b Nano Science and Technology Institute, University of Science and Technology of China, Suzhou 21500, China
Biofouling was defined as undesired accumulation of fouling organisms on the surface, and usually occurred on the ships, pipelines, medical devices, which could lead to huge economic losses. When an object was immersed in the sea water, a condition layer would form and cover the surface in the first few minutes [1-3]. During next few hours, marine microorganisms including bacteria and diatoms would settle on the surface as a reversible attachment [4], generating a friendly environment for the larval macroorganisms. As time went on, macroorganisms like barnacles and mussels started to colonize on the surface, and formed an irreversible biofouling community[5]. DLVO theory was applied to describe this process and according to it [5, 6], the interaction between surface and microorganism was the summation of their electrostatic and van der Waals interaction[5]. Initial adhesion of bacteria and diatoms influenced the settlement of macroorganism intensely and was a critical step to trigger marine biofouling. Extracellular polymeric substances (EPS) and cell wall material such as polysaccharides and bacterial nanofibers played significant roles for the development of biofouling[5]. Coarse surface of the substrate was easier attached by marine organisms [7], and bacteria were likely to aggregate on a positive charged surface. In order to restrain the formation of marine biofouling, the mechanism of bacteria initial settlement needed to be figured out.
Many experiments had been designed to investigate the bacteria settlement process on the surface and the influence thereafter [8-11]. Scanning electron microscopy (SEM) had been used to provide information of surface morphology and properties of the EPS [8]. But it could not be used to monitor in situ settlement process in aqueous circumstance. Atomic force microscopy (AFM) had been employed to study the strength of attachment Ulva spore [9-11]. Hydrodynamic methods had also been used to measure the adhesion strength of Ulva spores using a water jet apparatus [9]. However, AFM and hydrodynamic measurements information did not refer to the interfacial adhesion properties of the whole adhesion system. Transmission electron microscopy (TEM) imaging had been used to look at algal spore ultrastructure during both swimming freely and settled stages [12, 13]. Raman spectroscopy had also been used in study of microorganism, such as rapid detection analysis for free-cell, taxonomic comparison, characterization of microbes, differentiate the discrepancy between populations, and analyzing composition difference between cells grown on different cultures [8, 14-17]. Recently, Raman spectroscopy was used to compare cells in biofilms and planktonic cells [8]. Though TEM could really show the specific molecule interaction, it was obscure to reveal further reaction mechanism. Raman spectroscopy could offer the non-destructive analysis and could rapidly confirm cell properties, but it could only provide final state sample information.
Fourier transform infrared spectroscopy (FTIR) was an ideal approach to study the alive bacteria without destroy their cells. However, water could strongly absorb infrared radiation and would highly interfere the collected IR spectra. Therefore, attenuated total reflection infrared (ATR-IR) spectroscopy was introduced in this study because there was limited penetration of IR radiation into the aqueous medium and it was also a nondestructive detection method which provided an in situ approach to monitor the bacteria settlement process on the surface in aqueous environment [12]. Previously, ATR-FTIR spectroscopy had been used in rapidly detecting Bacillus globigii spores [11], exploring microbes adsorbing metal ions in industrial wastewater [18, 19], microbes adsorbing toxic ions and analyzing phosphate adsorption onto hematite [19]. In this paper, ATR-FTIR spectroscopy was applied to investigate the bio-reaction continuously during gram-positive Bacillus sp. and gram-negative Escherichia coli settlement on bare and Al2O3 coated ZnSe surface, respectively.
