b China University of Chinese Academy of Sciences, Beijing 100049, China
Polyhedral oligomeric silsesquioxanes, represented by the empirical formula (RSiO1.5)n, can be regarded as organic/inorganic hybrid materials at a molecular level [1-6]. Materials based on silsesquioxanes have been proposed for applications in highperformance materials. A typical silsesquioxane molecule that possesses a cubic rigid (n=8, T8) structure consisting of central inorganic core (Si8O12) and organic moieties (R) at each of the eight vertices is extensively studied for constructing hybrid polymers with novel architectures [7, 8], and for further enhancing the mechanical and thermal properties of the hybrid polymers [9-11]. With the flexible design of functional groups, a variety of morphologies and hierarchical structures such as tadpole-shaped [12, 13] and star-shaped [14, 15] polymers based on POSS has been prepared by controlled radical polymerization methods [14, 16-18]. Star polymers with well-defined architectures have attracted much attention because of their potential medical carriers [19-24]. These molecular hybrids composed of a hydrophobic silsesquioxane core and hydrophilic arms are studied as structural amphiphiles with self-organizability and morphological tenability [7, 25-28]. Thus, unique characteristics distinctly different from planar molecules can be expected due to such cage-structural features. However, there are few articles studying the synthesis and properties of POSS-containing star polymers.
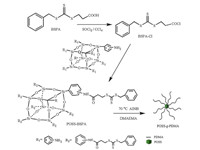
So far, there are many other reports about star polymer synthesis through "graft to" and "graft from" methods [29-31]. The advantage of the "graft to" method is that both the core or backbone and side chain can be firstly prepared by different living polymerization techniques, but the grafting density of resulting polymer brushes is often limited due to steric repulsion between bulky side chains. The "grafting from" approach enables preparation of star or brush-polymers with a high grafting density and a narrow molecular weight distribution [32]. In this paper, we mainly introduced a novel strategy for synthesis of a star polymer with octa-aminophenylsilsesquioxane (OA-POSS) incorporated into the polymer matrices to effectively form responsive functional materials as a nucleus of a micelle. The OA-POSS molecule was modified into a RAFT initiator (POSS-BSPA), which was further used to prepare star-shaped organic/inorganic hybrid polymer POSS-g-PDMA by "grafting from". Finally, the pH stimuli-response behavior of POSS-g-PDMA was also studied.
2. Experimental 2.1. MaterialsN, N-(Dimethylamino)ethyl methacrylate (DMAEMA) was purchased from Aladdin and purified by passing over a basic aluminum oxide column. Other regents of analytical grade were all obtained from Aldrich. Azobisisobuty-ronitrile (AIBN) was recrystallized twice from ethanol before use. Dioxane was used without further purification.
2.2. Synthesis of the POSS-g-PDMA3-Benzylsulfanylthiocarbonyl-sufanylpropionic acid (BSPA, RAFT agent), OA-POSS, and POSS-BSPA agent were synthesized in our laboratory (see Supporting information).
DMAEMA (1.05 g, 0.0067 mol), POSS-BSPA agent (19.5 mg, 0.0065 mmol), and AIBN (1.0 mg, 0.0061 mmol) were introduced into a Schlenk tube containing dioxane (3.0 mL). The reaction tube was degassed by three freeze-pump-thaw cycles. The polymerization was allowed to proceed under continuous stirring at 70 ℃. The polymerization was quenched by liquid nitrogen. The polymerization mixture was purified by precipitation into cold petroleum ether/diethyl ether (10:1) and arm polymer was also removed, yielding the POSS-containing star polymer POSS-g-PDMA.
2.3. Self-assembly of POSS-star polymer in aqueous solutionAll the samples were obtained by directly dissolving the polymers in aqueous solutions at pH 4.1 and formed 1 mg/mL solutions. The solutions were stirred at least for 24 h to ensure the system reached equilibrium. The pH of solutions was adjusted by hydrochloric acid and sodium hydroxide aqueous solutions. The different pH solutions of star polymers were stirred for 24 h and then measured.
