b Guangdong Innovative Research Team, State Key Laboratory of Luminescent Materials and Devices, South China University of Technology, Guangzhou 510641, China;
c Department of Chemistry, Institute for Advanced Study, Institute of Molecular Functional Materials, and State Key Laboratory of Molecular Neuroscience, The Hong Kong University of Science & Technology, Hong Kong, China
Porphyrin and its derivatives are one kind of the most intensively investigated heterocyclic organic compounds in more than a century [1, 2]. The macrocyclic topology and electronic structure makes porphyrin perfect in coordination with metal cations, highly aromatic, optically and electronically active yet highly stable in chemistry. It is well-known that porphyrins have shown fascinating performances for catalysts, molecular recognition, infra-red fluorescent sensors, building blocks of electron/energy transfer arrays, photodynamic therapy agents, and so on [3-11]. These promising applications are driving the continuous development of design and synthesis of functional porphyrins. Up to now, a variety of different structures and functions have been achieved by chemical modification with proper substituents at the meso- and/or β-positions of the porphine core [12-16]. Most porphyrin derivatives, no matter meso- or β-substituted ones, are prone to form aggregates in solution with high concentration and in solid states, due to the intrinsic strong intermolecular π-π interaction, which tends to heavily quench the fluorescence emission [17-19], a photophysical process termed as aggregation-caused quenching (ACQ) [20]. The ACQ effect greatly influences the optical properties and limits their practical applications as the active component in fluorescent sensors and photodynamic therapy agents.
To tackle this thorny problem, three strategies have been explored. The first strategy, also the classical one, is to prevent the luminogens from close packing thereby prohibits the ACQ effect. To this end, a few schemes, such as decorating porphyrin cores with dendritic branches, introducing porphyrins into metal organic frameworks, and burying porphyrins into hydrogels or silica matrix, have been attempted [21-28]. All these attempts have shown positive impact on the alleviation of ACQ effect. However, complex synthetic steps are necessary and the alleviation effect is still limited because molecular aggregation is a thermodynamically spontaneous process.
The second category is to make use of the advantageous molecular aggregation. It is well-accepted that the formation of J-aggregates can lead to enhanced and red-shifted emission for the π-conjugated luminogens. But pure J-aggregate of porphyrin derivatives have been rarely found, because there are no well-developed rules to guide the design of J-aggregate [29-33]. Consequently, the achievement of J- or other kinds of aggregates (e.g., H-aggregate and X-aggregate) depends on experimental condition, the chemical structure and property of the substituents and substitution modes.
A recently emerged strategy, or the third, is to take the advantage of the new concept of aggregation-induced emission (AIE) [34]. AIE is a phenomenological description of the unique emission property of organic dyes, which are weakly or nonemissive in dilute solution but highly emissive in aggregate/solid states. Tetraphenylethene (TPE) is a representative AIE-active luminogen or an AIE-gen that has been widely used in the construction of fluorescent materials [35, 36]. Up to now, it has been confirmed that linking one or more TPE moieties to an ACQcharacteristic luminogen may result in an AIE-active one. This strategy is successful in the cases of modifying anthracene, phenanthrene, pyrene, triphenylamine, borondipyrromethene, perylenebisimide, diketopyrrolopyrrole and so on [37-46]. It is a reasonable deduction that the modification of porphyrin with TPE moieties may convert the TPE-porphyrin conjugate from an ACQ to an AIE compound.
The decoration of porphyrin with TPE moieties was firstly reported by Bhosale and colleagues, but the authors had not addressed whether the obtained molecule was an AIE-active one or not [47]. Recently, Guo et al. reported the synthesis and aggregation-induced emission enhancement property of a porphyrin derivative (PorTPE) decorated with four TPE units at meso- positions of porphine core [48]. PorTPE emits red light in dilute THF (tetrahydrofuran) solution and its two Q-bands appear at 601 nm and 660 nm. It is noticed that the emission intensity of PorTPE nanoparticles at 660 nm is 4 times stronger than the counterpart porphyrin without TPE-modification. In addition, the nano-particles demonstrated high contrast fluorescent images when they were used for in vitro imaging HeLa cells.
