b College of Chemistry, Sichuan University, Chengdu 610064, China
The need for highly sensitive and selective determination of total sulfur in natural gas has never been greater because it poses a potential threat to people's health and cause damage to the expensive catalysts used in many processes of petroleum and natural gas industry even at its low concentrations [1-3]. Many methods have been established for sulfur detection in various samples, including coulometric titration (CT) [4], X-ray fluorescence (XRF) spectrometry [5], ultraviolent (UV) absorption spectrometry, inductively coupled plasma optical emission spectrometry (ICP-OES) [6]. Although these methods retain advantages of high sensitivity and low sample consumption, samples are usually digested one by one in a microwave device or a reaction vessel with high temperature, leading to low sample throughput and consumption of large amounts of chemicals [7-9]. Therefore, many efforts have been devoted to develop simpler techniques to determine total sulfur or sulfur containing compounds in samples. Recently, gas chromatography (GC) equipped with different detectors such as wet electrochemical detector, infrared detector, flame photometric detector (GC-FPD) [10] and sulfur chemiluminescence detector (GC-SCD) [11, 12], was widely used to accomplish the purpose of sulfur determination [13]. Among these GC detectors, SCD originally developed by Benner and Stedman [14] retains several unique advantages: 1) it provides high selectivity over non-sulfur containing compounds and high sensitivity for the determination of sulfur containing species, decreasing limits of detection down to a few picograms of sulfur, 2) it offers nearly sensitivity on a per mole of sulfur basis and not affected the analytes, 3) it retains a linear range with 4 orders of magnitude, 4) it is easy to operate and maintain [15]. Therefore, GC-SCD has rapidly been one of the most used techniques for the determination of sulfur compounds [16, 17].
The measurement principle of the SCD is the detection of the characteristic chemiluminescence (260-480 nm) arising from the reaction between SO and O3. In the available commercial SCD, the efficient conversion of sulfur containing compounds to SO can be accomplished through flame or furnace combustion chamber. In the flame SCD, sulfur compounds are firstly converted to SO in a flame ionization detector by using a hydrogen rich flame and then transported to the SCD reaction chamber wherein O3 is introduced to generate the characteristic chemiluminescence. The furnace SCD is similar to the flame SCD with the exception of the mode of combustion. In this SCD, sulfur containing compounds are converted to SO at 780 ℃ in a combustion device, which is consisted of two concentric ceramic tubes surrounding a heated furnace. A controller is used to maintain hydrogen and air flow rates at their optimal rates, respectively. Although both flame and furnace SCD can efficiently convert sulfur containing compounds to SO, hydrocarbons can be also converted to carbon monoxide (CO) during this conversion of sulfur containing compounds. Unfortunately, CO also produces strong chemiluminescence at the range of 260-480 nm in the presence of O3 and thus serious interference (the "ghost" peak) is inevitable when the analyzed sample contains high concentration of carbon. Therefore, there has been a surge in the development of a novel detector for highly sensitive, selective and interference-free determination of total sulfide in natural gas.
In this work, a novel detection system consisted of a flame ionization detector (FID) and a furnace SCD was developed to couple to GC for the sensitive and interference free determination of total sulfur in natural gas. In contrast to the conventional flame SCD, the product of the combusted sulfur containing compounds is not SO but SO2 in the proposed detection system because an oxygen rich flame was utilized as an alternative of hydrogen rich flame for FID. It is worth to note that the hydrocarbons contained in samples were completely combusted to CO2 in this system. Consequently, both the produced SO2 and CO2 were swept into the furnace SCD wherein SO2 was reduced to SO, whereas CO2 could not be reduced to CO because it is more stable than SO2. Therefore, only SO reacted with O3 and produced its characteristic chemiluminescence and realized highly selective determination of total sulfur in natural gas [18]. The proposed detection system not only retains the advantages of conventional SCD but also significantly reduces the interferences from carbon containing compounds of natural gas [19].