Characterization and instrumentation and other synthesis are listed in the Supporting information.
3. Results and discussion 3.1. Synthesis and characterization of silsesquioxane-cored starshaped hybrid polymerScheme 1 illustrates the synthetic routes of the POSS-BSPA and star POSS-g-(PDMA) polymer. BSPA can also be used in many RAFT polymerization systems, especially grafting modification [33, 34], although BSPA is not a very effective CTA. POSS was grafted to a RAFT agent via the reaction of the amino group of OA-POSS and BSPCl. Compared with the 1H NMR spectra of BSPA and OA-POSS in Fig. S1 in Supporting information, the peak shifts of methylene protons moved from 2.84 ppm in BSPA to 2.60 ppm in POSS-BSPA and the proton signal of the phenyl moved from 6.5-7.0 in OAPOSS to 8.00-7.50 ppm in POSS-BSPA and fused with the phenyl protons (-SCH2C6H5). In addition, the proton signal of the amino groups of OA-POSS at 4.7-5.1 ppm completely disappeared. From FT-IR spectra in Fig. S2 in Supporting information, compared with OA-POSS, two new absorption bands could be discerned at 1643 cm-1 and 1050 cm-1 in the spectrum of POSS-BSPA, which are assigned to the stretching vibration of carbonyl (-CONH-) and C=S, respectively. Based on 1HNMR and FTIR spectra, we concluded the POSS-BSPA agent was synthesized successfully. The POSS-BSPA macro-initiator was then used to prepare the POSScontaining inorganic/organic hybrid DMAEMA via a "grafting from" method according to Scheme 1.
|
Download:
|
| Scheme. 1. The synthesis process of POSS-BSPA and POSS-g-PDMA. | |
The SEC traces in Fig. S3 in Supporting information showed single and symmetrical peaks of POSS-g-PDMA and the polydispersities were in the range of 1.57-1.65. As reaction time increased, the chromatograms shift to lower elution volume. Furthermore, there was almost no shoulder observed. This indicated that irreversible terminations due to coupling reactions between the star or linear chain radicals did not occur for the star polymer [14, 31]. The main results of the polymerizations by SEC and 1H MNR are summarized in Table 1. From FT-IR spectra in Fig. S4 in Supporting information, characteristic peaks at 1720 cm-1 mainly attributed to the carbonyl of PDMA segment were observed, and the peak at 3700-3100 cm-1 was the stretching vibration of benzene ring of POSS and BSPA parts, and the other peaks were mainly fused with these of the PDMA segment.
|
|
Table 1 Results of the RAFT polymerization of DMAEMA using POSS-BSPA agent. |
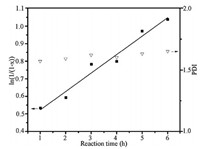
The kinetic plot of the star polymerization is shown in Fig. 1. A linear relationship exists between ln(1/(1 -χ)) (χ was the conversion of DMAEMA) and reaction time, indicating that the concentration of chain radicals is constant. Thus, the polymerization follows pseudo first-order kinetics.
|
Download:
|
| Figure 1. Pseudo-first-order kinetic plot and polydispersities (PDI) of the polymerization of DMAEMA in the presence of POSS-BSPA. | |
3.2. The self-assembly of the star polymers POSS-g-PDMA
The self-assembly of the resultant amphiphilic star polymers was also investigated in this work. Such stimuli-responsive star polymers have different hydrodynamic radii in the solution compared to the linear polymers. Amphiphilic polymers based on PDMA are well-known representatives in the study of the selfassembly of stimuli-responsive polymers. In this work, the starshaped inorganic/organic hybrid POSS-g-PDMA was similar to an amphiphilic block copolymer, since the POSS core was hydrophobic and the PDMA chain was hydrophilic.
