Coumarins, a class of compounds that contains a 1, 2-benzopyrone skeleton, constitute a major class of widely distributed O-heterocyclic natural products isolated from many fruits, vegetables, spices and medicinal plants [1]. More than 1300 coumarins have been identified as secondary metabolites from plants, bacteria, and fungi [2]. Naturally occurring coumarins possess a variety of biological activity, including antitumor activity [3], anti-inflammatory [4], anti-fungal activity [5], antihypertensive activity [6-8]. In addition, the intrinsic photophysical characteristics of coumarins and derivatives enable their use as biological and chemical probes, which are explored in the detection of ions [9], anions [10], proteins [11], DNA/RNA [12, 13] and being useful pH indicators [14]. Pyran is one of the most remarkable and important heterocyclic scaffolds, particularly 4H-pyrans have been used as valuable intermediates in modern organic synthesis due to the existence of the suitable functional groups of natural products [15]. It also possesses wide range of pharmacological activities and act as vasorelaxant [16], antitumor [17], antibacterial [18], antimicrobial [19] and anti-inflammatory agents [20].
One of the desirable goals in organic chemistry is assembly of simple and readily available precursor molecules into complex products in facile execution. Multicomponent reactions (MCRs) have gained considerable attention not only due to MCRs enable expedient and efficient assembly of molecules of structural complexity and diversity in one-pot operations in facile execution, but also with high atom economy, and selectivity. Multicomponent reactions (MCRs) have facilitated many fascinating and challenging transformations in organic synthesis [21]. These reactions cast off the process of isolation and purification of intermediates and decrease waste generation, therefore enhancing the greenness of transformations. MCRs have been emerging as powerful means in the synthesis of complex biologically and medicinally important compounds. In the course of our study on multicomponent reactions, we successfully finished synthesis of diversely substituted heterocyclic compounds [22, 23].
Cetyltrimethylammonium chloride (CTAC) is a kind of cationic surfactant and antiseptic, and also commonly used in shampoos and hair conditioners [24]. Commercially available surfactants (Fig. 1) can be used to promote multicomponent reactions, some traditional multicomponent reactions can be easily run to afford good to excellent yields with the addition of cationic surfactant, such as CTAC [25], CTAB (hexadecyl trimethylammonium bromide) [26], TEBA (triethylbenzyl ammonium chloride) [27], HDBAC (hexadecyl dimethyl benzyl ammonium chloride) [28].

|
Download:
|
| Figure 1. Structures of commercially available surfactants used in this study. | |
As far as we know coumarins were already used previously as constituents of MCRs [29-31]. To the best of our knowledge, however, no protocol access to fluorescent coumarin-4H-pyran conjugates has been reported yet. Accordingly, the aim of this work is to build a protocol for achieving a novel collection of coumarin-4H-pyran conjugates from synthetic coumarin β-ketoester derivatives in environmentally friendly way.
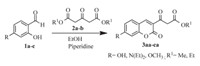
2. Results and discussionEncouraged by previous reports [32, 33], our initial investigation was focused on preparing various coumarin β-ketoester derivatives. These coumarin β-ketoester derivatives were synthesized successfully by using 4-hydroxysalicyl-aldehyde 1a, 4-diethylaminosalicylaldehyde 1b or 4-methoxysalicylaldehyde 1c and dimethyl 3-oxopentanedioate 2a or diethyl 3-oxopentanedioate 2b (Scheme 1).
|
Download:
|
| Scheme 1. Preparation of coumarin β-ketoester moieties. | |
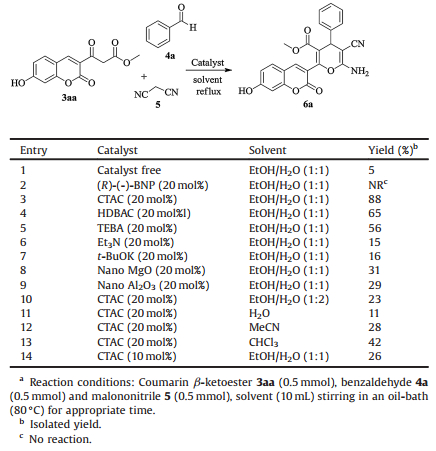
With the coumarin β-ketoesters in hand, we chose coumarin β-ketoester 3aa, benzaldehyde 4a and malononitrile 5 as model substrates to optimize the reaction conditions. At first, the model reaction was carried out without any catalyst in a mixture of EtOH and H2O (1:1), it was found that only a low yield of product was isolated after 24 h (entry 1, Table 1). This result suggests that a sort of catalyst is indispensable in this reaction. Based on the result, we decided to investigate the best reaction conditions for obtaining the desired product 6a by using various catalysts. In comparison with these catalysts, including (R)-(-)-BNP (R-(-)-1, 1'-binaphthyl-2, 2'-diylhydrogenphosphate), CTAC, HDBAC, TEBA, Nano MgO, Nano Al2O3, CTAC has proved to be the most efficient catalyst for the model reaction (entry 3, Table 1). In addition, when the catalytic amount of CTAC was 20 mol% we achieved the most excellent yield of product. However, when the catalytic amount of CATC was 10 mol%, the yield of product decreased drastically to 26% (entry 14, Table 1).
|
|
Table 1 Optimization of reaction conditions for the multicomponent reactions.a |
The use of water as a solvent for organic reactions offers several "green chemistry" benefits [34], thus we tested water and EtOH-H2O (1:2) mixture for the solvent of the model reaction, however, relatively lower yields were observed (entries 10 and 11, Table 1). On the other hand, EtOH-H2O (1:1) mixture proved its convenience as the solvent in the workup process rather than H2O did. Several other solvents including MeCN, CHCl3 were screened but inferior yields were obtained (entries 12 and 13, Table 1).
