Peptides are of great importance for supporting the increasing demand in biochemical and pharmaceutical communities as useful biological tools and promising therapies [1-5]. Precise structural design and modification, for instance, backbone diversification or labeling of specific moiety, produced functionalized non-proteinogenic peptides with enhanced biological activities or improved therapeutic capabilities compared to their natural counterparts [6-14]. Both genetically mediated modification and linear approach—solid phase peptide synthesis (SPPS) and even protein total chemical synthesis with non-natural amino acids had been developed in-depth and to a certain degree of sophistication [15-29]. However, late-stage chemical modification of peptides and proteins, which would be more flexible and expedient in both academic and industrial laboratories, still remains challenging, especially considering the strict chemo- and site selectivity with divers potentially reactive sites and functional groups [30, 31].
In recent years, with the sufficient and thorough cognition about the reaction mechanisms, transition-metal promoted functionalization of ubiquitous C-H bonds has been emerged as a powerful and tunable tool for the synthesis and modification of small molecules and even complex natural products [32-54]. On account of their atom and step-economy, high functional group toleration, accurate site selectivity, C-H functionalization appear to be an effective platform for the late-stage modification of peptides and proteins [55-61]. In this review, we will briefly introduce the progress achieved in transition-metal-catalyzed C–H functionalization for the late-stage modification of peptides and proteins.
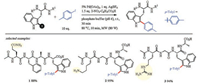
2. C-H bonds arylationIn the context of C–H functionalization of amino acids, tryptophan with an indole side chain, has much higher reactivity than others. Not only that, tryptophan also has a very low relative natural abundance (~1%). These features open up a broad opportunity for the late-stage modification of peptides and proteins through selective modification of tryptophan residues. In 2010, Lavilla, Albericio and co-workers reported an example of direct arylation of tryptophan peptides in aqueous using Pdcatalyst (Scheme 1) [62]. This reaction was accelerated by microwave (MW) heating and could be finished within 10 min. The toleration of various un-protected amino acid residues, such as, glutamine (1), lysine (2), and arginine (3), further demonstrated the utility of this method. Moreover, the high selectivity on tryptophan residue would not be influenced by the relative position (N-terminal, C-terminal or internal) in proteins.Thereafter, Pd-catalyzed site selective modification of tryptophan residues with aryl halides were further extended to an intramolecular version, and had been applied in the area of biaryl peptide stapling (Scheme 2) [63-65].
|
Download:
|
| Scheme 1. Pd-catalyzed intermolecular arylation of tryptophan peptides with iodobenzene. HPLC (high performance liquid chromatography) yields calculated by peak integrations | |

|
Download:
|
| Scheme 2. Pd-catalyzed intramolecular arylation of tryptophan residues for peptide stapling | |
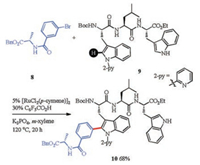
Not only aryl halides, but also arylboronic acids (ArB(OH)2) [66, 67], hypervalent aryl iodine reagents ([Ar2I]X) [67, 68], and aryldiazonium salts (ArN2X) [69] could be used in the Pd-catalyzed arylation of tryptophan peptides. While using ArB(OH)2 as aryl donor, extra oxidants were needed, such as, O2 (air could be used) or PhI(OAc)2. Ackermann and co-workers developed a metal-free arylation of tryptophan residues with hypervalent aryl iodine reagents ([Ar2I]OTs) [70]. Most recently, Ru-catalyzed pyridyl directed tryptophan peptide C–H arylation was also achieved (Scheme 3) [71].
|
Download:
|
| Scheme 3. Ru-catalyzed arylation of tryptophan peptides | |
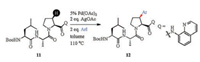
Over the past decades, Pd-catalyzed C(sp3)-H functionalization for the modification of amino acids or peptides enjoyed great success [72, 73]. With the sufficient and thorough cognition about the reaction mechanisms [74], a series of catalytic systems as well as novel directing groups [75] had been developed. These reactions are now been applied to more complex atmospheres. For example, in 2016, Kazmaier et al. described a proline peptide modification through Pd-catalyzed 8-amino quinolone directed β-arylation (Scheme 4) [76]. Most recently, Ackermann and co-workers reported an example of 1, 2, 3-triazole (Tzl) group directed C (sp3)-H arylation of peptides (Scheme 5) [77]. This reaction exhibited highly chemo- and positional selectivity in complex peptides, selectively arylated at alanine residue binding to Tzl group.
|
Download:
|
| Scheme 4. Pd-catalyzed 8-amino quinolone directed C-H arylation of peptides | |

|
Download:
|
| Scheme 5. Pd-catalyzed Tzl directed C–H arylation of peptides. a AcOH was used as solvent. b Hexafluoroisopropanol was used as solvent | |
The prior introduction and final removal of the directing groups would reduce the step-economy of late-stage modification strategies. Recently, non-directed (without external directing groups) C–H arylation reactions gained more and more attentions. For instance, Yu and co-workers reported an example of siteselective C(sp3)–H arylation of di-, tri-, and tetrapeptides (Scheme 6) [78]. The peptide backbone itself reacted as ligand to accelerate the C–H functionalization process.

|
Download:
|
| Scheme 6. Pd-catalyzed site-selective C(sp3)–H arylation of tetrapeptides | |
On the basis of Yu's backbone-assisted C-H arylation method [78], Albericio, Noisier and co-workers reported a late-stage peptide macrocyclization through Pd-catalyzed intramolecular linkage of N-terminal alanine and C-terminal phenylalanine residues (Scheme 7) [79]. The chemistry showed good functional group compatibility, various amino acid residues could be tolerated. The group of Wang also reported an analogous macrocyclization strategy, and applied this method to the total synthesis of key fragment in celogentin C [80]. In addition, cross dehydrogenative coupling (CDC) also provided access to the modification of amino acids and peptides. A large number of nucleophiles, such as indole [81-83], and arylboronic acid [84], could be used for the arylation at glycine residues with oxidative conditions (Scheme 8).

|
Download:
|
| Scheme 7. Pd-catalyzed C(sp3)–H arylation for late-stage peptide macrocyclization | |

|
Download:
|
| Scheme 8. CDC reaction for the arylation of peptides. TBHP = tert-butyl hydroperoxide | |
3. C-H bonds alkylation
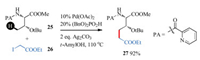
In 2012, Daugulis and co-workers reported an example of Pdcatalyzed C-terminal directed alkylation of side chain C(sp3)-H bond in amino acid derivatives [85-90]. Short after, Chen and coworkers realized a similar alkylation of amino acid derivatives with the N-terminal directing groups (Scheme 9) [91]. However, these reactions were limited to amino acid in a monomer, further exploration in this area were needed for expanding to the application in peptides.
|
Download:
|
| Scheme 9. Pd-catalyzed alkylation of side chain C(sp3)-H bond in amino acids | |
On the other hand, Rh-catalyzed cross-coupling provided opportunities for the modification of complex molecules. In 2004, Francis and co-workers reported an example of Rh-catalyzed bioconjugation (Scheme 10) [92]. This reaction was carried out smoothly in an aqueous solution (water/ethylene glycol = 80:20) with a low concentration (100 mmol/L). The component solvent improved the solubility of organic starting materials, and the addition of HONH2-HCl would enhance the catalyst reactivity through binding to Rh catalytic center and stabilizing the reaction intermediates. With above screened reaction conditions, tryptophan residues (sites: W7 and W14) on horse heart myoglobin were selectively modified (although N-alkylation and C2-alkylation mixtures were obtained). Moreover, with a lower pH (pH 1.5), the authors also accomplished the modification of tryptophan residue on subtilisin Carlsberg. With the discovery that N-(tert-butyl) hydroxylamine could promote the Rh-catalyzed carbenoid addition, tryptophan peptides modification conditions were further improved to a mild pH 6–7 [93].

