b University of Chinese Academy of Sciences, Beijing 100049, China;
c Beijing Institute of Graphic Communication, Beijing 102600, China
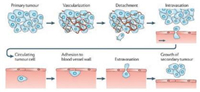
Tumor metastasis is a complex process responsible for > 90% of cancer-related deaths [1]. There are series of steps that comprise metastasis process, including cell detachment from primary tumor, migration/invasion, intravasation, circulating in blood system, adhesion to blood vessel wall, extravasation and formation of metastasis in secondary sites (Fig. 1) [2]. In addition to the genetic and biochemical factors, mechanochemistry as related to the mechanical regulation of molecular and cellular interactions plays crucial roles in these steps [3]. The study of how mechanical forces control the behavior of cells and tissues is a rapidly growing field in recent years [4].
|
Download:
|
| Fig. 1. The series of processes of tumor metastasis. Reproduced with permission [2]. Copyright 2018, Nature Publishing Group. | |
Major mechanical factors involved in tumor metastasis contain the mechanical properties of extracellular matrix (ECM), immunity cells, activated stroma cells, and blood system etc. [5, 6]. Only tumor cells that overcome the interaction forces from ECM and the effect of shear force will adhere to the vascular wall, subsequently fluid with vascular endothelium and successfully enter into the metastatic tissue [2]. In this review, we summarized the recent progress on the mechanical factors in tumor metastasis and the advanced methods to measure the mechanical behaviors of tumor cells.
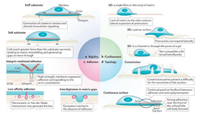
2. Mechanical factors in tumor metastasis 2.1. ECMECM plays a crucial role in modulating the interaction between cells, cells and microenvironment, regulating the morphologies and functions of cells. The major contributions of ECM in tumor metastasis are dynamic remodeling, physical scaffold for promoting the interaction between cells and providing the survival and differentiation signal [3]. The fibrotic ECM in tumor tissues potentiates the mechanotransduction in tumor cells, and thus controls or regulates the mechanical behaviors of tumor cells [7]. The stiffness, confinement, topological and geometric structure of ECM significantly influence the migration, adhesion, and contractility of tumor cells (Fig. 2) [8].
|
Download:
|
| Fig. 2. Physical variables of ECM influencing cell migration. Reproduced with permission [8]. Copyright 2018, Nature Publishing Group. | |
2.1.1. The stiffness of ECM
The stiffness of ECM is regulated by deposition, turnover, modification and crosslinking of collagen fibre, tendon protein and fibronectin. The enhancement of stiffness of ECM provides and promotes the cell migration and invasion [9]. Therefore, expanding researches focus on the investigation of guiding cell migration by the stiffness of ECM. Cells are able to follow gradients in the stiffness of their ECM, a process known as durotaxis which plays an important role in tumor metastasis [10]. For instance, the stiffness of ECM increases the growth and survival rate of tumor cells [11]. Therefore, people always detect tumor through the stiffness of tissue [12]. In addition, the enhancement of collagen expression, deposition and assembly can increase the stiffness of tissue [13]. During tumor development, the increase of matrix metalloproteinases (MMPs) [14] and lysyl oxidase (LOX) [15] can also increase the stiffness of ECM through ECM remodeling and crosslinking, leading to migration phonotype of cells [16].
The alteration of the stiffness of ECM is sensed by cell surface receptor integrinand transmitted to cytoskeleton through focal adhesion, leading to the contractility of actomyosin and promoting the migration and invasion of cells [17]. Thus, the malignancy development is enhanced owing to the promotion of the integrin-dependent mechanotransduction pathway. In addition, the physical properties of ECM regulate cell adhesion and its stability and dynamics [3]. Efficient cell migration requires cell contractile to balance the stiffness of ECM through adhesion strength [18]. The stiffness also promotes tumor metastasis through potentiating the epithelial-mesenchymal transition (EMT), which was considered as the major mechanism of tumor metastasis [19]. Previous research shows that, in human breast cancer, mechanical regulator TWIST1 increase the EMT through sensing the high stiffness of substrates, promoting the migration and invasion of cancer cells [20].
Recently, the major methods and materials utilized to adjust the stiffness of ECM for investigating their influences on mechanical behaviors of cancer cells include hydrazine-modified polyacrylamide (PA) hydrogels [20], substrate of polydimethylsiloxane (PDMS) [21], alginate [22], polyethylene glycoacid, collagen-agarose, hyaluronic matrices and soft fibrin gels etc. [23, 24]. All these approaches are limited in controlling the ligands density, stiffness ranges and matric activities. Therefore, it is also necessary to develop advanced materials which enable easily control over matrix physical properties for regulating cell migration and proliferation. Brendon Baker et al. combined polymer chemistry, electrospinning and soft lithography to formulate a protein-resistant, methacrylated dextran (DexMA) that could modulate the cell mechanical behaviors [25].
2.1.2. Confinement of ECMIn addition to the key variable of the stiffness of ECM, the confined ECM also influences the processes that drive cell migration [26]. However, it is difficult to model this kind of ECM to thoroughly investigate cell mechanical behaviors in complicatedly confined environment in vivo. Recently, researchers demonstrated that confinement of ECM indeed regulates many parameters of cell migration [27]. The cytoskeleton, other cellular structures or internal hydrostatic pressure in the confined environment pushes the cell to form filopodia or drive membrane to form blebs [8]. Therefore, the confinement can change the morphology of cells and promote the formation of bleb-like protrusion. With the alteration of confined environment, cell regulates or controls these parameters, leading to appearance of cell migration depended on cell contractility instead of cell adhesion. In the high strength of confinement, cell migration will change from epithelial phenotypes to mesenchymal migration, and spontaneously switch to a fast amoeboid migration phenotype [26, 28].
2.1.3. Topology structure of ECMDiscontinuous surfaces of ECM affect cell migration [8]. When cells migrate in the environment where the space is higher than a few hundred nanometers, the protrusion of the plasma membrane will be disrupted [8]. Another consequence of discontinuous environment is that the front of the cell are periods due to failure generation of traction forces positively feed-backed from substrates [8]. For example, by using multiphoton ablation lithography to pattern surfaces with nanoscale craters of various aspect ratios and pitches, Hojeong Jeon et al. showed that the surfaces altered the focal-adhesion size and distribution of cells, thus affecting cell morphology, migration and ultimately motivation [29]. Meanwhile, the fibrotic substrates can guide cell migration in the orientation of the substrates. Other topological structures also hamper the formation of protrusion, focal adhesion, and downstream signaling associated with them, preventing the efficient migration [30].