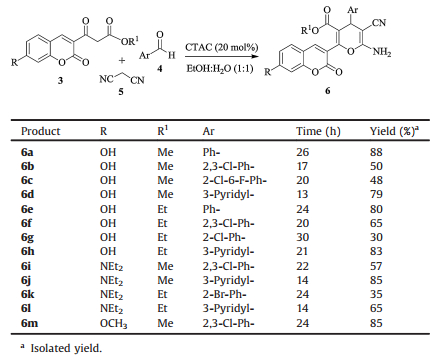
To demonstrate the generality and scope of this novel efficient strategy, synthesis of coumarin-4H-pyran conjugates was extended to various substituted aldehydes and coumarin β-ketoesters and from low to excellent yields were obtained (Table 2). The work-up procedure of the reaction is very convenient, after completion the reaction (monitored by TLC), the product was isolated from the reaction mixture simply by filtration and pure target compounds were obtained in considerably high yields after further recrystallization.
|
|
Table 2 Synthesis of coumarin-4H-pyran conjugates using CTAC as the catalyst. |
The 6-amino-5-cyano-4H-pyran ring formation of compounds 6a-6m was characterized by 1H NMR spectra exhibiting a typical singlet signal in a range of 4.02-5.16 ppm (s, 1H) for the CH originated from the aldehyde carbonyl moiety. The amino hydrogens of 4H-pyran could also be identified as a singlet ranging from 7.04 to 7.44 ppm. The proton at C-4 position of coumarin ring appeared as a singlet at 8.12-8.34 ppm.
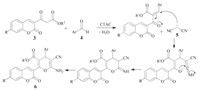
The plausible reaction mechanism that accounts for the multicomponent reaction is shown in Scheme 2. The effect of CTAC on this reaction can hardly be ascribed to the micellar system catalysis, because the concentration of CTAC (10 mmol/L) in the reaction solution is lower than the critical micelle concentration (125 mmol/L) [35] in a equal volume mixture of EtOH and H2O. Therefore, as in a great deal of classical multicomponent reactions, the mechanism is conceivable that CTAC with weak base character is an effective catalyst for aryl aldehyde undergoing Knoevenagel condensation with coumarin β-ketoester to give intermediate product, which acts as a Michael acceptor. The carbanion of malononitrile is generated by the abstraction of a proton from the active methylene group, which is stabilized by resonance. Then it attacks at the Knoevenagel adduct intermediate in Michael addition manner to produce an acyclic intermediate which might undergo intramolecular cyclization to give the desired coumarin-4H-pyran conjugate.
|
Download:
|
| Scheme 2. Plausible reaction mechanism for the formation of the coumarin-4H-pyran conjugates. | |
3. Conclusion
In summary, we have established an efficient, facile, ecofriendly, one-pot, three-component protocol for the synthesis of coumarin-4H-pyran conjugates in aqueous EtOH. The synthetic importance of coumarin-4H-pyran conjugates may be related to medicinal chemistry, since the products share structural and functional group properties of the biologically active molecules. The present methodology with mild reaction conditions, using of inexpensive and nontoxic commercially available cationic surfactant CTAC as a catalyst, operational simplicity and purification of the compounds by non-chromatographic methods offers the possibility of its use with cost-effective and environmentally friendlier ways for large-scale industrial syntheses as well.
4. Experimental 4.1. General informationReagents and solvents were from Aladdin, Acros or Energy Chemical. The progress of the reaction was monitored by TLC using analytical-grade silica gel plates (GF254). 1H NMR (400 MHz) and 13C NMR (100 MHz) were run on a Bruker Inova-400 instrument using DMSO-d6 as a solvent. Chemical shifts are given in ppm relative to TMS as an internal standard, and the coupling constants J values are given in Hz. IR spectra were recorded on a Bruker Vertex70 spectrophotometer using samples as KBr pellets. HRMS data were obtained using Thermo Scientific LTQ Orbitrap XL spectrometer. All the spectra of compounds 3 and 6 are deposited in Supporting information.
4.2. General procedure for the synthesis of compounds 3aa-3caTo a stirred solution of the appropriate β-ketoester 2 (10 mmol) and the corresponding salicylaldehyde 1 (10 mmol) in EtOH was added piperidine (2.5 mmol) dropwise. The mixture was stirred at room temperature for 24 h, and after completion of the reaction (monitored by TLC), the precipitate was filtered and washed with cold EtOH to afford the desired compound.
