b School of Materials Science and Engineering, TKL of Metal-and Molecule-Based Material Chemistry, Nankai University, Tianjin 300350, China
As an essential trace element in plants and animals, iron plays vital role during some biochemical processes at cell level such as oxygen transport and DNA copy [1-4]. As a result, iron deficiency may cause some diseases commonly during infancy, pregnancy and adolescence [5]. On the other hand, the excessive intake of iron from water as well as food may cause substantial harm to human tissue. Meanwhile, the high content of iron in water has negative effects to the production for plastic, textile, papermaking and brewing industry, which request a convenient, rapid and effective technique to detect the Fe content in water.
Luminescent spectral technique has been widely used in detecting molecules and metal ions in aqueous system [6-8], and the detection effective depends greatly on the intrinsic properties of luminophore. Of all luminescent materials, luminescent coordination polymers (LCPs) have gathered significant attention because of their advantages such as real-time monitoring, quick response, high sensitivity [9-13]. However many reported luminescent coordinatin polymers are of poor water stability and thus cannot be used directly in aqueous system.
Lanthanide ions exhibit strong fluorescent emission originating from the transiton of 4f electron [14, 15], and the larger stokes shift gives a visible light emssion, for example, thered and green emissions of Eu3+ and Tb3+, respectively. Meanwhile, the lanthanide-based coordinatin polymers (Ln-CPs) often exhibit enhanced latnanidecentered luminescent emission through the erengy transfer from organic ligands to lanthanide centers [16-18].Furthermoe, Ln-CPsare generally water stable and thus can be used in aqueous solution [19-21]. Recently, some Ln-CPs with free N-based coordination sites were reported, which may interact further with particular guestmetalions and improve response sensitivity [22-25].
In the past, we have focused on the detection of metal ions based on MOFs sensing [26], which however were normally carried out in non-aqueous system due to relatively poor water stability of MOFs used. Meanwhile, the organic ligands used in many previous work usually contain only carboxylic functional group, and should be replaced by bifunctional ligands involving additional nitrogen coordination sites, besides oxygen coordination sites, acting possibly as free sites for interacting with metal ions, which can improve effctively the detection sensitivity.
According to the principle of hardness and softness of acids and bases (HSAB), lanthanide metal ions interact more stronger with oxygen atom than nitrogen atoms [27]. Herein, Tb3+ was chosen as metal source to construct fluorescent coordination polymers of high water stability. As a result, a new luminesencent lanthanide coordination polymer [Tb(L)(HL)(H2O)·2H2O]n (NKU-115) with a two dimensional (2D) network structure was obtained under solvothermal condition based on 5-(4H-1, 2, 4-triazol-4-yl) benzene-1, 3-dicarboxylic acid (H2L [28], see Scheme 1) ligand. The assynthesized NKU-115 feature a strong green emission and exhibits sensitive detection of Fe3+ in aqueous.

|
Download:
|
| Scheme 1. The H2L ligand employed in the synthesis of NKU-115. | |
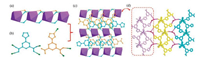
Single-crystal X-ray diffraction shows that NKU-115 crystallizes in the monoclinic crystal system of P21. The crystal data and structure refinement parameters of NKU-115 are listed in Table S1 in Supporting information, and the selected bond lengths and angles collected in Table S2 in Supporting information. The asymmetric unit of NKU-115 contains one Tb3+ ion, one L2-, one HL-, one coordination water and two lattice H2O (Fig. S1 in Supporting information). Tb3+ is surrounded by six carboxylic groups from two different H2L ligands, one O atom from coordinated H2O and one N atom from HL-, to form a dodecahedron (Fig. S2 in Supporting information). The Tb-Ocarboxyl bond lengths fall in the reasonable range of 2.335-2.510 Å [29]. Furthermore, Tb3+ ion was bridged with two adjacent ones by carboxylate group to form a terbium chain with a Tb· · ·Tb distance of 4.697 Å (Fig. 1a).
|
Download:
|
| Fig. 1. The single crystal structure of NKU-115: (a) the Tb3+ chain; (b) the two coordination modes of H2L; (c) the 2D layer over ab plane; and (d) the hydrogen interaction network seen along a axis. | |
On the other hand, two kinds of coordinated models were found for the ligand: one coordinates (L2-) toward Tb3+ only through two carboxylic groups, while the other (HL-) through one carboxylic group and triazole group (Fig. 1b). Bridging Tb chains by L2- and HL- ligand forms a 2D layer (Fig. 1c) that further interactes with adjacent layers through hydrogen-bonding to genearte a 3D network (Fig. 1d).
