b Shanghai Collaborative Innovation Center for Biomanufacturing Technology, Shanghai 200237, China
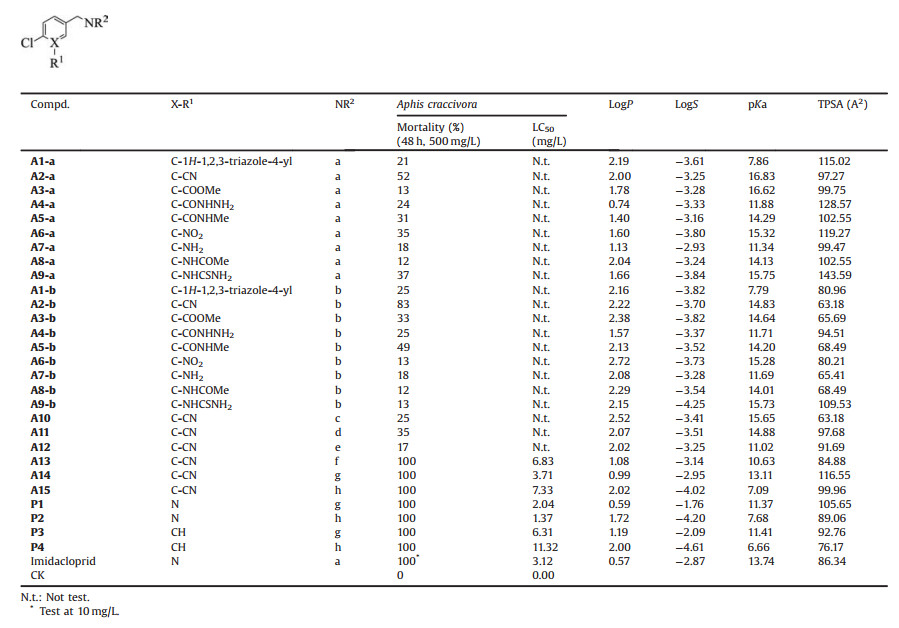
Water molecules in ligand binding pockets can bridge interactions between ligands and proteins through H-bonds, namely water-bridged H-bonds. Water bridges have been reported in numerous protein-ligand complex structures, and observed playing crucial roles in many cases of ligand-receptor recognitions [1-3]. If a water bridge is flexible in the binding pocket, it can be mimicked or replaced by other fragments. In drug discovery and optimization, such a strategy has been fully applied to get novel ligand scaffolds or improve binding affinities [4-7]. For example, by replacing the role of water bridge, a number of pharmaceutical instances have been successfully achieved in lead optimization of EGFR [8, 9], HIV-1 protease [10], etc.
In the cases of pesticides, water-bridged H-bonds also played key roles in the binding of insect nicotinic acetylcholine receptors (nAChRs) with neonicotinoids [11-14]. Neonicotinoids were agonists of insect nAChRs [14, 15], which occupied the largest insecticides market share of the world due to their excellent widespectrum insecticidal activity and safety to mammals [16-18], and gained continuous research concern [19-22]. In crystal structures of neonicotinoids with acetylcholine binding proteins (AChBP), good surrogates for the extracellular domain of nAChRs, a water molecule was captured next to the pyridine/thiazole nitrogen atom of neonicotinoids in the binding pocket, which bridged crucial Hbonding network with the relevant surrounding residues [23, 24]. The pyridine/thiazole nitrogen atom served as H-bond acceptor in the binding pocket (Fig. 1), whose role could also be replaced by a fluorine or a carbonyl oxygen of alkyl substituents in neonicotinoid analogues [11]. QSAR study revealed that the insecticidal activities of neonicotinoids were deeply correlated with the water-bridged H-bonds abilities [12]. When the water-bridged H-bonds were broken in neonicotinoids N-oxide analogues, the insecticidal activities would be weak [13].
|
Download:
|
| Fig. 1. Design strategy of neonicotinoid derivatives by mimicking water bridge | |
Although water-bridged H-bonds are reported in the recognition between neonicotinoids and nAChRs, few studies are focused on the water bridge itself. The detailed roles of water bridges on neonicotinoids are still open questions. It is still unknown whether the water bridge could be replaced by other structure fragments in pesticide molecular design. In this work, nine structure fragments that may mimic the roles of water molecule, including 1H-1, 2, 3- triazole, CN, COOMe, CONHNH2, CONHMe, NO2, NH2, NHCOMe and NHCSNH2 were selected to occupy the binding site of water bridge, and then twenty-four neonicotinoid compounds were designed, synthesized, bioassayed and modeled to capture the detailed roles of water bridge.
