2 Department of Chemistry, National Institute of Technology, Raipur, India
Poly(lactic acid) (PLA) [1, 2], produced by the ring-opening polymerization (ROP) of lactide (LA), is a leading biodegradable and biocompatible polyester and is very useful for biomedical and pharmaceutical applications [3-5]. Different metal initiators or catalysts have been used in the formation of PLA, such as aluminum [6], lithium [1], magnesium [7-9], iron [10], tin [11], titanium [12], or zinc [13, 14]. Comparatively, copper complexes with high electron transfer ability, moderate Lewis acidity and stability associated with reactive intermediates, should be the topic of in depth investigations. Copper is a biocompatible metal supporting the survival of life and to the best of our knowledge there have been few reports on copper initiators having nitrogencontaining polydentate ligands and their application in ROP of lactide. These include copper complexes derived from pyrazole [15], phenoxy-ketimine [16], salicylaldimine [17], and diketiminate [18] which are active toward polymerization of lactides and produce polymer with moderate number average molecular weights and narrow molecular weight distribution. Nowadays, all the commercial PLAs are synthesized using FDA-approved stannous octanoate as catalyst. Copper acetate has same catalytic characteristics as stannous octanoate and less toxic catalyst, which has been used for synthesis of PLA [19]. Despite extensive utility of salicylaldimine Schiff base complexes in many important chemical transformations [20], their application in ROP of L-lactide largely remains unexplored. Because of the relative scarcity of reports on the use of copper complexes as ROP initiators, we embarked on a study of these complexes in such processes. Here, we report the synthesis and use of tetra-coordinate salicylaldimine copper complex in L-lactide polymerization and block polymerization of PLA with poly(ethylene glycol)methyl ether (PLA-b-MePEG). This complex offers the advantage of being stable in air, and is also easier to prepare. In addition, the steric and electronic effects of ligands on polymerization activity were also investigated.
2. Experimental 2.1. Materials and instrumentsSyntheses were performed under a dry nitrogen atmosphere using a combination of a glove box and standard Schlenk techniques. All solvents were of analytical grade and were dried and distilled prior to use. Toluene and dichloromethane were dried and distilled from sodium benzophenone and P2O5, respectively. Anhydrous copper chloride and ethylene diamine (ED) were purchased from HiMedia Laboratories Pvt. Ltd., Mumbai, India, 2-hydroxy-3-methoxybenzaldehyde (HMB) and benzyl alcohol were procured form E. Merck, India. L-Lactide (LA) was used as received.
IR spectra were recorded on KBr pellet using Perkin-Elmer 1600 FTIR Spectrophotometer. The electronic spectra were recorded with Shimadzu 1601 PC UV-vis Spectrophotometer. TGA was carried out by using Perkin-Elmer Pyris, Diamond Thermal Analyzer under nitrogen atmosphere at a heating rate of 10 ℃/min. Atomic Absorption Spectra (AAS) was done by Perkin-Elmer 3100 Atomic Absorption Spectrometer at λmax of copper ion. Haraeus Carlo Ebra 1108 Elemental Analyzer was used for analyzing the composition of Schiff bases and its copper complexes. The NMR spectrum was recorded on an FT-NMRBrucker 300 MHz Spectrometer using DMSO-d6 as a solvent and tetramethylsilane (TMS) as an internal reference. The magnetic moment (μ) of metal complexes was measured using Vibrating Sample Magnetometer-155. The molecular weight of Schiff base, copper complex and PLA were determined using a Merck VAPRO 5600, Germany, Vapor Pressure Osmometer.
2.2. Cu-HMBED complex in ring opening polymerization of L-lactide and block copolymer PLA-b-MePEGA typical polymerization procedure was exemplified by the synthesis of PLA-150 ([LA]/[Cu] = 150) at room temperature. To a rapidly stirred solution of copper complex of N, N’-bis (2-hydroxy- 3-methoxy benzaldehyde) ethylenediamine (Cu-HMBED) (0.052 g, 0.133 mmol) in toluene (30 mL) was added L-lactide (2.88 g, 20 mmol) along with requisite amount of benzyl alcohol. A rise in the viscosity was observed and finally the stirring was ceased after 25 h. Volatile materials were removed under vacuum, and the residue was extracted with THF (30 mL). The extraction was dried again and the white precipitate was washed with n-hexane three times and dried under vacuum overnight, giving a crystalline white solid with yield 83% (2.4 g).