Thehigh phase purity of NKU-115 was unveiled bygood matching of the experimental powder X-ray diffraction (PXRD) pattern with the simulated one (Fig. S3 in Supporting information). Thermogravimetric analysis (TGA) reveals that NKU-115 was thermally stable up to 350 ℃ (Fig. S4 in Supporting information). Furthermore, the solvent stability was also investigated by immersing the crystal sample into some common solvents for 5 days, including N, N-dimethylformamide (DMF), N, N-dimethylacetamide (DMA), acetonitrile, ethanol and water. The power X-ray diffraction (PXRD) data of the recovered samples from the solution still coincide well with the simulated result (Fig. S5 in Supporting information), indicating the high solvent stability of NKU-115. Notably, the water stability of NKU-115 is of great interests that enable its application in aqueous solution for fluorescence detection.
The luminescence spectrum of solid NKU-115 were first investigated at room temperature. As shown in Fig. S6 (Supporting information), NKU-115 exhibits four characteristic emission peaks at 490, 545, 585 and 622 nm under excitation at 310 nm, all assignable to 5D4 → 7FJ (J = 6, 5, 4, 3) transitions of Tb3+ ion. The strongest green-light emission at 545 nm is attributed to magnetic-dipole induced 5D4 → 7F5 transition [30].
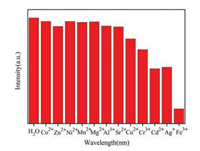
To investigate the influence of different metal ions on the fluorescence emission of NKU-115, 4 mg NKU-115 was dispersed in 3.6 mL aqueous solution to form a suspension solution by ultrasonication method. And then to the suspension system was added 0.4 mL aqueous solution of various metal cations (1 × 10-2 mol/L), including Ag+, Cd2+, Cu2+, Co2+, Cr3+, Zn2+, Ni2+, Mn2+, Mg2+, Al3+, Sr2+, and Fe3+. The variation of the strongest emissions at 545 nm were recorded. As clearly shown in Fig. 2, Fe3+ exhibits significant quenching effect on the emission of NKU-115, while Cr3+, Cd2+ and Ag+ have little effect and the other metal ions have no obvious effect. Notably, the quenching effect of Fe3+ may be clearly identified by naked eyes as shown in Fig. S7 (Supporting information), which facilitate the practical application of this sensing.
|
Download:
|
| Fig. 2. The emission intensities variation of the 5D4-7F5 transition (at 545 nm) for NKU-115 suspension upon the addition of different metal ions (in 10-2 mol/L) under the excitation of 310 nm. | |
The anti-interference experiments were also performed (Fig. S8 in Supporting information), and the results indicated that the presence of other metal ions exerts negligible influence on the effect of Fe3+. In order to assess the sensing sensitivity of NKU-115, the quantitative fluorescence titration was performed (Fig. 3a). Upon increasing the concentrations of Fe3+ from 6.6 × 10-5 mol/L to 2 ×10-3 mol/L, the luminescence intensity of NKU-115 decreased gradually.

|
Download:
|
| Fig. 3. (a) Stern-Volmer simulation of the concentration-dependent luminescence titration of NKU-115 upon increasing addition of Fe3+ (inset: the photo of the blank NKU- 115 suspension (left) and that upon addition of Fe3+ (right)); (b) Stern-Volmer simulation of I0/I of the NKU-115 suspension Fe3+ low concentration region). | |
The quenching effectivity was quantified by simulating the titration data with Stern-Volmer (SV) equation: I0/I = 1 + Ksv[M] [31]. Here, I0 and I are the luminescence intensities of NKU-115 before and after the addition of analyte, respectively; [M] is the concentration of analyte. As shown in Fig. S9 (Supporting information), the value of I0/I increases with adding Fe3+ concentrations. Meanwhile, a good linear correlation (Fig. 3b) was observed at low concentrations of Fe3+ (0-0.2 mmol/L, R2 = 0.991). And the slope Ksv, that often be used to estimate the sensing sensitivity, was estimated as 3.1 × 103 L/mol. In addition, the detection limit of Fe3+ is predicted to be about 1.611 × 10-3 mmol/L, based on the ratio of 3s/ Ksv (σ standard error) [32, 33]. In comparison with our previous work [26], the detection limit of NKU-115 is further improved about two order of magnitude, which may be ascribed to the introduction of open nitrogen site weak interacting with Fe3+ ion.