The synthesis of target compounds A1-a and A1-b was shown in Scheme S1 in Supporting information. The compound 2 was obtained in 80% yield from commercially available compound 1 as described in the literature [25]. Intermediate 3 was obtained in 65% yield through deoxidation of compound 2, then was subjected to a Sonogashira coupling reaction and desilylation to provide compound 4 in 91% yield. Conversion of compound 4 to compounds 6-a and 6-b was accomplished through chlorination and then aminolysis with relevant key intermediates a and b respectively. Lastly, the triazole substituted compounds A1-a and A1-b were gained by a modified Click reaction with excellent yields.
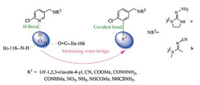
The synthesis of target compounds A2-a, A2-b was shown in Scheme 1. Intermediate 8 was obtained in 80% yield from compound 7 through a palladium-catalyzed ortho-halogenation. Based on compound 8, the target compounds were readily obtained through two steps involving a bromination reaction and a substitution reaction.
|
Download:
|
| Scheme 1. Synthesis route of cyano-substituted compounds A2-a and A2-b | |
The synthesis route of target compounds A3-a, A3-b, A4-a, A4- b, A5-a and A5-b was shown in Scheme S2 in Supporting information, and synthesis of target compounds A6-a, A6-b, A7- a, A7-b, A8-a, A8-b, A8-b, A9-a and A9-b were shown in Scheme S3 in Supporting information.
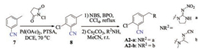
The synthesis of target compounds A10~A15 were depicted in Scheme 2. Compounds A10~A13 were gained through the similar routes with previous compounds A2-a and A2-b. Compounds A14 and A15 were prepared according to the procedures in our previous studies [26, 27]. Compound A14 containing dihydroxysubstituted pyrrole fused ring was generated by treatment A13 with methylglyoxal. Conjugated diene compound A15 was afforded Stirring the mixture of intermediate A13 and furfural in the presence of a catalyst amount of hydrogen chloride gas. Compounds P1 [26] and P2 [27] have been reported in our previous studies, which were also generated in this work for comparison. P3 and P4 were synthesized according to the procedures of P1 [26] and P2 [27], respectively.

|
Download:
|
| Scheme 2. Synthesis route of cyan-substituted compounds | |
Then insecticidal activities against Aphis craccivora of all compounds were evaluated by leaf-dip method (Supporting information). Compounds A2-a and A2-b with cyano group showed the best insecticidal activities as compared with the other compounds. The cyano group has been also successfully used in rational design of inhibitors of scytalone dehydratase [28], EGFR kinase [29] and p38α MAP kinase [30], through displacing the key water molecule in active sites. Accordingly, the cyano group was considered as the optimal fragment that could well mimic the role of water bridge and was chosen in further structure derivations.
Six cyano-substituted compounds A10~A15 with different scaffolds were synthesized and bioassayed. Insecticidal activities of compounds A13~A15 exhibited 100% mortality at 500 mg/L against cowpea aphids. Further bioassay showed that the LC50 values of two cis-nitromethylene compounds A14 and A15 were 3.71 mg/L and 7.33 mg/L, respectively, which were similar to that of imidacloprid (3.12 mg/L). Compounds A14 and A15 showed relative low insecticidal activities when compared with their corresponding chlorine pyridine prototypes P1 (2.04 mg/L) and P2 (1.37 mg/L), respectively. In addition, A14 and A15 displayed stronger insecticidal activities than the phenyl structures without cyano substitution P3 (6.31 mg/L) and P4 (11.32 mg/L), respectively. It indicated that the cyano substitution could contribute to the insecticidal activities, while the contribution was not as large as a pyridine ring.