2.1.4. Other parameters of ECMThe heterostructure of ECM in tumor tissue showed not only alteration in special structures and matrix concentrations, but also changes in geometric parameters. Junmin Lee et al. suggested that interfacial geometries modulate cell shapes and cell adhesion. They also showed that geometric features at the perimeter of tumor tissue prime a population of cells with a stem-cell-like phenotype [31]. In the mouse model, these geometric structures can activate the intracellular signaling and thus promote the cancer cell tumorigenicity, growth, and metastasis. In addition, the solubility, porosity and other mechanical properties are able to contribute to tumor metastasis [2].
2.2. CellsTumor tissue comprises tumor cells, stromal cells, and the ECM. The stromal cells are vascular endothelial cells, pericytes, fibroblasts, myofibroblasts, mesenchymal stem cells (MSCs) and various bone marrow-derived cells, such as macrophages, neutrophils, mast cells, et al. [6]. Many of the stromal cells recruited to the primary tumor promote primary tumor growth or metastasis [32]. We mainly summarize the contribution of cell mechanical forces to tumor malignancy.
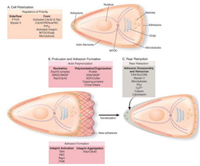
2.2.1. Tumor cellsThe mechanical properties of tumor cells modulate their own mechanical behaviors. In general, cell migration is usually conceptualized as a cyclic process regulated by mechanical properties of cells (Fig. 3) [33].
|
Download:
|
| Fig. 3. Steps in cell migration. Reproduced with permission [33]. Copyright 2018, Nature Publishing Group. | |
First, cells polarize and extend protrusions in the direction of migration. These polarization and protrusion are driven by actin polymerization. Second, the protrusion adheres to the ECM or adjacent cells via receptors that linked to the actin cytoskeleton. Third, the contractility generated by the cytoskeleton exerts force to the front focal adhesion, leading to the detachment of rear adhesion. Fourth, cell moves forward under strong cell traction force. Therefore, the mechanical properties of tumor cells directly influence the ability of cell motivation during the process of tumor metastasis.
At the initial metastasis process, cell adhesion and the alteration of cell morphology reduce the interaction forces among tumor cells in primary tumor, thus leading to the activation of EMT which increases tumor cell detachment from primary tumor and invasion to surrounding normal tissue [2]. The stress fiber and protrusion formed by intracellular actomyosin directly supply the contractility or traction force for cell migration on the substrates. Soft biological tissues can be described as viscoelastic materials [12]. Cells are composed of cytoplasm and cytoskeleton, which are complex composite system that behaves like an elastic material [34, 35]. Tumour cells undergo dramatic shape changes when they are entering into or exiting from the vascular system. These shape alterations are driven by cytoskeletal remodeling, which enable the tumor cells to penetrate through endothelial cell-cell junctions. The elastic formation enables the cell to rebound and keep round after entering to blood circulation system [2].
Meanwhile, the researches show that not only the elastic deformation of cytoplasm plays a crucial role in regulating the mechanical processes of cell intravasation or extravasation. The nucleus, the biggest and the more stiffness organelle of cells, also has modulation ability to elastic deformation and cytoskeleton remodeling [2]. For example, linkers of the nucleus and cytoskeleton complexes that span the nuclear envelope mediate physical connections between the nuclear lamina and the cytoskeleton, and thus influence tumor cell motility and invasion in three dimensional substrates [12].
In addition, the mechanical properties of tumor cells are different from those of the normal cells. Using atomic force microscope, Sarah Cross et al. reported that the stiffness of live metastatic cancer cells is more than 70% softer than the benign cells that line the body cavity [36]. These soft systems provide important mechanical support for elastic deformation of cells in ECM or blood circulation. These labile mechanical properties influence the adhesion abilities of cells under fluid share force. Only circulating tumor cells that overcome the effects of fluid shear are able to exit the circulation, adhere to the vascular wall of distant sites, and successfully invade the normal tissues to form metastasis foci [2].
Besides guiding cell migration, mechanical properties of tumor cells also contribute to ECM remodeling. Cells detached from the primary tumor begin to express MMPs on their surface, which promote the digestion of the collagen IVrich basement membrane, leading to the crosslinking of ECM and the alteration of the ECM stiffness, in turn, promote tumor migration [37]. Meanwhile, after leaving the primary tumor, motile tumor cells encounter the architecturally complex ECM which is rich in collagen I and fibronectin. Tumor cells adhering and moving on the ECM exert mechanical forces to ECM, increasing the mechanical properties of ECM through polymerization or remodeling of ECM.
Altogether, the cell-cell and cell-ECM interaction forces and the mechanical forces of tumor cells play a very important role during the series steps of cancer cell metastasis.
2.2.2. Macrophage cellsClinical and experimental evidences showed that tumor progression and metastasis are associated with macrophages [38]. A high density of tumor-associated macrophages correlated with poor prognosis. John Condeelis and Jeffrey Pollard enumerated six traits that are required for cells to develop malignant phenotypes [39]. These six characteristics all depend on the stroma cells interaction with malignant cells. They therefore summarized six aspects in malignant development promoted by macrophages, including inflammation, tumor cell invasion, matrix remodeling, intravasation, angiogenesis and seeding at distant sites [39].
Recent studies revealed that direct communications between macrophages and tumor cells lead to intravasation of tumor cells. Condeelis and Segall, using multiphoton imaging, revealed that macrophages enhancing tumor migration and invasion are supported by remarkable interactions between tumor cells and macrophages [40].
Macrophage motility and contractility can remodel ECM, which significantly influences tumor development and metastasis. For example, macrophages increased crosslinked collagen by raising the integrin-introduced mechanotransduction pathways [41]. Moreover, macrophages enhance the ability of tumor cell invasion and migration to surrounding tissues through altering the mechanical properties of ECM [39]. Meanwhile, fibrotic ECM promotes the formation of invadosomes and lamellae of macrophages, which are specialized actin-rich structures that combine adhesion and degradation of ECM to facilitate cell migration through surrounding ECM [42]. In vitro analysis indicates that invadosomes facilitate the MMP-mediated degradation of ECM substrates [42].
