b State Key Laboratory of Molecular Engineering of Polymers, Department of Macromolecular Science, Fudan University, Shanghai 200433, China
Supercapacitors' known as one of remarkable energy storage device' have attracted great attention in recent years' owing to its high power density' long cycle life' and high rate capability [1]. Based on the energy storage mechanism' supercapacitors store charges using either ion adsorption at the interface of electrode and electrolyte (electric double-layer capacitors' EDLCs) or fast surface redox reactions (pseudocapacitors) [2]. The commercial supercapacitors are mainly based on activated carbon with unsatisfactory specific capacitances (<120Fg-1) and relatively lower charge/discharge rates (<10Ag-1). It is a challenge to improve specific capacitance of the supercapacitor electrodes without sacrificing the rate performance or cycle life. Recently' three-dimensional graphene frameworks (3DGFs) such as foams' aerogels' and sponges' have come to be considered promising electrode materials for EDLCs' due to the highly interconnected pore structure' fast ion and mass transfer' high percolated electronic transport' low density' and easy access to adsorption sites [3]. However' pure 3DGF electrode usually give relatively low specific capacitance due to the limited capacitance contribution from the double layer storage mechanism [4]. On the other hand' π-functional conducting polymers' especially polyaniline (PANI) and polypyrrole (PPy)' that exhibit much higher theoretical capacitance than carbon materials' have been used as electrode materials for pseudocapacitors due to their superior redox properties and easy synthesis. [5] Unfortunately' their poor cycling stability limits the practical application owing to the swelling and shrinkage during the charge/discharge process [6]. Incorporating conducting polymers into the 3DGFs to construct three-dimensional graphene/conducting polymer (3DGCP) composites has the potential to take advantage of both components to achieve greatly improved overall electrochemical performance. As a typical example' using the 3D graphene hydrogel film prepared by filtration as a substrate' it was found that PANI had the capability to offer a combination of high capacitance' excellent rate performance and long-term cyclability [7]. Therefore' in recent years' tremendous efforts have been devoted to the development of synthetic methods for 3DGCP composites with various morphologies' structures and properties' in order to satisfy the requirements arising from energy storage applications. Although several reviews have involved introduction of 3DGCP composites' but a systematic summary focusing on their synthesis and applications in energy storage have not been reported [8]. In this short review' we present recent advances in the synthetic strategies of 3DGCP composites applied in supercapacitors, which could be roughly divided into four categories: (i) directly incorporating polymers during reduction self-assembly of graphene oxide (GO) sheets, (ii) in situ polymerization of monomers in pre-synthesized 3DGFs, (iii) one-step in situ polymerization of monomers and assembly of GO, (iv) template methods.
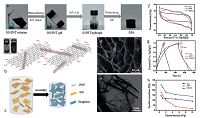
2. Directly incorporating polymers during reduction selfassembly of GOReduction self-assembly of GO is an effective way which induce chemically converted graphene sheets to cross-link with each other via partial π-π stacking interaction to form a 3D interconnected porous structure. Multiple reduction strategies, including chemical reduction, hydrothermal/solvothermal reduction, and electrochemical reduction, have been developed for the preparation of 3DGFs [9]. Direct incorporation of conducting polymers during reduction self-assembly process to synthesize 3DGCP composites is convenient and has gained increasing attention because these conducting polymers not only endow 3DGCP composites with novel electrochemical performance and characteristic, but also act as spacers to further minimize the agglomeration of graphene sheets [10]. Especially, nanostructured conducting polymers, such as nanotubes [11], nanowires [12], nanofibers [13], nanorods [14], have been often designed to incorporate with GO which then self-assembled into 3DGCP composites through a reduction route. For example, Ye et al. introduced pre-prepared one-dimensional hollow polypyrrole nanotubes (PNTs) into GO aqueous dispersion. As shown in Fig. 1, the PNTs may interact with more than one neighboring GO sheets via π-π stacking interactions. Then by a simple chemical reduction, the PNTs were successfully wrapped into graphene layers during the 3D self-assembly of reduced graphene oxide (rGO) and served as spacers to further prevent graphene from restacking and agglomeration. The as-prepared hierarchically porous 3D rGO/PNT framework could provide large accessible specific surface areas and abundant pore structures, and thus possessed high specific capacitance of 253 F g-1, which was superior to the pure 3DGFs and PNTs [11]. In the same way, a 3D rGO/PANI composite was also fabricated by a hydrothermal reduction process using GO precursor and pre-synthesized PNAI nanowires as the feedstock. The PANI nanowires could serve as spacers to effectively tailor the microstructures and electrochemical performances of 3D rGO/PANI composite, and the supercapacitor electrodes based on such material exhibited a high specific capacitance of 520 F g-1 and excellent cycling stability [12].