3aa: Yield 57%, bright yellow solid; 1H NMR (400 MHz, DMSOd6): δ 11.20 (s, 1H), 8.70 (s, 1H), 7.82 (d, 1H, J = 8.4 Hz), 6.88-6.86 (dd, 1H, J = 2, 8.4 Hz), 6.76 (d, 1H, J = 2.4 Hz), 4.01 (s, 2H), 3.62 (s, 3H).
3ab: Yield 65%, yellow solid; 1H NMR (400 MHz, DMSO-d6): δ 11.22 (s, 1H), 8.70 (s, 1H), 7.83 (d, 1H, J = 8.8 Hz), 6.89-8.86 (s, 1H), 6.77 (d, 1H, J = 2.4 Hz), 4.12-4.06 (m, 2H), 3.99 (s, 2H), 1.18-1.15 (m, 3H).
3ba: Yield 85%, yellow solid; 1H NMR (400 MHz, DMSO-d6): δ 8.50 (s, 1H), 7.42 (d, 1H, J = 9.2 Hz), 6.65-6.60 (m, 1H), 6.46 (d, 1H, J = 2.4 Hz), 4.12 (s, 2H), 3.75 (s, 3H), 3.50-3.44 (q, 4H), 1.27-1.23 (t, 6H, J = 7.2 Hz).
3bb: Yield 76%, deep yellow solid; 1H NMR (400 MHz, DMSOd6): δ 8.50 (s, 1H), 7.42 (d, 1H, J = 9.2 Hz), 6.65-6.62 (dd, 1H, J = 2.4, 8.8 Hz), 6.46 (d, 1H, 2.4 Hz), 4.24-4.18 (q, 2H), 3.50-3.44 (q, 4H), 1.32-1.22 (m, 6H).
3ca: Yield 70%, yellowish solid; 1H NMR (400 MHz, DMSO-d6): δ 8.75 (s, 1H), 7.93-7.88 (dd, 1H, J = 8.8, 16.8 Hz), 7.10-7.06 (m, 2H), 4.04 (s, 2H), 3.91 (s, 3H), 3.63 (s, 3H).
4.3. General procedure for the synthesis of compounds 6a-6mA mixture of coumarin β-ketoester 3 (0.5 mmol), the corresponding aldehyde 4 (0.5 mmol), malononitrile 5 (0.5 mmol) and CTAC (20 mol%) in EtOH-H2O (1:1, 10 mL) was taken in a 25 mL round-bottomed flask connected to a reflux condenser, and the mixture was stirred in an oil-bath (80 ℃). The progress of the reaction was monitored by TLC using acetone-chloroform (1:5) as an eluent. After completion of the reaction, the reaction mixture was cooled to room temperature. Then the precipitated product was filtered and washed with cold EtOH and pure product was obtained after further recrystallization from EtOH.
Methyl 6-amino-5-cyano-2-(7-hydroxy-2-oxo-2H-chromen-3-yl)-4-phenyl-4H-pyran-3-carboxylate (6a): Mp: 183-185 ℃; IR (KBr, cm-1): 3482, 3363, 3207, 2184, 1700, 1672, 1595, 1455, 1380, 1217; 1H NMR (400 MHz, DMSO-d6): δ 10.89 (d, 1H, J = 4.8 Hz), 8.28 (d, 1H, J = 4.8 Hz), 7.63 (d, 1H, J = 8.4 Hz), 7.34 (q, 2H), 7.28-7.24 (m, 3H), 7.05 (s, 2H), 6.88-6.85 (m, 1H), 6.79 (d, 1H, J = 2.4 Hz), 4.43 (s, 1H), 3.47-3.39 (m, 3H); 13C NMR (100 MHz, DMSO-d6): δ 165.3, 162.8, 159.2, 158.4, 155.7, 150.0, 144.3, 143.9, 130.8, 128.8, 127.4, 127.3, 127.2, 119.7, 117.1, 114.1, 111.0, 110.8, 102.3, 57.6, 56.2, 51.9, 18.7; HRMS (ESI) m/z, calcd. for C23H17N2O6 [M+H]+ 417.1081, found 417.1080.
Methyl 6-amino-5-cyano-4-(2, 3-dichlorophenyl)-2-(7-hydroxy-2-oxo-2H-chromen-3-yl)-4H-pyran-3-carboxylate (6b): Mp: 201-203 ℃; IR (KBr, cm-1): 3464, 3326, 2198, 1704, 1613, 1566, 1266, 1212; 1H NMR (400 MHz, DMSO-d6):δ 10.90 (s, 1H), 8.28 (s, 1H), 7.63 (d, 1H, J = 8 Hz), 7.55 (s, 1H), 7.38 (s, 2H), 7.17 (s, 2H), 6.86 (s, 1H), 6.79 (s, 1H), 5.08 (s, 1H), 3.35 (s, 3H); 13C NMR (100 MHz, DMSO-d6): δ 165.0, 162.8, 159.5, 158.4, 155.7, 151.0, 144.2, 144.1, 132.1, 130.9, 130.3, 129.5, 128.9, 128.7, 119.1, 116.9, 114.2, 110.7, 109.6, 102.4, 55.8, 52.0, 37.2; HRMS (ESI) m/z, calcd. for C23H15Cl2N2O6 [M+H]+ 485.0302, found 485.0301.