At early stages of tumor development, macrophages exhibit an M1 phenotype, while macrophages switch into alternatively activated M2 phenotype at late stages of tumor progression [43]. It has been demonstrated that M2 type is more important in tumor malignancy, which is favorable to tumor cell adhesion, migration and invasion [43]. Therefore, remodeling ECM by macrophage motility, mechanical properties and contractility enhance the fibrosis of collagen, and thus increases the mechanical behaviors of tumor cells [44]. Ran Afik et al., using a orthotopic colorectal cancer model, found that monocyte-derived tumor-associated M2 advance tumor development by the remodeling of its ECM composition and structure [45]. Recent research conducted by Sebastian Nielsen et al. showed that macrophage-secreted granulin supports pancreatic cancer metastasis through inducing liver fibrosis. They demonstrated that macrophages regulate the process of resident hepatic stellate cells changing into myofibroblasts that secrete periostin, resulting in a fibrotic microenvironment that sustains metastatic tumor growth [46].
2.2.3. FibroblastPrevious study showed that a second stromal component of importance to the development and progression of tumor metastasis is fibroblasts, especially cancer-associated fibroblasts (CAFs) which occupy a large proportion of tumor tissue [47]. The origins of CAFs are local activated and recruited fibroblasts, epithelial- or endothelial-to-mesenchymal transition, smooth muscle cells, pericytes, and mesenchymal stem cells or even tumor cells [48].
CAFs increase tissue stiffness and solid stress, promote tumor metastasis. The major contributions of CAFs are their abilities to alter the mechanical properties of ECM and remodel ECM through producing more ECM molecules, such as collagen-I, fibronectin, and glycosaminoglycans, [49]. In addition, CAFs increase the crosslinking of ECM fibro through secreting MMPs which digest ECM, thus alter the mechanical properties of ECM [50]. In higher stress condition, fibroblasts are able to differentiate into myofibroblasts which increase the mechanical forces of cells and their abilities of synthesized ECM components [49].
Fibroblasts directly exert physical forces to tumor tissues or cells, leading to the enhancement of ECM stiffness or cell forces in the periphery tumor, promoting tumor invasion. Some evidence indicates that CAFs overexpress N-cadherin which enables them to obtain migration phenotypes [51]. The increase of ECM stiffness influences the assembly, constitute and function of focal adhesions formed by N-cadherin. This N-cadherin transfers to OB-cadherin during the migration of CAFs, which involves the special interaction between CAFs and tumor cells [52]. The special interaction turns on the mechanotransduction of adjacent tumor cells, which affect the morphologies and migration of tumor cells, thus promotes the adhesion and migration of tumor cells.
Like the suggested mechanical model, collective CAF migration exerts mechanical stress at the front of migrating tumor cohort and changes the stress dynamics of tumor cells, leading to tumor cell movements toward regions of low cell density [49]. Altogether, the mechanical contributions of CAFs to tumor progression and metastasis are that CAFs, 1) obtained migration phonotypes of CAFs directly interacted with tumor cells, 2) specially adhered to ECM that increased ECM stiffness and remodeled ECM, 3) remodeled ECM through secreting collagen and MMPs, 4) formed clusters enhancing the tumor stiffness and mechanical stress.
2.2.4. Other cellsIn recent years, scientists are interested in the mechanical contribution of other stroma cells to tumor progression and metastasis. Lots of other vascular and constitute cells attract wide attention. In these cells, actin structures combining with adhesion of cells drive formation of lamellipodium, promoting cell migration on ECM [53]. The cell migration enhances stress, potentiating the stiffness of ECM. Tumor cells response these altered mechanical properties of ECM, which will promote tumor cell progression and metastasis. In vitro studies showed that lamellipodium facilitate ECM remodeling regulated by MMPs. Meanwhile, the major constitute N-WASP can promote the nucleation of actin and concentrate MMPs to lamellipodium, promoting lung tumor metastasis [53]. In addition, pericyte also potentiate tumor metastasis. For example, Vesselin Cooke et al., using genetic mouse model or pharmacological inhibitor, demonstrated that pericyte is associated with increased hypoxia, EMT, and Met receptor activation, which suppressed tumor growth but enhanced metastasis [54]. All these processes regulate the mechanical properties of tumor cells and contribute to tumor metastasis.
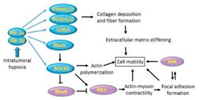
2.3. Other mechanical factors 2.3.1. Pressure of oxygenRapid tumor growth and abnormal tumor vessels result in generation of hypoxia regions within solid tumor. The low pressure of oxygen (PO2 < 10 mmHg) has tightly correlated with the risk of metastasis and mortality of cancer patient [55]. Oxygen concentration decreases from nearest blood vessel to deep tissue, leading to generation of intratumoural hypoxia region. The hypoxia regulates and affects ECM deposition, remodeling, degradation and crosslinking, which might potentiate tumor metastasis through influencing the mechanical properties of tumor tissue (Fig. 4) [56]. Researches revealed that tumor cells readily migrate along prolonged ECM under hypoxia condition [56]. Meanwhile, hypoxia can recruit other cells which produce and secrete collagen in this region to increase the stiffness of ECM, promoting tumor mobility.
|
Download:
|
| Fig. 4. Hypoxia induces collagen deposition and fiber formation that stiffen ECM. Reproduced with permission [56]. Copyright 2018, Elsevier. | |
2.3.2. Fluid stress
In circulatory system, tumor cells are subjected to haemodynamic forces and collisions with blood cells and endothelial cells [2]. All these stresses affect tumor cell survival and their ability to form metastatic foci. The major physical and mechanical parameters are the pattern of blood flow, the diameter of the blood vessels and the complex interplay between shear flow and intercellular adhesion [57]. These parameters activate the specific signal of tumor cells, driving rearrangement of cytoskeleton, and formation of adhesion complexes to adhere to vessel wall. Recent research demonstrated that shear force promotes the adhesion of tumor cells to collagen-rich ECM, which involves activation of Src signal pathway by shear force [58]. Shear force also enhanced the phosphoric FAK, which promotes the adhesion of colon cancer cell to ECM [59]. The adhesion and formation of metastasis foci happened under the moderate shear force [2]. Altogether, cells in circulatory system undergo a series of regulations and influences of in terms of mechanical forces.
