b Medical College, Tibet University, Lasa 850000, China
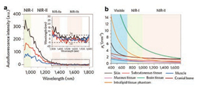
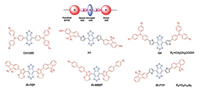
Fluorescence imaging has been widely used in biomedical field. At present, many fluorescence probes have been reported for pH sensitivity [1], imaging tumor hypoxia [2] and so on. NIR-Ⅱ (1000–1700 nm) fluorescence imaging is a new emerging imaging technology, providing deep tissue penetration, a high signal-to-noise ratio and high resolution due to lower tissue absorption, negligible tissue auto-fluorescence and reduced photon scattering [3]. The auto-fluorescence of the main organs of animals is also reduced as the emission wavelength increases, there is nearly zero tissue auto-fluorescence at the wavelength between 1500–1700 nm. Almost all biological tissues follow an inversely proportional wavelength relationship, that is, the longer the wavelength is, the less scattering will occur. Each tissue has associated scattering coefficient, but there is always a general trend of reduced scattering at longer wavelengths as shown in Fig. 1. To date, the main fluorophore units are composed of conjugated π system with a benzobisthiadiazole (BBTD) core and donor-acceptor-donor (D-A-D) structure (Scheme 1) [4-11]. In this highlight, we have focused on recent advances in the chemical design, synthesis and biomedical applications of small-molecule NIR-Ⅱ fluorophores without a BBTD core.
|
Download:
|
| Fig. 1. (a) Auto-fluorescence spectra of ex vivo mouse liver (black), spleen (red) and heart tissue (blue) under 808 nm excitation at different wavelength. (b) Scattering coefficients of different biological tissues in the different wavelength ranging from 400 to 1600 nm. Reproduced with permission [3]. Copyright 2018, Springer Nature. | |
|
Download:
|
| Scheme 1. Brief summary of D-A-D NIR-Ⅱ organic dyes. | |
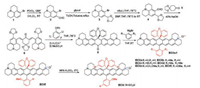
Organic fluorophores in NIR-Ⅰ region remain attractive due to their simple synthetic steps, facile modification and low toxicity. Rhodamine are widely used NIR-Ⅰ dyes that can be used to detect metal ions and changes in intracellular pH [12-15]. Recently, Youjun Yang et al. have designed and synthesized a new class of bright NIR organic fluorophores ECX as shown in Scheme 2, which originates from its bisbenzo-C-rhodamine unit [16]. The photo-physical properties of ECXa–f in over 20 kinds of organic solvents were studied. Both ECXb in CH2Cl2 and ECXi in water showed the maximal absorbing and emitting wavelengths at around 880/ 920 nm (Figs. 2a and b), respectively. And their emission bands tail beyond 1100 nm in different solvents. ECX exhibited high quantum yield with 7.6%–13.3% in CH2Cl2, especially ECXb reached up to 13.3% (Fig. 2c). Besides, ECX dyes displayed good photostability compared to a commercial naphthalocyaine as a reference. When exposed to 75 W halogen lamp for 25 min, the absorbance of naphthalocyaine decayed by 90%, 5%–10% for ECXa–f, and 26% for ECXi, demonstrating their better photostability than naphthalo-cyaine. Because of the different ortho-substituent of the bottom phenyl group, ECX dyes showed different fluorescence quantum yields, while their molar absorptivities were almost unaffected. ECX dyes were also chemically stable, among which ECXi showed excellent chemostabilities in aqueous solutions at different pH values, remaining unchanged unless the pH values went either below 3 or beyond 12 (Figs. 2d and e). In the presence of biological sulfides, reactive oxygen species and hydrogen peroxide, minimal changes were observed through the absorbance and fluorescence spectra of ECXi. Strong fluorescent signals of EXCb were observed from the Hela cells and the mouse in vivo (Figs. 2f and g).