We proceeded to explore the mechanism of the observed luminescence quenching. Firstly, the recovered NKU-115 sample from fluorescence quenching experiment, by centrifugation and then was washed with ethanol for several times, shows a nearly identical PXRD pattern to that of as-synthesized one (Fig. S10 in Supporting information), excluding possible framework collapse; Secondly, inductively coupled plasma spectroscopy (ICP) was utilized to monitor the content change of Fe and Tb of NKU-115 during the detection, and the results inidate that the content of Tb is much larger than that of Fe (Table S3 in Supporting information), suggesting the little effect of ion-exchange.
The non-zero content of Fe3+ may be caused by weak interaction between Fe3+ and the framework. It has been suggested that the free nitrogen and oxygen coordination sites of NKU-115 might weakly interact with Fe3+ [22-24], which may affect the luminescence emission of NKU-115. To verify this speculation, X-ray photoelectron spectroscopy (XPS) measurement was perforemed for the recovered NKU-115 as well as the as-synthesized one. As shownd in Fig. S11 (Supporting information), obvious shift of the N1s peak of the triazole-N atom was observed from the 397.0 eV of as-synthesized sample to the 397.5 eV of recovered one [22], while no obvious shift was observed for the O 1s peak of the carboxylic oxygen [34]. This verifies the weak interaction between the free nitrogen coordination sites with Fe3+ ion.
Energy transfer [35] and competitive absorption [36, 37] frequently contribut to the fluorescence qunching of MOFs. To calrify the role of these two factors in the fluorescence qunching of NKU-115, the UV-vis spectrium of Fe3+ was recorded and compared with the emission spectrum and UV-vis spectrium of NKU-115. As shown in Fig. S12 (Supporting information), no obvious spectral overlap between the absorption of Fe3+ and the emission of NKU-115 was observed, which excludes the influence of energy transfer mechanism. On contrary, intensive spectral overlap between the absorption of Fe3+ and the abosrption of NKU- 115 was observed as shown in Fig. S13 (Supporting information), which testifies that the competitive absorption mechanism exerts effects on the emission quchening.
In summary, a 2D coordination polymer (NKU-115) has been successfully constructed based on Tb3+ ion and a bifunctional ligand under solvothermal condition. Notably, NKU-115 is of high water stability and shows selective fluorescence quenching response toward Fe3+ ions in aqueous system, attributable to the interaction of the free nitrogen coordination sites with Fe3+, as well as competitive absorption. This research provides helpful ideas for designing MOF-based fluorescence materials for detecting metal ions in aqueous system.
AcknowledgmentsThis work was supported by the National Natural Science Foundation of China (Nos. 21531005 and 21673120) and the Natural Science Foundation of Tianjin (Nos. 15JCZDJC38800 and 16JCZDJC36900).
Appendix A. Supplementary dataSupplementary material related to this article can be found, in the online version, at doi:https://doi.org/10.1016/j.cclet.2018.06.014.
| [1] |
S. Chen, Z. Shi, L. Qin, H. Jia, H. Zheng, Cryst. Growth Des. 17 (2016) 67-72. |
| [2] |
N.R. Chereddy, S. Thennarasu, A.B. Mandal, Analyst 138 (2013) 1334-1337. DOI:10.1039/c3an36577h |
| [3] |
Y. Lin, X. Zhang, W. Chen, W. Shi, P. Cheng, Inorg. Chem. 56 (2017) 11768-11778. DOI:10.1021/acs.inorgchem.7b01790 |
| [4] |