|
Download:
|
| Figure 1. (a) Photographs of time-dependent formation process of 3D rGO/PNT composites. (b) Schematic illustration for the interactions between GO sheets and PNTs. (c) The formation process of 3D rGO/PNT composites. (d) SEM and (e) TEM images of 3D rGO/PPy material. (f) Cyclic voltammetry curves of 3DGFs (GA), PNTs, and 3D rGO/PNT (GPA) composites (g) Galvanostatic charge-discharge curves of GA, PNTs, and GPA at 1 Ag-1. (h) Specific capacitance of GA, PNTs, and GPA as a function of current density. Reproduced with permission from ref. [11]. Copyright 2014 American Chemical Society. | |
Flexible, light-weight and wearable supercapacitors have attracted significant attention in energy storage because of their potential application in portable electronic devices, flexible displays, electronic papers and mobile phones. 3DGCP composites are attractive candidates for high performance supercapacitors. In order to utilize them in real device, it is necessary to improve the capacitance and the structure stability of conducting polymers. To this end, Hu et al. recently designed a hierarchical 3D rGO/PANI nanofiber composite, in which all of PANI nanofibers were tightly wrapped inside reduced GO nanosheets skeletons, for flexible supercapacitors [13]. The overall preparation procedure was illustrated in Fig. 2. Firstly, GO solution and pre-synthesized PANI nanofibers were mixed together, and then PANI nanofibers with positive charge were GO wrapped by an electrostatic self-assembly when adjusting the pH of the solution to ~4. Finally, using GO wrapped PANI nanofibers as precursors, hydrothermal reductions were carried to form a 3D rGO/PANI porous architecture. The PANI nanofibers were wrapped by rGO nanosheets, resulting in an intimate contact between rGO nanosheets and PNAI nanofibers. The wrapping not only protected PANI during cycling but also offered rapid electron transfer between PANI nanofibers and rGO nanosheet networks. The as-fabricated film electrode with this unique structure showed a highest gravimetric specific capacitance of 921 F g-1 and volumetric capacitance of 391 F cm-3 at a discharge current density of 1 Ag-1. The film electrodes were further assembled with PVA/H2SO4 gel electrolyte to build the solid-state supercapacitors, which gave a high specific capacitance of211 Fg-1 (1 Ag-1), ahigharealcapacitanceof0.9 Fcm-2, anda volumetric capacitance of 25.6 F cm-3. The supercapacitors also exhibited outstanding rate capability (~75% retention at 20 A g-1) and excellent cycling stability(100% retention at10 A g-1 for 2000 cycles) under bending state without structural failure and performance loss.
