Supra-amphiphiles [1] refer to amphiphiles that are constructed by dynamic covalent bonds [2, 3] or non-π*covalent interactions such as hydrogen bonding, π-π interaction, host- guest interaction and charge transfer interaction [4-6]. Generally, the hydrophilic part and the hydrophobic part can be easily linked together via these dynamic connections, which avoids tedious syntheses. Moreover, the supra-amphiphiles are easier to be manipulated compared with amphiphiles fabricated by covalent bonds due to the dynamic nature of the linkage. They undergo selfassembly and disassembly processes in a controllable way with stimuli-responsive properties [7, 8].
Carbohydrates play an important role in a variety of biological processes, such as cell adhesion, proliferation, differentiation, recognition, inflammation and the immune response [9]. They are able to form hydrogen bonds, which makes them become important building blocks in supramolecular chemistry [10]. However, few works have been done to incorporate carbohydrates into supra-amphiphiles [11]. Zhang and co-workers constructed the first example of sugar-containing supra-amphiphile, i.e. supramolecular glycolipid based on host-enhanced charge transfer interaction [12]. However, as far as we know, supra-amphiphile of carbohydrate has not been achieved on the basis of dynamic covalent bond.
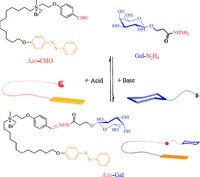
Herein, for the first time, construction of azobenzene-galactopyranoside (Azo-Gal) supra-amphiphile based on acylhydrazone dynamic covalent bond formed by the aldehyde of the azobenzenecontaining hydrophobic domain (Azo-CHO) and the hydrazine of the sugar moiety (Gal-N2H4) was reported (Fig. 1). Acylhydrazone bond is stable in neutral or alkaline environment, but it can be hydrolyzed under acidic condition. Besides, azobenzene is utilized as a model hydrophobic moiety since it is a wildly used building block for self-assembly and forms reversible complex with a-CD. The Azo-Gal supra-amphiphile self-assembles into fiber in mm scale, which is featured by light and pH responsiveness.
|
Download:
|
| Figure 1. An Azo-Gal supra-amphiphile based on a dynamic covalent bond. | |
2. Experimental 2.1. Materials and experiments
Ethyl 3-hydroxypropanoate was purchased from Maya Chemical and used as received. D-Galactose was purchased from Bangcheng Chemical and used as received. Trichloroacetonitrile, dichloromethane (DCM) and N, N-dimethylformamide (DMF) were distilled before use. Unless specially mentioned, all other chemicals were purchased from J & K Chemical and used as received. The reactions were monitored and the Rf values were determined using analytical thin-π*layer chromatography (TLC). The TLC plates were visualized by immersion into 5% sulfuric acid solution in ethanol followed by heating using hot air heater. Column chromatography was carried out on silica gel (200-300 mesh).
1HNMRand 13CNMR spectrawere recorded on anAVANCE III HD 400 MHz spectrometer. Thematrix-assisted laser desorption ionization time-of-flight mass spectrometry (Maldi-TOF MS) measurement was performed using a Perspective Biosystem Voyager DE-STR MALDI-TOF MS (Applied Biosystems, Framingham, MA). Transmission electron microscopy (TEM) images were taken on Tecnai G2 operating at 200 kV. Atomic force microscope (AFM) was carried out on a Bruker Multimode VIII SPMequipped with a J scanner. Dynamic light scattering studies (DLS) were conducted using Zetasizer Nano-ZS from Malvern Instruments. UV-vis spectroscopy was recorded in a conventional quartz cell (light path 10 mm) on a Perkin-π*Elmer Lambda 35 spectrophotometer.