|
Download:
|
| Scheme 10. Rh-catalyzed C-H alkylation of tryptophan residues on myoglobin | |
On the basis of Francis's Rh2(OAc)4-catalyzed peptide modification [92], Ball and co-workers developed an example of dirhodium metallopeptide catalyzed side chain modification (Scheme 11) [94]. The core thought of this strategy is the increase of resides selectivity via coiled coil molecular recognition. The highly efficiency of dirhodium metallopeptide catalyst led to an enhanced reaction rate, more accurate site selectivity and much lower amount of diazo reagents. More importantly, this strategy extended the scope of Rh-catalyzed peptide modification to tyrosine and phenylalanine residues, which were recognized unreactive on previous works. For the tyrosine residue, etherification through O-H insertion occurred. For the phenylalanine residue, aromatic or benzylic C-H insertion was not clear, formation of a norcaradiene through dearomatizing cyclopropanation was also likely to happen.
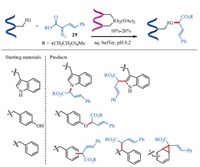
|
Download:
|
| Scheme 11. Dirhodium metallopeptide catalyzed side chain modification. FG = functional group | |
CDC alkylation reactions were wildly used for the synthesis of non-natural α-amino acids, with divers transition-metal-catalysts, such as, Cu [84, 95, 96], Ni [97] and so on. The asymmetric version of this reaction was also accomplished with a BOX (30)/Cu catalytic system (Scheme 12) [98]. Most recently, Correa et al. extended this reaction to the oxidative coupling of peptides [82]. In dipeptides and tripeptides with various amino acid fragments, the alkylation selectivity occurred at N-aryl group directed terminal glycine residues. In addition, similar products could also be obtained through Pd- or Ru- catalyzed alkylation of peptides, which went through an enolization process [99, 100].
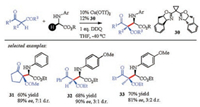
|
Download:
|
| Scheme 12. Cu/BOX catalyzed asymmetric CDC reaction for the synthesis of α-alkyl α-amino acids. DDQ = 2, 3-dichloro-5, 6-dicyano-1, 4-benzoquinone | |
Baran and co-workers described an example of C-H methylation of heteroarenes which could be used for the modification of biologically active molecules [101]. PSMS (Zinc bis(phenylsulfonylmethanesulfinate)) were developed for achieving this goal (Scheme 13). With the simple reaction conditions: 1.5 equiv. PSMS, 5 equiv. TBHP, in PhCF3/H2O at room temperature, (phenylsulfonyl) methylation occurred. And the subsequent desulfonylation revealed the methylated desired product. For instance, methylated derivative 37 of BQ-123 (36) could be easily prepared with this protocol.
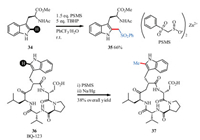
|
Download:
|
| Scheme 13. C-H methylation of tryptophan peptides | |
4. C-H bonds alkynylation
The introduction of alkynyl groups provided valuable modification for peptides and proteins, especially considering various further manipulations, for instance, click chemistry for late-stage labeling or conjugating [102, 103]. Recently, Waser et al. reported an example of Au-catalyzed direct C2 position alkynylation of tryptophan peptides using the hypervalent iodine reagent TIPSEBX 40 (Scheme 14) [104, 105]. Furthermore, Hansen and coworkers accomplished the chemo- and region- selective alkynylation of tryptophan in robust proteins, and also showed the further synthetic utility in Cu-catalyzed azide-alkyne cycloaddition (Cu-AAC) reaction [106].
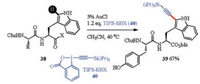
|
Download:
|
| Scheme 14. Au-catalyzed alkynylation of tryptophan peptides | |
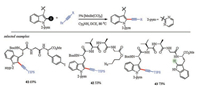
Most recently, Ackermann and co-workers reported the first Mn-catalyzed C-H alkynylations [107]. This reaction provided an efficient method for the modification of peptides (Scheme 15). The toleration of aryl iodide and alkyl azido demonstrated the robustness of this cross-coupling reaction. Moreover, it offered potential opportunities for further transformations, such as Huisgen reaction [108] and transition-metal-catalyzed crosscoupling at halogen groups [109-111].
|
Download:
|
| Scheme 15. Mn-catalyzed C-H alkynylation of peptides. TIPS = triisopropylsilyl | |
5. C-F bonds formation
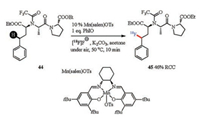
Late-stage fluorination of peptides would bring about interesting biological activities and physical properties, such as a series of positive influences on metabolic stability, lipid solubility and binding property [112-114]. In addition, 18F radiolabeling of peptides would also provide wide applications in positron emission tomography (PET) imaging technology [86, 115-117]. Recently, Groves and co-workers reported an easy handling Mncatalyzed late-stage radiofluorination of benzylic C-H bonds (Scheme 16) [118]. In this reaction, Mn(salen)OTs was selected as effective F-transfer catalyst which enabled labeling of various bioactive molecules including angiotensin-converting enzyme (44). Visible-light-promoted benzylic C-H fluorination could also been applied to the late-stage modification of phenylalanine peptides [119]. Pd-catalyzed stereoselective fluorination of α-amino acids [120-122] in conjugation with peptide synthesis provided another alternative strategy to access fluorinated peptides. Additionally, Doyle and co-workers described a Cu-catalyzed radiofluorination through the insertion of H-F into α-diazocarbonyl compounds, which provided an effective route to 18F-labeled peptides [123].
|
Download:
|
| Scheme 16. Mn-catalyzed C-H fluorination. RCC = radiochemical conversion | |
In addition, with Li's CDC methods, glycine peptides could also been alkynylated with slightly modified reaction conditions [84, 124].
6. C-Cl bonds formationMany other halogen-containing peptides (e.g., Cl, Br) could also been achieved nowadays. For instance, in 2016, Chen's research group reported an example of visible-light-promoted chlorination of 3° aliphatic C-H (Scheme 17) [125]. This reaction was carried out under mild reaction conditions: 0.1 mol% Ru(bpy)3Cl2 as photocatalyst, 2 equiv. Zhdankin azidoiodane reagent 46, 4 equiv. LiCl, in HFIP (hexafluoro-2-propanol) at room temperature. The authors showed the application of this chlorination to dipeptide. In addition, this reaction was also successful for the C-Br bonds formation.