Methyl 6-amino-4-(2-chloro-6-fluorophenyl)-5-cyano-2-(7-hydroxy-2-oxo-2H-chromen-3-yl)-4H-pyran-3-carboxylate (6c): Mp: 193-195 ℃; IR (KBr, cm-1): 3324, 2190, 1719, 1617, 1572, 1215; 1H NMR (400 MHz, DMSO-d6): δ 10.88 (s, 1H), 8.16 (s, 1H), 7.66 (d, 1H, J = 8.4 Hz), 7.36 (s, 2H), 7.24-7.18 (m, 3H), 6.85 (d, 1H, J = 8.4 Hz), 6.77 (s, 1H), 5.16 (s, 1H), 3.46-3.35 (m, 3H); 13C NMR (100 MHz, DMSO-d6): δ 165.1, 162.8, 160.0, 158.1, 155.7, 151.0, 143.7, 133.9, 131.0, 130.0, 129.9, 126.2, 119.2, 117.0, 115.5, 114.1, 110.7, 107.6, 102.3, 56.2, 53.3, 51.8, 18.7; HRMS (ESI) m/z, calcd. for C23H15ClFN2O6 [M+H]+ 469.0597, found 469.0596.
Methyl 6-amino-5-cyano-2-(7-hydroxy-2-oxo-2H-chromen-3-yl)-4-(pyridin-3-yl)-4H-pyran-3-carboxylate (6d): Mp: 238-240 ℃; IR (KBr, cm-1): 3350, 2999, 2191, 1720, 1668, 1563, 1211; 1H NMR (400 MHz, DMSO-d6): δ 10.90 (s, 1H), 8.51-8.47 (m, 2H), 8.29 (s, 1H), 7.70-7.67 (m, 1H), 7.62 (d, 1H, J = 8.4 Hz), 7.42-7.39 (dd, 1H, J = 8.4, 8 Hz), 7.17 (s, 2H), 6.88-6.85 (dd, 1H, J = 2, 8.4 Hz), 6.79 (d, 1H, J = 2 Hz), 4.50 (s, 1H), 3.37 (s, 3H); 13C NMR (100 MHz, DMSOd6): δ 165.1, 162.8, 159.4, 158.5, 155.7, 150.7, 148.7, 148.5, 144.1, 139.9, 134.9, 130.9, 124.1, 119.5, 117.0, 114.2, 110.7, 109.9, 102.4, 56.8, 52.0, 37.0; HRMS (ESI) m/z, calcd. for C22H16N3O6 [M+H]+ 418.1034, found 418.1034.
Ethyl 6-amino-5-cyano-2-(7-hydroxy-2-oxo-2H-chromen-3-yl)-4-phenyl-4H-pyran-3-carboxylate (6e): Mp: 204-206 ℃; IR (KBr, cm-1):3477, 3356, 3179, 2986, 2900, 2181, 1925, 1887, 1721, 1671, 1639, 1594, 1565, 1455, 1215; 1H NMR (400 MHz, DMSO-d6): δ 10.87 (s, 1H), 8.25 (s, 1H), 7.63-7.61 (d, 1H, J = 8.8 Hz), 7.28-7.23 (m, 5H), 7.04 (s, 2H), 6.87-6.84 (dd, 1H, J = 2, 8.4 Hz), 6.79-6.78 (d, 1H, J = 2 Hz), 4.42 (s, 1H), 3.87-3.78 (m, 2H), 0.85-0.81 (t, 3H, J = 7.2 Hz); 13C NMR (100 MHz, DMSO-d6): δ 164.7, 162.7, 159.2, 158.5, 155.6, 150.0, 144.4, 143.9, 130.8, 128.8, 127.4, 127.2, 119.8, 117.4, 114.1, 111.1, 110.7, 102.3, 60.6, 57.5, 13.6; HRMS (ESI) m/z, calcd. for C24H19N2O6 [M+H]+ 431.1238, found 431.1235.
Ethyl 6-amino-5-cyano-4-(2, 3-dichlorophenyl)-2-(7-hydroxy-2-oxo-2H-chromen-3-yl)-4H-pyran-3-carboxylate (6f): IR (KBr, cm-1): 3324, 2191, 1712, 1621, 1220; 1H NMR (400 MHz, DMSOd6): δ 10.88 (s, 1H), 8.26 (s, 1H), 7.63 (d, 1H, J = 8.4 Hz), 7.56-7.54 (dd, 1H, J = 2.6, 6.8 Hz), 7.41-7.37 (m, 2H), 7.16 (s, 2H), 6.88-6.85 (m, 1H), 6.80 (d, 1H, J = 2 Hz), 5.09 (s, 1H), 3.85-3.82 (m, 2H), 0.82-0.78 (t, 3H, J = 7.2 Hz); 13C NMR (100 MHz, DMSO-d6): δ 164.3, 162.8, 159.3, 158.5, 155.7, 151.2, 144.3, 143.9, 132.1, 130.8, 130.4, 129.4, 128.9, 128.7, 119.1, 117.2, 114.1, 110.7, 109.7, 102.3, 60.6, 56.2, 55.7, 18.7, 13.5; HRMS (ESI) m/z, calcd. for C24H17Cl2N2O6 [M+H]+ 499.0458, found 499.0458.