2.2. SynthesisThe synthesis details of A1-A3 and G1-G3 are shown in Supporting information.
Synthesis of Azo-CHO: A2 (130 mg, 0.67 mmol), A3 (461 mg, 1 mmol) were added to acetonitrile and the mixture was refluxed overnight. After cooling down to room temperature, the solvent was evaporated under vacuum and the crude product was purified by column chromatography using EtOH/DCM (1:20) as eluent to yield 300 mg (70%) of Azo-CHO as yellow powder. 1H NMR (400 MHz, CDCl3): δ 9.91 (s, 1H), 8.03-7.79 (m, 6H), 7.57-7.36 (m, 3H), 7.03 (dd, 4H, J = 25.8, 8.8 Hz), 4.65 (s, 2H), 4.35 (s, 2H), 4.04 (t, 2H, J = 6.5 Hz), 3.75-3.55 (m, 6H), 1.94-1.77 (m, 4H), 1.56-1.21 (m, 17H). 13C NMR (101 MHz, CDCl3): δ 190.62, 161.70, 152.74, 146.81, 132.16, 130.32, 129.03, 124.73, 122.51, 114.93, 114.71, 114.13, 68.34, 62.71, 62.56, 52.12, 43.95, 29.48, 29.45, 29.42, 29.38, 29.31, 29.22, 29.16, 26.29, 25.98, 22.96. Maldi-TOF MS: calcd. for C35H48N3O3 + 558.37, found 558.37.
Synthesis of G4: A mixture of G3 (1 g, 2 mmol), ethyl 3- hydroxypropanoate (0.186 g, 1.6 mmol) in dry DCM (15 mL) was stirred at -35 ℃ for 30 min under Ar atmosphere. Trimethylsilyl trifluoromethanesulfonate (TMSOTf, 140 mL, 0.77 mmol) was added dropwise to the mixture while stirring. Then the mixture was stirred at -35 ℃ until the complete disappearance of the starting material G3 (using the TLC to monitor the reaction process). Then the reaction was quenched by adding triethylamine. After the reaction mixture was recovered to the ambient temperature, it was concentrated under vacuum. The product was purified by column chromatography using EtOAc/petroleum ether (1:2) as eluent to yield 0.3 g (42.5%) of G4 as white solid. 1 H NMR (400MHz, CDCl3): δ 5.39 (dd, 1H, J = 3.4, 1.1 Hz), 5.18 (dd, 1H, J = 10.5, 7.9 Hz), 5.01 (dd, 1H, J = 10.5, 3.4 Hz), 4.52 (d, 1H, J = 8.0 Hz), 4.19-4.10 (m, 5H), 3.96-3.81(m, 2H), 2.65-2.56(m, 2H), 2.15 (s, 3H), 2.05 (d, 6H, J = 0.8 Hz), 1.98 (s, 3H), 1.27 (t, 3H, J = 7.1 Hz). 13C NMR (101 MHz, CDCl3): δ 171.02, 170.39, 170.25, 170.15, 169.46, 101.53, 70.85, 70.65, 68.64, 67.00, 65.54, 61.24, 60.58, 34.83, 20.71, 20.66, 20.58, 14.17.
Synthesis of Gal-N2H4: A mixture of G4 (0.7 g, 1.56 mmol), 85% hydrazine hydrate (0.59 g, 15.7 mmol) in ethanol was stirred at ambient temperature for 1 d until the complete disappearance of the raw material of G4 and appearance of the product (using the TLC to monitor the reaction process). The mixture was concentrated under vacuum and was purified by column chromatography using water/acetonitrile (1:2) as eluent to yield 0.2 g (48.8%) of Gal-N2H4 as white solid. 1H NMR (400 MHz, D2O):δ 4.26 (d, 1H, J = 7.9 Hz), 4.01 (dt, 1H, J = 10.8, 6.0 Hz), 3.85-3.77 (m, 2H), 3.64 (qd, 2H, J = 11.7, 6.1 Hz), 3.54 (ddd, 2H, J = 13.4, 8.9, 3.8 Hz), 3.37 (dd, 1H, J = 9.9, 7.9 Hz), 2.42 (t, 2H, J = 6.0 Hz). 13C NMR (101 MHz, D2O):δ 172.78, 102.83, 75.10, 72.64, 70.60, 68.57, 65.80, 60.95, 34.36. Maldi-TOF MS: calcd. for C9H18N2O7 266.11, found 266.29 (289.29-Na+23).