Physical-chemical properties (logP, logS, pKa and TPSA) of all compounds were calculated (methods were shown in Supporting information) and then analyzed between the new cyano-substituted compounds versus imidacloprid. As shown in Table 1, the calculated logP, logS, pKa and TPSA values of new compounds are similar to those of their prototypes without cyano group (P1–P4). Accordingly, the difference in insecticidal activities could not be resulted only from the pesticide-like properties.
|
|
Table 1 Physicochemical property and insecticidal activities of the target compounds and imidacloprid against cowpea aphids (Aphis craccivora) |
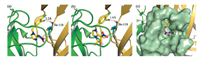
Docking studies were performed on compounds A2-a and A14 with Ac-AChBP to illustrate the difference of insecticidal activities and detailed interactions between compounds and target (method was shown in Supporting information). As displayed in Fig. 2, a N-H…N H-bond, instead of the water bridged H-bond network, was observed between residue Ile-118 and the cyano groups of A2- a and A14, respectively. The binding pose of A2-a was very similar to imidacloprid and aligned well with imidacloprid (Fig. 2c). In the crystal structures of imidacloprid with AChBP, the water molecule could act as both donor and acceptor, and bridged two H-bonds with Ile-106 and Ile-118, respectively [23, 24]. Although the insecticidal activities indicated that the cyano group was the optimal group mimicking the water bridge among all nine tested fragments, it could act only as H-bond acceptor and formed a single H-bond with target, which might be one reason why the new cyano-substituted compounds displayed relative low insecticidal activities when compared with their corresponding leads.
|
Download:
|
| Fig. 2. The docking poses of compounds within Ac-AChBP. The binding poses of A2-a (a), A14 (b), and molecular alignment diagram of A2-a and imidacloprid (c) | |
On the other hand, the fragments 1H-1, 2, 3-triazole, CONHNH2, CONHMe, NHCSNH2 and NHCOMe could act both as H-bond donor and acceptor, however, they were larger than the cyano in size. The weak activities of such fragment-substituted compounds indicated that the active water site might be small in volume. In the complex crystal structure of AChBP-imidacloprid, the water bridge located in a deep inner site of the binding pocket [23, 24], implying that the water site was difficult to be occupied by other groups, even by the cyano group. It illustrated that the water bridge was essential for high insecticidal activities of neonicotinoids.
In summary, to explore the key roles of water bridges in neonicotinoids recognition with receptor, nine fragments including 1H-1, 2, 3-triazole, CN, COOMe, CONHNH2, CONHMe, NO2, NH2, NHCOMe and NHCSNH2 were introduced into the molecular design of neonicotinoids, and then twenty-four neonicotinoid compounds were synthesized, bioassayed and evaluated. The cyano-substituted compounds showed better insecticidal activities than other fragment-substituted compounds, thus the cyano group was considered as the optimal fragment mimicking water bridge. Although, the cyano-substituted compounds displayed similar pesticide-like properties and binding poses with their corresponding prototype structures, they showed relative weak insecticidal activities. It revealed that the water bridge might be stable in the active site and was difficult to replace by other groups, which illustrated again the significance of water-bridged H-bonds in the recognition of neonicotinoids with insect nAChRs.
AcknowledgmentsWe thank for the financial supports from the National Natural Science Foundation of China (Nos. 21572059, 21172070), Innovation Program of Shanghai Municipal Education Commission (No. 201701070002E00037), and the Fundamental Research Funds for the Central Universities.