|
Download:
|
| Figure 2. (a) Schematic illustration of the preparation of the flexible hybrid hydrogel film electrodes with continuous 3D rGO nanosheet network skeletons embedded with rGO- wrapped PANI nanofibers. (b) Comparison of gravimetric specific capacitances and (c) comparison of the volumetric specific capacitance of the as-prepared hydrogel film electrodeswith different mass loadings ofPANI at different current densities. (d) Cycling stability of the solid-state device at a current density of10 Ag-1 under bending state. The inset shows the last 10 galvanostatic charge/discharge curves for the device. (e) Cyclic voltammetry curves of the 3D rGO/PANI (50%) hybrid-based flexible solid-state supercapacitor at different bending angles with a scan rate of10 mV s-1. The insets show a photograph and a schematic diagram of the flexible solid-state device under the bending state. (f) The energy and power densities of the flexible supercapacitor in comparison with landmark all-solid-state supercapacitors. (g) Photograph of an electronic LED tree (2.5 V) powered by three supercapacitors in series bended with a "W" shape. Reproduced with permission from ref. [13]. Copyright 2016 Nature Publishing Group. | |
3. In situ polymerization of monomers in pre-synthesized 3DGFs
3DGCP composites can be also prepared by in situ oxidative polymerization or electro-polymerization (electrochemical deposition) of monomers in pre-synthesized 3DGFs [15]. The method does not require the complicated functionalization and dispersion of individual GO sheets, which is beneficial for overcoming any process incompatibility and thus, the composites exhibit superior mechanical properties and electrical conductivities. Not limited to reduction self-assembly of GO, chemical vapor deposition (CVD) [16], thermolytic cracking [17], and in situ unzipping carbon nanotubes [18], etc. have been also used to produce the pre-synthesized 3DGFs with more controlled and uniform morphologies and structures. Compared with in situ oxidative polymerization of monomer that require firstly synthesizing 3DGCP composites followed by slurry coating on conductive substrates [19], a convenient electrochemical deposition strategy can be used to produce 3DGCP electrodes directly. Moreover, the electrochemical deposition technique have been also found effective for making the ordered structures and as-fabricated hybrid electrodes have shown excellent electrochemical performance. For example, using a 3D graphene foam by template-directed growth as a free-standing working electrode, Yu et al. prepared 3D graphene/PANI composite electrode through an one-step electrochemical deposition in which highly ordered polyaniline nanocone arrays were tightly attached to the surface of the 3D graphene electrode (Fig. 3a-f). The as-prepared electrode possessed high specific capacitance of 751 Fg-1 in 1 mol L-1 HClO4 at a current density of 1.0 A g-1, and remain 93.2% of initial capacitance after 1000 charging/discharging cycles [20]. Recently, a novel 3D bicontinuous nanotubular graphene/PPy composite has been also developed by electrodepositing PPy on CVD synthesized nanotubular graphene (Fig. 3g-i).The nanotubular hybrid composites used as free standing electrodes in supercapacitor showed high specific capacitance of 509 Fg-1, excellent cycle stability and outstanding rate performance. Moreover, all-solid-state flexible supercapacitor fabricated by the hybrid material and PVA/H2SO4 gel electrolyte showed the specific capacitance as high as 514 F g-1 at a discharge current density of 0.2 A g-1 and the maximum energy density of 21.6 W h kg-1 at power density of 32.7 kW kg-1. The outstanding supercapacitor performance of the 3D nanotubular graphene/PPy composites benefited from the high electric conductivity and excellent electrochemical and mechanical stability of the nanoporous graphene support which had unique bicontinuous open porosity [21]. In addition, two-step consecutive electrochemical deposition process has also been used for fabrication of 3DGCP composites. Taking 3D rGO/APNI as an example, 3DGFs were firstly grown directly on the electrode by electrochemical reduction of a concentrated GO dispersion, and then the conducting polymers were electrochemically deposited onto this 3D rGO matrix, yielding 3D rGO/APNI porous composite materials. The as-prepared 3D rGO/APNI composites had a conductive graphene network as the matrix, onto which the second component is homogeneously coated, and exhibited a high specific capacitance of716 F g-1 at 0.47 Ag-1 [15].

|
Download:
|
| Figure 3. (a) One-step electrodeposition process ofPANI on the surface of 3D graphene network. (b) SEM images of vertically aligned PANI nanocone arrays on the surface of 3D graphene with electrode-position time of 7500 s. (c) Schematic illustration of ion diffusion and charge transfer for the 3D graphene/PANI nanocone array electrode. (d) SEM images of vertically aligned PANI nanocone arrays on the surface of 3D graphene with lower resolution. (e) Scan-rate dependent specific capacitance and high-rate capacitance percentage of 3D graphene/PANI nanocone array electrodes with different electrode-position time. (f) Charge-discharge cycles of 3D graphene/PANI nanocone array electrodes. Reproduced with permission from ref. [20]. Copyright 2015 Elsevier Ltd. (g) Schematic of preparation steps of 3D nanotubular graphene/PPy composite by chemical vapor deposition. (h) STEM image of 3D nanotubular graphene/PPy composite. (i) Power density versus energy density of 3D nanotubular graphene/PPy composite and recently reported data from the literature. Reproduced with permission from ref. [21]. Copyright 2016 Elsevier Ltd. | |
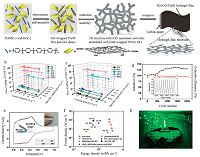