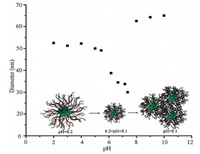
The self-assembly solutions of POSS-g-PDMA in Fig. 2 were measured by dynamic light scattering (DLS). As shown in Fig. 2, Dh was about 52 nm at pH < 5.5, and the value (Dh) of micelle decreased as the pH increased from 6.2 to 8.1. This trend proved the reversible change of the aggregate structure between "shrink" and "stretch" of the complex micelle in response to alkali or acid stimulation (inset of Fig. 2). However, Dh was increased when pH was beyond 8.1 because PDMA became hydrophobic and the solutions would become unstable. The unique properties of POSSg-PDMA are not observed for their linear counterparts in the our experiment, in which there was no alkali or acid stimulation because of no diameter change from DLS in different pH solutions at 25 ℃ [7]. We tentatively attributed the self-assembly into micelle of star polymer to the interaction of hydrophobic POSS cage with hydrophilic PDMA arms and the strong tendency toward densely packing due to its high symmetry [35].
|
Download:
|
| Figure 2. The diameter change in aqueous solution at different pH at 25 ℃. | |
To investigate the self-assembly morphology of POSS-g-PDMA, AFM and high-resolution TEM were applied because they could directly reveal the morphology. The AFM images of the POSS-gPDMA micelles are shown in Fig. 3, and we can calculate the size of the micelle to be about 23 nm at pH 2.1 3.2, 6.2, and 7.3 at 25 ℃. The diameters measured by DLS in aqueous solution at 25 ℃ were bigger than those measured by AFM, since DLS data directly reflected the dimension of micelles in solution where the PDMA chains could be well dispersed in water. However, for AFM measurement, the micellar solution was spray-coated on the mica surface, where POSS-g-PDMA micelles sharply shrunk during the process of water evaporation, which resulted in a smaller diameter. In addition, the theoretical diameter of the micelles could be calculated according to the molecular weight of POSS-g-PDMA. The calculated result showed the molecular diameter was about 30 nm for a single POSS-g-PDMA (Mn=72 920, determined by 1H NMR), which is smaller than that measured by DLS and larger than that of micelle by AFM. From the TEM image in Fig. 3, it can be seen that the size of the micelle was about 20 nm at pH 3.2 and 6.2, which was almost the same as the results obtained by AFM at pH 3.2 and 6.2 within the allowable error. As for pH > 8.1, it was difficult to be characterized by AFM and TEM for well dispersed individual micelles because micelles would aggregate under the condition (see AFM images in Fig. S6 in Supporting information).

|
Download:
|
| Figure 3. The AFM and HRTEM images of the POSS-g-PDMA self-assembly micelles in aqueous solution at pH 2.1 (a), 3.2 (b, e), 6.2 (c, f) and 7.3 (d). | |
The ζ-potential is a good measure of colloidal stability. Table 2 gives the ζ-potentials under different pH conditions. The zpotentials of POSS-g-PDMA micelles were observed to increase as pH decreased. The values of ζ-potentials were about 59, 61, and 60 mV at pH 3.0, 4.1 and 5.5, 40 and 37 mV at pH 6.2 and 7.3, and were 18 and 13 mV at pH 8.1 and 9.2. We could conclude that the core-shell structures were formed, they had different stability at different pH and was unstable at pH > 8.1 while being stable at pH < 6.2. The tertiary amine groups in PDMA shell were cationized when solutions were acidic in the scheme inset of Fig. 2. The cationic shells repelled each other, rendering this solution more stable. On the contrary, under basic condition, there was no repulsive interaction and the stability of solution was markedly reduced. This analysis from the ζ-potentials shows an agreement with that from the data of DLS.