S. Fakih, X. Kong, H.L. Collins, et al., J. Med. Chem. 51 (2008) 4539-4552. DOI:10.1021/jm8001247 |
| [5] |
C. Brugnara, Clin. Chem. 49 (2003) 1573-1578. DOI:10.1373/49.10.1573 |
| [6] |
H. Nawaz, W. Tian, J. Zhang, et al., ACS Appl. Mater. Interfaces 10 (2018) 2114-2121. DOI:10.1021/acsami.7b17342 |
| [7] |
J.M. Jung, S. YoulLee, C. Kim, Sensor. Actuat. B-Chem. 251 (2017) 291-301. |
| [8] |
A. George, H. Gopalakrishnan, S. Mandal, Sensor. Actuat. B-Chem. 243 (2017) 332-337. |
| [9] |
S. Mukherjee, W.P. Lustig, N.D. Rudd, et al., Chem. Soc. Rev. 46 (2017) 3242-3285. DOI:10.1039/C6CS00930A |
| [10] |
X. Sun, Y. Wang, Y. Lei, Chem. Soc. Rev. 44 (2015) 8019-8061. DOI:10.1039/C5CS00496A |
| [11] |
J.R. Li, X.H. Bu, R.H. Zhang, Inorg. Chem. 43 (2004) 237-244. DOI:10.1021/ic034772i |
| [12] |
X.H. Bu, W. Weng, J.R. Li, W. Chen, R.H. Zhang, Inorg. Chem. 41 (2002) 413-415. DOI:10.1021/ic010605b |
| [13] |
W. Weng, X.H. Bu, M. Du, et al., Inorg. Chem. 41 (2002) 1007-1010. DOI:10.1021/ic010932j |
| [14] |
D. Banerjee, Z. Hu, J. Li, Dalton Trans. 43 (2014) 10668-10685. DOI:10.1039/C4DT01196A |
| [15] |
J.G. Bunzli, Chem. Rev. 110 (2010) 2729-2755. DOI:10.1021/cr900362e |
| [16] |
K. Binnemans, Chem. Rev. 109 (2009) 4283-4374. DOI:10.1021/cr8003983 |
| [17] |
J. Kido, Chem. Rev. 102 (2002) 2357-2368. DOI:10.1021/cr010448y |
| [18] |
M.D. Allendorf, C.A. Bauer, R.K. Bhakta, R.J.T. Houk, Chem. Soc. Rev. 38 (2009) 1330-1352. DOI:10.1039/b802352m |
| [19] |
J.N. Hao, B. Yan, Chem. Commun. 53 (2015) 7737-7740. |
| [20] |
H.H. Chen, B. Yan, Zhou You, J. Mater. Chem. A 2 (2014) 13691-13697. DOI:10.1039/C4TA01297F |
| [21] |
X.H. Zhou, L. Li, H.H. Li, et al., Dalton Trans. 42 (2013) 12403-12409. DOI:10.1039/c3dt51081f |
| [22] |
B. Chen, L. Wang, Y. Xiao, et al., Angew. Chem. Int. Ed. 48 (2009) 500-503. DOI:10.1002/anie.v48:3 |
| [23] |
J.Q. Liu, G.P. Li, W.C. Liu, et al., ChemPlusChem 81 (2016) 1299-1304. DOI:10.1002/cplu.v81.12 |
| [24] |
D.K. Singha, P. Mahata, Inorg. Chem. 54 (2015) 6373-6379. DOI:10.1021/acs.inorgchem.5b00688 |
| [25] |
J. Zhao, Y.N. Wang, W.W. Dong, et al., Inorg. Chem. 55 (2016) 3265-3271. DOI:10.1021/acs.inorgchem.5b02294 |
| [26] |
X.L. Zhao, D. Tian, X.H. Bu, et al., Dalton Trans. 45 (2016) 1040-1046. DOI:10.1039/C5DT03283K |
| [27] |
R. Pearson, J. Am. Chem. Soc. 85 (1963) 3533-3539. DOI:10.1021/ja00905a001 |
| [28] |
Z. Yao, Z. Zhang, L. Liu, et al., Chem.-Eur. J. 22 (2016) 5676-5683. DOI:10.1002/chem.201505107 |
| [29] |
H. Xu, H.C. Hu, C.S. Cao, B. Zhao, Inorg. Chem. 54 (2015) 4585-4587. DOI:10.1021/acs.inorgchem.5b00113 |
| [30] |
Q. Cheng, Y. Dong, M. Kang, P. Zhang, J. Lumin. 156 (2014) 91-96. DOI:10.1016/j.jlumin.2014.07.019 |
| [31] |
S.K. Sahoo, D. Sharma, R.K. Bera, G. Crisponi, J.F. Callan, Chem. Soc. Rev. 41 (2012) 7195-7227. DOI:10.1039/c2cs35152h |
| [32] |
L. Li, J.Y. Zou, S.Y. You, et al., Dalton Trans. 46 (2017) 16432-16438. DOI:10.1039/C7DT03545D |
| [33] |
B. Hou, D. Li, Y. Lan, et al., Inorg. Chem. 55 (2016) 10580-10586. DOI:10.1021/acs.inorgchem.6b01809 |
| [34] |
G. Ji, J. Liu, X. Gao, et al., J. Mater. Chem. A 5 (2017) 10200-10205. DOI:10.1039/C7TA02439H |
| [35] |
B.P. Mehl, C.A. Kent, L. Ma, et al., J. Am. Chem. Soc. 132 (2010) 12767-12769. DOI:10.1021/ja102804s |
| [36] |
Z. Guo, H. Xu, S. Su, et al., Chem. Commun. 47 (2011) 5551-5553. DOI:10.1039/c1cc10897b |
| [37] |
K. Cao, F. Shi, W.M. Zhang, S. Zang, T.C.W. Mak, Chem.-Eur. J. 21 (2015) 15705-15712. DOI:10.1002/chem.v21.44 |
 2019, Vol. 30
2019, Vol. 30 