Ethyl 6-amino-4-(2-chlorophenyl)-5-cyano-2-(7-hydroxy-2-oxo-2H-chromen-3-yl)-4H-pyran-3-carboxylate (6g):IR (KBr, cm-1): 3321, 2983, 2191, 1712, 1618, 1219; 1H NMR (400 MHz, DMSO-d6):δ 10.87 (s, 1H), 8.26 (s, 1H), 7.63 (d, 1H, J = 8.8 Hz), 7.44-7.34 (m, 3H), 7.29-7.25 (m, 1H), 7.08 (s, 2H), 6.87-6.85 (dd, 1H, J = 2.4, 8.4 Hz), 6.79 (d, 1H, J = 2.4 Hz), 5.01 (s, 1H), 3.84-3.34 (m, 2H), 0.83-0.80 (t, 3H, J = 7.2 Hz); 13C NMR (100 MHz, DMSO-d6): δ 164.4, 162.7, 159.3, 158.5, 155.7, 150.9, 143.8, 141.6, 132.2, 130.8, 130.1, 129.6, 128.9, 128.1, 119.2, 117.3, 114.1, 110.7, 110.1, 102.3, 60.6, 56.2, 13.5; HRMS (ESI) m/z, calcd. for C24H18ClN2O6 [M+H]+ 465.0848, found 465.0846.
Ethyl 6-amino-5-cyano-2-(7-hydroxy-2-oxo-2H-chromen-3-yl)-4-(pyridin-3-yl)-4H-pyran-3-carboxylate (6h): IR (KBr, cm-1): 3377, 2989, 2191, 1721, 1614, 1565, 1212; 1H NMR (400 MHz, DMSO-d6): δ 10.89 (s, 1H), 8.51 (d, 1H, J = 2.4 Hz), 8.48-8.47 (dd, 1H, J = 1.6, 4.8 Hz), 8.32 (s, 1H), 7.70-7.68 (m, 1H), 7.68-7.60 (m, 1H), 7.43-7.39 (dd, 1H, J = 4.8, 7.6 Hz), 7.16 (s, 2H), 6.88-6.85 (m, 1H), 6.79 (d, 1H, J = 2 Hz), 4.50 (s, 1H), 3.85-3.82 (m, 2H), 0.84-0.81 (t, 3H, J = 7.2 Hz); 13C NMR (100 MHz, DMSO-d6): δ 164.5, 162.8, 159.3, 158.5, 155.7, 150.8, 148.7, 148.4, 144.0, 140.0, 135.1, 130.8, 124.1, 119.5, 117.2, 114.2, 110.7, 110.0, 102.3, 79.4, 60.7, 56.7, 37.1, 13.6; HRMS (ESI) m/z, calcd. for C23H18N3O6 [M+H]+ 432.1190, found 432.1188.
Methyl 6-amino-5-cyano-4-(2, 3-dichlorophenyl)-2-(7-(diethylamino)-2-oxo-2H-chromen-3-yl)-4H-pyran-3-carboxylate (6i): IR (KBr, cm-1): 2972, 1721, 1617, 1572, 1505, 1350; 1H NMR (400 MHz, DMSO-d6): δ 8.12 (s, 1H), 7.56-7.50 (m, 2H), 7.40-7.33 (m, 2H), 7.14 (s, 2H), 6.78-6.75 (dd, 1H, J = 2, 8.8 Hz), 6.59 (d, 1H, J = 2 Hz), 5.05 (s, 1H), 3.49-3.37 (m, 4H), 2.08 (s, 3H), 1.15-1.12 (t, 6H, J = 7.2 Hz); 13C NMR (100 MHz, DMSO-d6): δ 165.3, 159.6, 158.9, 156.7, 151.8, 151.4, 144.4, 143.9, 132.1, 130.5, 130.2, 129.4, 128.8, 128.7, 119.2, 113.0, 109.8, 109.0, 107.1, 96.5, 55.8, 51.8, 44.4, 37.3, 12.5; HRMS (ESI) m/z, calcd. for C27H24Cl2N3O5 [M+H]+ 540.1088, found 540.1087.