|
|
Table 2 ζ-potential values in different pH aqueous solutions. |
4. Conclusion
In this paper, the star polymer POSS-g-PDMA was successfully synthesized via RAFT polymerization; the polymer can selfassemble into a hierarchical structure in aqueous solution. Driven by the hydrophobic and rigid silesequioxane cube, the star polymers self-assemble into the micelles with the POSS as micellar cores, and the PDMAEMA chains stretching as coronas. The micelles aggregated into two different morphologies, "shrink" or "stretch", in aqueous solution, under the different acid-base stimuli. According to DLS characterization, the average diameter of the micelles was about 52 nm at pH < 5.5 and it would lessen at 6.2 < pH < 8.1 with pH increasing, however, the diameter would become bigger at pH > 8.1. The average diameter was 20 nm by AFM at pH < 8.1, because the samples of micelle were spraycoated on the substrate and dried away from solutions. Star polymers well could be applicated in nanocarriers and nanocapsules and material modification and the works will be discussed in our next study.
AcknowledgmentsThis research was supported by the Guangdong Province Research Project (No. 2011A091000007). And we are also grateful to the National Natural Science Foundation of China for its financial support (No. 21174162).
Appendix A. Supplementary dataSupplementary material related to this article can be found, in the online version, at http://dx.doi.org/10.1016/j.cclet.2016.04.018.
| [1] | H.L. Cai, K. Xu, X. Liu, Z.E. Fu, M.C. Chen, A facile synthesis of octa (carboxyphenyl) silsesquioxane. Dalton Trans. 41 (2012) 6919–6921. DOI:10.1039/c2dt30378g |
| [2] | S.S. Choi, A.S. Lee, H.S. Lee, Synthesis and characterization of ladder-like structured polysilsesquioxane with carbazole group. Macromol. Res. 19 (2011) 261–265. DOI:10.1007/s13233-011-0304-3 |
| [3] | D. Gnanasekaran, K. Madhavan, B.S.R. Reddy, Developments of polyhedral oligomeric silsesquioxanes (POSS), POSS nanocomposites and their applications: a review. J. Sci. Ind. Res. 68 (2009) 437–464. |
| [4] | A.S.S. Lee, S.S. Choi, K.Y. Baek, S.S. Hwang, Thiol-ene photopolymerization of welldefined hybrid graft polymers from a ladder-like polysilsesquioxane. Macromol. Res. 23 (2015) 60–66. DOI:10.1007/s13233-015-3004-6 |
| [5] | D.A. Loy, J.H. Small, K. J., Shea, Nanostructure in Polysilsesquioxanes. Polym. Prepr. (Am. Chem. Soc., Div. Polym. Chem.) 46 (2005) 61. |
| [6] | S. Skaria, S.R. Schricker, Synthesis and characterization of inorganic-organic hybrid materials derived from polysilsesquioxanes (POSS). J. Macromol. Sci. Part A: Pure Appl. Chem. 47 (2010) 381–391. DOI:10.1080/10601321003659440 |
| [7] | W.A. Zhang, A.H.E. Müller, Architecture, self-assembly and properties of welldefined hybrid polymers based on polyhedral oligomeric silsequioxane (POSS). Prog. Polym. Sci. 38 (2013) 1121–1162. DOI:10.1016/j.progpolymsci.2013.03.002 |
| [8] | K. Tanaka, Y. Chujo, Advanced functional materials based on polyhedral oligomeric silsesquioxane (POSS). J. Mater. Chem. 22 (2012) 1733–1746. DOI:10.1039/C1JM14231C |