|
Download:
|
| Scheme 17. C-H chlorination of dipeptides. Bpy = 2, 2'-bipyridine | |
7. C-O bonds formation
In 2006, Corey et al. reported a pioneering work of Pd-catalzed C–H acetoxylation of amino acids [126]. Compared with the hydroxylation of amino acid in a monomer, the analogous modification for peptides were lagged far behind. Until 2014, Yu and co-workers accomplished the site-selective C–H acetoxylation of tripeptides (Scheme 18) [78]. However, the narrow scope of alanine at the N-terminus at this stage partially limited the utilization of this transformation.

|
Download:
|
| Scheme 18. Pd-catalyzed C-H acetoxylation | |
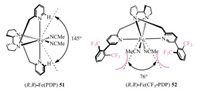
The group of White reported an example of catalyst controlled site-selectivity aliphatic C–H oxidation with interesting simple small-molecule, non-heme iron catalyst (Scheme 19) [127, 128]. This reaction was applied to the oxidation of a dipeptide (53). With the Fe-catalyst 51 which has a large 145° bite angle, the oxidation of C-H on norvaline and valine showed no obvious selectivity (the ratio of product 54/55 = 24 : 27). While the Fe-catalyst 52 with a much smaller 76° bite angle, diverted a much higher reactivity toward 2° over 3° C-H sites to the oxidant. This condition afforded a selectivity of 51:6 for oxidizing the norvaline methylene group, and provided 54 as the major product. The better site-selectivity control for 2° over 3° C-H sites when using Fe-catalyst 52 relied on the non-bonding interactions between the substrate and the catalyst. By comparison, with the Fe-catalyst 51, the selectivity was dictated by the competing electronic and steric effects [129].
|
Download:
|
| Scheme 19. Fe-catalyzed site-selective C-H hydroxylation. Method A: iterative addition of 5% Fe catalyst 51, AcOH (0.5 equiv.), H2O2 (1.2 equiv.), MeCN. Method B: AcOH (0.5 equiv.), MeCN, slow addition of 25% Fe catalyst 52 and H2O2 (5.0 equiv.) over 1 h. Ns = 4-nitrobenzenesulfonyl | |
Most recently, Shi, Gao and co-workers also reported an example of transition-metal-free hydroxylation of aliphatic C-H in dipeptides [130]. In this reaction, Na2S2O8 was used as oxidant, while the introduced oxygen atom came from solvent H2O. In addition, a series of transition-metal-free oxidative methods using trifluoromethyl(methyl)-dioxirane (TFDO) or dimethyldioxirane (DMDO) were also developed for the modification of peptides [131-135].
8. C-N bonds formationIt has a long history for the modification of amino acids through Pd-catalyzed C-H activation and subsequent new C-N bond formation, however, scarcely any successfully applied to complex peptide substrates [136-138]. Most recently, Shi et al. achieved an example of Pd-catalyzed intramolecular amidation of dipeptides at phenylalanine moiety (Scheme 20) [139]. In this reaction, the use of Tf (triflyl) as N-protecting group was crucial for obtaining the desired C-N coupling.

|
Download:
|
| Scheme 20. Pd-catalyzed intramolecular amidation of dipeptides. MsOH = methanesulfonic acid | |
An example of Pd-catalyzed C-H activation carbonylative cyclization of amines was also developed for the late-stage modification of peptides (Scheme 21) [140]. In this reaction, N- (2-pyridyl)sulfonyl (N-SO2Py) was selected as directing group, while Mo(CO)6 was used as air-stable solid carbonyl source. This reaction provided an expedient access to functionalized γ-lactams form amino acid and peptide derivatives.

|
Download:
|
| Scheme 21. Pd-catalyzed C-H activation carbonylative cyclization of amines. BQ = 1, 4-benzoquinone | |
Another practical C-N forming reaction was reported by Chen's group [125]. Chen and co-workers used a simple radicalmediated reaction system: Ru(bpy)3Cl2, Zhdankin azidoiodane reagent 46, and visible-light irradiation, which was quite effective for the C-H azidation of peptides (Scheme 22). It should be pointed out that, Fe-catalyzed [141, 142] and Mn-catalyzed [143] site-selective late-stage azidation of complex natural products also provided efficient access to nitrogen-containing complex natural product derivatives.

|
Download:
|
| Scheme 22. Radical-mediated C-H azidation of dipeptides | |
9. C-B bonds formation
Boron-containing compounds are important synthons during the construction of C-N, C-O and especially C-C bonds with Nobel Prize winning Suzuki reactions [144-150]. Additionally, through boron neutron capture therapy (BNCT) or hydrogen bonding, these compounds were now found broad applications in cancer treatment and enzyme inhibition [151].
C-H borylation was an attractive field, Hartwig, Miyaura, Smith et al. reported pioneering works in Ir-catalyzed borylation area [152, 153]. These findings were nowadays widely applied to the synthesis of boron-containing non-natural amino acids [154-156]. Recently, Elhammer and co-workers described a successful application to complex cyclic peptide: C-H borylation, transformation of boron moiety to bromide or iodide and subsequent Suzuki coupling, on a phenylalanine structure in natural product Aureobasidin A (62) (Scheme 23) [157]. In this transformation, borylation occurred only on one of the two phenylalanine residues. With this method, a series of biaryl derivatives were obtained for in vitro activities testing.

|
Download:
|
| Scheme 23. Ir-catalyzed borylation of peptides. Dtbpy = 4, 4'-di-tert-butyl bipyridine. COD = cis, cis-1, 5-cyclooctadiene | |
In addition, Shi and co-workers reported a challenging C(sp3)-H borylation using Pd catalyst. In this reaction, iPr2S played important role for preventing the formation of palladium black, and O2 was used as environment friendly oxidant [158]. Recently, Shi's group accomplished the Pd-catalyzed intermolecular C(sp3)- H silylation of amino acids, nicely supplement the means of Pdcatalyzed C-H activation and synthesis of non-natural amino acids [159, 160].
10. Other late-stage diversificationsRecent years, numerous elegant works for the residue-specific modification other than C–H functionalization were also reported [161, 162]. For example, Buchwald and Pentelute reported the development of organometallic palladium reagents for the modification of cysteine or lysine peptides with C-S or C-N bond formation [163, 164]. Baran, MacMillan and others developed various catalytic systems (Ni, Fe, visible-light, etc.) for peptide decarboxylative diversification with C-C or C-B bond construction [165-172]. Jackson realized the Negishi coupling of alanine derived organozinc for the synthesis of non-natural amino acids, and afterwards been improved by reductive cross-coupling strategies [113, 173-176].