The molecular weights (Mn and Mw) and polydispersity index (Mw/Mn) were determined by using gel permeation chromatography (GPC) manufactured by Waters. The GPC instrument was equipped with a Waters 1525 Binary HPLC pump. Two columns, namely, Waters Styragel HR4 7.8×300 mm WAT10573, and “Waters Styragel HR3 7.8×300 mm WAT04422300 were used in a series for separation using THF as solvents. THF was used as the mobile phase. The flow rate of THF was 1.0 ml/min. An ELS detector, Waters 2420, was used for detection of different molecular weight fractions. Polystyrene standards with a low dispersity were used to generate a calibration curve. The GPC chromatograms were analyzed through Breeze version 3.3 software.
For block polymer PLA-b-MePEG, a mixture of Cu-HMBED (0.05 mmol), PEG (Mw = 2000, 0.800 g, 0.400 mmol) and L-lactide (1.728 g, 6.0 mmol) in toluene (20.0 mL) was stirred at room temperature for 4 h, during which an increase in viscosity was observed. After the reaction was quenched by the addition of a methanol solution (5.0 mL), and n-hexane (100.0 mL) was then added to the above mixture to give white solid. The white solid was dissolved in CH2Cl2 (10.0 mL) and then n-hexane (100 mL) was added to give white crystalline solid and was then dried under vacuum. Yield: 78.0% (1.719 g).
3. Results and discussionThe copper complex (Cu-HMBED) was synthesized (Scheme 1) by refluxing the methanolic solution of Schiff base and copper chloride in a round bottom flask at 60 ℃ for 7 h as per the modified synthetic rout reported in the research paper [21]. All reactions were performed under nitrogen atmosphere. Finally, copper complex was recrystallized in methanol and dried in a vacuum desiccator. The TGA of HMBED Schiff base showed a weight loss of 50.4 wt% at 500 ℃, but its copper (II) ion complex showed a weight loss of 37.0 wt%, which indicates that Cu-HMBED complex was more stable in comparison to ligand. Due to the formation of copper complex, there was a considerable difference in IR bands for > C55N and > C-O groups and also two new absorption bands at 566 cm-1 and 442 cm-1 come into sight due to the formation of Cu-O and Cu-N bonds in Cu-HMBED complex. And also, vanishing of phenolic OH band between 2800 and 3000 cm-1 of HMBED Schiff base was a confirmation for the formation of Cu-HMBED.

|
Download:
|
| Scheme. 1. Synthesis of Cu-HMBED complex. | |
The formation of Cu-HMBED showed hypsochromic shift in π→π * transition from 264 nm to 254 nm, and for the n→π* transition from 330 nm to 269 nm and also at 322 nm and 383 nm showed C→T transition and d→d transition, respectively. These electronic transitions correspond to t2g 6eg 3 configurations for copper(II) ion in this complex. The magnetic moment (μ) of Cu-HMBED complex was found to be 1.84 BM, which indicated that it was paramagnetic and square planar structure with dsp2 hybridization.
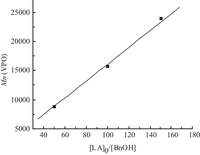
By comparing the polymerization results listed in Table 1, several structure-activity trends may be drawn. Experimental results indicate that compound Cu-HMBED is an efficient catalyst for ROP of L-lactide in the presence of BnOH, when [M]0/[I]0 ratio is ranging from 50 to 150. The polymerization is well controlled and is demonstrated by the linear relationship between Mn and [M]0/ [BnOH]0 ratio (Fig. 1). This indicates that PLA is the only product and no side products. It is interesting to note that compound Cu- HMBED catalyzes ROP of L-lactide with benzyl alcohol (BnOH) shows immortally. The ‘immortal’ character was examined using 2-4 equiv. ratios of benzyl alcohol as the chain transfer agent (entries 4, 5). It was found that for polymerization without the use of BnOH, there is almost negligible conversion (<5%), (Table 1, entry 1) but the use of BnOH changes drastically the activities of the catalyst. And also use of more amount of BnOH (Table 1, entries 4 and 5) the reaction time decreases from 24 h to 15 h. This may be due to initiator actively participating in the polymerization reaction. The molecular weight of the polymers was also affected by the ratio of BnOH used. By the addition of two or four equiv. benzyl alcohol in polymerization reactions, the molecular weights became half or one fourth, respectively.