Here, we report the synthesis and photophysical property of a novel TPE-modified tetraphenylporphyrin (TPP). In the present work, the TPE units are linked to TPP core through ester group. Such a molecular design has at least three advantages. (ⅰ) The esterification reaction catalyzed by N, N-dicyclohexylcarbodi-imide has higher efficiency than the reported Suzuki coupling reaction between tetra-(para-bromophenyl)-porphyrin and boronic acid modified TPE. (ⅱ) The ester linkage breaks the conjugation between the porphyrin core and TPE moieties, thus the resultant shows the expected high emission efficiency both in solution and solid. (ⅲ) The efficient red emission of the derivative is much higher than TPP.
The synthetic route to target compound of 5, 10, 15, 20-tetrakis-(benzene-4, 1-diyl)tetrakis-(4-(1, 2, 2-triphenylvinyl)benzoate)-por-phyrin (4(TPE-COO)-TPP) is shown in Scheme 1. The experimental details are described in the Supporting information. The final product is a purple solid and the total yield is 68.7%. The chemical structure and purity of 4(TPE-COO)-TPP were characterized with multiple spectroscopic methods and the data are summarized in Fig. S1 in Supporting information. But the overall yield of PorTPE reported by Guo et al. is only about 5.6% [48]. In addition to the high yield, 4(TPE-COO)-TPP also has a higher thermal stability than the pristine TPP solid (Fig. S2 in Supporting information).

|
Download:
|
| Scheme 1. Synthetic route to 4(TPE-COO)-TPP. | |
We firstly examined the photophysical properties of the obtained 4(TPE-COO)-TPP. Its absorption and emission spectra in dilute tetrahydrofuran (THF) solution (1 × 10-6 mol/L) and some other solvents were measured and the results are depicted in Fig. 1, Figs. S3 and S4 in Supporting Information. As depicted in Fig. 1A (fw = 0), the B band of TPP appears at 416 nm and the discrete Q band appears at 513, 547, 592 and 650 nm, which are coincident to the results reported by Kim et al. [26]. The absorption features of 4 (TPE-COO)-TPP are nearly identical to TPP. A sharp and strong B band peaked at 418 nm and a group of weak and successive four-peaked band appears at 515, 550, 592 and 650 nm (Fig. 1C, fw = 0). The slight red-shift of absorption spectrum suggests very limited contribution of the TPE units to TPP core. Besides the absorption features for TPP moiety, there is a broad and moderate band ranged from 300 nm to 380 nm and peaked at around 325 nm, which is assigned to the absorption of TPE units.

|
Download:
|
| Fig. 1. Absorption (A) and photoluminescence (PL) (B) spectra of TPP in THF/water mixtures with different water fractions (fw, % by volume). Absorption (C) and PL (D) spectra for 4(TPE-COO)-TPP in THF/water mixtures with different fw values. Concentration of solute: [4(TPE-COO)-TPP] = [TPP] = 1.0 × 10-6 mol/L; Excitation wavelength (λex): 514 nm. | |
In dilute THF solution, the photoluminescence (PL) spectrum of 4(TPE-COO)-TPP (Fig. 1D, fw = 0) is very similar to that of TPP (Fig. 1B, fw = 0), a main emission band centered at 655 nm and a secondary peak at around 715 nm. Such a special emission feature stems from the unique electronic structure of porphyrin, which has two bonding orbitals and two degenerate anti-bonding orbitals. Accordingly, there are two sets of radiative transitions allowing porphyrin and its derivatives to show a primary (e.g., 655 nm) and a secondary emission peak (e.g., 715 nm). All of the above results indicate that the indirect linking of TPE units has little influence on the optical property of the TPP core in dilute solution.