Methyl 6-amino-5-cyano-2-(7-(diethylamino)-2-oxo-2H-chromen-3-yl)-4-(pyridin-3-yl)-4H-pyran-3-carboxylate (6j): IR (KBr, cm-1): 3342, 2978, 2189, 1727, 1617, 1579, 1410, 1349; 1H NMR (400 MHz, DMSO-d6): δ 8.50-8.46 (m, 2H), 8.13 (s, 1H), 7.68-7.66 (m, 1H), 7.50 (d, 1H, J = 9.2 Hz), 7.42-7.36 (dd, 1H, J = 4.8, 4.8 Hz), 7.14 (s, 2H), 6.78-6.75 (dd, 1H, J = 2.4, 2.4 Hz), 6.60 (d, 1H, J = 2 Hz), 4.47 (s, 1H), 3.49-3.40 (m, 4H), 3.34 (s, 3H), 1.16-1.12 (t, 6H, J = 7.2 Hz); 13C NMR (100 MHz, DMSO-d6): δ 165.4, 159.5, 158.9, 156.6, 151.8, 151.1, 148.6, 148.4, 144.0, 140.0, 134.9, 130.5, 124.1, 119.6, 113.0, 109.8, 109.1, 107.1, 96.5, 56.8, 51.8, 44.4, 37.2, 12.5; HRMS (ESI) m/z, calcd. for C26H25N4O5 [M+H]+ 473.1820, found 473.1816.
Ethyl 6-amino-4-(2-bromophenyl)-5-cyano-2-(7-(diethyl amino)-2-oxo-2H-chromen-3-yl)-4H-pyran-3-carboxylate (6k): IR (KBr, cm-1): 3396, 2979, 2201, 1714, 1616, 1589, 1347; 1H NMR (400 MHz, DMSO-d6):δ 8.11 (s, 1H), 7.58 (d, 1H, J = 8 Hz), 7.51 (d, 1H, J = 8.8 Hz), 7.42-7.34 (m, 2H), 7.20-7.16 (m, 1H), 7.05 (s, 2H), 6.77-6.74 (dd, 1H, J = 2, 1.6 Hz), 6.59 (d, 1H, J = 2 Hz), 5.00 (s, 1H), 3.83-3.78 (m, 2H), 3.51-3.35 (m, 4H), 1.24-1.04 (m, 6H), 0.86-0.83 (t, 3H, J = 7.2 Hz); 13C NMR (100 MHz, DMSO-d6): δ 164.6, 159.3, 158.9, 156.6, 151.7, 151.2, 143.7, 143.5, 132.8, 130.4, 130.1, 129.1, 128.7, 122.9, 119.2, 113.5, 109.8, 109.7, 107.1, 96.5, 60.4, 56.4, 56.2, 44.3, 38.5, 18.7, 13.7, 12.5; HRMS (ESI) m/z, calcd. for C28H27BrN3O5 [M+H]+ 564.1129, found 564.1122.
Ethyl 6-amino-5-cyano-2-(7-(diethylamino)-2-oxo-2H-chromen-3-yl)-4-(pyridin-3-yl)-4H-pyran-3-carboxylate (6l): Mp: 193-195 ℃; IR (KBr, cm-1): 3361, 2978, 2186, 1722, 1616, 1583, 1513, 1410, 1345; 1H NMR (400 MHz, DMSO-d6): δ 8.50 (d, 1H, J = 2 Hz), 8.48-8.460 (dd, 1H, J = 1.6, 4.8 Hz), 8.12 (s, 1H), 7.68-7.66 (dd, 1H, J = 2, 6 Hz), 7.50 (d, 1H, J = 8.8 Hz), 7.42-7.38 (dd, 1H, J = 4.8, 8 Hz), 7.14 (s, 2H), 6.77-6.74 (dd, 1H, J = 2.4, 8.8 Hz), 6.60 (d, 1H, J = 2.4 Hz), 4.47 (s, 1H), 3.88-3.81 (m, 2H), 3.48-3.43 (m, 4H), 1.15-1.12 (t, 6H, J = 7.2 Hz), 0.88-0.84 (t, 3H, J = 7.2 Hz); 13C NMR (100 MHz, DMSO-d6): δ 164.7, 159.4, 158.9, 156.5, 151.7, 151.1, 148.6, 148.2, 143.7, 139.9, 134.8, 130.2, 123.9, 119.4, 113.3, 109.6, 109.4, 108.9, 107.0, 96.4, 60.3, 56.8, 44.2, 37.1, 13.5, 12.4; HRMS (ESI) m/z, calcd. for C27H27Cl2N4O5 [M+H]+ 487.1976, found 487.1972.
Methyl 6-amino-5-cyano-4-(2, 3-dichlorophenyl)-2-(7-methoxy-2-oxo-2H-chromen-3-yl)-4H-pyran-3-carboxylate (6m): Mp: 216-218 ℃; IR (KBr, cm-1): 3458, 3318, 2198, 1732, 1608, 1553, 1423, 1368; 1H NMR (400 MHz, DMSO-d6): δ 8.34 (s, 1H), 7.74-7.72 (d, 1H, J = 8.8 Hz), 7.57-7.55 (dd, 1H, J = 2.4, 2.8 Hz), 7.42-7.36 (m, 2H), 7.19 (s, 2H), 7.11 (d, 1H, J = 2.4 Hz), 7.05-7.02 (dd, 1H, J = 2.8, 2.4 Hz), 5.09 (s, 1H), 3.89 (s, 3H), 3.45-3.37 (t, 3H, J = 7.2 Hz); 13C NMR (100 MHz, DMSO-d6): δ 164.9, 163.8, 159.4, 158.3, 155.6, 150.9, 144.1, 143.8, 132.2, 130.5, 130.3, 129.5, 128.9, 128.7, 119.0, 118.0, 113.5, 111.7, 109.7, 100.9, 56.3, 56.2, 55.8, 52.0, 37.2, 18.7; HRMS (ESI) m/z, calcd. for C24H17Cl2N2O6 [M+H]+ 499.0458, found 499.0457.
