, et al., Electrochim. Acta 152 (2015) 443-455. DOI:10.1016/j.electacta.2014.11.165 |
| [49] |
Y. Zhang, C. Li, X. Liu, et al., RSC Adv. 6 (2016) 79593-79601. DOI:10.1039/C6RA17477A |
| [50] |
Q. He, T. Xu, H. Qian, et al., J. Power Sources 278 (2015) 590-598. DOI:10.1016/j.jpowsour.2014.12.135 |
| [51] |
N. Li, T. Yan, Z. Li, et al., Energy Environ. Sci. 5 (2012) 7888-7892. DOI:10.1039/c2ee22050d |
| [52] |
M. Ingratta, E.P. Jutemar, P. Jannasch, Macromolecules 44 (2011) 2074-2083. DOI:10.1021/ma102879w |
| [53] |
J. Pang, H. Zhang, X. Li, Z. Jiang, Macromolecules 40 (2007) 9435-9442. DOI:10.1021/ma070080s |
| [54] |
X.F. Li, F.P.V. Paoloni, E.A. Weiber, Z.H. Jiang, P. Jannasch, Macromolecules 45 (2012) 1447-1459. DOI:10.1021/ma201599p |
| [55] |
K. Firouz Tadavani, A. Abdolmaleki, M.R. Molavian, et al., Energy Fuel. 31 (2017) 11460-11470. DOI:10.1021/acs.energyfuels.7b01065 |
| [56] |
H.Y. Jung, J.K. Park, Electrochim. Acta 52 (2007) 7464-7468. DOI:10.1016/j.electacta.2007.06.035 |
| [57] |
L. Gao, T. Kong, G. Guo, Y. Huo, Int. J. Hydrogen Energy 41 (2016) 20373-20384. DOI:10.1016/j.ijhydene.2016.08.048 |
| [58] |
X. Liu, Z. Yang, Y. Zhang, et al., Int. J. Hydrogen Energy 42 (2017) 10275-10284. DOI:10.1016/j.ijhydene.2017.02.128 |
 2019, Vol. 30
2019, Vol. 30 