2.3. Preparation of Azo-Gal supra-amphiphileMixing Azo-CHO and Gal-N2H4 together in the molar ratio of 1:1 in DMSO for 3 days until the complete formation of Azo-Gal and then stored as original solution. Ultrasound or heating accelerates the formation speed.
2.4. Self-assembly of Azo-Gal supra-amphiphileDeionized water (8 mL) was added into the DMSO solution of Azo-Gal (1 mL, 1 mg/mL) dropwise by using a syringe pump at the rate of 20 mm/h under vigorous stirring. Then the solution was dialyzed (MWCO 1000) against deionized water to remove the extra DMSO. Concentration of the solution was fixed at 0.1 mg/mL by adding extra deionized water.
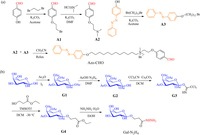
3. Results and discussion 3.1. Preparation of the Azo-Gal supra-amphiphileFirst, the two new precursors of Azo-Gal, Azo-CHO and Gal- N2H4 were synthesized separately. As shown in Scheme 1, Azo- CHO was prepared via four steps. First, 4-hydroxybenzaldehyde reacted with 1, 2-dibromoethane in the presence of K2CO3 affording compound A1. Then A1 was treated with dimethylamine hydrochloride in the presence of K2CO3 to afford compound A2. Meanwhile, compound A3 was synthesied by reacting 4-phenylazophenol with 1, 12-dibromododecane in the presence of K2CO3. In the end, through the reaction between A2 and A3, the final product Azo-CHO was formed. On the other hand, Gal-N2H4 was synthesized via five steps following the classical glycosylation strategy. First all of the hydroxy groups of galactose were protected by acetyl groups. After selectively deprotecting the acetyl group on the anomeric carbon followed by reacting with trichloroacetonitrile, galactosyl trichloroacetimidate was synthesized. Compound G4 was prepared via glycosylation of ethyl 3-hydroxypropanoate with the galactosyl trichloroacetimidate and was transformed to the final product of Gal-N2H4 with hydrazine hydrate in ethanol.
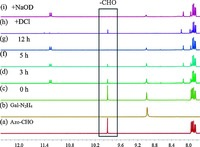
After mixing the synthesized Azo-CHO and Gal-N2H4 in DMSO, the dynamic covalent bond formed, which was confirmed by 1H NMR. The spectrum of the mixture of Azo-CHO and Gal-N2H4 in equimolar amount showed obvious differences from those of the two compounds themselves (Fig. 2). When the mixing time was increased, intensity of the signal at 9.91 ppm corresponding to the aldehyde group in Azo-CHO decreased gradually. Concomitantly a new peak at 11.2 ppm gradually became stronger, indicating formation of the acylhydrazone bond [13]. After addition of 5.0 mL DCl solution (20 wt% in D2O), the signal of the aldehyde group at 9.91 ppm appeared again with disappearance of the peak at 11.2 ppm, indicating hydrolyzation of the acylhydrazone bond. Then, upon changing the condition back to basic by adding 2.75 mL NaOD solution (40 wt% in D2O), the dynamic covalent bond was regenerated with the reappearance of the acylhydrazone peak at 11.2 ppm as well as the disappearance of the aldehyde peak at 9.91 ppm. This result fully supported the formation of acylhydrazone bond and its response to pH. In addition, the acylhydrazone bond formation was also supported by Maldi-TOF MS result (Fig. S1 in Supporting information).