In aggregation state, the absorption and emission behaviors of 4 (TPE-COO)-TPP and TPP demonstrate differences from those observed in dilute solution, as displayed in Fig. 1, which show the UV-vis absorption and PL spectra in THF and water mixture solutions with different water fractions. Since THF and water are good and poor solvents for both TPP and 4(TPE-COO)-TPP respectively, aggregates will form in the mixture of THF and water with proper fw values. The absorption features of TTP in THF/water mixtures are identical to that in pure THF solution when fw is lower than 80%. When fw is equal to or higher than 80%, the sharp B-band becomes blunt and its intensity decreases evidently (open circles and open squares in Fig. 1A). Meanwhile, the absorption maximum red shifts from 416 nm to about 418 nm. These feature changes indicate the existence of interaction between porphyrin chromo-phores, thereby the formation of molecular aggregation can be deduced. The Q-band peaked at around 513, 547, 592 and 650 nm shows a little decrease in absorption intensity and small red-shift of absorption maximum, which are in accordance with the changes of B-band and suggest the aggregate formation.
As a derivative of TPP, the changes in absorption features of 4 (TPE-COO)-TPP in THF/water mixtures demonstrate a similar trend to TPP, except the weak and broad absorption band in the range from 300 nm to 380 nm, which is attributed to TPE moieties. The decrease in absorption intensity, the red-shift of absorption bands (both B-band and Q-band) and the broadening of the sharp B-band with increasing water fraction can be observed (Fig. 1C). However, the details have some differences. For 4(TPE-COO)-TPP, the threshold of the absorption feature changes occurs at the fw value of 50%, while this value is 80% for TPP. We ascribe the threshold difference to the fact that 4(TPE-COO)-TPP has lower water solubility than TPP due to the four peripheral hydrophobic TPE units on TPP core, which leads to the formation of aggregates at a relative lower water fraction.
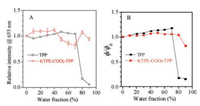
Similar to the emission features observed in the pure THF solution, both 4(TPE-COO)-TPP and TPP in THF/water mixture solutions show two emission bands, a main band peaked at around 655 nm and a pronounced side band peaked at around 715 nm. With the increasing of water fraction from 0 to 50%, the emission intensity of TPP shows a slight enhancement, and then recedes with further increasing of fw. But it undergoes a steep decrease when fw approaches to 80% (comparing the open square and open circle symbols in Fig. 1B) or higher (90%, solid circle, Fig. 1B). This trend is quantitatively shown in Fig. 2 by plotting the emission intensity at 655 nm vs. fw. According to the experimental data displayed in Fig. 1A, the dramatically reduced emission intensity corresponds to the formation of molecular aggregates. Thus TPP is a typical ACQ-molecule.
|
Download:
|
| Fig. 2. (A) Plot of PL intensity of TPP and 4(TPE-COO)-TPP (the error bars are depicted based on 6 experimental results) at 655 nm vs. fw. (B) Variation of Φ/Φ0 of TPP and 4(TPE-COO)-TPP with fw. Φ: quantum yield at a certain water fraction; Φ0: quantum yield in pure THF solution. The Φ values were evaluated assuming a value of 13.3% for TPP in benzene as standard, upon this value, Φ0 of TPP in pure THF solution was estimated to be 10.4%. Concentration of TPP and 4(TPE-COO)-TPP was 10 mmol/L. | |
In comparison with TPP, the emission intensity of 4(TPE-COO)- TPP shows different dependence on fw. As shown by the data in Figs. 1D and 2A, the PL intensity has a small enhancement when fw changes from 0 to 40% and then goes down with increasing fw from 40% to 70%. This turning point is coincident with the decrease in absorbance change of 4(TPE-COO)-TPP (Fig. 1C). Further increasing fw from 70% to 80%, the relative emission intensity at 655 nm is boosted up again. These observations can be explained as following: TPP is a strong electron donor and TPE with a carboxylate group can be viewed as weak electron acceptor relative to TPP, and the TPE moieties can twist around the TPP core. With increasing the solvent polarity, the twisted intermolecular charge transfer (TICT) takes place and the PL is partially quenched. As a proof, the robust absorption B-band of 4(TPE-COO)-TPP in dilute N, N-dimethyl-formamide solution shows a red-shift in comparison with that in dioxane solution (Fig. S3). When fw is further increased, the 4(TPE-COO)-TPP molecules form aggregates and the TICT process is restricted thus the emission intensity exhibits a certain enhancement. The emission intensity of 4 (TPE-COO)-TPP in THF/water mixture with fw of 80% retains 90% of that in pure THF solution. Based on the experimental data shown in Fig. 1C, molecular aggregates have formed when fw is higher than 40% (see open circle and square symbols). These results indicate that the attachment of TPE-moieties onto TPP core via ester groups has evidently changed the ACQ behaviour of TPP.