|
Download:
|
| Figure 1. Polymerization of L-lactide catalyzed by Cu-HMBED in CH2Cl2 at room temperature showing linear relationship between Mn and [M]0/[BnOH]0 ratio. | |
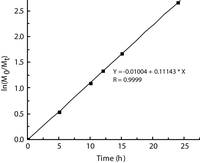
Our polymerization results are much superior to those reported for copper complexes with elaborate ligands [15-18]. Literature report for the polymerization of L-lactide using three different copper phenoxy-ketimine complexes at 160 ℃ found to be Mn = 2200 4500 and 6500 g/mol, respectively [16] suggests our results are superior in terms of Mn (Table 1). This may be due to faster propagation than initiation has occurred. The kinetic studies for the ROP of lactide has been observed in the presence of BnOH in the ratio [LA]/[Cu-HMBED]/[BnOH] = 100/1/1 (Fig. 2). This plot implies that the polymerization reaction obeys first-order kinetics with the value of kapp for L-lactide polymerization was found to be 0.11143 h-1.
|
Download:
|
| Figure 2. Kinetics plot for lactide conversion in time. | |
In the mechanism, the copper complex (Cu-HMBED) has produced active species by combining with lacide and benzyl alcohol and subsequently formed PLA through rearrangement with benzyl ester in one end (Scheme 2) and during the formation of active species, the benzyl alcohol has been activated by forming hydrogen bond with the oxygen atom of Cu-HMBED complex [23]. The block polymer PLA-b-MePEG has shown their potential applications in the medical field. Due to their biodegradable, biocompatible, and permeable properties, they can be used as a delivery medium for a controlled release of drugs [24]. This block polymer with various PLA: PEG ratios, molecular weights, structure, and solubility can be synthesized and have attracted interest for a variety of biomedical applications such as drug carriers for targeted drug delivery system. Recently, a tin complex with PEG has shown activity toward the ROP of L-lactide [24]. A very important task for developing new catalytic systems is to make the catalyst more biocompatible and less toxic with the purpose of biomedical applications. Most important the PDI of copolymer must be narrow. Therefore, we have used Cu-HMBED complex with BnOH, PEG, for the polymerization of L-lactide. The result is shown in Table 1 (entry 6).
|
Download:
|
| Scheme. 2. Synthesis of PLA. [(Fig. 2)TD$FIG] Fig. 2. Kinetics plot for lactide conversion in time. [(Fig. 1)TD$FIG] Fig. 1. Polymerization of L-lactide catalyzed by Cu-HMBED in CH2Cl2 at room temperature showing linear relationship between Mn and [M]0/[BnOH]0 ratio. | |
|
|
Table 1 Polymerization of L-lactide by copper complex (Cu-HMBED) in presence of benzyl alcohol. |
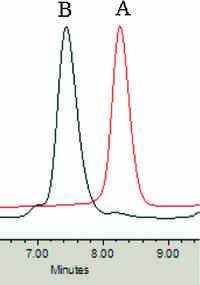
The PLA-b-MePEG was obtained by stirring the mixture of copper complex Cu-HMBED, MePEG and L-lactide of equivalent amount in toluene at room temperature for 4 h. The 1H NMR spectrum of PLA-b-MePEG (Fig. 3) show a triplet of methylene protons (3.65 ppm), a typical quartet of methine proton (5.16 ppm), and a doublet of methyl protons (1.58 ppm) show the block nature of the PLA-b-MePEG polymer. It is further verified by the GPC graph. Fig. 4 shows that the peak A for the MePEG (Mn = 2000, PDI = 1.06) appeared at 8.18 min and after formation of copolymer PLA-b-MePEG, the peak B appears at 7.31 min and the MePEG peak was disappeared. Thermal degradation of PLA and PLA-b-MePEG was investigated by TGA. It can be concluded that the PLA is thermally stable up to a temperature of 200 ℃, whereas PLA-b-MePEG is found to be stable up to 250 ℃. This may be attributed to the high molecular weight of PLA-b-MePEG [25].