|
Download:
|
| Figure 2. 1H NMR spectra (400 MHz, DMSO-d6) of (a) Azo-CHO, (b) Gal-N2H4, (c)-(g) mixtures of (a) and (b) in the molar ratio of 1:1 with an increasing ultrasound time, (h) after addition of DCl to (g), (i) after addtion of NaOD to (h). | |
|
Download:
|
| Scheme. 1. Synthetic routes of (a) Azo-CHO and (b) Gal-N2H4. | |
3.2. Self-assembly of the Azo-Gal supra-amphiphile
The amphiphilic Azo-Gal was assembled by selective solvent method. The obtained assemblies were first characterized by TEM and AFM. One dimentional fibrillar structure was observed with their length in micrometer scale and their width in nanometer scale under TEM (Fig. 3a). As shown in the AFM image in Fig. 3b, height of the fibers was found around 150 nm. This result indicates that the fiber was formed by multilayers of the Azo-Gal molecules. Under TEM and AFM, the fibers were found in similar size and morphology with relatively sharp endings, indicating the morphology was quite stable and reproducible. Finally, DLS was performed proving existence of the fibers in aqueous solution (Fig. S2 in Supporting information).

|
Download:
|
| Figure 3. (a) TEM and (b) AFM images of the self-assembled Azo-Gal supraamphiphile. | |
|
Download:
|
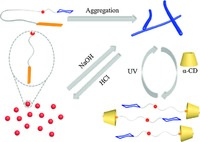
| Scheme. 2. The cartoon illustration of the dual-responsive self-assembly and disassembly of the Azo-Gal supra-amphiphile. | |
3.3. Dual-stimuli responsiveness of the self-assembly
It is known that the trans and cis conformations of azobenzene can be transformed to each other by UV or visible light [14]. Previous works by us and other research groups suggested that the assembly of the azobenzene-containing molecules into nanoobjects can be regulated by photo irradiation and vice versa [15-18]. So at first we expected possible morphology transformation of the fiber upon irradiation of UV light. UV-vis spectroscopy was first applied to investigate the photo responsiveness of the Azo-Gal supra-amphiphile (Fig. 4a). Upon UV irradiation at 365 nm for 0.5 h, the absorption band at around 325 nm of Azo-Gal in water decreased remarkably compared to that before irradiation, and concomitantly the band at around 430 nm increased slightly. The absorption band at 325 nm is ascribed to π-π* transition of trans-azobenzene while the 430 nm absorption band is attributed to n-π* transition of the cis one [19]. This result clearly shows the transformation of trans-isomer of the Azo-Gal supra-amphiphile to cis. After exposing the sample to ambient light, gradual increase of the absorption peak at 325 nm as well as decrease of peak at 430 nm were observed, suggesting the transformation of cisisomer to trans. The above results proved that the reversible photo isomerization ability of azobenzene was remained in Azo-Gal. However, UV irradiation (365 nm, 600 s) did not induce any significant change of the fiber morphology, as shown in Fig. 4b. We suppose that after the assembly formed, the Azo-Gal supraamphiphile packed quite compactly, thus the photo isomerization of azobenzene group had no obvious effect on the shape of selfassembled aggregates.
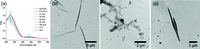
|
Download:
|
| Figure 4. (a) UV-vis spectra of Azo-Gal before and after 0.5 h irradiation with UV light (365 nm) as well as the time-dependent interconversion of cis- to trans-Azo-Gal under ambient light. The concentration of Azo-Gal is 0.05 mg/mL. TEM images of Azo-Gal: (b) (c) after UV irradiation with UV light (365 nm); (c) mixed with a-CD (1:1 molar ratio); (d) after irradiation with UV light (365 nm). | |
a-CD and azobenzene is a well-known host-guest pair in supramolecular chemistry.Only trans-azobenzene canbe recognized by a-CDto forma stable complex, while the cis one could not [20, 21]. In order to further explore the effect of a-CD on the self-assembly of Azo-Gal, TEM experiment was performed. After addition of a- CD in a molar ratio of 1:1 to Azo-Gal, the fibrillar aggregates disassembled (Fig. 4c). As shown in Fig. S3 in Supporting information, the 1H NMR clearly showed the formation of inclusion complex between a-CD and azobenzene. Before adding a-CD, the hydrophobic azobenzene group was in the core of the Azo-Gal assemblies, thus the signals of azobenzene group could hardly been observed. However, after the addition of a-CD in equal amount, signals of azobenzene group appeared as a result of the inclusion between a-CD and azobenzene and the disassembly of the self-assembled aggregates, which was consistent with literature [20]. Thus the decrease of hydrophobicity of the amphiphilic Azo-Gal supra-amphiphile after addition of a-CD induced the dissociation of the fiber. Moreover, when the above system was irradiated by UV light, it was found that the fibrillar structures formed again due to the dissociation of the a-CD/azobenzene complex (Fig. 4d).