The bacteria samples were cultured as following method. Gram-positive Bacillus sp. bacteria (MCCC1A00791) was supplied by Marine Culture Collection of China (MCCC) and cultured in sterile CM 0471-2216E media. The CM 0471-2216E media were prepared by dissolving 5 g peptone, 1 g yeast extract, 1 g beef extract and 0.01 g FePO4 in 1000 mL ASW (artificial sea water). Then 100 mL CM 0471-2216E media were sterilized in autoclave at 120 ℃ for 20 min. A single bacterial colony was picked from nutrition agar plate to 100 mL sterile CM 0471-2216E media. The sterile media with the single bacterial strain were shaken at 30 ℃ at 120 rpm for 24 h. To collect clean bacteria, Bacillus sp. suspension was rinsed with sterile deionized water for 3 times through centrifugation with a rotational speed of 3000 rpm for 5 min then re-suspended in sterile deionized water. Gram-negative Escherichia coli bacteria (ATCC25922) were chosen to compare adsorption of bacteria with different cell wall properties. The nutrient medium was prepared by dissolving 10 g peptone, 10 g NaCl and 5 g yeast extract in 1000 mL deionized water. The same way to sterilize and transfer bacteria to 100 mL sterile nutrient medium. The sterile media with Escherichia coli were shaken at 37 ℃ at 120 rpm for 24 h. Escherichia coli suspension was rinsed by sterile deionized water to collect clean bacteria. The morphology of both bacteria was imaged by a metalloscope (Leica DM2500M) with enlargement factor of 500 after they well settled on the surface.
ATR-FTIR had been selected to investigate the bacteria. The ATRFTIR spectra were collected using a standard ATR liquid trough plate (PN 022-19XX) arrangement from Pike equipped with ZnSe IRE (80 × 10 × 4 mm3, 45°, 17 reflections). ZnSe was selected for this experiment because of its relatively high refractive index (~2.4) and low solubility product constant in water (Ksp = 3.6 ×10-26). The refractive index of bacteria samples was usually less than ZnSe crystal, and the angle of incidence was 45° which was greater than the critical angle, therefore, the total reflection of infrared radiation occurred in the ZnSe IRE, and the interface reaction information was carried out by the evanescent wave (Fig. 1). Bare and Al2O3 coated ZnSe were conducted to get kinetic ATR-FTIR spectra of Bacillus sp. and Escherichia coli attached on the surface. ZnSe surface almost carried no electric charges and alumina surface carries positive charges in water at pH 5–7, so we could discover mechanism of bacteria adhere on surfaces with different property. 2 mL bacteria suspension with a concentration of 5 ×108 cells/mL was dripped to the trough plate with bared ZnSe using a pipette. The concentration of bacteria was determined at the wavelength of 600 nm by a microplate reader SpectraMax190 (Molecular Devices, U.S.A.). After that, the same procedure was repeated on a surface with 100 nm thickness alumina film which was made by deposition of alumina suspension. The alumina suspension was prepared by adding 20 mg alumina to 30 mL ethanol and ultrasonicated for 30 min. A 200 mL aliquot of the suspension was deposited evenly on one side of the ZnSe crystal using a pipette and the solvent was then allowed to evaporate. After all solvent evaporated, coated IRE was flushed by sterile deionized water to remove loose alumina. Background spectrum was collected at the time zero after bacteria suspension was dripped to trough plate. The spectra of bacteria were collected kinetically with an interval of 15 min. All spectra were collected at 4 cm-1 resolution with 100 co-adding scans.

|
Download:
|
| Fig. 1. Schematic of internal reflection of IR radiation in an internal reflection element (IRE). dp represents the penetration depth. | |
Bacillus sp. bacteria suspension 50 mL was dripped onto the bare ZnSe IRE followed by flowing dry air at 30 ℃ until water completely evaporated. Then, the transmittance IR spectrum of dried bacteria on ZnSe was collected using the bare ZnSe IRE as a reference. 20 mL high concentration Bacillus sp. suspension was dripped on a 13 mm diameter ZnSe window, another ZnSe window was using to cover on, then ZnSe window was put on the FTIR holder to collect spectra. The transmittance IR spectrum of the wet bacteria was recorded to get the live bacteria in aqueous environment, and an open beam spectrum was used as a background. All spectra were collected at 4 cm-1 resolution with 100 co-adding scans without baseline correction.