The electrochemical deposition has been proved to be an effective and green approach to prepare 3DGCP composites, and is attractive for electrochemical applications, since high surface area and low electrolyte transport resistance make these electro- synthesized composites suitable for high-performance electrode materials in electrochemical devices. However, the shortcoming of electrodepositing polymers on 3DGFs is that only a thin layer of conducting polymers can be coated on the surface of graphene, resulting in small areal capacitance. Thus, it is highly desirable to fill the macro-sized pores with pseudocapacitive materials with smaller pores in order to boost its areal capacitance. To this end, Zhang etal. designed and constructed a bicontinuous macroporous graphene foam composed of few-layered graphene sheets, and used it as a highly conductive platform to grow mesoporous polyaniline via electrodeposition and inkjet techniques [22]. The preparation procedure was illustrated in Fig. 4. PANI thin film was first coated on 3DGF by a galvanostatic electro-polymerization method at a current density of 2 mA cm-2. Then, aniline monomers were inkjet printed into the pores of the PANI-coated graphene foam and subsequently oxidative polymerized to produce porous PANI hydrogel with the help of phytic acid. The coating PANI thin layer on the surface of 3DGF via electrodeposition was of importance for changing the hydrophobic surface to a hydrophilic one and for the subsequent filling of the mesoporous PANI network into the macroporous graphene foam. The inkjet method was effective to fill the aniline monomer with a defined volume into the pores of graphene foam in order to regulate the loading of porous PNAI. When used as electrode materials for supercapacitors, the 3D graphene/PANI network with high porosity rendered a large areal capacitance of over 1700 mF cm-2,which was over two times enhancement in comparison with the pure graphene foam and PANI thin layer coated one. The ultrahigh areal capacitance could attributed to the following features: (1) The good hydrophilicity of the samples facilitated the penetration of electrolytes into the inner surface. (2) The good electrical conductivity of the the graphene foam facilitated the electrical transfer among the electrode materials, leading to low internal resistance. (3) The porous PANI filling into the pores of the graphene foam provided large pesudocapacitance on the basis of the redox reaction.

|
Download:
|
| Figure 4. (a) Illustration of the preparation procedure of PANI-filled 3DGF via a two-step method. (b and c) SEM images of PANI-coated 3DGF foam via electrochemical polymerization for deposition times of 300 s. (d and e) SEM images for deposition times of 1800 s. (f and g) SEM images for deposition times of 3600 s. (h) Cycle voltammetry curves, (i) charge-discharge curves, (j) and areal capacitances of PNAI-coated 3DGF foam with different deposition times and PANI-filled 3DGF foam. (k) Cycling stability of 3D graphene/PANI foam. Reproduced with permission from ref. [22]. Copyright 2014 American Chemical Society. | |
4. One-step in situ polymerization of monomers and assembly of GO
To simplify the preparation process, one-step in-situ polymerization of monomers and assembly of GO have also been developed to construct 3DGCP composites. For example, 3D rGO/PPy composite hydrogel electrodes were fabricated by a one-step hydrothermal process in the presence of KMnO4. In this case, pyrrole (Py) monomers were polymerized with KMnO4 as an oxidant to form PPy on the graphene surface under heating condition. Meanwhile, GO sheets were thermally reduced and self-assembled into a 3D porous structure [23]. It is well known that the aniline can be polymerized via oxidization under an acidic condition using a variety of oxidants. On the other hand, GO sheets can be chemically reduced into rGO by certain reductants including aniline. Based on these, Shen et al. fabricated 3D rGO/ PANI composites through one-step in situ redox reaction between GO and aniline. The redox reaction could take place instantly at room temperature [24]. In another similar case, Sun et al. developed 3D rGO/PPy hybrid aerogels by a spontaneous assembly (i.e. in situ redox) method in the absence of any other reductants or oxidizing agents at room temperature (Fig. 5) [25]. The redox reaction between GO and Py resulted in reduction of GO and polymerization of Py in the meantime. The as-prepared 3D rGO/ PPy aerogels had a microporous network with interconnected pores with a wide size distribution due to randomly self-assembled intertwisted rGO nanosheets. The solid walls of the pores were thin, which demonstrated that the stacking of rGO nanosheets was inhibited by PPy remarkably. The large surface areas and pore volumes made the 3D rGO/PPy aerogels exhibited outstanding electrochemical supercapacitor performance. Thus, one-step assembly through redox reaction of monomers and GO is environmentally friendly, low cost, and easy to scale-up. In addition, a one-step electrochemical co-deposition method could also be employed to prepare 3DGCP composite hydrogel in a mixture dispersion of GO and monomers, in which GO sheets were electrochemically reduced to form a rGO hydrogel layer on the substrate electrode, and monomers were polymerized and homogeneously coated on the surfaces of rGO sheets at positive potential. The composites are porous, conductive and electroactive, and thus can be directly used as electrode materials for supercapacitors without using binders and conductive fillers, exhibiting high areal specific capacitances and good electrochemical stability [26]. For example, 3D rGO/PPy composite films were electrochemically deposited from the aqueous solutions containing Py monomer and sulfonated graphene at a constant potential of 0.7 V, and the prepared composite electrode possessed high specific capacitance of 285 F g-1 at a current density of 0.5 A g-1, and remained 92% of initial capacitance after 800 charging/discharging cycles at a charging/discharging rate of 0.5 Ag-1 [27].