2. Results and discussion 2.1. Preliminary studies and validation of the mechanism of GC-FIDSCDThe obvious interference of hydrocarbons on the determination of total sulfur in natural gas using GC-SCD was previously identified [20]. However, the proposed GC-FID-SCD system is different to the commercial GC-SCD. In order to indentify the interference of hydrocarbons and insight the mechanism of hydrocarbons interferences elimination by the GC-FID-SCD, hydrogen sulfide and high purity methane were selected as representative compounds of sulfur containing compounds and hydrocarbons, respectively. 1 mL of 10 μmol/mol H2S in nitrogen and 1 ml of pure methane were injected into the GC-FID-SCD system and directly analyzed with FID turning on or off, as shown in Fig. S1. (see the Supporting information) Fig. S1a shows both H2S and CH4 produce obvious chemiluminescence responses when the FID was turned off, confirming that hydrocarbons influence the chemiluminescence response of sulfur compounds. Although the response from CH4 is lower than that arising from H2S, the concentration of hydrocarbons in natural gas is much higher than that of total sulfur. Therefore, the interference of hydrocarbons cannot be ignored when GC-SCD was used to determine total sulfur in natural gas. On the contrary, Fig. S1b shows the response from CH4 is disappeared when the oxygen rich flame of FID is ignited. However, the response of H2S has no change and then the interference of hydrocarbons is eliminated under these operation conditions.
On the basis of the previous works [21], the reasons resulted in these phenomenons can be speculated as following: H2S and CH4 were converted to SO and CO in the furnace of SCD with hydrogen rich heating atmosphere, respectively, when the FID was turn off. Thus, SO and CO react with ozone to generate the intermediates of SO2* and CO2*, respectively. These intermediates will emit the chemiluminescence as they return to the ground states, resulting in such interference of hydrocarbons on the determination of total sulfur. When the FID using oxygen rich flame was turned on, H2S and CH4 were firstly combusted to SO2 and CO2. Since it is very stable, CO2 cannot be reduced to CO by H2 in the heated furnace of the SCD and thus cannot produce response in the presence of O3. Compared to CO2, SO2 can be easily reduced to SO by H2 and finally react with O3 for the generation of chemiluminescence. Therefore, the interference arising from hydrocarbons was eliminated when GC-FID-SCD was used to determine the total sulfur in natural gas. To further validate this speculation, 10 μmol/mol of SO2 in nitrogen, pure CO and pure CO2 were successively injected into the GC-FID-SCD for their analysis with FID turning on or off. The results are summarized in Fig. 1. Fig. 1a shows that obvious responses can be detected for both CO and SO2 in the case of turning off FID, whereas only SO2 generated response when FID was turned on (Fig. 1b). Moreover, No response of CO2 was detected regardless of whether the FID was turned on or off. These results confirm the following conclusions: 1) CO can produces chemiluminescence in the presence of O3; 2) CO (equal to hydrocarbons) can be converted to CO2 by FID using oxygen rich flame; 3) CO2 cannot react with O3 for the generation of chemiluminescence and it also cannot be reduced to CO by H2 in the heated furnace of SCD, thus the interferences of coexisting hydrocarbons were eliminated when GC-FID-SCD was used to determine total sulfur in natural gas; 4) although the sulfur containing compounds are converted to SO2 in FID, their responses can be easily detected because SO2 can be reduced to SO in the followed heated chamber of SCD.

|
Download:
|
| Fig. 1. Investigation of the mechanism of GC-FID-SCD. Response of CO, CO2 and SO2 obtained by GC-SCD (a) and by GC-FID-SCD (b). | |
2.2. Optimization of the hydrogen flow rate for SCD
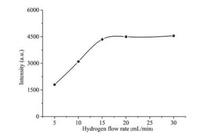
In order to ensure total carbon and sulfur contained in natural gas can be completely combusted to CO2 and SO2, the flow rates of H2 and air used in FID was performed as those recommended by the manufacturer and then fixed at 40 and 400 mL/min, respectively. The flow rate of H2 used in SCD plays a very important role in the proposed analytical system, which acts not only as combustion gas in SCD but also as reductive gas to efficiently reduce the produced SO2 to SO prior to reaction with O3. Therefore, the flow rate of the hydrogen was carefully investigated by injecting 1 mL of 10 μmol/mol H2S and fixing the air flow rate at 5 mL/min. The results are summarized in Fig. 2 and show that response was increased significantly with increasing the flow rate of H2 throughout the range 1-20 mL/min, followed by a plateau at higher concentrations. SO2 cannot be efficiently reduced to SO when the flow rate of H2 is lower than 20 mL/min. Since the high flow rate of H2 can damage the ceramic tube, thus 20 mL/min of H2 was used in subsequent experiments. Other operation parameters are summarized in Table S1. (in Supporting information)
|
Download:
|
| Fig. 2. The effect of hydrogen flow rate on the intensity of GC-SCD. | |
2.3. Effect of sulfur compounds
The previous works [22] demonstrated that SCD can offer nearly sensitivity on a per mole of sulfur basis, which is not only the biggest advantage of SCD but also the basis to ensure successful determination of total sulfur with GC-SCD equipped with a nonstationary phase column. To evaluate the effect of sulfur compounds on the response obtained by the proposed system, several compounds contained equal molar sulfur were analyzed, including hydrogen sulfide, sulfur dioxide, methyl mercaptan, carbon disulfide, dimethyldisulfide, carbonyl sulfide and thiophene, as shown in Fig. S2. (in Supporting information) The results indicate that there are no significant differences ( < 5%) between the responses obtained for these tested sulfur containing compounds, which implies the developed analytical system not only eliminates the interferences of hydrocarbons but also remains the advantages of commercial SCD. Therefore, the developed GC-FIDSCD is more suitable for the determination of total sulfur in natural gas than conventional GC-SCD.