Peptone, beef extract and ethanol were obtained from Sinopharm Chemical Reagent limited corporation. Yeast extract and alumina powder (30 nm) were supplied by Aladdin. Artificial sea water was prepared according to ASTMD 1141-98, all involved reagents in ASW were obtained from Aladdin. ZnSe windows were acquired from PIKE Technologies Incorporation. All the solvents and reagents were used as received without any further purification. All spectra were collected on an ABB MB 3000 series Fourier transform infrared (FTIR) spectrometer equipped with a DTGS detector.
The morphology of bacteria was captured by an optical microscope. Fig. 2 demonstrated the status of Bacillus sp. and Escherichia coli after they settled on bare ZnSe surface. Both Bacillus sp. and Escherichia coli were rod-shaped bacteria. Their lengths were both around 2.5–3 mm, and both of them were agglomerate when they adhered on the surface.
|
Download:
|
| Fig. 2. The status of Bacillus sp. and Escherichia coli on the ZnSe internal reflection element. | |
Fig. 3 demonstrated the transmittance IR spectra of both bacteria, and the major peaks of bacteria were assigned and listed in Table 1 [18, 20]. The distinguished peaks were at around 1640 cm-1, 1550 cm-1 and bands between 900 cm-1 and 1150 cm-1. The peaks at 1640 cm-1 and 1550 cm-1 were amide Ⅰ and amide Ⅱ vibrations of proteins, respectively, and the peaks from 900 cm-1 to 1150 cm-1 were complex composite of various overlapping peaks including C-O, C-O-P, and C-O-C stretching vibration, which were from the functional groups of cell polysaccharides and phosphatide. Though it was difficult to make a distinction between them, the intensity change of the peak was still used to monitor the settlement progress of the bacteria. The peak at 1735 cm-1 was assigned to C=O stretching in carboxylic acid group at the C terminal of the protein, carboxylic acid in the cytoplasm, and lipids in the bacteria membrane.
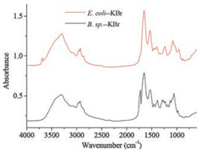
|
Download:
|
| Fig. 3. Transmittance IR spectra of Bacillus sp. KBr pellet and Escherichia coli KBr pellet. | |
|
|
Table 1 Major IR absorption peaks assignments of bacteria. |
FTIR-ATR spectra of both Bacillus sp. and Escherichia coli under different conditions showed distinguished shift of amide bands (Fig. 4). The amide Ⅰ bands of Bacillus sp. in aqueous environment on bare ZnSe surface and Al2O3 coated ZnSe surface appeared at 1633 cm-1 and 1634 cm-1, and the amide Ⅱ bands appeared at 1548 cm-1 and 1551 cm-1, respectively. Compared with dried Bacillus sp. on bare ZnSe, the amide Ⅰ bands red shifted about 7–8 cm-1 to the low wavenumber, but the amide Ⅱ bands blue shifted about 8–11 cm-1 to the high wavenumber. The similar case happened on Escherichia coli. Because there were few proteins in the bacteria cell walls and cell membranes, the amide bands shift indicated that the water passed through the cell walls and cell membranes of the bacteria and led to the secondary structure of proteins changed by forming new hydrogen bonds [21]. The secondary structure change of the protein was reversible, because the amide bands went back to their original position when the water evaporated. The band at 1735 cm-1 in the dry status also blue shifted 8 cm-1 when Bacillus sp. were in aqueous environment on both bare and Al2O3 coated ZnSe surface, because of ionization of the carboxylic acid group at the C terminal of the protein. However, no carbonyl IR bands were observed in the spectra of Escherichia coli. under both ATR mode and transmittance mode. The "disappeared" carbonyl band could not be simply attributed the less amount of the protein in the cell wall to the bacteria, because the transmittance mode of IR would give the complete information of the bacteria.