11. Conclusion and outlookRecent developed C-H functionalization reactions became effective means for the late-stage modification of peptides and proteins. A view of progress toward side chain modification with C-C and C-X (C-F, Cl, O, N, B, etc.) bonds formation is outlined in this review. Although considerable achievements had been made in this field, several challenges still remain, or rather, points could be improved. First, milder reaction conditions are required for extensive use in bioconjugation: pH 6–8, temperature < 37 ℃, low concentration, and aqueous solvent preferred [177-180]. Second, artificial structural entities should be avoided, especially the protecting and directing groups which need prior introduction and final removal. Third, more efforts should be devoted to the practical applications in large peptides and proteins rather than model short peptides. Fourth, powerful tools for the modification of aliphatic C-H on peptides and proteins are urgently needed [181]. Fifth, while the unique reactivity and advantages for the modification of tryptophan residues, more choices with accurate selectivity on other residues in large peptides also deserve more attentions. We expected that in cooperation with photocatalysis [182-188], electrochemistry [189-192], continuous-flow manufacturing [193-195], and even molecule-making machine [196], metalcatalyzed C-H functionalization could be brought to a new stage, and would be a better choice for the late-stage modification of peptides and proteins in a very near future.
AcknowledgmentsWe are grateful for the support from the National Key R & D Program of China (No. 2017YFA0505400), the National Natural Science Foundation of China (Nos. 21572214, 21702200).
| [1] |
K. Fosgerau, T. Hoffmann, Drug Discov. Today 20 (2015) 122-128. DOI:10.1016/j.drudis.2014.10.003 |
| [2] |
B. Albada, N. Metzler-Nolte, Chem. Rev. 116 (2016) 11797-11839. DOI:10.1021/acs.chemrev.6b00166 |
| [3] |
T. Uhlig, T. Kyprianou, F.G. Martinelli, et al., EuPA Open Proteom. 4 (2014) 58-69. DOI:10.1016/j.euprot.2014.05.003 |
| [4] |
A.A. Kaspar, J.M. Reichert, Drug Discov. Today 18 (2013) 807-817. DOI:10.1016/j.drudis.2013.05.011 |
| [5] |
D.J. Craik, D.P. Fairlie, S. Liras, et al., Chem. Biol. Drug. Des. 81 (2013) 136-147. DOI:10.1111/cbdd.2012.81.issue-1 |
| [6] |
N. Krall, F.P. da Cruz, O. Boutureira, et al., Nat. Chem. 8 (2015) 103-113. |
| [7] |
M. Yang, J. Li, P.R. Chen, Chem. Soc. Rev. 43 (2014) 6511-6526. DOI:10.1039/C4CS00117F |
| [8] |
K. Lang, J.W. Chin, Chem. Rev. 114 (2014) 4764-4806. DOI:10.1021/cr400355w |
| [9] |
Y. Gong, L. Pan, Tetrahedron Lett. 56 (2015) 2123-2132. DOI:10.1016/j.tetlet.2015.03.065 |
| [10] |
X. Chen, Y.W. Wu, Org. Biomol. Chem. 14 (2016) 5417-5439. DOI:10.1039/C6OB00126B |
| [11] |
H. Liu, X. Li, Acta Chim. Sinica 72 (2014) 1197-1198. DOI:10.6023/A14110820 |
| [12] |
J.B. Li, S. Tang, J.S. Zheng, et al., Acc. Chem. Res. 50 (2017) 1143-1153. DOI:10.1021/acs.accounts.7b00001 |
| [13] |
Y. Lin, J. Wang, Y. Lu, Sci. China Chem. 57 (2014) 346-355. DOI:10.1007/s11426-014-5063-5 |
| [14] |
H.K. Cui, Y. Guo, Y. He, et al., Angew. Chem. Int. Ed. 52 (2013) 9558-9562. DOI:10.1002/anie.v52.36 |
| [15] |
J.M. Chalker, G.J.L. Bernardes, B.G. Davis, Acc. Chem. Res. 44 (2011) 730-741. DOI:10.1021/ar200056q |
| [16] |
J. Altenbuchner, M. Siemann-Herzberg, C. Syldatk, Curr. Opin. Biotechnol. 12 (2001) 559-563. DOI:10.1016/S0958-1669(01)00263-4 |
| [17] |
H. Xiao, A. Chatterjee, S.H. Choi, et al., Angew. Chem. Int. Ed. 52 (2013) 14080-14083. DOI:10.1002/anie.201308137 |
| [18] |
Y. Huang, L. Liu, Sci. China Chem. 58 (2015) 1779-1781. |
| [19] |
Q.Q. He, G.M. Fang, L. Liu, Chin. Chem. Lett. 24 (2013) 265-269. DOI:10.1016/j.cclet.2013.03.013 |
| [20] |
X. Wang, X. Wang, J. Hu, et al., Acta Chim. Sinica 73 (2015) 699-704. DOI:10.6023/A15030205 |
| [21] |
C.Z. Tao, Z.T. Zhang, J.W. Wu, et al., Chin. Chem. Lett. 25 (2014) 532-534. DOI:10.1016/j.cclet.2014.01.035 |
| [22] |
F. Ni, C. Fu, X. Gao, et al., Sci. China Chem. 58 (2015) 374-382. DOI:10.1007/s11426-015-5321-1 |
| [23] |
S.B.H. Kent, Annu. Rev. Biochem. 57 (1988) 957-989. DOI:10.1146/annurev.bi.57.070188.004521 |
| [24] |
P. Dawson, T. Muir, I. Clark-Lewis, et al., Science 266 (1994) 776-779. DOI:10.1126/science.7973629 |
| [25] |
S.B.H. Kent, Chem. Soc. Rev. 38 (2009) 338-351. DOI:10.1039/B700141J |
| [26] |
G.M. Fang, Y.M. Li, F. Shen, et al., Angew. Chem. Int. Ed. 50 (2011) 7645-7649. DOI:10.1002/anie.201100996 |
| [27] |
G.M. Fang, J.X. Wang, L. Liu, Angew. Chem. Int. Ed. 51 (2012) 10347-10350. DOI:10.1002/anie.201203843 |