|
Download:
|
| Figure 3. 1H NMR spectrum of PLA-b-MePEG. | |
|
Download:
|
| Figure 4. GPC curve of PLA-b-MePEG copolymer: (peak A for MePEG and peak B for PLA-b-MePEG copolymer). | |
4. Conclusion
In conclusion, the copper complex Cu-HMBED was synthesized and characterized successfully for their structures and, is good catalyst to initiate the ring opening polymerization of L-lactide in the presence of benzyl alcohol and synthesis of block polymer PLAb- MePEG. All the PLA produced by ROP of L-lactide show moderate molecular weight with above 90% conversion rate.
Acknowledgment The authors are thankful to CSIR & UGC,New Delhi,India for funding.The authors are also grateful to Ravenshaw University and NIT,Raipur for providing research facilities.| [1] | Sutar A.K, Maharana T, Dutta S, Chen C.T, Lin C.C, Ring-opening polymerization by lithium catalysts:an overview. Chem. Soc. Rev 39 (2010) 1724–1746. DOI:10.1039/B912806A |
| [2] | Wu J, Yu T.L, Chen C.T, Lin C.C, Recent developments in main group metal complexes catalyzed/initiated polymerization of lactides and related cyclic esters. Coord. Chem. Rev 250 (2006) 602–626. DOI:10.1016/j.ccr.2005.07.010 |
| [3] | Maharana T, Mohanty B, Negi Y.S, Melt-solid polycondensation of lactic acid and its biodegradability. Prog. Polym. Sci 24 (2009) 99–124. |
| [4] | Jeong B, Bae Y.H, Lee D.S, Kim S.W, Biodegradable block copolymers as injectable drug-delivery systems. Nature 388 (1997) 860–862. DOI:10.1038/42218 |
| [5] | Gref R, Minamitake Y, Peracchia M.T, Biodegradable long-circulating polymeric nanospheres. Science 263 (1994) 1600–1603. DOI:10.1126/science.8128245 |
| [6] | A. Routaray, N. Nath, T. Maharana, A.K. Sutar, Ring-opening polymerization of lactide by aluminium catalyst, Catal. Sci. Technol. (2015), http://dx.doi.org/10.1039/C5CY00454C. |
| [7] | Gao Y, Dai Z, Zhang J, Trinuclear and tetranuclear magnesium alkoxide clusters as highly active initiators for ring-opening. Inorg. Chem 53 (2014) 716–726. DOI:10.1021/ic401459a |
| [8] | Wojtaszak J, Mierzwicki K, Szafert S, Gulia N, Ejfler J, Homoleptic aminophenolates of Zn, Mg and Ca. Synthesis, structure, DFT studies and polymerization activity in ROP of lactides. Dalton Trans 43 (2014) 2424–2436. DOI:10.1039/C3DT52868E |
| [9] | Chen C.T, Hung C.C, Chang Y.J, Peng K.F, Chen M.T, Magnesium and zinc complexes containing pendantpyrazolylephenolate ligands as catalysts for ring opening polymerization of cyclic esters. J. Organomet. Chem 73 (2013) 81–89. |
| [10] | Bnesser A.B, Li B, Byers J.A, Redox-controlled polymerization of lactide catalyzed by bis (imino) pyridine iron bis (alkoxide) complexes. J. Am. Chem. Soc 135 (2013) 16553–16560. DOI:10.1021/ja407920d |
| [11] | Lee E.J, Lee K.M, Jang J, Characteristics of silica-supported tin(Ⅱ) methoxide catalysts for ring-opening polymerization (ROP) of L-lactide. J. Mol. Catal. A:Chem 385 (2014) 68–72. DOI:10.1016/j.molcata.2014.01.008 |