3. Intracellular mechano-sensorsTumor tissues are usually stiffer than normal tissues. Therefore, tumor cells can response through cellular mechano-sensors extrinsic mechanical forces and translate them into cellular functions, which further control or modulate tumor development. A number of sensory elements exist in tumor cells, such as integrin, notch receptor, stretch-sensitive ion channels, which are able to probe and detect external forces [60]. Among these mechano-sensory complexes, integrin is the most important receptors for sensing and regulating mechanical properties of cells and ECM [61, 62]. They recruit focal adhesion complexes as integrated units, and provide mechanical links between actomyosins and ECM [63]. The units serve as signal transduction bridges when cells response to external the physical forces [64]. The external forces in turn influence the morphologies, adhesions, and migrations of cells through inducing assemblies of actomyosin skeletons.
Focal adhesion complex consists of integrin and other signal proteins, including vinculin, talin, filamin, and focal adhesion kinase. Those mechano-sensors are thought to regulate the mechanical properties of the cells through structure altercations, assemblies and disassemblies. In general, the incident of ECM stiffness modulating tumor malignancy mainly depends on the activation of integrin-dependent mechano-transduction pathway.
Cadherin are capable of responding to mechanical cues from other cells and regulating adhesion between two cells [65]. Cadherins are transmembrane Ca2+-dependent adhesion receptors that sustain the overall state of adhesion between epithelial cells. There are several cadherin isoforms, including E-cadherin, P-cadherin, and N-cadherin, which form adherens junctions to transmit forces between the actomyosin cytoskeleton and transmembrane proteins of the cadherin receptors [66, 67]. Xavier Trepat and his coworkers showed that concentrations of cadherins and catenins predict the intercellular forces and their interaction rates. They identified P-cadherin predicts levels of intercellular forces, E-cadherin predicts the rate at which intercellular forces build up [68]. The results of in vitro experiments indicated that the under force, cadherin-based adhesions will transmit the force form cadherin-catenin complex to actin filaments [65].
Notch receptors, a transmembrane protein, are normally triggered or activated by ligands a neighboring cell, which is responsible for intercellular communication, migration and subsequent metastasis of tumor cells [69, 70]. Mechanical forces facilitate the receptors conformational changes which are required for the activation of Notch signal [71]. Recent research showed that mechanical forces have the potential to activate Notch signal [72]. Xuefeng Wang et al., using tension gauge tether (TGT), determined the single-molecule forces required for mechanical signaling in cells. They concluded that Notch activation requires either no tension or a tension below 12 pN [69].
Stretch-sensitive ion channels can also sense mechanical force and transmit the force between the cells. Forces have been shown to open mechanosensitive calcium ion channels mainly through altering the molecules conformation. Piezo1 and Piezo2 are essential components of cation channels activated by distinct mechanical force [73]. Recent research showed that the typical force to induce Piezo1 conformation change is almost 10 pN [74]. Altogether, these mechano-sensors response to the external mechanical forces and further transmit the force to the cytoskeleton to activate additional mechanosensitive signaling pathways, regulating and controlling mechanical behaviors of tumor cells.
4. Measurement of mechanical forces of tumor cellsScientists are interested in characterizing and measuring the mechanical forces of tumor cells. The major forces mediating tumor mechanical behaviors are forces of cell-ECM adhesion, cell-cell adhesion and mechanical forces generated by plasma membrane, actomyosin networks and nucleus [75-78]. Researchers have developed many advanced methods and tools to measure tumor cellular forces, such as traction microscopy, AFM, micropillars, optical tweezers, which can reveal the mechanisms of cell migration and adhesion.
4.1. Traction microscopyIn tumor metastasis, cell physical properties including contractility, adhesion and cell stiffness, and nucleus mechanical properties are influenced by the stiffness of ECM. Traction microscopy has been developed to measure the forces generated by cells on two- and three-dimensional substrates through measuring deformations of the surrounding materials [79-81]. For example, Xavier Trepat and his coworkers recently using fourier transform traction microscopy and optogenetic tools measured the contractile forces which influence cell migration, division, morphogenesis, and cancer invasion [82]. Jeffrey Fredberg and coworkers using monolayer traction microscopy measured cell adhesive stress within intact cellular layer [83]. This traction microscopy record the traction force exerted by cells onto substrates from the displacement of fluorescent bead in deformable gel substrates [84]. However, this technology has been limited by the need of advanced software and important experimental caveats [85]. For example, a small amount of measurement noise in the displacement images can lead to large errors in the force.
4.2. MicropillarsMicropillar systems have commonly been used to study forces generated by cells, which provide a more direct measurement of forces [86]. In micropillar systems, each pillar has a known spring constant and can be examined independently. The forces acting on a pillar are directly proportional to the deformation of the pillar. Thus, cell forces exerted on the elastic pillars can be readily measured from pillar displacements. Micropillars are typically made of polydimethylsiloxane (PDMS), which allows only a limited variation in the Young's modulus. For the precise measurement of cell adhesion force, some scientists adjusted the Young's modulus over a wide range hydrogel instead of PDMS. They design this new tool to measure cell traction forces transmitted through integrins [86, 87]. Furthermore, magnetic beads can be inserted into pillars to provide additional mechanical stimuli. For example, Christopher Chen et al. using a magnetic field to induce torque in the nanowires deflected the magnetic posts and imparted force of individual cell adhesion. They found that applying a step force led to an increase in focal adhesion size and revealed the dynamic relationship between external and internal force and their interactions [88].
The advantages of this approach are (1) cell forces can be easily calculated from displacements without a reference images like traction microscopy, (2) pillar displacement only depends on the force exerted to that pillar. Adjustment of the stiffness of the micropillar allows the generation of heterogeneous mechanical substrates. However, there are also disadvantages. For example, cell adhesions are likely to be affected by the discontinuous substrates, and micro-fabricationrestricts the stiffness range. Although some efforts focus on the developing advanced approaches for enhancing the stiffness ranges, it is difficult to produce effective substrates with stiffness as such in physiological conditions.