Appendix A. Supplementary dataSupplementary material related to this article can be found, in the online version, at doi:https://doi.org/10.1016/j.cclet.2018.05.013.
| [1] |
J. Yin, J.C. Mobarec, P. Kolb, D.M. Rosenbaum, Nature 519 (2015) 247-250. DOI:10.1038/nature14035 |
| [2] |
M. Nagae, A. Ikeda, M. Hane, et al., J. Biol. Chem. 288 (2013) 33784-33796. DOI:10.1074/jbc.M113.496224 |
| [3] |
M. Adler, D.D. Davey, G.B. Phillips, et al., Biochemistry 39 (2000) 12534-12542. DOI:10.1021/bi001477q |
| [4] |
S.B.A. Beer, N.P.E. Vermeulen, C. Oostenbrink, Curr. Top. Med. Chem. 10 (2010) 55-56. DOI:10.2174/156802610790232288 |
| [5] |
J. Seo, J. Igarashi, H.Y. Li, et al., J. Med. Chem. 50 (2007) 2089-2099. DOI:10.1021/jm061305c |
| [6] |
G. Rastelli, Z.Q. Tian, Z. Wang, D. Myles, Y. Liu, Bioorg. Med. Chem. Lett. 15 (2005) 5016-5021. DOI:10.1016/j.bmcl.2005.08.013 |
| [7] |
S. Ness, R. Martin, A.M. Kindler, et al., Biochemistry 39 (2000) 5312-5321. DOI:10.1021/bi992505b |
| [8] |
A. Wissner, M.B. Floyd, S.K. Rabindran, et al., Bioorg. Med. Chem. Lett. 12 (2002) 2893-2897. DOI:10.1016/S0960-894X(02)00598-X |
| [9] |
C.J. Torrance, P.E. Jackson, E. Montgomery, et al., Nat. Med. 6 (2000) 1024-1028. DOI:10.1038/79534 |
| [10] |
P.Y.S. Lam, P.K. Jadhav, C.J. Eyermann, et al., Science 263 (1994) 380-384. DOI:10.1126/science.8278812 |
| [11] |
I. Ohno, M. Tomizawa, K.A. Durkin, J.E. Casida, S. Kagabu, J. Agric. Food Chem. 57 (2009) 2436-2440. DOI:10.1021/jf803985r |
| [12] |
S. Xia, J.G. Cheng, Y. Feng, et al., Chin. J. Chem. 32 (2014) 324-334. DOI:10.1002/cjoc.201400112 |
| [13] |
W.W. Zhang, Y.F. Fan, Y. Tao, et al., Chin. J. Chem. 30 (2012) 357-361. DOI:10.1002/cjoc.v30.2 |
| [14] |
M. Tomizawa, J. Casida, Acc. Chem. Res. 42 (2009) 260-269. DOI:10.1021/ar800131p |
| [15] |
K. Matsuda, S.D. Buckingham, D. Kleier, et al., Trends Pharmacol. Sci. 22 (2001) 573-580. DOI:10.1016/S0165-6147(00)01820-4 |
| [16] |
P. Jeschke, R. Nauen, M.E. Beck, Angew. Chem. Int. Ed.. 52 (2013) 9464-9485. DOI:10.1002/anie.v52.36 |
| [17] |
X.S. Shao, P.W. Lee, Z.W. Liu, et al., J. Agric. Food Chem. 59 (2011) 2943-2949. DOI:10.1021/jf103499x |
| [18] |
M. Tomizawa, J.E. Casida, Annu. Rev. Pharmacol. Toxicol. 45 (2005) 247-268. DOI:10.1146/annurev.pharmtox.45.120403.095930 |
| [19] |
C.W. Sun, T. Fang, J. Wang, Z.B. Hao, S.B. Nan, J. Agric. Food Chem. 60 (2012) 9553-9561. DOI:10.1021/jf3024479 |
| [20] |
W.C. Su, Y.H. Zhou, Y.Q. Ma, et al., J. Agric. Food Chem. 60 (2012) 5028-5034. DOI:10.1021/jf300616x |
| [21] |
Q.Q. Hou, Y.F. Jing, X.S. Shao, Chin. Chem. Lett. 28 (2017) 1723-1726. DOI:10.1016/j.cclet.2017.05.016 |
| [22] |
D.D. Zhang, S.X. Cui, Z.P. Xu, D.M. Li, Z.Z. Tian, Chin. Chem. Lett. 28 (2017) 1743-1745. DOI:10.1016/j.cclet.2017.05.002 |
| [23] |
M. Ihara, T. Okajima, A. Yamashita, et al., Invertebr. Neurosci. 8 (2008) 71-81. DOI:10.1007/s10158-008-0069-3 |
| [24] |
T.T. Talley, M. Harel, R.E. Hibbs, et al., Proc. Natl. Acad. Sci. U. S. A. 105 (2008) 7606-7611. DOI:10.1073/pnas.0802197105 |
| [25] |
L. Kraszkiewicz, M. Sosnowski, L. Skulski, Synthesis 7 (2006) 1195-1199. |
| [26] |
Z.J. Ye, L.N. Shi, X.S. Shao, et al., J. Agric. Food. Chem. 61 (2013) 312-319. DOI:10.1021/jf3044132 |
| [27] |
X.S. Shao, Z. Li, X.H. Qian, X.Y. Xu, J. Agric. Food Chem. 57 (2009) 951-957. DOI:10.1021/jf803305f |
| [28] |
J.M. Chen, S.L. Xu, Z. Wawrzak, G.S. Basarab, D.B. Jordan, Biochemistry 37 (1998) 17735-17744. DOI:10.1021/bi981848r |
| [29] |
A. Wissner, D.M. Berger, D.H. Boschelli, et al., J. Med. Chem. 43 (2000) 3244-3256. DOI:10.1021/jm000206a |
| [30] |
C.J. Liu, S.T. Wrobleski, J. Lin, et al., J. Med. Chem. 48 (2005) 6261-6270. DOI:10.1021/jm0503594 |
 2019, Vol. 30
2019, Vol. 30