| [9] | K. Tanaka, S. Adachi, Y. Chujo, Structure-property relationship of octa-substituted POSS in thermal and mechanical reinforcements of conventional polymers. J. Polym. Sci. Part A: Polym. Chem. 47 (2009) 5690–5697. DOI:10.1002/pola.v47:21 |
| [10] | J. Wu, P.T. Mather, POSS polymers: physical properties and biomaterials applications. Polym. Rev. 49 (2009) 25–63. DOI:10.1080/15583720802656237 |
| [11] | W.C. Zhang, X.M. Li, X.Y. Guo, R.J. Yang, Mechanical and thermal properties and flame retardancy of phosphorus-containing polyhedral oligomeric silsesquioxane (DOPOPOSS)/polycarbonate composites. Polym. Degrad. Stab. 95 (2010) 2541–2546. DOI:10.1016/j.polymdegradstab.2010.07.036 |
| [12] | A. Franczyk, H.K. He, J. Burdyńska, Synthesis of high molecular weight polymethacrylates with polyhedral oligomeric silsesquioxane moieties by atom transfer radical polymerization. ACS Macro Lett. 3 (2014) 799–802. DOI:10.1021/mz5003799 |
| [13] | W.A. Zhang, L. Liu, X.D. Zhuang, Synthesis and self-assembly of tadpoleshaped organic/inorganic hybrid poly (N-isopropylacrylamide) containing polyhedral oligomeric silsesquioxane via RAFT polymerization. J. Polym. Sci. Part A: Polym. Chem. 46 (2008) 7049–7061. DOI:10.1002/pola.v46:21 |
| [14] | Y.S. Ye, W.C. Shen, C.Y. Tseng, Versatile grafting approaches to star-shaped POSS-containing hybrid polymers using RAFT polymerization and click chemistry. Chem. Commun. 47 (2011) 10656–10658. DOI:10.1039/c1cc13412d |
| [15] | W.A. Zhang, A.H.E. Müller, A "click chemistry" approach to linear and star-shaped telechelic POSS-containing hybrid polymers. Macromolecules 43 (2010) 3148–3152. DOI:10.1021/ma902830f |
| [16] | M.Z. Asuncion, M. Ronchi, H. Abu-Seir, R.M. Laine, Synthesis, functionalization and properties of incompletely condensed "half cube" silsesquioxanes as a potential route to nanoscale Janus particles. C.R. Chim. 13 (2010) 270–281. DOI:10.1016/j.crci.2009.10.007 |
| [17] | K.Y. Mya, H.B. Gose, T. Pretsch, M. Bothe, C.B. He, Star-shaped POSS-polycaprolactone polyurethanes and their shape memory performance. J. Mater. Chem. 21 (2011) 4827–4836. DOI:10.1039/c0jm04459h |
| [18] | X. Wang, V. Ervithayasuporn, Y.H. Zhang, Y. Kawakami, Reversible self-assembly of dendrimer based on polyhedral oligomeric silsesquioxanes (POSS). Chem. Commun. 47 (2011) 1282–1284. DOI:10.1039/C0CC03359F |
| [19] | H.T.T. Duong, K. Jung, S.K. Kutty, Nanoparticle (star polymer) delivery of nitric oxide effectively negates pseudomonas aeruginosa biofilm formation. Biomacromolecules 15 (2014) 2583–2589. DOI:10.1021/bm500422v |
| [20] | C.S. Fischer, C. Jenewein, S. Mecking, Conjugated star polymers from multidirectional Suzuki-Miyaura polymerization for live cell imaging. Macromolecules 48 (2015) 483–491. DOI:10.1021/ma502294n |
| [21] | C.C. Lee, J.A. MacKay, J.M.J. Fréchet, F.C. Szoka, Designing dendrimers for biological applications. Nat. Biotechnol. 23 (2005) 1517–1526. DOI:10.1038/nbt1171 |
| [22] | Y. Zhou, H. Li, Y.W. Yang, Controlled drug delivery systems based on calixarenes. Chin. Chem. Lett. 26 (2015) 825–828. DOI:10.1016/j.cclet.2015.01.038 |