To eliminate the variances in PL measurement, the emission quantum efficiency (Φ) has been measured for both TPP and 4 (TPE-COO)-TPP in THF/water mixtures and the results obtained in a typical experiment. For TPP, the quantum efficiency in pure THF solution (Φ0) is 10.4%. The Φ value increases slowly with increasing fw from 0 to 70% and the maximum of 12.2% has been recorded at fw of 70%. Then Φ drops drastically down to about 1.4% when fw is over 80%. At fw of 90%, Φ reduces to 1.1%, which is only about 10% of Φ0, which further confirmed the ACQ characteristic of TPP. For 4(TPE-COO)-TPP, Φ0 is about 14.4% in dilute THF solution. Φ undergoes slight increase with increasing fw from 0 to 60% and a maximum value of about 15% has been recorded at fw of 60%. Further increasing fw to 70%, the emission efficiency decreases slowly. When fw is 90%, Φ changes to 12%, which is 85% of its original value (Φ0). This is evidently distinct from the behaviour of TPP, which can only retain 10% of its emission efficiency in pure THF solution (Fig. 2B).
Due to the highly hydrophobic property, 4(TPE-COO)-TPP molecules precipitate quickly in a THF/water mixture with high water fraction. As a consequent, we cannot check the Φ value when fw is higher than 90%. Alternatively, we directly measured the emission efficiency of solid samples of TPP and 4(TPE-COO)-TPP using integrating sphere technique. The Φ value of 4(TPE-COO)-TPP solid powder is detected to be 7.5% (data on Fig. 3A), while the emission from TPP crystal is too weak to be detected (< 0.1%, the limit of the detection instrument). No single crystal of 4 (TPE-COO)-TPP was obtained after trying for months. Consequently, we only compared the Φ values for the amorphous sample of TPP with 4(TPE-COO)-TPP. The measured data was only 2.1%, evidently lower than that of 4(TPE-COO)-TPP. These results confirm the successful modification of the ACQ property of TPP by linking bulky TPE units to TPP core.

|
Download:
|
| Fig. 3. Photographs for solid powder of 4(TPE-COO)-TPP taken under UV-light (A, λex: 365 nm) and daylight (C). Photographs for solid powder sample of TPP taken under UV-light (B, λex: 365 nm) and day-light (D), respectively. | |
The photographs of 4(TPE-COO)-TPP and TPP powder samples taken under UV-light and day-light are also displayed in Fig. 3. Under UV-light, intense red fluorescence (Fig. 3A) is emitted from the purple powder of 4(TPE-COO)-TPP (Fig. 3C). But no recordable fluorescence (Fig. 3B) has been detected from the dark-blue purple powder of TPP (Fig. 3D) under UV-light. In comparison with PorTPE [48], 4(TPE-COO)-TPP has not shown typical AIE activity. Checking the emission spectrum of 4(TPE-COO)-TPP solid powder displayed in Fig. S4A, it is found that 4(TPE-COO)-TPP shows moderate PL efficiency and similar emission features in dilute solution, aggregates and solid powder. We tentatively associated such emission behaviour of 4(TPE-COO)-TPP with the effects of TPE moieties on TPP chromophore.
On one hand, in dilute solution, the rotational and vibrational movements of the phenyl groups in TPE moieties cannot exhaust the excitation energy and have no effects on emission quenching of the porphyrin luminogen because the TPE units are indirectly linked to the porphyrin core. As a result, the Φ of 4(TPE-COO)-TPP is 14.4%, a little higher than that of TPP (10.4%). And on the other hand, in aggregate and/or solid states, the propeller shape and bulky size of the TPE moieties obstacle the close packing of adjacent porphyrin cores and thereby reduce the fluorescence quenching caused by intermolecular π-π interaction. Meanwhile, the rotational and vibrational movements are restricted in aggregate and/or solid states. Thus, 4(TPE-COO)-TPP exhibits good emission efficiency in aggregate (Φ = 12%, fw = 90%) and in solid (Φ = 7.5%, amorphous powder).