|
Download:
|
| Scheme 2. Synthetic steps of ECX dyes. | |

|
Download:
|
| Fig. 2. Absorption and emission wavelength of ECXb in CH2Cl2 (a) and those of ECXi in H2O (b). (c) Fluorescent characteristics of ECX dyes with different substituent groups. (d) The absorption spectra of ECXi in aqueous solutions at different pH values. (e) Absorbance of an ECXi solution in water at 620 nm (blue) and 880 nm (red) at different pH values, respectively. (f) Bright field (upper) and fluorescent images of Hela cells (bottom). (g) The fluorescent image of the nude mouse after the injection of ECXb through tail vein. Reproduced with permission [16]. Copyright 2018, John Wiley and Sons. | |
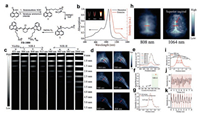
Fan Zhang and his team have synthesized a new dye FD-1080 through rational modifications on cyanine as shown in Fig. 3a [17], with its maximal excitation and emission wavelengths at 1064 nm and 1080 nm in the NIR-Ⅱ region (Fig. 3b), respectively. The excitation wavelength of the previous reported NIR-Ⅱ dyes is in the NIR-Ⅰ region. FD-1080 was the first organic NIR-Ⅱ dye with its excitation wavelength at 1064 nm. Irradiated at continuous laser, it exhibited superior photostability to the clinically approved ICG dye. The quantum yield of FD-1080 in FBS solvent can be increased up to 5.94%. FD-1080 could interact with the hydrophobic structure inside the FBS protein, leading to the binding of proteins to dyes and hindering the aggregation, which results in the brighter fluorescence. The longer excitation wavelength was, the less diminished scattering of photons became. At an excitation wavelength of 1064 nm, the sharp edges of the capillary tube even at a 5 mm immersion depth could be resolved when FD-1080 solution was immersed in an intralipid solution. To study the effect of different excitation wavelengths on imaging performance in vivo, varied excitation wavelengths were chosen from 655 nm to 1064 nm (Fig. 3c). Compared with the excitation at a shorter wavelength, the left hind limb vascular angiography could be better under the excitation of 1064 nm (Figs. 3d and e). The SBR obtained by imaging at 1064 nm excitation was much higher than for the shorter wavelength excitations (4.32 under 1064 nm excitation vs. 1.9–2.2 under shorter wavelength excitation) as shown in Fig. 3f. A small vessel with a FWHW of 0.47 mm was measured with 1064 nm NIR-Ⅱ excitation in vivo imaging (Fig. 3g). The same superior sagittal sinus vessel resolved by 1064 nm NIR-Ⅱ excitation in the mouse brain showed an apparent width (i.e., FWHM) of approximately 1.43 mm, while under 808 nm NIR-Ⅰ excitation was only 0.65 mm (Figs. 3h and i). Dynamic bioimaging such as respiratory rate was further detected with FD-1080-FBS complexes under 1064 nm excitation. The respiration rate of the awake mice was 124 breaths/min and that of the anesthetized mice was 71 breaths/min (Figs. 3j and k).
|
Download:
|
| Fig. 3. (a) Synthetic steps of FD-1080. (b) Absorbance and fluorescence wavelength of FD-1080. (c) NIR fluorescence images (1300 nm long pass filter) under excitation from 655 nm to 1064 nm of a capillary tube filled with FD-1080 solution immersed at different depth in 1% intralipid. (d) Fluorescent images of FD-1080 complexes in the mouse left hindlimb vasculature under varied excitation wavelengths. (e) Fluorescence signals intensity curves with different excitation wavelengths. (f) Fluorescent images of (d) with their corresponding signal background ratio (SBR) analysis. (g) The fluorescence intensity profiles (black dots) and Gaussian fit across a red line of interest under 1064 nm excitation. High-magnification vessel imaging of brain under different excitation wavelengths (h) with the vessel FWHM width analysis shown in (i). The respiratory rate of the awake (j) and anaesthetized (k) mouse. Reproduced with permission [17]. Copyright 2018, John Wiley and Sons. | |
In summary, fluorescence imaging in NIR-Ⅱ region is a promising tool to play a significant role in future in vivo imaging, early diagnosis of tumors and surgical navigation, which has received more and more attention. The traditional NIR-Ⅱ dye is based on the D-A-D structure design. Herein, two novel NIR-Ⅱ dyes following non-D-A-D architecture are successfully synthesized, enriching the structure of the NIR-Ⅱ probes. They exhibited excellent high quantum yield, chemo-/photo-stability, low ten-dency to aggregate and low cytotoxicity, demonstrating its good performance in deeper penetration depth and higher resolution in vivo imaging. Although the success of NIR-Ⅱ fluorophores has enriched our understanding and application in the field of NIR-Ⅱ bio-imaging, it is still a long way to go before research meets quality clinical care.