2.4. Figures of merit (Calibration curve and sensitivity)A typical calibration curve was established using the developed GC-FID-SCD under optimal experimental conditions, as shown in Fig. 3. Hydrogen sulfide was used as the test compound to establish the calibration curve. Linear correlation coefficient for this calibration curve is 0.999. The limit of detection (LOD), defined as the analyte concentration equivalent to three standard deviations of 11 measurements of a blank solution, was better than 0.5 μmol/mol for total sulfur based on a 1 mL sample volume. Linear range can be extended to 20 μmol/mol and is limited by saturation of the detector. It is worth to note that the linear range can be changed with changing the negative voltage supplied to the photomultiplier tube of SCD. Table 1 summarizes figures of merit characterizing the current analytical systems and compares its performance with that of similar published analytical methods, which shows the LOD is comparable or better than those obtained by MAS, ICP-OES, XRF, GC-FPD or GC-FPD. Its absolute LOD is much better than all the previously reported results. The reproducibility of this analytical system was demonstrated using 1 mL of a real natural gas and a certified reference material (containing 1 μmol/mol of total sulfur), respectively. Results are summarized in Fig. S3 (in Supporting information) and show that reproducibility (expressed as the relative standard deviations, RSDs) obtained after repeated 7 measurements are 0.9% and 1.0% for the real natural gas and the certified reference material, respectively.

|
Download:
|
| Fig. 3. Calibration curve of total sulfur. | |
|
|
Table 1 Comparison of performance with other methods. |
2.5. Sample analysis
In order to evaluate the accuracy of the proposed analytical system, two real natural gas samples collected from two natural gas companies near our institute were analyzed. Analytical results are summarized in Table 2 and show that good spike recoveries of analytes (96%-98%) were achieved for these samples, confirming the accuracy of the proposed system for the determination of total sulfur in real natural gas samples. The utility of the proposed technique was further demonstrated by the determination of total sulfur in two Certificate Reference Materials (GBW 06332 and GBW (E) 061320). The t-test showed that the analytical results obtained by the proposed method were not significantly different from the certified values at the 95% level of confidence.
|
|
Table 2 Determination of total sulfur in CRM and natural gas. |
3. Conclusion
A simple and novel analytical system coupling a flame ionization detector to a sulfur chemiluminescence detector was developed for the determination of total sulfur in natural gas by GC equipped with a non-stationary phase column. The proposed analytical system showed good analytical performance for the determination of total sulfur in various samples. Compared to conventional techniques, this system not only remains the advantages of conventional GC-SCD but also eliminates the serious interferences arising from the hydrocarbons, extending its promising potential application to petrochemical industry.
4. Experimental 4.1. ReagentsAll pure gas used in this work were of at least 99.999%. The used high purity dry air, hydrogen and nitrogen were obtained from Dongfeng Gas Co. (Chengdu, China). High purity methane (CH4), carbon monoxide (CO), carbon dioxide (CO2), ethylene (C2H6) and propylene (C3H8) were purchased from Institute of Guangming Chemistry (Dalian, China). 10 μmol/mol hydrogen sulfide (H2S) in nitrogen, 10 μmol/mol carbonyl sulfide (COS) in nitrogen, 10 μmol/mol methyl mercaptan in nitrogen, 10 μmol/mol sulfur dioxide (SO2) in nitrogen, 10 μmol/mol carbon disulfide in nitrogen, 10 μmol/mol thiophene in nitrogen, 10 μmol/mol dimethyl disulfide in nitrogen and other chemicals were purchased from National Institute of Measurement and Testing Technology (Chengdu, China). Certified reference materials (GBW 06332 and GBW (E) 061320) and real natural gas were obtained from the National Institute of Measurement and Testing Technology (Chengdu, China) and PetroChina Southwest Oil & Gasfield Company (Chengdu, China), respectively.