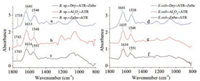
|
Download:
|
| Fig. 4. ATR-FTIR spectra of bacteria on surfaces: (a) Dried Bacillus sp. on bare ZnSe, (b) Bacillus sp. in aqueous environment on Al2O3 coated ZnSe surface, (c) Bacillus sp. in aqueous environment on bare ZnSe, (d) Dried Escherichia coli on bare ZnSe, (e) Escherichia coli in aqueous environment on Al2O3 coated ZnSe surface, (f) Escherichia coli in aqueous environment on bare ZnSe. | |
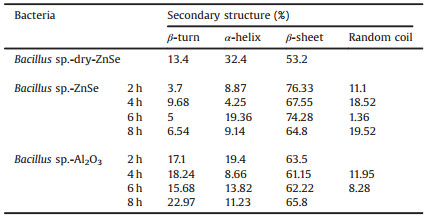
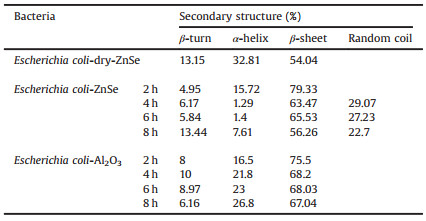
The second derivative of IR spectra was used to distinguish the secondary structure of protein under different conditions (Fig. 5) [21], and the calculated results showed in Tables 2 and 3. In order to eliminate water bands affect, each spectrum above subtract a certain amount of water spectrum until the bands 1750–2000 cm-1 become a straight line before all spectra were conducted to the second derivative of IR spectra [22]. A straight base line in the bands from 1750 cm-1 to 2000 cm-1 is a criterion to make sure that water affection was removed [22]. The calculation demonstrated that the protein of both Bacillus sp. and Escherichia coli had similar ratio of their secondary structure when they were on bare ZnSe surface in dry. However, when the bacteria settled on bare ZnSe surface in aqueous environment, α-helix and β-turn's ratio dropped rapidly and β-sheet increased, random coil could be distinguished. When the bacteria settled on Al2O3 coated ZnSe surface, β-turn's ratio of Bacillus sp. increased, but its ratio of Escherichia coli decreased. And their α-helix and β-sheet showed similar trend as on bare ZnSe. It was noticed that the secondary structure's ratio change was relevant to the amide bands shift in their IR spectra. Fig. 6 demonstrated the wavenumber change of amide Ⅰ bands when both bacteria settled progress on bare ZnSe and Al2O3 coated ZnSe surface. The trend of the change of the secondary structure was coincident with the shift of the amide bands. Therefore, the shift of the amide bands was caused by the proteins' secondary structure change, but what caused the proteins' secondary structure change, the hydrogen bond or the interaction with the surface? The IR spectra of Bacillus sp. in transmittance mode were used to investigate the main reason to cause the amide bands shift. In Fig. 7, the spectra clearly showed the amide bands shift when the bacteria were in water. Therefore, the band shift could be attributed to the new hydrogen bonds between the protein and water. However, the amide Ⅰ and Ⅱ bands of dry Bacillus sp. were located at 1647 cm-1 and 1535 cm-1 in the transmittance mode, and they were located at 1641 cm-1 and 1540 cm-1 in ATR mode. The amide bands of the bacteria in aqueous environment were located at 1642 cm-1 and 1546 cm-1 in transmittance mode. As shown in Fig. 4, the amide Ⅰ and Ⅱ bands position in ATR mode were at 1634 cm-1 and 1551 cm-1 on bare ZnSe, but they were at 1633 cm-1 and 1548 cm-1 on Al2O3 coated ZnSe surface, respectively. It implied that not only the hydrogen bonds interacted with the protein of the bacteria, but also the surface. Another evidence was the different positions of carbonyl bands in Figs. 4 and 7. In transmittance mode, the carbonyl bands appeared at 1735 cm-1, no matter the bacteria were in dry or wet status, but in ATR mode, the carbonyl bands appeared at 1743 cm-1 when the bacteria settled on the surface. The band shift of carbonyl bands was triggered by the interaction between the bacteria and the surface.