| [28] |
Z. Wang, W. Xu, L. Liu, et al., Nat. Chem. 8 (2016) 698-704. DOI:10.1038/nchem.2517 |
| [29] |
M. Pan, S. Gao, Y. Zheng, et al., J. Am. Chem. Soc. 138 (2016) 7429-7435. DOI:10.1021/jacs.6b04031 |
| [30] |
C.D. Spicer, B.G. Davis, Nat. Commun. 5 (2014) 4740. DOI:10.1038/ncomms5740 |
| [31] |
O. Boutureira, G.J.L. Bernardes, Chem. Rev. 115 (2015) 2174-2195. DOI:10.1021/cr500399p |
| [32] |
T.W. Lyons, M.S. Sanford, Chem. Rev. 110 (2010) 1147-1169. DOI:10.1021/cr900184e |
| [33] |
T. Gensch, M.N. Hopkinson, F. Glorius, et al., Chem. Soc. Rev. 45 (2016) 2900-2936. DOI:10.1039/C6CS00075D |
| [34] |
H. Sun, N. Guimond, Y. Huang, Org. Biomol. Chem. 14 (2016) 8389-8397. DOI:10.1039/C6OB01258B |
| [35] |
K.M. Engle, T.S. Mei, M. Wasa, et al., Acc. Chem. Res. 45 (2012) 788-802. DOI:10.1021/ar200185g |
| [36] |
Y. Segawa, T. Maekawa, K. Itami, Angew. Chem. Int. Ed. 54 (2015) 66-81. DOI:10.1002/anie.201403729 |
| [37] |
G. Rouquet, N. Chatani, Angew. Chem. Int. Ed. 52 (2013) 11726-11743. DOI:10.1002/anie.v52.45 |
| [38] |
W. Ma, P. Gandeepan, J. Li, et al., Org. Chem. Front. 4 (2017) 1435-1467. DOI:10.1039/C7QO00134G |
| [39] |
Q.Z. Zheng, N. Jiao, Chem. Soc. Rev. 45 (2016) 4590-4627. DOI:10.1039/C6CS00107F |
| [40] |
P. Tao, Y. Jia, Sci. China Chem. 59 (2016) 1109-1125. DOI:10.1007/s11426-016-0058-7 |
| [41] |
K. Wang, F. Hu, Y. Zhang, et al., Sci. China Chem. 58 (2015) 1252-1265. DOI:10.1007/s11426-015-5362-5 |
| [42] |
X. Yang, G. Shan, L. Wang, et al., Tetrahedron Lett. 57 (2016) 819-836. DOI:10.1016/j.tetlet.2016.01.009 |
| [43] |
Y. Rao, G. Shan, X. Yang, Sci. China Chem. 57 (2014) 930-944. |
| [44] |
J. Wang, Sci. China Chem. 57 (2014) 1057. |
| [45] |
Z. Wang, P. Xie, Y. Xia, Chin. Chem. Lett. 29 (2018) 47-53. DOI:10.1016/j.cclet.2017.06.018 |
| [46] |
M.E. Wei, L.H. Wang, Y.D. Li, et al., Chin. Chem. Lett. 26 (2015) 1336-1340. DOI:10.1016/j.cclet.2015.08.009 |
| [47] |
J. Ding, Y. Guo, L.Y. Shao, et al., Chin. Chem. Lett. 27 (2016) 1617-1621. DOI:10.1016/j.cclet.2016.04.007 |
| [48] |
L. Xiang, K. Yang, Q.L. Song, Chin. Chem. Lett. 28 (2017) 517-520. DOI:10.1016/j.cclet.2016.11.023 |
| [49] |
Y.Y. Jiang, X. Man, S. Bi, Sci. China Chem. 59 (2016) 1448-1466. DOI:10.1007/s11426-016-0330-3 |
| [50] |
Z.Y. Xu, H.Z. Yu, Y. Fu, Sci. China Chem. 60 (2017) 165-166. |
| [51] |
Y. Wang, H. Zhou, K. Xu, et al., Chin. Chem. Lett. 28 (2017) 92-96. DOI:10.1016/j.cclet.2016.05.011 |
| [52] |
D. Zhou, C. Wang, M. Li, et al., Chin. Chem. Lett. 29 (2018) 191-193. DOI:10.1016/j.cclet.2017.06.007 |
| [53] |
T.J. Gong, B. Xiao, W.M. Cheng, et al., J. Am. Chem. Soc. 135 (2013) 10630-10633. DOI:10.1021/ja405742y |
| [54] |
B. Xiao, Z.J. Liu, L. Liu, et al., J. Am. Chem. Soc. 135 (2013) 616-619. DOI:10.1021/ja3113752 |
| [55] |
J.F. Hartwig, M.A. Larsen, ACS Central Sci. 2 (2016) 281-292. DOI:10.1021/acscentsci.6b00032 |
| [56] |
J. Wencel-Delord, F. Glorius, Nat. Chem. 5 (2013) 369-375. DOI:10.1038/nchem.1607 |
| [57] |
M. Jbara, S.K. Maity, A. Brik, Angew. Chem. Int. Ed. 56 (2017) 10644-10655. DOI:10.1002/anie.201702370 |
| [58] |
D.Y. Chen, S.W. Youn, Chem. Eur. J. 18 (2012) 9452-9474. DOI:10.1002/chem.201201329 |
| [59] |
Y. Liang, N. Jiao, Sci. China Chem. 60 (2017) 105-106. DOI:10.1007/s11426-016-0441-6 |
| [60] |
H. Yan, C. Zhu, Sci. China Chem. 60 (2017) 214-222. DOI:10.1007/s11426-016-0399-5 |
| [61] |
Y. Hu, C. Wang, Sci. China Chem. 59 (2016) 1301-1305. DOI:10.1007/s11426-016-0164-8 |
| [62] |
J. Ruiz-Rodriguez, F. Albericio, R. Lavilla, Chem. Eur. J. 16 (2010) 1124-1127. DOI:10.1002/chem.v16:4 |
| [63] |
H. Dong, C. Limberakis, S. Liras, et al., Chem. Commun. 48 (2012) 11644-11646. DOI:10.1039/c2cc36962a |
| [64] |
L. Mendive-Tapia, S. Preciado, J. García, et al., Nat. Commun. 6 (2015) 7160. DOI:10.1038/ncomms8160 |
| [65] |
L. Mendive-Tapia, A. Bertran, J. García, et al., Chem. Eur. J. 22 (2016) 13114-13119. DOI:10.1002/chem.201601832 |
| [66] |
T.J. Williams, A.J. Reay, A.C. Whitwood, et al., Chem. Commun. 50 (2014) 3052-3054. DOI:10.1039/C3CC48481E |
| [67] |
A.J. Reay, T.J. Williams, I.J.S. Fairlamb, Org. Biomol. Chem. 13 (2015) 8298-8309. DOI:10.1039/C5OB01174D |
| [68] |
Y. Zhu, M. Bauer, L. Ackermann, Chem. Eur. J. 21 (2015) 9980-9983. DOI:10.1002/chem.201501831 |
| [69] |
A.J. Reay, L.A. Hammarback, J.T.W. Bray, et al., ACS Catal. 7 (2017) 5174-5179. DOI:10.1021/acscatal.6b03121 |
| [70] |
Y. Zhu, M. Bauer, J. Ploog, et al., Chem. Eur. J. 20 (2014) 13099-13102. DOI:10.1002/chem.201404603 |
| [71] |
A. Schischko, H. Ren, N. Kaplaneris, et al., Angew. Chem. Int. Ed. 56 (2017) 1576-1580. DOI:10.1002/anie.201609631 |