The interchromophore interaction can be reflected on the absorption features. As shown in Fig. S4B, the absorption maximum of the B-band for TPP solid film appears at around 430 nm, which is 14 nm red-shift compared with TPP in THF solution. The evident red-shift indicates the existence of strong inter-chromophore interaction. For 4(TPE-COO)-TPP solid film, the peak of B-band appears at 420 nm, which is 2 nm red-shift relative to that recorded in THF solution (Fig. 1C). So small spectral shift indicates weak intermolecular interaction.
In principle, for the large p-conjugated molecules, strong interactions between chromophores, on one hand, are beneficial to form excimers or exciplexes, and on the other hand, they can provide more channels than the cases that only have weak or no interchromophore interactions for an excited molecule to decay to its ground state. As a result, in the former case, the life-time will become longer as compared the aggregates with the dilute solution. While in the latter case, the life time will be shortened due to the competition between the non-radiative with the irradiative decays. Thus the intermolecular interaction can be monitored by transient fluorescence spectroscopy. Based on this consideration, we measured the transient fluorescence spectra of TPP and 4(TPE-COO)-TPP in pure THF solvent and their aggregates formed in THF/water mixture solution at fw of 90%. The transient fluorescence spectra cannot be comparatively investigated on TPP and 4TPE-TPP solids because the emission of TPP solid is too weak to record analytical emission signals. The experimental data are displayed in Fig. 4.

|
Download:
|
| Fig. 4. Time-resolved fluorescence decay traces of TPP (A) and 4(TPE-COO)-TPP (B) in pure THF solution and in THF/water mixture at an fw of 90%. [4(TPE-COO)-TPP]: 10-6 mol/L; λex: 416 nm; pulse width: 58.4 ps. | |
In dilute THF solution, the life-time of TPP's emission is 10.22 ns and this parameter reduces to 5.39 ns in aggregation state. The 47% life-time reduction indicates the existence of substantial interchromophore interactions. According to the feature changes observed in the absorption and emission spectra of TPP, there are no obvious evidence indicate the formation of excimer or exciplexes. Therefore, the evidently shortened life-time can be associated with the presence of non-radiative decay channels in the aggregates of TPP molecules. For 4(TPE-COO)-TPP, the life time in THF solution is 9.61 ns and it becomes 8.42 ns in aggregation state. The life-time reduction is only 12%, indicating the presence of relative weak interchromophore interactions. The measurement data are reasonable because the modification of TPP with four bulky TPE units prevents the 4(TPE-COO)-TPP molecules from close packing in aggregation states. These results are also in good agreement with the spectral feature changes discussed previously (Figs. 2 and 3).
Weak interaction between porphyrin cores (π-π interaction) and bulky size effect of the TPE moiety will result in loose packing of 4(TPE-COO)-TPP molecules. Therefore, it is expected that 4 (TPE-COO)-TPP molecules tends to form amorphous solid. On the contrary, in TPP solid, well-ordered molecular packing is expected. To confirm the inferences above, X-ray diffraction (XRD) measurements were carried out to the solid powders of the as-prepared TPP and 4(TPE-COO)-TPP samples, and the data are displayed in Fig. 5. There exist a series of sharp and strong reflection peaks in the diffraction pattern of TPP powder sample, indicating that the sample is a highly crystalline state. Distinct from TPP, there is no strong diffraction peaks can be observed for 4(TPE-COO)-TPP powder sample and spin-coating film (Figs. 5b and c). The profile appearing at around 20° suggests the amorphous solid, and the small and blunt peak appearing at around 27.5° indicates the existence of ordered molecular packing in a certain short range, or the existence of micro-crystalline in the solid sample. The XRD-pattern for solid powder collected from THF/water mixture with 70% water fraction also suggests the mainly existence of amorphous state and co-existence of small fraction of micro-crystalline (Fig. 5d). When the non-solvent/solvent mixture system was changed to ethanol/chloroform, for an instance, the solid powder collected from ethanol/chloroform in a 1:1 volume ratio contained mainly microcrystalline (Fig. 5e). In this system, the aggregate formation process was slow and there was a long time for the molecules to form regular packing. The above results and analyses validate our above inferences.