AcknowledgmentWe are grateful for the financial support from the National Natural Science Foundation of China (No. 21462041), NaturalScience Foundation for Distinguished Young Scholars of Xinjiang Uyghur Autonomous Region (No. Qn2015jq002), and the Tianshan Talent Cultivation Project of Xinjiang Uyghur Autonomous Region.
Appendix A. Supplementary dataSupplementary data associated with this article can be found, in the online version, at http://dx.doi.org/10.1016/j.cclet.2016.12.016.
| [1] | M.R. Abenavoli, C.D. Santis, M. Sidari, et al., Influence of coumarin on the net nitrate uptake in durum wheat. New Phytol. (2001) 619–627. |
| [2] | C.H. Hwang, B.U. Jaki, L.L. Klein, et al., Chlorinated coumarins from the polypore mushroom Fomitopsis officinalis and their activity against Mycobacterium tuberculosis. J. Nat. Prod. (2013) 1916–1922. |
| [3] | Y. Li, T. Zhang, S.J. Schwartz, D. Sun. New developments in Hsp90 inhibitors as anti-cancer therapeutics:mechanisms. clinical perspective and more potential. Drug Resist. Update (2009) 17–27. |
| [4] | K.C. Fylaktakidou, H.L.D. Ke, D.N. Nicolaides. Natural and synthetic coumarin derivatives with anti-inflammatory/antioxidant activities. Curr. Pharm. Des. (2004) 3813–3833. |
| [5] | S. Sardari, Y. Mori, K. Horita, et al., Synthesis and antifungal activity of coumarins and angular furanocoumarins. Bioorg. Med. Chem. (1999) 1933–1940. |
| [6] | K.M. Amin, F.M. Awadalla, A.A.M. Eissa, S.M. Abou-Seri, G.S. Hassan. Design. synthesis and vasorelaxant evaluation of novel coumarin-pyrimidine. Bioorg. Med. Chem. (2011) 6087–6097. |
| [7] | S. Rost, A. Fregin, V. Ivaskevicius, et al., Mutations in VKORC1 cause warfarin resistance and multiple coagulation factor deficiency type 2. Nature (2004) 537–541. |
| [8] | T. Wang, C. Wang, N. Zhou, X. Pan, H. He. Synthesis and vasorelaxation evaluation of novel biphenyl-furocoumarin derivatives. Med. Chem. Res. (2015) 2417–2431. |
| [9] | V.K. Gupta, N. Mergu, L.K. Kumawat, A.K. Singh. Selective naked-eye detection of magnesium (Ⅱ) ions using a coumarin-derived fluorescent probe. Sens. Actuators B:Chem. (2015) 216–223. |
| [10] | M. Yoo, S. Park, H.J. Kim. Highly selective detection of cyanide by 2-hydroxyphenylsalicylimine of latent fluorescence through the cyanidecatalyzed imine-to-oxazole transformation. Sens. Actuators B:Chem. (2015) 788–793. |
| [11] | H. Wang, L.E. Guo, X.M. Li, et al., Coumarin-based turn-on fluorescence probes for highly selective detection of Pi in cell culture and Caenorhabditis elegans. Dyes Pigments (2015) 293–298. |
| [12] | D. Ye, L. Wang, H. Li, J. Zhou, D. Cao. Synthesis of coumarin-containing conjugated polymer for naked-eye detection of DNA and cellular imaging. Sens. Actuators B:Chem. (2013) 234–243. |
| [13] | T. Sarwar, S.U. Rehman, M.A. Husain, H.M. Ishqi, M. Tabish. Interaction of coumarin with calf thymus DNA:deciphering the mode of binding by in vitro studies. Int. J. Biol. Macromol. (2015) 9–16. |
| [14] | S. Zhu, W. Lin, L. Yuan. Development of a ratiometric fluorescent pH probe for cell imaging based on a coumarin-quinoline platform. Dyes Pigments (2013) 465–471. |
| [15] | Y. Gu. Multicomponent reactions in unconventional solvents:state of the art. Green Chem. (2012) 2091–2128. |
| [16] | F. Hernández, A. Sánchez, P. Rendón-Vallejo, et al., Synthesis. ex vivo and in silico studies of 3-cyano-2-pyridone derivatives with vasorelaxant activity. Eur. J. Med. Chem. (2013) 669–676. |
| [17] | J.L. Wang, D. Liu, Z.J. Zhang, et al., Structure-based discovery of an organic compound that binds Bcl-2 protein and induces apoptosis of tumor cells. Proc. Natl. Acad. Sci. U. S. A. (2000) 7124–7129. |