| [1] |
J.R. Hou, D. Jin, B. Chen, et al., Chin. Chem. Lett. 28 (2017) 1681-1687. DOI:10.1016/j.cclet.2017.03.037 |
| [2] |
F.L. Song, R. Liang, J.D. Deng, et al., Chin. Chem. Lett. 28 (2017) 1997-2000. DOI:10.1016/j.cclet.2017.08.023 |
| [3] |
G.S. Hong, A.L. Antaris, H.J. Dai, Nat. Biomed. Eng. (2017) 0010. |
| [4] |
A.L. Antaris, H. Chen, K. Cheng, et al., Nat. Mater. 15 (2016) 235-242. DOI:10.1038/nmat4476 |
| [5] |
J. Yang, Q. Xie, H. Zhou, et al., J. Proteome Res. 17 (2018) 2428-2439. DOI:10.1021/acs.jproteome.8b00181 |
| [6] |
A.L. Antaris, H. Chen, S. Diao, et al., Nat. Commun. 8 (2017) 15269. DOI:10.1038/ncomms15269 |
| [7] |
Y. Sun, C.R. Qu, H. Chen, et al., Chem. Sci. 7 (2016) 6203-6207. DOI:10.1039/C6SC01561A |
| [8] |
Y. Sun, M.M. Ding, X.D. Zeng, et al., Chem. Sci. 8 (2017) 3489-3493. DOI:10.1039/C7SC00251C |
| [9] |
Y. Sun, X.D. Zeng, Y.L. Xiao, et al., Chem. Sci. 9 (2018) 2092-2097. DOI:10.1039/C7SC04774F |
| [10] |
Y. Xu, M. Tian, H. Zhang, et al., Chin. Chem. Lett. 29 (2018) 1093-1097. DOI:10.1016/j.cclet.2018.03.032 |
| [11] |
X.D. Zeng, Y.L. Xiao, J.C. Lin, et al., Adv. Healthcare Mater. 7 (2018) 1800589. DOI:10.1002/adhm.v7.18 |
| [12] |
D. Wu, Y.Z. Shen, J.H. Chen, et al., Chin. Chem. Lett. 28 (2017) 1979-1982. DOI:10.1016/j.cclet.2017.07.004 |
| [13] |
X. Li, R. Zhao, Y. Yang, et al., Chin. Chem. Lett. 28 (2017) 1258-1261. DOI:10.1016/j.cclet.2016.12.029 |
| [14] |
J. Wen, P. Xia, Z. Zheng, et al., Chin. Chem. Lett. 28 (2017) 2005-2008. DOI:10.1016/j.cclet.2017.09.014 |
| [15] |
M. Hong, A. Liu, Y. Xu, et al., Chin. Chem. Lett. 27 (2016) 989-992. DOI:10.1016/j.cclet.2016.03.027 |
| [16] |
Z.H. Lei, X.R. Li, X. Luo, et al., Angew. Chem. Int. Ed. 56 (2017) 2979-2983. DOI:10.1002/anie.201612301 |
| [17] |
B.H. Li, L.F. Lu, M.Y. Zhao, et al., Angew. Chem. Int. Ed. 57 (2018) 7483-7487. DOI:10.1002/anie.201801226 |
 2018, Vol. 29
2018, Vol. 29 