As above described, the dynamic covalent bond used here is pH-responsive, which can be fabricated under neutral or alkaline conditions and hydrolyzed in acidic environment. To explore the pH responsiveness of the Azo-Gal assemblies, TEM and DLS experiments were carried out. When changing the self-assembled Azo-Gal solution to acidic condition (2 mL Azo-Gal solution, 20 mL 20 wt% HCl solution), the fibers transformed into nanospheres as a result of the hydrolysis of the acylhydrazone bond (Fig. 5a). Since the Gal-N2H4 has very good water solubility and cannot form aggregates in water itself, the corresponding spherical aggregates was formed by Azo-CHO. When adjusting the solution back to slightly alkaline (2 mL Azo-Gal solution, 10.92 mL 40 wt% NaOH solution), the morphology returned to fiber again because of the formation of the dynamic covalent bond (Fig. 5b). DLS results were in consistent with the observations under TEM (Fig. 5c).

|
Download:
|
| Figure 5. TEM images of Azo-Gal aggregates (a) in acidic environment and (b) then changing back to slightly alkaline environment; (c) DLS of Azo-Gal supra-amphiphile assemblies (blue), after adding HCl (red) and then after adding NaOH (black). | |
4. Conclusion
In summary, we have successfully constructed a dual-responsive Azo-Gal supra-amphiphile on the basis of acylhydrazone dynamic covalent bond in water. The amphiphilic Azo-Gal selfassembled into fibers that can be regulated by addition of a-CD and pH (Scheme 2). This dual-responsive Azo-Gal supra-amphiphile will enrich the family of supra-amphiphiles and have potential applications in biomedical fields, such as drug delivery and controlled release.
Acknowledgments National Natural Science Foundation of China (Nos. 91527305,21474020,91227203 and 51322306) and the Innovation Program of the Shanghai Municipal Education Commission are acknowledged for their financial support.| [1] | Wang C, Wang Z.Q, Zhang X, Amphiphilic building blocks for self-assembly:from amphiphiles to supra-amphiphiles. Acc. Chem. Res 45 (2012) 608–618. DOI:10.1021/ar200226d |
| [2] | Minkenberg C.B, Florusse L, Eelkema R, Koper G.J.M, van Esch J.H, Triggered selfassembly of simple dynamic covalent Surfactants. J. Am. Chem. Soc 131 (2009) 11274–11275. DOI:10.1021/ja902808q |
| [3] | Wang G.T, Wang C, Wang Z.Q, Zhang X, Bolaform superamphiphile based on a dynamic covalent bond and its self-assembly in water. Langmuir 27 (2011) 12375–12380. DOI:10.1021/la203040e |
| [4] | Xing P.Y, Sun T, Hao A.Y, Vesicles from supramolecular amphiphiles. RSC Adv 3 (2013) 24776–24793. DOI:10.1039/c3ra44569k |
| [5] | Versluis F, Tomatsu I, Kehr S, Shape and release control of a peptide decorated vesicle through pH sensitive orthogonal supramolecular interactions. J. Am. Chem. Soc 131 (2009) 13186–13187. DOI:10.1021/ja9026264 |
| [6] | Jeon Y.J, Bharadwaj P.K, Choi S.W, Lee J.W, Kim K, Supramolecular amphiphiles:spontaneous formation of vesicles triggered by formation of a charge-transfer complex in a host. Angew. Chem. Int. Ed 41 (2002) 4807. |