4.3. Atomic force microscope (AFM)AFM has advantages in measurement of cellular force, which has been developed to measure the force of cell adhesion at single-molecule levels [89]. In this approach, beads or cells are attached to a micrometer-scale cantilever of AFM using them as probes. The cantilever bending, which is caused by the interaction of ligands and receptors, is proportional to the forces of cell-cell and cell-ECM interactions [85]. Therefore, AFM is widely used as multifunctional tool to investigate mechanical behaviors of cells, even under near-physiological conditions [90]. For example, Bryan Marshall et al. using AFM and flow-chamber experiments showed that dual response of catch bonds involving cell-adhesion molecules to force, which provides a mechanism for regulating cell adhesion under conditions of variable mechanical stress. They revealed that increasing the force first prolonged then shortened the lifetimes of adhesion molecules, revealing both catch and slip bond behavior [91].
AFM has also been used in in vitro study of the mechanical properties of cytoskeletal proteins like talin [92]. These measuring methods of the interaction forces or the conformation alterations of macromolecules require purified molecules. However, the purifying process alters the structures and binding sites of molecules, leading the alteration of the mechanical force as in vivo. Therefore, scientists developed AFM to detect micromechanical modulus of tumor cells [93] or the direct the interaction force between two cells [89, 91, 94]. Most important, AFM has been used in ex vivo mechanical analyses of cancer cells obtained from patients [36]. By using AFM, researchers found that the stiffness of metastatic cancer cells is more than 70% softer than that of normal cells lie in the body cavities. They showed that different cancer types displayed similar mechanical properties which can be used to distinguish cancer cells from normal ones.
4.4. FIRMS development and applicationAll these methods can efficiently measure cellular forces. However, each method has its own limitations, such as the advanced software and important experimental caveats for traction microscopy, the microfabrication technologies and uniform surface substrates for micropillar, the high throughout and wide variations in stiffness of cantilevers for AFM [85]. Scientists endeavor to develop much more accurate and effective methods and technologies to measure the mechanical behaviors of tumor cells. For example, molecular fluorescence sensors [95], laser ablation [96], force inference etc [97]. In addition to these technologies of in vivo measuring the mechanical properties of cells, some advanced methods have overcome the limitations and are able to be used in vivo to detect the mechanical behaviors of tissues [98].
Recently, force-induced remnant magnetization spectroscopy (FIRMS) technique has been developed to measure the binding force between the target receptor molecules and their corresponding ligand molecules. This approach has a few advantages, such as simple fabrication of substrates, high throughput, short time measurement, etc. FIRMS uses an atomic magnetometer to measure magnetization of ligand-conjugated magnetic particles as a function of an external force. By gradually increasing the force amplitude, different noncovalent bonds will undergo dissociation at different forces. The dissociation is indicated by a decrease in magnetization, because the dissociated magnetic particles undertake Brownian motion that leads to the randomization of their magnetic dipoles. A FIRM spectrum will be obtained by taking the derivative of the magnetization with regard to force amplitude, in which peak positions indicate the binding forces of the interactions and peak amplitudes represent the abundance of the corresponding interactions force.
FIRMS utilized the supersensitive atomic magnetometer to directly monitor the magnetic particles. Therefore, applications of FIRMS have been demonstrated with antibody-antigen dissociation and cellular recognition through ligand-receptor interactions. During detection of the interaction force of magnetically labeled molecules, previous report showed that FIRMS on the use of atomic magnetometer can achieve high performance in terms of sensitivity, spatial resolution, and molecular specificity. For example, sequence-specific binding forces for DNA duplexes can be detected through FIRMS technique [99]. The well-defined binding forces of DNA oligomers, with a narrow force distribution of 1.8 pN, can be obtained through FIRMS investigation, directly resolving two DNA duplexes with a single base-pair difference in the same sample [99]. We previously demonstrated that the application of FIRMS in imaging of human T cells quantitatively revealed several different binding forces, for example, nonspecific binding force between the magnetic particles and the cell surface and specific binding force between antibodies and their antigens [100].
In addition, we showed that FIRMS utilized to detect the interaction force through successfully imaging of magnetic nanoparticles, which need to fulfill two criteria, high sensitivity in detecting a DC magnetic field and high capability of resolving spatial information at a long detection distance [101]. These parameters are very important for enhanced applicability in biomedical research.
Based on these advantages of measuring interaction force among molecules and cells, and imaging magnetic nanoparticles (NPs) in spatial information, we made great efforts to use FIRMS to investigate the mechanical behaviors of tumor cells in the process of metastasis. Comparing the previous techniques used to investigate the mechanical properties of tumor cells, besides the advantages above mentioned, FIRMS is also a high throughput platform. Therefore, we endeavor to develop FIRMS as a great platform to investigate the mechanical behaviors (adhesion force and migration rate) of tumor cells (Fig. 5) to reveal the detailed mechanical mechanisms of tumor metastasis.
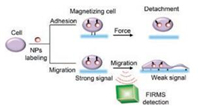
|
Download:
|
| Fig. 5. Schematic illustration of methods using FIRMS to measure the cell adhesion force and migration rate. | |
5. Future perspective and conclusions
The important mechanical processes of tumor metastasis are not fully understood. How cells sense and respond to the mechanical cues from external environment and how many external factors influence the mechanical behaviors of tumor cells are also unclear. The metastasis process is a complex, multistep process. The behaviors of tumor cells, at each step of the cascade, are controlled and modulated by mechanical forces. Therefore, forces coming from stroma cells and the ECM play important roles in controlling cell function [102]. It is difficult to study the integral influences of mechanical forces exerted on the tumor cells. It is clear that the mechanical behaviors of tumor cells depend on mechanical properties of neighbor cells, ECM, and other external forces or pressures. In recent years, a few of studies focus on the influences of the ECM stiffness and the immune cells on tumor physical behavior. However, the detailed physical interaction or mechanical contribution to in vivo tumor metastasis is still on challenging, especially the physical properties of tumor cells and physical interactions between tumor cells. Meanwhile, the technology and method measuring the physical force in vitro or in vivo showed many limitations. There is a pressing need to develop newly and advanced technology or methods which have the ability to reveal the details of cancer mechanochemistry, the study of mechanosensing and signaling pathways of mechanical cues that activate the tumor mechanical behaviors.