|
Download:
|
| Fig. 5. X-ray diffraction patterns of the solid samples of as prepared TPP powder sample (a), and spin-coating film (b), as prepared solid powder (c), aggregates formed in THF/water mixture with fw = 70% (d) as well as the aggregates formed in 1:1 ethanol/chloroform mixture (e) for 4(TPE-COO)-TPP, respectively. | |
The amorphous state of 4(TPE-COO)-TPP was further confirmed by the scanning electronic microscope (SEM) images shown in Fig. S5A in Supporting information. The morphology of the asprepared powders of 4(TPE-COO)-TPP is mostly composed of fine powders mixing with some lamella-shaped objects. Carefully treating the aggregates formed in THF/water mixture with fw of 90% and SEM observations were carried out. The morphology of the aggregates is characterized by feeble sheets in size of several tens micrometers in length and several nanometers in thickness (Fig. S5B in Supporting information). These microstructures cannot afford enough information about 3-dimentional periodic structure. As a result, serial sharp diffraction peaks have not been recorded on the XRD pattern, and the blunt peak at 27.5° may be associated with the diffraction in direction of the sheet growth. In contrast to 4(TPE-COO)-TPP, the SEM image of the as-prepared TPP powder sample displays typical large sized and regularly-shaped blocks (Fig. S5C in Supporting information). The morphology of the aggregates formed in THF/water mixture with fw of 90% presents a profile of micro-prisms (Fig. S5D in Supporting information). The degree of order of the microstructures observed in Fig. S5D is higher than that in Fig. S5B. These morphological observations are in good consistence with the respective XRD patterns and spectral features.
In summary, the experimental results indicate that the fluorescence efficiency (Φ) of TPP can be greatly enhanced by modification the TPP core with the representative AIE-gen of TPE. It is noted that the modification reaction takes place in mild condition and the overall yield is as high as 68.7%. The obtained 4(TPE-COO)-TPP emits red fluorescence with a maximum at 655 nm and an absolute Φ of 7.5% in amorphous solid. Since TPP is a typical ACQ luminogen with very low emission efficiency in crystal solid, this modification is very significant. Distinct from most of the TPE-modified ACQ luminogens, 4(TPE-COO)-TPP demonstrates good red-emission in solution, aggregates and solids. Such a balanced emission in different states may help porphyrin derivatives to find versatile applications in different situations. Considering that porphyrin is a family of luminescent materials that have drawn great research efforts, the information disclosed in the present work is useful for the development of novel and promising porphyrin-based opto/electronic materials.
AcknowledgmentsThis work was financially supported by the Key Project of the Ministry of Science and Technology of China (No. 2013CB834704), the National Natural Science Foundation of China (No. 51573158), the Research Grants Council of Hong Kong (Nos. 16301614, N_HKUST604/14 and N_HKUST-620/11). Prof. Qin and Tang thank the support of Guangdong Innovative Research Team Program (No. 201101C0105067115).
Appendix A. Supplementary dataSupplementary data associated with this article can be found, in the online version, at https://doi.org/10.1016/j.cclet.2018.10.020.