| [18] | J. Li, Y.P. Sui, J.J. Xin, et al., Synthesis of biscoumarin and dihydropyran derivatives with promising antitumor and antibacterial activities. Bioorg. Med. Chem. Lett. (2015) 5520–5523. |
| [19] | T.S. Kamatchi, P. Kalaivani, P. Poornima, et al., New organometallic ruthenium (ii) complexes containing chelidonic acid (4-oxo-4H-pyran-2, 6-dicarboxylic acid):synthesis. structure and in vitro biological activity. RSC Adv. (2014) 2004–2022. |
| [20] | G.M. Giblin, C.T. O'Shaughnessy, A. Naylor, et al., Discovery of 2-[(2, 4-dichlorophenyl) amino]-N-[(tetrahydro-2H-pyran-4-yl)methyl]-4-(trifluoromethyl)-5-pyrimidinecarboxamide. a selective CB2 receptor agonist for the treatment of inflammatory pain. J. Med. Chem. (2007) 2597–2600. |
| [21] | A. Dömling. Recent developments in isocyanide based multicomponent reactions in applied chemistry. Chem. Rev. (2006) 17–89. |
| [22] | W. Liju, K. Ablajan, F. Jun. Rapid and efficient one-pot synthesis of spiro[indoline-3, 4'-pyrano[2.3-c] pyrazole] derivatives catalyzed by L-proline under ultrasound irradiation. Ultrason. Sonochem. (2015) 113–118. |
| [23] | A. Keyume, Z. Esmayil, L. Wang, F. Jun. Convenient DABCO-catalyzed one-pot synthesis of multi-substituted pyrano[2.3-c] pyrazole dicarboxylates. Tetrahedron (2014) 3976–3980. |
| [24] | Y.S. Nanayakkara, D.W. Armstrong. A liquid drop RC filter apparatus for detection. Anal. Bioanal. Chem. (2011) 2669–2678. |
| [25] | K.R. Reddy, K. Rajanna, K. Uppalaiah. Environmentally benign contemporary Friedel-Crafts acylation of 1-halo-2-methoxy naphthalenes and its related compounds under conventional and nonconventional conditions. Tetrahedron Lett. (2013) 3431–3436. |
| [26] | T.S. Jin, J.C. Xiao, S.J. Wang, T.S. Li. Ultrasound-assisted synthesis of 2-amino-2-chromenes with cetyltrimethylammonium bromide in aqueous media. Ultrason. Sonochem. (2004) 393–397. |
| [27] | D. Albanese, A. Donghi, D. Landini, V. Lupi, M. Penso. Environmentally benign sequential synthesis of 3, 4-dihydro-2H-1.4-benzoxazines under phase transfer catalysis conditions. Green Chem. (2003) 367–369. |
| [28] | K. Ablajan, W. Liju, A. Tuoheti, Y. Kelimu. An efficient four-component one-pot synthesis of 6-amino-4-aryl-3-methyl-2, 4-dihydropyrano[2.3-C] pyrazole-5-carbonitriles under phase-transfer catalyst. Lett. Org. Chem. (2012) 639–643. |
| [29] | A. Benazzouz, M. Makhloufi-Chebli, N. Khatir-Hamdi, et al., A facile synthesis of new coumarin-3.4-dihydropyrimidin-2(1H)-ones/thiones dyads. Tetrahedron (2015) 3890–3894. |
| [30] | P.P. Ghosh, G. Pal, S. Paul, A.R. Das. Design and synthesis of benzylpyrazolyl coumarin derivatives via a four-component reaction in water:investigation of the weak interactions accumulating in the crystal structure of a signified compound. Green Chem. (2012) 2691–2698. |
| [31] | H. Wang, X. Liu, X. Feng, Z. Huang, D. Shi. GAP chemistry for pyrrolyl coumarin derivatives:a highlyefficientone-potsynthesis undercatalyst-freeconditions. Green Chem. (2013) 3307–3311. |
| [32] | F. Vitório, T.M. Pereira, R.N. Castro, et al., Synthesis and mechanism of novel fluorescent coumarin-dihydropyrimidinone dyads obtained by the Biginelli multicomponent reaction. New J. Chem. (2015) 2323–2332. |
| [33] | T. Sugino, K. Tanaka. Solvent-free coumarin synthesis. Chem. Lett. (2001) 110–111. |
| [34] | M.B. Gawande, V.D.B. Bonifacio, R. Luque, P.S. Branco, R.S. Varma. Benign by design:catalyst-free in-water. on-water green chemical methodologies in organic synthesis. Chem. Soc. Rev. (2013) 5522–5551. |
| [35] | D. Li, H. Li, Y. Fu, et al., Critical micelle concentrations of cetyltrimethylammonium chloride and their influence on the periodic structure of mesoporous silica. Colloid J. (2008) 747–752. |
 2017, Vol. 28
2017, Vol. 28