|
Download:
|
| Figure 5. (a) Schematic illustration of the formation mechanism of 3D rGO/PPy aerogels by one-step in situ redox reaction. (b and c) SEM images of 3D rGO/PPy aerogels with different magnifications. (d and e) Nitrogen adsorption-desorption isotherm of 3D rGO/PPy aerogels and the corresponding pore-size distributions. (f) Cycle voltammetry curves (5 mV s-1) of 3D rGO/PPy aerogels and 3DGF aerogel. (g) Cycle voltammetry curves of 3D rGO/PPy aerogels at different scan rates. (h) Variation in the specific capacitance of 3D rGO/PPy aerogels and 3DGF aerogel as a function of current density. (i) Nyquist plots of 3D rGO/PPy aerogels and 3DGF aerogel (the insert shows the equivalent circuit model of Nyquist plots). Reproduced with permission from ref. [25]. Copyright 2014 The Royal Society of Chemistry. | |
5. Template method
Hollow micro/nanostructured materials have been recognized as one type of promising materials in energy-related fields. Directly mixing GO with various polymer templates is an convenient and efficient route to hollow 3DGCP composites. For highly conductive pathways by bridging individual PANI hollow spheres could offer highly conductive pathways by bridging individual PANI hollow spheres together, thus facilitating the rate and cycling performance of supercapacitor. The specific capacitance of 3D rGO/PANI hybrids could reach 614 F g-1 at a current density of 1 A g-1, and retained 90% after 500 charging/discharging cycles at a current density of 1 A g-1, indicating a good cycling stability [28]. For another case, using polymethyl methacrylate (PMMA) colloidal particles as a template, Trung et al. produced hollow 3D rGO/PANI composite electrodes by assembling GO sheets on the surface of PMMA colloidal particles, followed by the synthesis of PANI and removal of the core PMMA particles (Fig. 6a-c). The supercapacitor based on the 3D rGO/PANI electrodes showed a specific capacitance of 331 Fg-1 at a current density of 1Ag-1 and excellent cycling stability with a 14% loss of the capacitance after 500 charging/ discharging cycles. Furthermore, due to the mechanical stability of 3D rGO/PANI composites, the 3D rGO/PANI electrodes could be also used for flexible supercapacitors, which exhibited a specific capacitance of 182 Fg-1 at the bending state with good cycling stability [29]. Recently, a novel 3D rGO/PANI hybrid hollow sphere has been also designed and fabricated via layer-by layer (LBL) assembly of negatively-charged rGO and positively charged PANI on PS microsphere, followed by the removal of the PS template (Fig. 6d-f). When used as the electrode materials for supercapacitor, the specific capacitance of the 3D rGO/PANI hybrid hollow sphere reached 381 F g-1 at a current density of 4.0 A g-1, and maintained 83% after 1000 charging/discharging cycles. The high specific capacitance of the3D rGO/PANI hybrid hollow sphere could be attributed to its unique hollow structure which provides a large accessible surface area and facilitate the charge and ion transport. In addition, its specific capacitance could be facilely tailored by changing the assembly cycle number [30].