| [1] |
L.E. Webb, E.B. Fleischer, J. Am. Chem. Soc. 87 (1965) 667-669. DOI:10.1021/ja01081a058 |
| [2] |
T.D. Lash, J. Porphyrins Phthalocyanines 15 (2011) 1094-1115. |
| [3] |
L. Chen, L. Yang, D. Jiang, J. Am. Chem. Soc. 132 (2010) 9138-9143. DOI:10.1021/ja1028556 |
| [4] |
T. Carofiglio, E. Lubian, I. Menegazzo, et al., J. Org. Chem. 74 (2009) 9034-9043. DOI:10.1021/jo901749w |
| [5] |
E. Chekmeneva, C.A. Hunter, M.J. Packer, et al., J. Am. Chem. Soc. 130 (2008) 17718-17725. DOI:10.1021/ja803434z |
| [6] |
Y.B. Ding, W.H. Zhu, Y. Xie, Chem. Rev. 117 (2017) 2203-2256. |
| [7] |
N.V.S. Dinesh, K. Bhupathiraju, W. Rizvi, et al., Org. Biomo. Chem 14 (2016) 389-408. |
| [8] |
T. Bessho, S.M. Zakeeruddin, C.Y. Yeh, et al., Angew. Chem. Int. Ed. 49 (2010) 6646-6649. DOI:10.1002/anie.201002118 |
| [9] |
S.Y. Ku, C.D. Liman, J.E. Cochran, et al., Adv. Mater. 23 (2011) 2289-2293. DOI:10.1002/adma.201100028 |
| [10] |
S. Li, K. Chang, K. Sun, et al., ACS Appl. Mater. Interfaces 8 (2016) 3624-3634. DOI:10.1021/acsami.5b07995 |
| [11] |
L. Lu, C. He, W. Lin, J. Am. Chem. Soc. 137 (2015) 7600-7603. DOI:10.1021/jacs.5b04069 |
| [12] |
P. Rothempund, J. Am. Chem. Soc. 57 (1935) 2009-2011. |
| [13] |
R. Yerushalmi, R. Scherz, M.E. van der Boom, J. Am. Chem. Soc. 126 (2004) 2700-2701. DOI:10.1021/ja038918o |
| [14] |
G. de Luca, G. Pollicino, A. Romeo, et al., Chem. Mater. 18 (2006) 5429-5436. DOI:10.1021/cm060896s |
| [15] |
H.F. Zhang, Y.J. Han, Y.J. Guo, et al., J. Mater. Chem. 22 (2012) 23900-23905. DOI:10.1039/c2jm35379b |
| [16] |
J. Yang, Z. Wang, K.L. Hu, et al., ACS Appl. Mater. Interfaces 7 (2015) 11956-11964. DOI:10.1021/acsami.5b01946 |
| [17] |
P. Muthukumar, J.S. Abraham, Sensor. Actuat. B-Chem. 159 (2011) 238-244. |
| [18] |
H. Zhou, L. Baldini, J. Hong, et al., J. Am. Chem. Soc. 128 (2006) 2421-2425. DOI:10.1021/ja056833c |
| [19] |
J.P. Celli, B.Q. Spring, I. Rizvi, et al., Chem. Rev. 110 (2010) 2795-2838. DOI:10.1021/cr900300p |
| [20] |
T. Förster, K. Kasper, Z. Phys. Chem. (Muenchen, Ger.) 1 (1954) 275-277. DOI:10.1524/zpch.1954.1.5_6.275 |
| [21] |
S. Li, K. Chang, K. Sun, et al., ACS Appl. Mater. Interfaces 8 (2016) 3624-3634. DOI:10.1021/acsami.5b07995 |
| [22] |
X. Shen, F. He, J. Wu, et al., Langmuir 27 (2011) 1739-1744. DOI:10.1021/la104722q |
| [23] |
J.L. Grimland, C. Wu, R.R. Ramoutar, et al., Nanoscale 3 (2011) 1451-1455. DOI:10.1039/c0nr00834f |
| [24] |
A.V. Kondrashina, R.I. Dmitriev, S.M. Borisov, et al., Adv. Funct. Mater. 22 (2012) 4931-4939. DOI:10.1002/adfm.v22.23 |
| [25] |
W.D. Jang, N. Nishiyama, G.D. Zhang, et al., Angew. Chem. Int. Ed. 44 (2005) 419-423. DOI:10.1002/(ISSN)1521-3773 |