| [7] | Zhou Q.Z, Jiang H.J, Chen R, A triply-responsive pillar[6] arene-based supramolecular amphiphile for tunable formation of vesicles and controlled release. Chem. Commun 50 (2014) 10658–10660. DOI:10.1039/C4CC05031B |
| [8] | Shao L, Zhou J, Hua B, Yu G.C, A dual-responsive supra-amphiphile based on a water-soluble pillar[7] arene and a naphthalene diimide-containing guest. Chem. Commun 51 (2015) 7215–7218. DOI:10.1039/C5CC00937E |
| [9] | Bertozzi C.R, Kiessling L.L, Chemical glycobiology. Science 291 (2001) 2357–2364. DOI:10.1126/science.1059820 |
| [10] | Delbianco M, Bharate P, Varela-Aramburu S, Seeberger P.H, Carbohydrates in supramolecular chemistry. Chem. Rev 116 (2015) 1693–1752. |
| [11] | Kang Y.T, Liu K, Zhang X, Supra-amphiphiles:a new bridge between colloidal science and supramolecular chemistry. Langmuir 30 (2014) 5989–6001. DOI:10.1021/la500327s |
| [12] | Yang L.L, Yang H, Li F, Zhang X, Supramolecular glycolipid based on hostenhanced charge transfer interaction. Langmuir 29 (2013) 12375–12379. DOI:10.1021/la402973g |
| [13] | Lehn J.M, Dynamers:dynamic molecular and supramolecular polymers. Prog. Polym. Sci 30 (2005) 814–831. DOI:10.1016/j.progpolymsci.2005.06.002 |
| [14] | Kumar G.S, Neckers D.C, Photochemistry of azobenzene-containing polymers. Chem. Rev 89 (1989) 1915–1925. DOI:10.1021/cr00098a012 |
| [15] | Rahman M.L, Hegde G, Sarkar S.M, Yusoff M.M, Synthesis and photoswitching properties of azobenzene liquid crystals with a pentafluorobenzene terminal. Chin. Chem. Lett 25 (2014) 1611–1614. DOI:10.1016/j.cclet.2014.06.025 |
| [16] | Wu J, Lu X.M, Shan F, Guan J.F, Lu Q.H, Photoresponding ionic complex containing azobenzene chromophore for use in birefringent film. Chin. Chem. Lett 25 (2014) 15–18. DOI:10.1016/j.cclet.2013.11.008 |
| [17] | Wei K.C, Li J, Liu J.H, Chen G.S, Jiang M, Reversible vesicles of supramolecular hybrid nanoparticles. Soft Matter 8 (2012) 3300–3303. DOI:10.1039/c2sm25178g |
| [18] | Li Z.Q, Zhang Y.M, Chen H.Z, Zhao J, Liu Y, Hierarchical organization of spherical assembly with reversibly photocontrollable cross-links. J. Org. Chem 78 (2013) 5110–5114. DOI:10.1021/jo400772j |
| [19] | Zhao J, Zhang Y.M, Sun H.L, Chang X.Y, Liu Y, Multistimuli-responsive supramolecular assembly of cucurbituril/cyclodextrin pairs with an azobenzene-containing bispyridinium guest. Chem. Eur. J 20 (2014) 15108–15115. DOI:10.1002/chem.201404216 |
| [20] | Wang Y, Ma N, Wang Z, Zhang X, Photocontrolled reversible supramolecular assemblies of an azobenzene-containing surfactant with alpha-cyclodextrin. Angew. Chem. Int. Ed 46 (2007) 2823–2826. DOI:10.1002/(ISSN)1521-3773 |
| [21] | Liao X.J, Chen G.S, Liu X.X, Photoresponsive pseudopolyrotaxane hydrogels based on competition of host-guest interactions. Angew. Chem. Int. Ed 49 (2010) 4409–4413. DOI:10.1002/anie.201000141 |
 2016, Vol. 27
2016, Vol. 27 