|
Download:
|
| Figure 6. (a) Schematic illustration of the fabrication of 3D rGO/PANI hybrid hollow balls using PMMA particles as template. (b) SEM and (c) TEM images of 3D rGO/PANI hollow balls. Reproduced with permission from ref. [28]. Copyright 2014 Elsevier Ltd. (d) Schematic illustration of the fabrication procedure of 3D rGO/PANI hollow sphere by layer- by layer assembly technology. (e) SEM and (f) TEM images of 3D rGO/PANI multi-layer hollow sphere. Reproduced with permission from ref. [29]. Copyright 2015 Elsevier Ltd. | |
6. Summary and perspectives
Benefiting from the rapid development of synthesis methods in recent years, a number of 3DGCP composites with different structures, morphologies and properties have been prepared. This review article summarizes recent advances in synthesis of 3DGCP composites applied in supercapacitors. The rapid growth of this field assures us that 3DGCP composites will be a new generation of powerful materials in energy storage fields. With more synthetic strategies being developed, a wide range of advanced 3DGCP composites with elaborated structures can be readily prepared on a large scale for supercapacitor applications. Beyond this exciting progress to date, we believe that there are considerable challenges and opportunities remaining for continued investigation. Firstly, the fundamental 3D assembly mechanism between graphene sheets and conducting polymers, particularly, the interactions between the two components, need to be further studied for promoting their electrochemical performance. Secondly, the stability of conducting polymers should also be further improved by design and construction of new 3DGCP composites, because it is much needed for real-world applications. Thirdly, 3DGCP composites simultaneously exhibit flexibility, strechability, compressibility have not been realized but are highly desired for smart and wearable electronic products. Finally, economic, green and scalable production of 3DGCP composites is a critical issue for their practical widespread applications.
Acknowledgments This work was supported by The Program for Professor of Special Appointment (Eastern Scholar) at Shanghai Institutions of Higher Learning (No. TP2015002), the National Natural Science Foundation of China (No. 51403099).| [1] | (a) P. Simon, Y. Gogotsi, Materials for electrochemical capacitors, Nat. Mater. 7(2008) 845-854; (b) Y.X. Xu, G.Q. Shi, X.F. Duan, Self-assembled three-dimensional graphene macrostructures: synthesis and applications in supercapacitors, Acc. Chem. Res. 48(2015) 1666-1675; (c) L.B. Zhang, S.R. Yang, J.Q. Wang, Y. Xu, X.Z. Kong, A facile preparation and electrochemical properties of nickel based compound-graphene sheet composites for supercapacitors, Chin. Chem. Lett. 26(2015) 522-528. |
| [2] | (a) X.H. Cao, Z.Y. Yin, H. Zhang, Three-dimensional graphene materials: preparation, structures and application in supercapacitors, Energy Environ. Sci. 7(2014) 1850-1865; (b) Y.L. Shao, M.F. EI-Kady, L.J. Wang, et al., Graphene-based materials for flexible supercapacitors, Chem. Soc. Rev. 44(2015) 3639-3665. |
| [3] | (a) M.H. Yu, Y.C. Huang, C. Li, et al., Building three-dimensional graphene frameworks for energy storage and catalysis, Adv. Funct. Mater. 25(2015) 324-330; (b) Y.X. Xu, C.Y. Chen, Z.P. Zhao, et al., Solution processable holey graphene oxide and its derived macrostructures for high-performance supercapacitors, Nano Lett. 15(2015) 4605-4610. |
| [4] | S. Mao, G.H. Lu, J.H. Chen. Three-dimensional graphene-based composites for energy applications. Nanoscale 7 (2015) 6924–6943. DOI:10.1039/C4NR06609J |