| [26] |
Y. Lee, Y.J. Kim, S. Kim, et al., J. Mater. Chem. 19 (2009) 5643-5647. DOI:10.1039/b906587n |
| [27] |
Z. Fang, K.Y. Pu, et al., Macromolecules 41 (2008) 8380-8387. DOI:10.1021/ma801874z |
| [28] |
F.J. Lovell, R. Aron, K. Ng Kenneth, et al., Biomacromolecules 12 (2011) 3115-3118. DOI:10.1021/bm200784s |
| [29] |
K. Nakata, T. Kobayashibcde, E. Tokunaga, Phys. Chem. Chem. Phys. 13 (2011) 17756-17767. DOI:10.1039/c1cp21964b |
| [30] |
S. Biswas, H.Y. Ahn, M.V. Bondar, et al., Langmuir 28 (2012) 1515-1522. DOI:10.1021/la203883k |
| [31] |
N. Venkatramaiah, B. Ramakrishna, R. Venkatesan, et al., New J. Chem. 37 (2013) 3745-3754. DOI:10.1039/c3nj00482a |
| [32] |
M. Pérez-Morales, G. Miguel, H.J. Bolink, et al., J. Mater. Chem. 19 (2009) 4255-4260. DOI:10.1039/b902066g |
| [33] |
L.M. Scolaro, A. Romeo, M.A. Castricianoa, et al., Chem. Commun. (Camb.) (2005) 3018-3020. |
| [34] |
J. Mei, N.L.C. Leung, R.T.K. Kwok, et al., Chem. Rev. 115 (2015) 11718-11940. DOI:10.1021/acs.chemrev.5b00263 |
| [35] |
B.Z. Tang, X. Zhan, G. Yu, et al., J. Mater. Chem. 11 (2001) 2974-2978. DOI:10.1039/b102221k |
| [36] |
Y. Dong, J.W.Y. Lam, A. Qin, et al., Appl. Phys. Lett. 91 (2007) 11111-9. DOI:10.1063/1.2753723 |
| [37] |
W.Z. Yuan, P. Lu, S. Chen, et al., Adv. Mater. 22 (2010) 2159-2163. DOI:10.1002/adma.v22:19 |
| [38] |
Z.J. Zhao, P. Lu, J.W.Y. Lam, et al., Chem. Sci. 2 (2011) 672-675. DOI:10.1039/C0SC00521E |
| [39] |
Z.J. Zhao, S.M. Chen, J.W.Y. Lam, et al., J. Mater. Chem. 21 (2011) 7210-7216. DOI:10.1039/c0jm04449k |
| [40] |
W.Z. Yuan, Y.Y. Gong, S. Chen, et al., Chem. Mater. 24 (2012) 1518-1528. DOI:10.1021/cm300416y |
| [41] |
Q. Zhao, S. Zhang, Y. Liu, et al., J. Mater. Chem. 22 (2012) 7387-7394. DOI:10.1039/c2jm16613e |
| [42] |
Q. Zhao, X.A. Zhang, W. Qiang, et al., Chem. Commun. (Camb.) 48 (2012) 11671-11673. DOI:10.1039/c2cc36060h |
| [43] |
Q. Zhao, K. Li, S. Chen, et al., J. Mater. Chem. 22 (2012) 15128-15135. DOI:10.1039/c2jm31368e |
| [44] |
R. Hu, C.F.A.G. Gméz-Duran, J.W.Y. Lam, et al., Chem. Commun. (Camb.) 48 (2012) 10099-10102. DOI:10.1039/c2cc35188a |
| [45] |
M.H. Chua, Y. Ni, M. Garai, et al., Chem.-Asian J. 10 (2015) 1631-1634. DOI:10.1002/asia.v10.8 |
| [46] |
X.Y. Shen, Y.J. Wang, H. Zhang, et al., Chem. Commun. (Camb.) 50 (2014) 8747-8750. DOI:10.1039/C4CC03024A |
| [47] |
A. Rananaware, R.S. Bhosale, K. Ohkubo, et al., J. Org. Chem. 80 (2015) 3832-3840. DOI:10.1021/jo502760e |
| [48] |
B. Guo, X.L. Cai, S.D. Xu, et al., J. Mater. Chem. B-Mater. Biol. Med. 4 (2016) 4690-4695. DOI:10.1039/C6TB01159D |
 2019, Vol. 30
2019, Vol. 30 