| [23] | X.J. Loh, Z.X. Zhang, K.Y. Mya, Efficient gene delivery with paclitaxel-loaded DNA-hybrid polyplexes based on cationic polyhedral oligomeric silsesquioxanes. J. Mater. Chem. 20 (2010) 10634–10642. DOI:10.1039/c0jm01461c |
| [24] | F. Wang, T.K. Bronich, A.V. Kabanov, R.D. Rauh, J. Roovers, Synthesis and evaluation of a star amphiphilic block copolymer from poly (ε-caprolactone) and poly (ethylene glycol) as a potential drug delivery carrier. Bioconjugate Chem. 16 (2005) 397–405. DOI:10.1021/bc049784m |
| [25] | T. Hirai, M. Leolukman, C.C. Liu, One-step direct-patterning template utilizing self-assembly of POSS-containing block copolymers. Adv. Mater. 21 (2009) 4334–4338. DOI:10.1002/adma.v21:43 |
| [26] | B.B. Jiang, W. Tao, X. Lu, A POSS-based supramolecular amphiphile and its hierarchicalself-assemblybehaviors. Macromol.RapidCommun 33 (2012) 767–772. |
| [27] | W.A. Zhang, B. Fang, A. Walther, A.H.E. Müller, Synthesis via RAFT polymerization of tadpole-shaped organic/inorganic hybrid poly (acrylic acid) containing polyhedral oligomeric silsesquioxane (POSS) and their self-assembly in water. Macromolecules 42 (2009) 2563–2569. DOI:10.1021/ma802803d |
| [28] | W.A. Zhang, J.Y. Yuan, S. Weiss, Telechelic hybrid poly (acrylic acid) s containing polyhedral oligomeric silsesquioxane (POSS) and their self-assembly in water. Macromolecules 44 (2011) 6891–6898. DOI:10.1021/ma201152t |
| [29] | W. Wu, W.G. Wang, J.S. Li, Star polymers: advances in biomedical applications. Prog. Polym. Sci. 46 (2015) 55–85. DOI:10.1016/j.progpolymsci.2015.02.002 |
| [30] | Y.Y. Cui, Y.E. Ren, X.X. Liu, Synthesis of methyl methacrylate star-branched polymer with divinylbenzene as a linking agent via controlled/living photopolymerization. Chin. Chem. Lett. 23 (2012) 985–988. DOI:10.1016/j.cclet.2012.06.026 |
| [31] | J.B. Li, J. Ren, Y. Cao, W.Z. Yuan, Synthesis of biodegradable pentaarmed star-block copolymersviaanasymmetricBIS-TRIScorebycombinationofROPandRAFT:from star architectures to double responsive micelles. Polymer 51 (2010) 1301–1310. DOI:10.1016/j.polymer.2010.01.037 |
| [32] | S.S. Sheiko, B.S. Sumerlin, K. Matyjaszewski, Cylindrical molecular brushes: synthesis, characterization, and properties. Prog. Polym. Sci. 33 (2008) 759–785. DOI:10.1016/j.progpolymsci.2008.05.001 |
| [33] | M.H. Stenzel, T.P. Davis, Star polymer synthesis using trithiocarbonate functional b-cyclodextrin cores (reversible addition-fragmentation chain-transfer polymerization). J. Polym. Sci. Part A: Polym. Chem. 40 (2002) 4498–4512. DOI:10.1002/(ISSN)1099-0518 |
| [34] | X.W. Xu, J.L. Huang, Synthesis and characterization of amphiphilic copolymer of linear poly (ethylene oxide) linked with [poly (styrene-co-2-hydroxyethyl methacrylate)-graft-poly (e-caprolactone)] using sequential controlled polymerization. J. Polym. Sci. Part A: Polym. Chem. 44 (2006) 467–476. DOI:10.1002/(ISSN)1099-0518 |
| [35] | Y.X. Xing, J. Peng, K. Xu, Polymerizable molecular silsesquioxane cage armored hybrid microcapsules with in situ shell functionalization. Chem. Eur. J. 22 (2016) 2114–2126. DOI:10.1002/chem.201504473 |
 2016, Vol. 27
2016, Vol. 27 