| [5] | K.H.S. Lessa, Y. Zhang, G.A. Zhang, F. Xiao, S. Wang. Conductive porous sponge-like ionic liquid-graphene assembly decorated with nanosized polyaniline as active electrode material for supercapacitor. J. Power Sources 302 (2016) 92–97. DOI:10.1016/j.jpowsour.2015.10.036 |
| [6] | P.P. Yu, X. Zhao, Z.L. Huang, Y.Z. Li, Q.H. Zhang. Free-standing three-dimensional graphene and polyaniline nanowire arrays hybrid foams for high-performance flexible and lightweight supercapacitors. J. Mater. Chem. A 2 (2014) 14413–14420. DOI:10.1039/C4TA02721C |
| [7] | Y.F. Wang, X.W. Yang, L. Qiu, D. Li. Revisiting the capacitance of polyaniline by using graphene hydrogel films as a substrate: the importance of nano-architecturing. Energy Environ. Sci. 6 (2013) 477–481. DOI:10.1039/C2EE24018A |
| [8] | (a) Y.Q. Sun, G.Q. Shi, Graphene/polymer composites for energy applications, J. Polym. Sci. B 51(2013) 231-253; (b) J. Chen, C. Li, G.Q. Shi, Graphene materials for electrochemical capacitors, J. Phys. Chem. Lett. 4(2013) 1244-1253; (c) Y.Q. Sun, Q. Wu, G.Q. Shi, Graphene based new energy materials, Energy Environ. Sci. 4(2011) 1113-1132. |
| [9] | (a) Y.X. Xu, Z.Y. Lin, X. Zhong, et al., Holey graphene frameworks for highly efficient capacitive energy storage, Nat. Commun. 5(2014) 4554-4561; (b) Y.X. Xu, Z.Y. Lin, X. Zhong, et al., Solvated graphene frameworks as highperformance anodes for lithium-ion batteries, Angew. Chem. Int. Ed. 54(2015) 5345-5350; (c) Y. Zhao, C.G. Hu, Y. Hu, et al., A versatile, ultralight, nitrogen-doped graphene framework, Angew. Chem. Int. Ed. 51(2012) 11371-11375; (d) Y.X. Xu, Z.Y. Lin, X.Q. Huang, et al., Flexible solid-state supercapacitors based on three-dimensional graphene hydrogel films, ACS Nano 7(2013) 4042-4049; (e) Y.X. Xu, Z.Y. Lin, X.Q. Huang, et al., Functionalized graphene hydrogel-based high-performance supercapacitors, Adv. Mater. 25(2013) 5779-5784. |
| [10] | H. Zhou, T. Ni, X.T. Qing, et al. One-step construction of graphene-polypyrrole hydrogels and their superior electrochemical performance. RSC Adv. 4 (2014) 4134–4139. DOI:10.1039/C3RA44647F |
| [11] | S.B. Ye, J.C. Feng. Self-assembled three-dimensional hierarchical graphene/polypyrrole nanotube hybrid aerogel and its application for supercapacitors. ACS Appl. Mater. Interfaces 6 (2014) 9671–9679. DOI:10.1021/am502077p |
| [12] | F. Yang, M.W. Xu, S.J. Bao, H. Wei, H. Chai. Self-assembled hierarchical graphene/polyaniline hybrid aerogels for electrochemical capacitive energy storage. Electrochim. Acta 137 (2014) 381–387. DOI:10.1016/j.electacta.2014.06.017 |
| [13] | (a) N.T. Hu, L.L. Zhang, C. Yang, et al., Three-dimensional skeleton networks of graphene wrapped polyaniline nanofibers: an excellent structure for high-performance flexible solid-state supercapacitors, Sci. Rep. 6(2016) 19777; (b) S. Li, K.W. Shu, C. Zhao, et al., One-step synthesis of graphene/polypyrrole nanofiber composites as cathode material for a biocompatible zinc/polymer battery, ACS Appl. Mater. Interfaces 6(2014) 16679-16686. |
| [14] | X.B. Liu, P.B. Shang, Y.B. Zhang, et al. Three-dimensional and stable polyanilinegrafted graphene hybrid materials for supercapacitor electrodes. J. Mater. Chem. A 2 (2014) 15273–15278. DOI:10.1039/C4TA03077J |