| [72] |
A.F.M. Noisier, M.A. Brimble, Chem. Rev. 114 (2014) 8775-8806. DOI:10.1021/cr500200x |
| [73] |
X. Lu, B. Xiao, R. Shang, et al., Chin. Chem. Lett. 27 (2016) 305-311. DOI:10.1016/j.cclet.2015.12.021 |
| [74] |
Y. Dang, S. Qu, J.W. Nelson, et al., J. Am. Chem. Soc. 137 (2015) 2006-2014. DOI:10.1021/ja512374g |
| [75] |
M. Fan, D. Ma, Angew. Chem. Int. Ed. 52 (2013) 12152-12155. DOI:10.1002/anie.201306583 |
| [76] |
B. Mondal, B. Roy, U. Kazmaier, J. Org. Chem. 81 (2016) 11646-11655. DOI:10.1021/acs.joc.6b01963 |
| [77] |
M. Bauer, W. Wang, M.M. Lorion, et al., Angew. Chem. Int. Ed. 57 (2018) 203-207. DOI:10.1002/anie.201710136 |
| [78] |
W. Gong, G. Zhang, T. Liu, et al., J. Am. Chem. Soc. 136 (2014) 16940-16946. DOI:10.1021/ja510233h |
| [79] |
A.F.M. Noisier, J. García, I.A. IonuÛ, et al., Angew. Chem. Int. Ed. 56 (2017) 314-318. DOI:10.1002/anie.201608648 |
| [80] |
J. Tang, Y. He, H. Chen, et al., Chem. Sci. 8 (2017) 4565-4570. DOI:10.1039/C6SC05530C |
| [81] |
S. Zhu, M. Rueping, Chem. Commun. 48 (2012) 11960-11962. DOI:10.1039/c2cc36995h |
| [82] |
M. San Segundo, I. Guerrero, A. Correa, Org. Lett. 19 (2017) 5288-5291. DOI:10.1021/acs.orglett.7b02567 |
| [83] |
X. Wu, D. Zhang, S. Zhou, et al., Chem. Commun. 51 (2015) 12571-12573. DOI:10.1039/C5CC03706A |
| [84] |
L. Zhao, O. Baslé, C.J. Li, Proc. Nat. Acad. Sci. 106 (2009) 4106-4111. DOI:10.1073/pnas.0809052106 |
| [85] |
L.D. Tran, O. Daugulis, Angew. Chem. Int. Ed. 51 (2012) 5188-5191. DOI:10.1002/anie.201200731 |
| [86] |
S.M. Ametamey, M. Honer, P.A. Schubiger, Chem. Rev. 108 (2008) 1501-1516. DOI:10.1021/cr0782426 |
| [87] |
K. Chen, F. Hu, S.Q. Zhang, et al., Chem. Sci. 4 (2013) 3906-3911. DOI:10.1039/c3sc51747k |
| [88] |
K. Chen, B.F. Shi, Angew. Chem. Int. Ed. 53 (2014) 11950-11954. DOI:10.1002/anie.201407848 |
| [89] |
S.Y. Zhang, Q. Li, G. He, et al., J. Am. Chem. Soc. 135 (2013) 12135-12141. DOI:10.1021/ja406484v |
| [90] |
Q. Yang, S.D. Yang, ACS Catal. 7 (2017) 5220-5224. DOI:10.1021/acscatal.7b01779 |
| [91] |
S.Y. Zhang, G. He, W.A. Nack, et al., J. Am. Chem. Soc. 135 (2013) 2124-2127. DOI:10.1021/ja312277g |
| [92] |
J.M. Antos, M.B. Francis, J. Am. Chem. Soc. 126 (2004) 10256-10257. DOI:10.1021/ja047272c |
| [93] |
J.M. Antos, J.M. McFarland, A.T. Iavarone, et al., J. Am. Chem. Soc. 131 (2009) 6301-6308. DOI:10.1021/ja900094h |
| [94] |
B.V. Popp, Z.T. Ball, J. Am. Chem. Soc. 132 (2010) 6660-6662. DOI:10.1021/ja101456c |
| [95] |
J. Xie, Z.Z. Huang, Angew. Chem. Int. Ed. 49 (2010) 10181-10185. DOI:10.1002/anie.201004940 |
| [96] |
L. Zhao, C.J. Li, Angew. Chem. Int. Ed. 47 (2008) 7075-7078. DOI:10.1002/anie.v47:37 |
| [97] |
K. Li, Q. Wu, J. Lan, et al., Nat. Commun. 6 (2015) 8404. DOI:10.1038/ncomms9404 |
| [98] |
G. Zhang, Y. Zhang, R. Wang, Angew. Chem. Int. Ed. 50 (2011) 10429-10432. DOI:10.1002/anie.201105123 |
| [99] |
S. Datta, A. Bayer, U. Kazmaier, Org. Biomol. Chem. 10 (2012) 8268-8275. DOI:10.1039/c2ob26351c |
| [100] |
A. Bayer, U. Kazmaier, J. Org. Chem. 79 (2014) 8491-8497. DOI:10.1021/jo501731y |
| [101] |
J. Gui, Q. Zhou, C.M. Pan, et al., J. Am. Chem. Soc. 136 (2014) 4853-4856. DOI:10.1021/ja5007838 |
| [102] |
M. Meldal, C.W. Tornøe, Chem. Rev. 108 (2008) 2952-3015. DOI:10.1021/cr0783479 |
| [103] |
J.F. Lutz, Angew. Chem. Int. Ed. 46 (2007) 1018-1025. DOI:10.1002/(ISSN)1521-3773 |
| [104] |
J.P. Brand, J. Charpentier, J. Waser, Angew. Chem. Int. Ed. 48 (2009) 9346-9349. DOI:10.1002/anie.v48:49 |
| [105] |
G.L. Tolnai, J.P. Brand, J. Waser, Beilstein J. Org. Chem. 12 (2016) 745-749. DOI:10.3762/bjoc.12.74 |
| [106] |
M.B. Hansen, F. Hubálek, T. Skrydstrup, et al., Chem. Eur. J. 22 (2016) 1572-1576. DOI:10.1002/chem.201504462 |
| [107] |
Z. Ruan, N. Sauermann, E. Manoni, et al., Angew. Chem. Int. Ed. 56 (2017) 3172-3176. DOI:10.1002/anie.201611118 |
| [108] |
L. Li, S. Ding, Y. Yang, et al., Chem. Eur. J. 23 (2017) 1166-1172. DOI:10.1002/chem.201605034 |
| [109] |
T. Willemse, W. Schepens, H. Vlijmen, et al., Catalysts 7 (2017) 74. DOI:10.3390/catal7030074 |
| [110] |
M. Kaiser, C. Siciliano, I. Assfalg-Machleidt, et al., Org. Lett. 5 (2003) 3435-3437. DOI:10.1021/ol035178f |
| [111] |
O. Skaff, K.A. Jolliffe, C.A. Hutton, J. Org. Chem. 70 (2005) 7353-7363. DOI:10.1021/jo051076m |
| [112] |
X. Lu, Y. Wang, B. Zhang, et al., J. Am. Chem. Soc. 139 (2017) 12632-12637. DOI:10.1021/jacs.7b06469 |
| [113] |
X. Lu, J. Yi, Z.Q. Zhang, et al., Chem. Eur. J. 20 (2014) 15339-15343. DOI:10.1002/chem.v20.47 |
| [114] |