AcknowledgmentsThis work was supported by grants from the National Natural Science Foundation of China (Nos. 21778055, 21573250, 61501039), Beijing Natural Science Foundation (Nos. L172048, 2162017), Beijing Municipal Education Commission(No. KM201710015005), the Chinese Academy of Sciences (Nos. QYZDB-SSW-SLH024, YZ201424), and the Program for Young Outstanding Scientists of ICCAS (No. Y41Z011).
| [1] |
P.S. Steeg, Nat. Med. 12 (2006) 895-904. DOI:10.1038/nm1469 |
| [2] |
D. Wirtz, K. Konstantopoulos, P.C. Searson, Nat. Rev. Cancer 11 (2011) 512-522. DOI:10.1038/nrc3080 |
| [3] |
K.R. Levental, H. Yu, L. Kass, et al., Cell 139 (2009) 891-906. DOI:10.1016/j.cell.2009.10.027 |
| [4] |
Nat. Cell Biol. 19 (2017) 579.
|
| [5] |
K.B. Hotary, E.D. Allen, P.C. Brooks, et al., Cell 114 (2003) 33-45. DOI:10.1016/S0092-8674(03)00513-0 |
| [6] |
J.A. Joyce, J.W. Pollard, Nat. Rev. Cancer 9 (2009) 239-252. DOI:10.1038/nrc2618 |
| [7] |
X.C. Zhang, L. Han, Biophys. Reports 2 (2016) 45-54. DOI:10.1007/s41048-016-0030-7 |
| [8] |
G. Charras, E. Sahai, Nat. Rev. Mol. Cell Biol. 15 (2014) 813-824. DOI:10.1038/nrm3897 |
| [9] |
C. Frantz, K.M. Stewart, V.M. Weaver, J. Cell Sci. 123 (2010) 4195-4200. DOI:10.1242/jcs.023820 |
| [10] |
S.V. Plotnikov, A.M. Pasapera, B. Sabass, et al., Cell 151 (2012) 1513-1527. DOI:10.1016/j.cell.2012.11.034 |
| [11] |
C.M. Lo, H.B. Wang, M. Dembo, et al., Biophys. J. 79 (2000) 144-152. DOI:10.1016/S0006-3495(00)76279-5 |
| [12] |
D.T. Butcher, T. Alliston, V.M. Weaver, Nat. Rev. Cancer 9 (2009) 108-122. DOI:10.1038/nrc2544 |
| [13] |
S. Ramaswamy, K.N. Ross, E.S. Lander, et al., Nat. Genet. 33 (2003) 49-54. DOI:10.1038/ng1060 |
| [14] |
S. Jodele, L. Blavier, J.M. Yoon, et al., Cancer Metastasis Rev. 25 (2006) 35-43. DOI:10.1007/s10555-006-7887-8 |
| [15] |
C.G. Colpaert, P.B. Vermeulen, S.B. Fox, et al., Breast Cancer Res. Treat. 81 (2003) 137-147. DOI:10.1023/A:1025702330207 |
| [16] |
A.J.V.D. Slot, E.A.V. Dura, E.C.D. Wit, et al., Biochim. Biophys. Acta (BBA)-Mol. Basis Dis. 1741 (2005) 95-102. DOI:10.1016/j.bbadis.2004.09.009 |
| [17] |
C.K. Miranti, J.S. Brugge, Nat. Cell Biol. 4 (2002) 83-90. DOI:10.1038/ncb734 |
| [18] |
R. Madan, M.B. Smolkin, R. Cocker, et al., Hum. Pathol. 37 (2006) 9-15. DOI:10.1016/j.humpath.2005.09.024 |
| [19] |
L. Przybyla, J.M. Muncie, V.M. Weaver, Ann. Rev. Cell Dev. Biol. 32 (2016) 527-554. DOI:10.1146/annurev-cellbio-111315-125150 |
| [20] |
S.C. Wei, L. Fattet, J.H. Tsai, et al., Nat. Cell Biol. 17 (2015) 678-688. DOI:10.1038/ncb3157 |
| [21] |
R. Sunyer, V. Conte, J. Escribano, et al., Science 353 (2016) 1157-1161. DOI:10.1126/science.aaf7119 |
| [22] |
O. Chaudhuri, S.H. Parekh, W.A. Lam, et al., Nat. Methods 6 (2009) 383-387. DOI:10.1038/nmeth.1320 |
| [23] |
J. Liu, Y. Tan, H. Zhang, et al., Nat. Mater. 11 (2012) 734-741. DOI:10.1038/nmat3361 |
| [24] |
A.D. Doyle, N. Carvajal, A. Jin, et al., Nat. Commun. 6 (2015) 8720. DOI:10.1038/ncomms9720 |
| [25] |
B.M. Baker, B. Trappmann, W.Y. Wang, et al., Nat. Mater. 14 (2015) 1262-1268. DOI:10.1038/nmat4444 |
| [26] |
Y.J. Liu, M. Le Berre, F. Lautenschlaeger, et al., Cell 160 (2015) 659-672. DOI:10.1016/j.cell.2015.01.007 |
| [27] |
F. Lautenschläger, M. Piel, Curr. Opin. Cell Biol. 25 (2013) 116-124. DOI:10.1016/j.ceb.2012.10.017 |
| [28] |
V. Ruprecht, S. Wieser, A. Callan-Jones, et al., Cell 160 (2015) 673-685. DOI:10.1016/j.cell.2015.01.008 |
| [29] |
H. Jeon, S. Koo, W.M. Reese, et al., Nat. Mater. 14 (2015) 918-923. DOI:10.1038/nmat4342 |
| [30] |
M.J. Paszek, N. Zahir, K.R. Johnson, et al., Cancer Cell 8 (2005) 241-254. DOI:10.1016/j.ccr.2005.08.010 |
| [31] |
J. Lee, A.A. Abdeen, K.L. Wycislo, et al., Nat. Mater. 15 (2016) 856-862. DOI:10.1038/nmat4610 |
| [32] |
D. Hanahan, L.M. Coussens, Cancer Cell 21 (2012) 309-322. DOI:10.1016/j.ccr.2012.02.022 |
| [33] |