|
Download:
|
| Fig. 5. Secondary derivative IR spectra of Bacillus sp. and Escherichia coli under different conditions. | |

|
Download:
|
| Fig. 6. Amide Ⅰ bands shift progress of Bacillus sp. and Escherichia coli on bare ZnSe and Al2O3 coated ZnSe surfaces. | |

|
Download:
|
| Fig. 7. IR spectra of dry and wet Bacillus sp. | |
|
|
Table 2 Calculated percentage of protein secondary structure of Bacillus sp. |
|
|
Table 3 Calculated percentage of protein secondary structure of Escherichia coli. |
In the settlement progress on the surfaces, except gravity and buoyancy on the bacteria, the main forces would be the interactions between the bacteria and surfaces. There were few functional groups on ZnSe surface, but there were a lot of hydroxyl groups on Al2O3 surface. Hence, because the isoelectric point of Al2O3 was about pH 9, when the Al2O3 exposed to the suspension of the bacteria, the Al2O3 coated ZnSe surface was expected to carry positive charges which were opposite to the charges on the bacteria. In a word, the interaction between the bacteria and Al2O3 was greater than it between the bacteria and ZnSe. The broad bands abound 1085 cm-1, which were assigned to the stretching vibrations of C-O, C-O-P, C-O-C and P-O in bacteria cell walls, were used to monitor the relative amount of the bacteria on the surfaces. Fig. 8 showed the kinetic progress of the both bacteria settling on the surfaces. Bacillus sp. appeared on the both surfaces rapidly and the amount of the bacteria saturated under ATR mode in about 2 h. The maximum amount of Bacillus sp. on Al2O3 coated ZnSe surface was about 20% higher than on ZnSe surface. Escherichia coli showed the similar trend as Bacillus sp. on the both surfaces, but the maximum amount of the bacteria on Al2O3 coated ZnSe surface was about 3 times higher than on ZnSe surface. Because Al2O3 carried opposite charges to the bacteria, it was understandable that the bacteria preferred to settle on Al2O3 surface. However, the maximum amount of the bacteria on the surfaces suggested that the cell wall property of Escherichia coli was different from that of Bacillus sp. The externus structure of Bacillus sp. contained about 90% peptidoglycan and 10% teichoic acid, and Escherichia coli had peptidoglycan, lipopolysaccharide, phospholipids with saturated fatty acid and some enzymes [23, 24]. Because of the thicker peptidoglycan in Bacillus sp. cell wall, the settlement behavior on Al2O3 coated ZnSe surface was similar with on bare ZnSe surface. Escherichia coli had very thin peptidoglycan layer, so the other active components in the cell wall played significant roles and were inclined to bind with Al2O3.

|
Download:
|
| Fig. 8. Kinetic progress of Bacillus sp. and Escherichia coli settling on bare ZnSe and Al2O3 coated ZnSe surfaces. | |
In conclusion, ATR-FTIR was used to study the settlement process of gram-positive bacteria Bacillus sp. and gram-negative bacteria Escherichia coli on bare ZnSe surface and Al2O3 coated ZnSe surface. When the both bacteria were in aqueous environment, their amide Ⅰ and amide Ⅱ bands in the ATR-FTIR spectra were red shifted and blue shifted comparing with the dry bacteria, respectively. The hydrogen bonds between the water and the protein in the bacteria led to the protein secondary structure change, which cause the amide bands shift. The interaction between the surface and the bacteria also played significant role in the band shift. Due to the different components of the cell wall of Bacillus sp. and Escherichia coli, Escherichia coli was inclined to settle on Al2O3 coated ZnSe surface, but Bacillus sp. did not show the clear trend to settle on both surface. And the research indicated that ATR-FTIR could be an appropriate approach in protein secondary structure study and it could be also used to monitor the protein interaction with other chemicals.