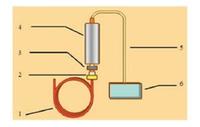
4.2. InstrumentationA schematic diagram of the GC-FID-SCD setup is shown in Fig. 4. An Agilent 355 SCD furnace sulfur chemilumninescence detector (Agilent Technologies, USA) was directly mounted on top of the flame ionization detector of an Agilent 7890B gas chromatograph (Agilent Technologies, USA) for the analysis of total sulfur in natural gas. In common gas chromatographic analysis, a stationary phase column is usually needed to separate the components prior to analysis. However, a non-stationary phase column was used in the current work because our aim was to analyze total sulfur in natural gas. In order to alleviate the adsorption of sulfur containing compounds, all the pressure reducing valves used in this system were treated by Restek Company (USA) using their sulfur deactivation technique.
|
Download:
|
| Fig. 4. Schematic of the GC-FID-SCD. 1. Non-stationary phase column; 2. Flame ionization detector; 3. Temperature controller; 4. Ceramic tube; 5. Transfer tube; 6. Sulfur chemiluminescence detector. | |
4.3. Analysis procedure
The compounds of sulfur in natural gas or in other hydrocarbon gases were injected into the 1 mL sampling loop through a deactivation valve and tube. Then the mixed gas passed through the non-stationary phase column and was further transported to the FID of GC. Sulfur containing compounds and hydrocarbons were burned to SO2, CO2 and H2O, respectively. SO2 together with CO2 and H2O were swept into the furnace SCD and then reduced to SO. Finally, the generated SO reacted with O3 and produced characteristic chemiluminescence.
4.4. Safety considerationsGenerally, sulfur containing compounds are extremely toxic. Moreover, some unknown products may be generated during the whole analytical procedure. Thus, essential safety must be taken into account during all manipulations and then an adequate ventilation/exhaust system must be required.
AcknowledgementWe gratefully acknowledge the National Science and Technology Supporting Plan (No. 2013BAK12B04) for financial support.
Appendix A. Supplementary dataSupplementary data associated with this article can be found, in the online version, at http://dx.doi.org/10.1016/j.cclet.2017.04.014.
| [1] | D.S. Medina, Y.D. Liu, L.M. Wang, J.S. Zhang. Detection of sulfur dioxide by cavity ring-down spectroscopy. Environ. Sci. Technol. 45 (2011) 1926–1931. DOI:10.1021/es103739r |
| [2] | X. Yan. Detection by ozone-induced chemiluminescence in chromatography. J. Chromatogr. A 842 (1999) 267–308. DOI:10.1016/S0021-9673(99)00177-6 |
| [3] | Q.Y. Tan, H.M. Lv, H.M. Liu. Research and evaluation of test methods of trace quantities of sulfur in light liquid petroleum products. China Meas. Technol. 33 (2007) 57–60. |
| [4] | E. Barendrecht, W. Martens. Completely automatic, coulometric titration apparatus for process use. Determination of sulfur dioxide in gases with concentrations ranging from 0.1 to 100% by volume. Anal. Chem 34 (1962) 138–142. DOI:10.1021/ac60181a042 |
| [5] | V. Šprta, B. Knob, P. Janoš. X-ray fluorescence determination of total sulfur in fly ash. Fresenius' J. Anal. Chem. 364 (1999) 705–708. DOI:10.1007/s002160051418 |
| [6] | R.E. Santelli, E.P. Oliveira, M.A. Bezerra, et al., Total sulfur determination in gasoline, kerosene and diesel fuel using inductively coupled plasma optical emission spectrometry after direct sample introduction as detergent emulsions. Spectrochim. Acta Part B 63 (2008) 800–804. DOI:10.1016/j.sab.2008.04.020 |
| [7] | S.J. Christopher, T.W. Vetter. Application of microwave-induced combustion and isotope dilution strategies for quantification of sulfur in coals via sectorfield inductively coupled plasma mass spectrometry. Anal. Chem. 88 (2016) 4635–4643. DOI:10.1021/acs.analchem.5b03981 |