| [12] | Chen H.Y, Liu M.Y, Sutar A.K, Lin C.C, Synthesis and structural studies of heterobimetallic alkoxide complexes supported by bis(phenolate) ligands:efficient atalysts for ring-opening polymerization of L-lactide. Inorg. Chem 49 (2010) 665–674. DOI:10.1021/ic901938e |
| [13] | Chen H.Y, Peng Y.L, Huang T.H, Comparative study of lactide polymerization by zinc alkoxide complexes with a beta-diketiminato ligand bearing different substituents. J. Mol. Catal. A:Chem 339 (2011) 61–71. DOI:10.1016/j.molcata.2011.02.013 |
| [14] | Fliedel C, Vila-Viçosa D, Calhorda M.J, Dagorne S, Aviles T, Dinuclear zinc-Nheterocyclic carbene complexes for either the controlled ring-opening polymerization of lactide or the controlled degradation of polylactide under mild conditions. Chem. Catal. Chem 6 (2014) 1357–1367. |
| [15] | Appavoo D, Omondi B, Guzei I.A, Bis(3,5-dimethylpyrazole) copper(Ⅱ) and zinc(Ⅱ) complexes as efficient initiators for the ring opening polymerization of ecaprolactone and D L-lactide. Polyhedron 69 (2014) 55–60. DOI:10.1016/j.poly.2013.11.011 |
| [16] | John A, Katiyar V, Pang K, Ni(Ⅱ) and Cu(Ⅱ) complexes of phenoxy-ketimine ligands:Synthesis, structures and their utility in bulk ring-opening polymerization (ROP) of L-lactide. Polyhedron 26 (2007) 4033–4044. DOI:10.1016/j.poly.2007.04.039 |
| [17] | Bhunora S, Mugo J, Bhaw-Luximon A, The use of Cu and Zn salicylaldimine complexes as catalyst precursors in ring opening polymerization of lactides:ligand effects on polymer characteristics. Appl. Organomet. Chem 25 (2011) 133–145. DOI:10.1002/aoc.v25.2 |
| [18] | Whitehorne T.J.J, Schaper F, Square-planar Cu (Ⅱ) diketiminate complexes in lactide polymerization. Inorg. Chem 52 (2013) 13612–13622. DOI:10.1021/ic402133c |
| [19] | Gowda R.R, Chakraborty D, Copper acetate catalyzed bulk ring opening polymerization of lactides. J. Mol. Catal. A:Chem 349 (2011) 86–93. DOI:10.1016/j.molcata.2011.08.024 |
| [20] | Gupta K.C, Sutar A.K, Catalytic activities of Schiff base transition metal complexes. Coord. Chem. Rev 252 (2008) 1420–1450. DOI:10.1016/j.ccr.2007.09.005 |
| [21] | Sutar A.K, Das Y, Pattnaik S, Novel polystyrene-anchored zinc complex:efficient catalyst for phenol oxidation. China J. Catal 35 (2014) 1701–1708. DOI:10.1016/S1872-2067(14)60113-3 |
| [22] | Alamri H, Zhao J, Pahovnik D, Hadjichristidis N, Phosphazene-catalyzed ringopening polymerization of e-caprolactone:influence of solvents and initiators. Polym. Chem 55 (2014) 471–5478. DOI:10.1016/j.polymer.2013.12.022 |
| [23] | Hsueh M.L, Huang B.H, Wu J, Lin C.C, Synthesis, characterization, and catalytic studies of lithium complexes:efficient initiators for ring-opening polymerization of l-lactide. Macromolecules 38 (2005) 9482–9487. DOI:10.1021/ma050600o |
| [24] | Zhang Z, Feng S.S, Nanoparticles of poly(lactide)/vitamin E TPGS copolymer for cancer chemotherapy:synthesis, formulation, characterization and in vitro drug release. Biomaterials 27 (2006) 262–270. DOI:10.1016/j.biomaterials.2005.05.104 |
| [25] | Jamshidi K, Hyon S, Ikada Y, Thermal characterization of polylactides. Polymer 29 (1988) 2229–2234. DOI:10.1016/0032-3861(88)90116-4 |
 2016, Vol. 27
2016, Vol. 27 