J. Xu, E.A. Ahmed, B. Xiao, et al., Angew. Chem. Int. Ed. 54 (2015) 8231-8235. DOI:10.1002/anie.201502308 |
| [115] |
M.E. Phelps, Proc. Nat. Acad. Sci. 97 (2000) 9226-9233. DOI:10.1073/pnas.97.16.9226 |
| [116] |
P.W. Miller, N.J. Long, R. Vilar, et al., Angew. Chem. Int. Ed. 47 (2008) 8998-9033. DOI:10.1002/anie.v47:47 |
| [117] |
J.J. Dai, C. Fang, B. Xiao, et al., J. Am. Chem. Soc. 135 (2013) 8436-8439. DOI:10.1021/ja404217t |
| [118] |
X. Huang, W. Liu, H. Ren, et al., J. Am. Chem. Soc. 136 (2014) 6842-6845. DOI:10.1021/ja5039819 |
| [119] |
D.D. Bume, C.R. Pitts, R.T. Jokhai, et al., Tetrahedron 72 (2016) 6031-6036. DOI:10.1016/j.tet.2016.08.018 |
| [120] |
R.Y. Zhu, K. Tanaka, G.C. Li, et al., J. Am. Chem. Soc. 137 (2015) 7067-7070. DOI:10.1021/jacs.5b04088 |
| [121] |
Q. Zhang, X.S. Yin, K. Chen, et al., J. Am. Chem. Soc. 137 (2015) 8219-8226. DOI:10.1021/jacs.5b03989 |
| [122] |
J. Miao, K. Yang, M. Kurek, et al., Org. Lett. 17 (2015) 3738-3741. DOI:10.1021/acs.orglett.5b01710 |
| [123] |
E.E. Gray, M.K. Nielsen, K.A. Choquette, et al., J. Am. Chem. Soc. 138 (2016) 10802-10805. DOI:10.1021/jacs.6b06770 |
| [124] |
S.A. Girard, T. Knauber, C.J. Li, Angew. Chem. Int. Ed. 53 (2014) 74-100. DOI:10.1002/anie.v53.1 |
| [125] |
Y. Wang, G.X. Li, G. Yang, et al., Chem. Sci. 7 (2016) 2679-2683. DOI:10.1039/C5SC04169D |
| [126] |
B.V.S. Reddy, L.R. Reddy, E.J. Corey, Org. Lett. 8 (2006) 3391-3394. |
| [127] |
P.E. Gormisky, M.C. White, J. Am. Chem. Soc. 135 (2013) 14052-14055. DOI:10.1021/ja407388y |
| [128] |
T. Nanjo, de Lucca E.C., M.C. White, J. Am. Chem. Soc. 139 (2017) 14586-14591. DOI:10.1021/jacs.7b07665 |
| [129] |
T.J. Osberger, D.C. Rogness, J.T. Kohrt, et al., Nature 537 (2016) 214-219. DOI:10.1038/nature18941 |
| [130] |
X. Li, X. Che, G.H. Chen, et al., Org. Lett. 18 (2016) 1234-1237. DOI:10.1021/acs.orglett.5b03690 |
| [131] |
R. Curci, L. D'Accolti, C. Fusco, Acc. Chem. Res. 39 (2006) 1-9. DOI:10.1021/ar050163y |
| [132] |
C. Annese, L. D'Accolti, M. De Zotti, et al., J. Org. Chem. 75 (2010) 4812-4816. DOI:10.1021/jo100855h |
| [133] |
M.R. Rella, P.G. Williard, J. Org. Chem. 72 (2007) 525-531. DOI:10.1021/jo061910n |
| [134] |
R. Saladino, M. Mezzetti, E. Mincione, et al., J. Org. Chem. 64 (1999) 8468-8474. DOI:10.1021/jo990185w |
| [135] |
C. Annese, I. Fanizza, C.D. Calvano, et al., Org. Lett. 13 (2011) 5096-5099. DOI:10.1021/ol201971v |
| [136] |
G. He, Y. Zhao, S. Zhang, et al., J. Am. Chem. Soc. 134 (2012) 3-6. DOI:10.1021/ja210660g |
| [137] |
G. He, S.Y. Zhang, W.A. Nack, et al., Angew. Chem. Int. Ed. 52 (2013) 11124-11128. DOI:10.1002/anie.201305615 |
| [138] |
Q. Zhang, K. Chen, W. Rao, et al., Angew. Chem. Int. Ed. 52 (2013) 13588-13592. DOI:10.1002/anie.201306625 |
| [139] |
M. Yang, X. Jiang, Z.J. Shi, Org. Chem. Front. 2 (2015) 51-54. DOI:10.1039/C4QO00282B |
| [140] |
E. Hernando, J. Villalva, A. Manu Martinez, et al., ACS Catal. 6 (2016) 6868-6882. DOI:10.1021/acscatal.6b01987 |
| [141] |
A. Sharma, J.F. Hartwig, Nature 517 (2015) 600-604. DOI:10.1038/nature14127 |
| [142] |
R.R. Karimov, A. Sharma, J.F. Hartwig, ACS Central Sci. 2 (2016) 715-724. DOI:10.1021/acscentsci.6b00214 |
| [143] |
X. Huang, T.M. Bergsten, J.T. Groves, J. Am. Chem. Soc. 137 (2015) 5300-5303. DOI:10.1021/jacs.5b01983 |
| [144] |
A. Suzuki, Angew. Chem. Int. Ed. 50 (2011) 6722-6737. DOI:10.1002/anie.201101379 |
| [145] |
C.T. Yang, Z.Q. Zhang, Y.C. Liu, et al., Angew. Chem. Int. Ed. 50 (2011) 3904-3907. DOI:10.1002/anie.201008007 |
| [146] |
C.T. Yang, Z.Q. Zhang, H. Tajuddin, et al., Angew. Chem. Int. Ed. 51 (2012) 528-532. DOI:10.1002/anie.201106299 |
| [147] |
J. Yi, J.H. Liu, J. Liang, et al., Adv. Synth. Catal. 354 (2012) 1685-1691. DOI:10.1002/adsc.201200136 |
| [148] |
L. Li, T. Gong, X. Lu, et al., Nat. Commun. 8 (2017) 345. DOI:10.1038/s41467-017-00363-4 |
| [149] |
W. Su, T.J. Gong, X. Lu, et al., Angew. Chem. Int. Ed. 54 (2015) 12957-12961. DOI:10.1002/anie.v54.44 |
| [150] |
W. Su, T.J. Gong, Q. Zhang, et al., ACS Catal. 6 (2016) 6417-6421. DOI:10.1021/acscatal.6b02039 |
| [151] |
D.B. Diaz, A.K. Yudin, Nat. Chem. 9 (2017) 731-742. DOI:10.1038/nchem.2814 |
| [152] |
T. Ishiyama, J. Takagi, J.F. Hartwig, et al., Angew. Chem. Int. Ed. 41 (2002) 3056-3058. DOI:10.1002/1521-3773(20020816)41:16<3056::AID-ANIE3056>3.0.CO;2-# |
| [153] |
J.Y. Cho, M.K. Tse, D. Holmes, et al., Science 295 (2002) 305-308. DOI:10.1126/science.1067074 |
| [154] |
F.M. Meyer, S. Liras, A. Guzman-Perez, et al., Org. Lett. 12 (2010) 3870-3873. DOI:10.1021/ol1015674 |
| [155] |
H. Audi, E. Rémond, M.J. Eymin, et al., Eur. J. Org. Chem. (2013) 7960-7972. |