| [8] | C. López García, M. Becchi, M.F. Grenier-Loustalot, O. Païsse, R. Szymanski. Analysis of aromatic sulfur compounds in gas oils using GC with sulfur chemiluminescence detection and high-resolution MS. Anal. Chem. 74 (2002) 3849–3857. DOI:10.1021/ac011190e |
| [9] | D. Tsikas, K. Chobanyan, -Jü rgens. Quantification of carbonate by gas chromatography-mass spectrometry. Anal. Chem. 82 (2010) 7897–7905. DOI:10.1021/ac1007688 |
| [10] | K.A. Goode. Gas chromatographic determination of sulphur compounds in north sea natural gases by a flame-photometric detector. J. Inst. Petrol 56 (1970) 33. |
| [11] | R.X. Hua, Y.Y. Li, W. Liu, et al., Determination of sulfur-containing compounds in diesel oils by comprehensive two-dimensional gas chromatography with a sulfur chemiluminescence detector. J. Chromatogr. A 1019 (2003) 101–109. DOI:10.1016/j.chroma.2003.08.048 |
| [12] | R.L. Shearer. Development of flameless sulfur chemiluminescence detection:application to gas chromatography. Anal. Chem. 64 (1992) 2192–2196. DOI:10.1021/ac00042a030 |
| [13] | T. Nagahata, H. Kajiwara, S. Ohira, K. Toda. Simple field device for measurement of dimethyl sulfide and dimethylsulfoniopropionate in natural waters, based on vapor generation and chemiluminescence detection. Anal. Chem. 85 (2013) 4461–4467. DOI:10.1021/ac303803w |
| [14] | R.L. Benner, D.H. Stedman. Universal sulfur detection by chemiluminescence. Anal. Chem. 61 (1989) 1268–1271. DOI:10.1021/ac00186a018 |
| [15] | R.L. Benner, D.H. Stedman. Field evaluation of the sulfur chemiluminescence detector. Environ. Sci. Technol. 24 (1990) 1592–1596. DOI:10.1021/es00080a021 |
| [16] | P.L. Burrow, J.W. Birks. Flow tube kinetics investigation of the mechanism of detection in the sulfur chemiluminescence detector. Anal. Chem. 69 (1997) 1299–1306. DOI:10.1021/ac960937b |
| [17] | M. Li, D.X. Yuan, Q.L. Li, X.Y. Jin. Sequential analysis of dimethyl sulfur compounds in seawater. Chin. Chem. Lett. 18 (2007) 99–102. DOI:10.1016/j.cclet.2006.11.005 |
| [18] | H.C.K. Chang, L.T. Taylor. Sulfur-selective chemiluminescence detection after packed-capillary-column high-performance liquid chromatography. Anal. Chem. 63 (1991) 486–490. DOI:10.1021/ac00005a020 |
| [19] | S. Chen, Z.J. Chen, W. Ren, H.W. Ai. Reaction-based genetically encoded fluorescent hydrogen sulfide sensors. J. Am. Chem. Soc. 134 (2012) 9589–9592. DOI:10.1021/ja303261d |
| [20] | T. Matsukami, A. Machino, H. Sugimoto. Determination of the total sulfur content using a sulfur chemiluminescence detector. Anal. Sci. 10 (1994) 453–456. DOI:10.2116/analsci.10.453 |
| [21] | M.L. Yuan, T.Y. Jiang, L.P. Du, M.Y. Li. Luminescence of coelenterazine derivatives with C-8 extended electronic conjugation. Chin. Chem. Lett. 27 (2016) 550–554. DOI:10.1016/j.cclet.2016.02.011 |
| [22] | H. Shi, L.T. Taylor, E.M. Fujinari, X. Yan. Sulfur-selective chemiluminescence detection with packed column supercritical fluid chromatography. J. Chromatogr. A 779 (1997) 307–313. DOI:10.1016/S0021-9673(97)00455-X |
| [23] | M.D. Huang, H. Becker, - Ross, S. Florek, et al., Determination of sulfur forms in wine including free and total sulfur dioxide based on molecular absorption of carbon monosulfide in the air-acetylene flame. Anal. Bioanal. Chem. 390 (2008) 361–367. DOI:10.1007/s00216-007-1669-1 |
| [24] | V. Vitvitsky, R. Banerjee. H2S analysis in biological samples using gas chromatography with sulfur chemiluminescence detection. Methods Enzymol. 554 (2015) 111–123. DOI:10.1016/bs.mie.2014.11.013 |
| [25] | D.A. Clay, C.H. Rogers, R.H. Jungers. Determination of total sulfur in gasoline by gas chromatography with a flame photometric detector. Anal. Chem. 49 (1977) 126–128. DOI:10.1021/ac50009a039 |
| [26] | H.J. Rath, J. Wimmer. Gas chromatographic analysis of traces of hydrogen sulfide in hydrogen. Chromatography 13 (1980) 513–514. DOI:10.1007/BF02261623 |
 2017, Vol. 28
2017, Vol. 28