AcknowledgmentThis research was supported by the National Natural Science Foundation of China (No. 21675165).
| [1] |
M.E. Callow, J.A. Callow, J.D. Pickett-Heaps, R. Wetherbee, Phycologia 36 (1997) 15.
|
| [2] |
S.M. Bennett, J.A. Finlay, N. Gunari, et al., Biofouling 26 (2010) 235-246. |
| [3] |
M.P. Schultz, J.A. Bendick, E.R. Holm, W.M. Hertel, Biofouling 27 (2011) 87-98. DOI:10.1080/08927014.2010.542809 |
| [4] |
Y.H. An, R.J. Friedman, J. Biomed. Mater. Res. 43 (1998) 338-348. DOI:10.1002/(ISSN)1097-4636 |
| [5] |
K. Hori, S. Matsumoto, Biochem. Eng. J. 48 (2010) 424-434. DOI:10.1016/j.bej.2009.11.014 |
| [6] |
K.C. Marshall, R. Stout, R. Mitchell, J. Gen. Microbiol. 68 (1971) 337-348. DOI:10.1099/00221287-68-3-337 |
| [7] |
S.R. Suwarno, S. Hanada, T.H. Chong, et al., Desalination 387 (2016) 1-13. DOI:10.1016/j.desal.2016.02.029 |
| [8] |
V.A. Henry, J.L. Jessop, T.L. Peeples, Anal. Bioanal. Chem. 409 (2017) 1441-1449. DOI:10.1007/s00216-016-0077-9 |
| [9] |
J.A. Finlay, M.E. Callow, M.P. Schultz, G.W. Swain, J.A. Callow, Biofouling 18 (2002) 251-256. DOI:10.1080/08927010290029010 |
| [10] |
J.A. Callow, S.A. Crawford, M.J. Higgins, P. Mulvaney, R. Wetherbee, Planta 211 (2000) 641-647. DOI:10.1007/s004250000337 |
| [11] |
H. Li, C.P. Tripp, Appl. Spectrosc. 62 (2008) 963-967. DOI:10.1366/000370208785793317 |
| [12] |
L. Petrone, R. Easingwood, M.F. Barker, A.J. McQuillan, J. R. Soc. Interface 8 (2011) 410-422. DOI:10.1098/rsif.2010.0316 |
| [13] |
E.C. Henry, K.M. Cole, J. Phycol. 18 (1982) 550-569. DOI:10.1111/jpy.1982.18.issue-4 |
| [14] |
R. Patzold, M. Keuntje, A. Anders-von Ahlften, Anal. Bioanal. Chem. 386 (2006) 286-292. DOI:10.1007/s00216-006-0663-3 |
| [15] |
L.P. Choo-Smith, K. Maquelin, T. van Vreeswijk, et al., Appl. Environ. Microb. 67 (2001) 1461-1469. DOI:10.1128/AEM.67.4.1461-1469.2001 |
| [16] |
M. Wagner, N.P. Ivleva, C. Haisch, R. Niessner, H. Horn, Water Res. 43 (2009) 63-76. DOI:10.1016/j.watres.2008.10.034 |
| [17] |
W.E. Huang, R.I. Griffiths, I.P. Thompson, M.J. Bailey, A.S. Whiteley, Anal. Chem. 76 (2004) 4452-4458. DOI:10.1021/ac049753k |
| [18] |
S.J. Parikh, J. Chorover, Langmuir 22 (2006) 8492-8500. DOI:10.1021/la061359p |
| [19] |
E.J. Elzinga, J.H. Huang, J. Chorover, R. Kretzschmar, Environ. Sci. Technol. 46 (2012) 12848-12855. DOI:10.1021/es303318y |
| [20] |
W. Jiang, A. Saxena, B. Song, et al., Langmuir 20 (2004) 11433-11442. DOI:10.1021/la049043+ |
| [21] |
J. Kong, S. Yu, Acta Biochem. Biophys. Sin. 39 (2007) 549-559. DOI:10.1111/abbs.2007.39.issue-8 |
| [22] |
A.C. Dong, P. Huang, W.S. Caughey, Biochemistry 29 (1990) 3303-3308. DOI:10.1021/bi00465a022 |
| [23] |
T. Kaneda, Microbiol. Rev. 55 (1991) 288-302. |
| [24] |
P. Demchick, A.L. Koch, J. Bacteriol. 178 (1996) 768-773. DOI:10.1128/jb.178.3.768-773.1996 |
 2019, Vol. 30
2019, Vol. 30