A.J. Ridley, M.A. Schwartz, K. Burridge, et al., Science 302 (2003) 1704-1709. DOI:10.1126/science.1092053 |
| [34] |
M. Bergert, A. Erzberger, R.A. Desai, et al., Nat. Cell Biol. 17 (2015) 524-529. DOI:10.1038/ncb3134 |
| [35] |
L. Deng, X. Trepat, J.P. Butler, et al., Nat. Mater. 5 (2006) 636-640. DOI:10.1038/nmat1685 |
| [36] |
S.E. Cross, Y.S. Jin, J. Rao, et al., Nat. Nanotechnol. 2 (2007) 780-783. DOI:10.1038/nnano.2007.388 |
| [37] |
K. Hotary, X.Y. Li, E. Allen, et al., Genes Dev. 20 (2006) 2673-2686. DOI:10.1101/gad.1451806 |
| [38] |
J.W. Pollard, Nat. Rev. Cancer 4 (2004) 71-78. DOI:10.1038/nrc1256 |
| [39] |
J. Condeelis, J.W. Pollard, Cell 124 (2006) 263-266. DOI:10.1016/j.cell.2006.01.007 |
| [40] |
J. Condeelis, R.H. Singer, J.E. Segall, Ann. Rev. Cell Dev. Biol. 21 (2005) 695-718. DOI:10.1146/annurev.cellbio.21.122303.120306 |
| [41] |
J.B. Wyckoff, Y. Wang, E.Y. Lin, et al., Cancer Res. 67 (2007) 2649-2656. DOI:10.1158/0008-5472.CAN-06-1823 |
| [42] |
S. Linder, C. Wiesner, M. Himmel, Annu. Rev. Cell Dev. Biol. 27 (2011) 185-211. DOI:10.1146/annurev-cellbio-092910-154216 |
| [43] |
B.Z. Qian, J.W. Pollard, Cell 141 (2010) 39-51. DOI:10.1016/j.cell.2010.03.014 |
| [44] |
S. Sangaletti, C.E. Di, S. Gariboldi, et al., Cancer Res. 68 (2008) 9050-9059. DOI:10.1158/0008-5472.CAN-08-1327 |
| [45] |
R. Afik, E. Zigmond, M. Vugman, et al., J. Exp. Med. (2016) 2315-2331. |
| [46] |
S.R. Nielsen, V. Quaranta, A. Linford, et al., Nat. Cell Biol. 18 (2016) 549-560. DOI:10.1038/ncb3340 |
| [47] |
D.F. Quail, J.A. Joyce, Nat. Med. 19 (2013) 1423-1437. DOI:10.1038/nm.3394 |
| [48] |
M. Mutoh, M. Takahashi, K. Wakabayashi, Curr. Pharm. Des. 12 (2006) 2375-2382. DOI:10.2174/138161206777698972 |
| [49] |
G.S. Karagiannis, T. Poutahidis, S.E. Erdman, et al., Mol. Cancer Res. 10 (2012) 1403-1418. DOI:10.1158/1541-7786.MCR-12-0307 |
| [50] |
C. Gaggioli, S. Hooper, C. Hidalgo-Carcedo, et al., Nat. Cell Biol. 9 (2007) 1392-1400. DOI:10.1038/ncb1658 |
| [51] |
R. Kalluri, Nat. Rev. Cancer 16 (2016) 582-598. DOI:10.1038/nrc.2016.73 |
| [52] |
B. Hinz, P. Pittet, J. Smithclerc, et al., Mol. Biol. Cell 15 (2004) 4310-4320. DOI:10.1091/mbc.e04-05-0386 |
| [53] |
L. Blanchoin, R. Boujemaa-Paterski, C. Sykes, et al., Physiol. Rev. 94 (2014) 235-263. DOI:10.1152/physrev.00018.2013 |
| [54] |
V.G. Cooke, V.S. LeBleu, D. Keskin, et al., Cancer Cell 21 (2012) 66-81. DOI:10.1016/j.ccr.2011.11.024 |
| [55] |
P. Vaupel, Adv. Exp. Med. Biol. 645 (2009) 241-246. DOI:10.1007/978-0-387-85998-9 |
| [56] |
G.L. Semenza, Biochim. Biophys. Acta 1863 (2016) 382-391. DOI:10.1016/j.bbamcr.2015.05.036 |
| [57] |
I.J. Fidler, S. Yano, R. Zhang, et al., Lancet Oncol. 3 (2002) 53-57. DOI:10.1016/S1470-2045(01)00622-2 |
| [58] |
K. Lawler, G. O'Sullivan, A. Long, et al., Cancer Sci. 100 (2009) 1082-1087. DOI:10.1111/cas.2009.100.issue-6 |
| [59] |
S.A. Von, P. Gassmann, K.M. Fisch, et al., Am. J. Pathol. 166 (2005) 585-596. DOI:10.1016/S0002-9440(10)62280-8 |
| [60] |
E.K. Paluch, C.M. Nelson, N. Biais, et al., BMC Biol. 13 (2015) 47-61. DOI:10.1186/s12915-015-0150-4 |
| [61] |
P. Kanchanawong, G. Shtengel, A.M. Pasapera, et al., Nature 468 (2010) 580-584. DOI:10.1038/nature09621 |
| [62] |
Y. Zhou, S. Chakraborty, S. Liu, Theranostics 1 (2011) 58-82. DOI:10.7150/thno/v01p0058 |
| [63] |
L. Ning, S. Lü, Z. Yan, et al., Acta Mech. Sin. 31 (2015) 248-258. DOI:10.1007/s10409-015-0407-8 |
| [64] |
J.D. Humphrey, E.R. Dufresne, M.A. Schwartz, Nat. Rev. Mol. Cell Biol. 15 (2014) 802-812. |
| [65] |
C.D. Buckley, J. Tan, K.L. Anderson, et al., Science 346 (2014) 1254211. DOI:10.1126/science.1254211 |
| [66] |
N. Borghi, M. Sorokina, O.G. Shcherbakova, et al., Proc. Natl. Acad. Sci. U. S. A. 109 (2012) 12568-12573. DOI:10.1073/pnas.1204390109 |
| [67] |
J. Maître, C.P. Heisenberg, Curr. Biol. 23 (2013) R626-R633. DOI:10.1016/j.cub.2013.06.019 |
| [68] |