| [156] |
V.A. Kallepalli, F. Shi, S. Paul, et al., J. Org. Chem. 74 (2009) 9199-9201. DOI:10.1021/jo901822b |
| [157] |
P.G.M. Wuts, L.J. Simons, B.P. Metzger, et al., ACS Med. Chem. Lett. 6 (2015) 645-649. DOI:10.1021/acsmedchemlett.5b00029 |
| [158] |
L.S. Zhang, G. Chen, X. Wang, et al., Angew. Chem. Int. Ed. 53 (2014) 3899-3903. DOI:10.1002/anie.201310000 |
| [159] |
Y. Yang, C. Wang, Sci. China Chem. 58 (2015) 1266-1279. DOI:10.1007/s11426-015-5375-0 |
| [160] |
Y.J. Liu, Y.H. Liu, Z.Z. Zhang, et al., Angew. Chem. Int. Ed. 55 (2016) 13859-13862. DOI:10.1002/anie.201607766 |
| [161] |
J.N. deGruyter, L.R. Malins, P.S. Baran, Biochemistry 56 (2017) 3863-3873. DOI:10.1021/acs.biochem.7b00536 |
| [162] |
S. Sengupta, G. Mehta, Tetrahedron Lett. 58 (2017) 1357-1372. DOI:10.1016/j.tetlet.2017.02.069 |
| [163] |
E.V. Vinogradova, C. Zhang, A.M. Spokoyny, et al., Nature 526 (2015) 687-691. DOI:10.1038/nature15739 |
| [164] |
H.G. Lee, G. Lautrette, B.L. Pentelute, et al., Angew. Chem. Int. Ed. 56 (2017) 3177-3181. DOI:10.1002/anie.201611202 |
| [165] |
L.R. Malins, Pept. Sci. (2018). DOI:10.1002/pep1002.24049 |
| [166] |
S. Bloom, C. Liu, D.K. Kolmel, et al., Nat. Chem. 10 (2018) 205-211. |
| [167] |
S.J. McCarver, J.X. Qiao, J. Carpenter, et al., Angew. Chem. Int. Ed. 56 (2017) 728-732. DOI:10.1002/anie.201608207 |
| [168] |
C. Li, J. Wang, L.M. Barton, et al., Science 356 (2017) eaam7355. DOI:10.1126/science.aam7355 |
| [169] |
T. Qin, J. Cornella, C. Li, et al., Science 352 (2016) 801-805. DOI:10.1126/science.aaf6123 |
| [170] |
W.M. Cheng, R. Shang, Y. Fu, ACS Catal. 7 (2016) 907-911. |
| [171] |
X. Lu, B. Xiao, L. Liu, et al., Chem. Eur. J. 22 (2016) 11161-11164. DOI:10.1002/chem.201602486 |
| [172] |
Y. Jin, M. Jiang, H. Wang, et al., Sci. Rep. 6 (2016) 20068. DOI:10.1038/srep20068 |
| [173] |
H.J.C. Deboves, C.A.G.N. Montalbetti, R.F.W. Jackson, J. Chem. Soc. Perkin Trans. 1 (2001) 1876-1884. |
| [174] |
X. Lu, B. Xiao, Z. Zhang, et al., Nat. Commun. 7 (2016) 11129. DOI:10.1038/ncomms11129 |
| [175] |
K.M.M. Huihui, J.A. Caputo, Z. Melchor, et al., J. Am. Chem. Soc. 138 (2016) 5016-5019. DOI:10.1021/jacs.6b01533 |
| [176] |
X. Wang, Y. Dai, H. Gong, Top. Curr. Chem. 374 (2016) 43. DOI:10.1007/s41061-016-0042-2 |
| [177] |
J.S. Zheng, S. Tang, Y.K. Qi, et al., Nat. Protoc. 8 (2013) 2483-2495. DOI:10.1038/nprot.2013.152 |
| [178] |
Y.M. Li, Y.T. Li, M. Pan, et al., Angew. Chem. Int. Ed. 53 (2014) 2198-2202. DOI:10.1002/anie.201310010 |
| [179] |
J.S. Zheng, M. Yu, Y.K. Qi, et al., J. Am. Chem. Soc. 136 (2014) 3695-3704. DOI:10.1021/ja500222u |
| [180] |
S. Tang, Y.Y. Si, Z.P. Wang, et al., Angew. Chem. Int. Ed. 54 (2015) 5713-5717. DOI:10.1002/anie.201500051 |
| [181] |
J. He, M. Wasa, K.S.L. Chan, et al., Chem. Rev. 117 (2017) 8754-8786. DOI:10.1021/acs.chemrev.6b00622 |
| [182] |
J.R. Chen, D.M. Yan, Q. Wei, et al., ChemPhotoChem 1 (2017) 148-158. DOI:10.1002/cptc.201700008 |
| [183] |
J. Twilton, C. Le, P. Zhang, et al., Nat. Rev. Chem. 1 (2017) 0052. DOI:10.1038/s41570-017-0052 |
| [184] |
J.R. Chen, X.Q. Hu, L.Q. Lu, et al., Acc. Chem. Res. 49 (2016) 1911-1923. DOI:10.1021/acs.accounts.6b00254 |
| [185] |
M.N. Hopkinson, A. Tlahuext-Aca, F. Glorius, Acc. Chem. Res. 49 (2016) 2261-2272. DOI:10.1021/acs.accounts.6b00351 |
| [186] |
J.C. Tellis, C.B. Kelly, D.N. Primer, et al., Acc. Chem. Res. 49 (2016) 1429-1439. DOI:10.1021/acs.accounts.6b00214 |
| [187] |
J. Zhang, Y. Chen, Acta Chim. Sinica 75 (2017) 41-48. DOI:10.6023/A16080416 |
| [188] |
Z. Feng, T. Zeng, J. Xuan, et al., Sci. China Chem. 59 (2016) 171-174. DOI:10.1007/s11426-015-5548-x |
| [189] |
E.J. Horn, B.R. Rosen, Y. Chen, et al., Nature 533 (2016) 77-81. DOI:10.1038/nature17431 |
| [190] |
M. Yan, Y. Kawamata, P.S. Baran, Chem. Rev. 117 (2017) 13230-13319. DOI:10.1021/acs.chemrev.7b00397 |
| [191] |
S. Tang, Y. Liu, A. Lei, Chem. 4 (2018) 27-45. DOI:10.1016/j.chempr.2017.10.001 |
| [192] |
Z.W. Hou, Z.Y. Mao, Y.Y. Melcamu, et al., Angew. Chem. Int. Ed. 57 (2018) 1636-1639. DOI:10.1002/anie.v57.6 |
| [193] |
M. Movsisyan, E.I.P. Delbeke, J.K.E.T. Berton, et al., Chem. Soc. Rev. 45 (2016) 4892-4928. DOI:10.1039/C5CS00902B |
| [194] |
D. Cambié, C. Bottecchia, N.J.W. Straathof, et al., Chem. Rev. 116 (2016) 10276-10341. DOI:10.1021/acs.chemrev.5b00707 |
| [195] |
B. Gutmann, D. Cantillo, C.O. Kappe, Angew. Chem. Int. Ed. 54 (2015) 6688-6728. DOI:10.1002/anie.201409318 |
| [196] |
J. Li, S.G. Ballmer, E.P. Gillis, et al., Science 347 (2015) 1221-1226. DOI:10.1126/science.aaa5414 |
 2018, Vol. 29
2018, Vol. 29 