E. Bazellieres, V. Conte, A. Elosegui-Artola, et al., Nat. Cell Biol. 17 (2015) 409-420. DOI:10.1038/ncb3135 |
| [69] |
X. Wang, T. Ha, Science 340 (2013) 991-994. DOI:10.1126/science.1231041 |
| [70] |
S. Artavanistsakonas, M.D. Rand, R.J. Lake, Science 284 (1999) 770-776. DOI:10.1126/science.284.5415.770 |
| [71] |
C.C. DuFort, M.J. Paszek, V.M. Weaver, Nat. Rev. Mol. Cell Biol. 12 (2011) 308-319. |
| [72] |
W.R. Gordon, D. Vardar-Ulu, G. Histen, et al., Nat. Struct. Mol. Biol. 14 (2007) 295-300. DOI:10.1038/nsmb1227 |
| [73] |
B. Coste, J. Mathur, M. Schmidt, et al., Science 330 (2010) 55-60. DOI:10.1126/science.1193270 |
| [74] |
J. Wu, R. Goyal, J. Grandl, Nat. Commun. 7 (2016) 12939-12949. DOI:10.1038/ncomms12939 |
| [75] |
D. Riveline, E. Zamir, N.Q. Balaban, et al., J. Cell Biol. 153 (2001) 1175-1185. DOI:10.1083/jcb.153.6.1175 |
| [76] |
S. Yonemura, Y. Wada, T. Watanabe, et al., Nat. Cell Biol. 12 (2010) 533-542. DOI:10.1038/ncb2055 |
| [77] |
B. Sinha, D. Köster, R. Ruez, et al., Cell 144 (2011) 402-413. DOI:10.1016/j.cell.2010.12.031 |
| [78] |
J. Swift, I.L. Ivanovska, A. Buxboim, et al., Science 341 (2013) 1240104. DOI:10.1126/science.1240104 |
| [79] |
L.A. Lautscham, C. Kammerer, J.R. Lange, et al., Biophys. J. 109 (2015) 900-913. DOI:10.1016/j.bpj.2015.07.025 |
| [80] |
R.J. Bloom, J.P. George, A. Celedon, et al., Biophys. J. 95 (2008) 4077-4088. DOI:10.1529/biophysj.108.132738 |
| [81] |
M. Dembo, Y.L. Wang, Biophys. J. 76 (1999) 2307-2316. DOI:10.1016/S0006-3495(99)77386-8 |
| [82] |
L. Valon, A. Marin-Llaurado, T. Wyatt, et al., Nat. Commun. 8 (2017) 14396-14406. DOI:10.1038/ncomms14396 |
| [83] |
J.A. Park, J.H. Kim, D. Bi, et al., Nat. Mater. 14 (2015) 1040-1048. DOI:10.1038/nmat4357 |
| [84] |
S.J. Han, Y. Oak, A. Groisman, et al., Nat. Methods 12 (2015) 653-656. DOI:10.1038/nmeth.3430 |
| [85] |
P. Roca-Cusachs, V. Conte, X. Trepat, Nat. Cell Biol. 19 (2017) 742-751. DOI:10.1038/ncb3564 |
| [86] |
J.L. Tan, J. Tien, D.M. Pirone, et al., Proc. Natl. Acad. Sci. U. S. A. 100 (2003) 1484-1489. DOI:10.1073/pnas.0235407100 |
| [87] |
S. Rahmouni, A. Lindner, F. Rechenmacher, et al., Adv. Mater. 25 (2013) 5869-5874. DOI:10.1002/adma.201301338 |
| [88] |
N.J. Sniadecki, A. Anguelouch, M.T. Yang, et al., Proc. Natl. Acad. Sci. U. S. A. 104 (2007) 14553-14558. DOI:10.1073/pnas.0611613104 |
| [89] |
J. Helenius, C.P. Heisenberg, H.E. Gaub, et al., J. Cell Sci. 121 (2008) 1785-1791. DOI:10.1242/jcs.030999 |
| [90] |
Z. Liu, Y. Liu, Y. Chang, et al., Nat. Methods 13 (2016) 143-146. DOI:10.1038/nmeth.3689 |
| [91] |
B.T. Marshall, M. Long, J.W. Piper, et al., Nature 423 (2003) 190-193. DOI:10.1038/nature01605 |
| [92] |
M. Yao, B.T. Goult, B. Klapholz, et al., Nat. Commun. 7 (2016) 11966-11977. DOI:10.1038/ncomms11966 |
| [93] |
X. Tang, T.B. Kuhlenschmidt, J. Zhou, et al., Biophys. J. 99 (2010) 2460-2469. DOI:10.1016/j.bpj.2010.08.034 |
| [94] |
W. Zhao, M. Cai, H. Xu, et al., Nanoscale 5 (2013) 3226-3229. DOI:10.1039/c3nr00553d |
| [95] |
K.E. Kubow, R. Vukmirovic, L. Zhe, et al., Nat. Commun. 6 (2015) 8026-8037. DOI:10.1038/ncomms9026 |
| [96] |
S. Porazinski, H. Wang, Y. Asaoka, et al., Nature 521 (2015) 217-221. DOI:10.1038/nature14215 |
| [97] |
J.L. Maître, H. Berthoumieux, S.F. Krens, et al., Science 338 (2012) 253-256. DOI:10.1126/science.1225399 |
| [98] |
O. Campàs, T. Mammoto, S. Hasso, et al., Nat. Methods 11 (2014) 183-189. DOI:10.1038/nmeth.2761 |
| [99] |
L. De Silva, L. Yao, Y. Wang, et al., J. Phys. Chem. B 117 (2013) 7554-7558. DOI:10.1021/jp403817b |
| [100] |
L. Yao, S. Xu, Angew. Chem. Int. Ed. 50 (2011) 4407-4409. DOI:10.1002/anie.201007297 |
| [101] |
L. Yao, S. Xu, Angew. Chem. Int. Ed. 48 (2009) 5679-5682. DOI:10.1002/anie.v48:31 |
| [102] |
T. Iskratsch, H. Wolfenson, M.P. Sheetz, Nat. Rev. Mol. Cell Biol. 15 (2014) 825-833. |
 2019, Vol. 30
2019, Vol. 30 