| [15] | (a) K.W. Chen, L.B. Chen, Y.Q. Chen, H. Bai, L. Li, Three-dimensional porous graphene-based composite materials: electrochemical synthesis and application, J. Mater. Chem. 22(2012) 20968-20976; (b) K. Chi, Z.Y. Zhang, J.B. Xi, et al., Freestanding graphene paper supported threedimensional porous graphene-polyaniline nanocomposite synthesized by inkjet printing and in flexible all-solid-state supercapacitor, ACS Appl. Mater. Interfaces 6(2014) 16312-16319; (c) S.B. Kulkarni, U.M. Patil, I. Shackery, et al., High-performance supercapacitor electrode based on a polyaniline nanofibers/3D graphene framework as an efficient charge transporter, J. Mater. Chem. A 2(2014) 4989-4998. |
| [16] | (a) X.H. Cao, Y.M. Shi, W.H. Shi, et al., Preparation of novel 3D graphene networks for supercapacitor applications, Small 7(2011) 3163-3168; (b) F. Yavari, Z.P. Chen, A.V. Thomas, et al., High sensitivity gas detection using a macroscopic three-dimensional graphene foam network, Sci. Rep. 1(2011) 166; (c) L. Zhang, D. DeArmond, N.T. Alvarez, et al., Beyond graphene foam, a new form of three-dimensional graphene for supercapacitor electrodes, J. Mater. Chem. A 4(2016) 1876-1886. |
| [17] | (a) D.Q. Fan, Y. Liu, J.P. He, Y.W. Zhou, Y.L. Yang, Porous graphene-based materials by thermolytic cracking, J. Mater. Chem. 22(2012) 1396-1402; (b) X. Zhang, K.S. Ziemer, K. Zhang, et al., Large-area preparation of high-quality and uniform three-dimensional graphene networks through thermal degradation of graphene oxide-nitrocellulose composites, ACS Appl. Mater. Interfaces 7(2015) 1057-1064; (c) W.W. Liu, H. Li, Q.P. Zeng, et al., Fabrication of ultralight three-dimensional graphene networks with strong electromagnetic wave absorption properties, J. Mater. Chem. A 3(2015) 3739-3747; (d) M.Q. Xue, D. Chen, X.S. Wang, J.T. Chen, G.F. Chen, Carbonized poly(vinylidene fluoride)/graphene oxide with three-dimensional multiscale-pore architecture as an advanced electrode material, J. Mater. Chem. A 3(2015) 7715-7718. |
| [18] | (a) Q.Y. Peng, Y.B. Li, X.D. He, et al., Graphene nanoribbon aerogels unzipped from carbon nanotube sponges, Adv. Mater. 26(2014) 3241-3247; (b) Y.Y. Zhang, Z. Zhen, Z.L. Zhang, et al., In-situ synthesis of carbon nanotube/ graphene composite sponge and its application as compressible supercapacitor electrode, Electrochim. Acta 157(2015) 134-141. |
| [19] | H.L. Liu, Y. Wang, X.L. Gou, et al. Three-dimensional graphene/polyaniline composite material for high-performance supercapacitor applications. Mater. Sci. Eng. B 178 (2013) 293–298. DOI:10.1016/j.mseb.2012.12.002 |
| [20] | M. Yu, Y.X. Ma, J.H. Liu, S.M. Li. Polyaniline nanocone arrays synthesized on threedimensional graphene network by electrodeposition for supercapacitor electrodes. Carbon 87 (2015) 98–105. DOI:10.1016/j.carbon.2015.02.017 |
| [21] | H. Kashani, L.Y. Chen, Y. Ito, et al. Bicontinuous nanotubular graphene-polypyrrole hybridforhighperformanceflexiblesupercapacitors. NanoEnergy 19 (2016) 391–400. |
| [22] | J.T. Zhang, J. Wang, J.E. Yang, et al. Three-dimensional macroporous graphene foam filled with mesoporous polyaniline network for high areal capacitance. ACS Sustain. Chem. Eng. 2 (2014) 2291–2296. DOI:10.1021/sc500247h |
| [23] | X.Y. Gu, Y. Yang, Y. Hu, et al. Facile fabrication of graphene-polypyrrole-Mn composites as high-performance electrodes for capacitive deionization. J. Mater. Chem. A 3 (2015) 5866–5874. DOI:10.1039/C4TA06646D |
| [24] | W.Z. Shen, Y.M. Wang, J. Yan, H.X. Wu, S.W. Guo. Enhanced electrochemical performance of lithium iron(II) phosphate modified cooperatively via chemically reduced graphene oxide and polyaniline. Electrochim. Acta 173 (2015) 310–315. DOI:10.1016/j.electacta.2015.05.071 |
| [25] | R. Sun, H.Y. Chen, Q.W. Li, Q.J. Song, X.T. Zhang. Spontaneous assembly of strong and conductive graphene/polypyrrole hybrid aerogels for energy storage. Nanoscale 6 (2014) 12912–12920. DOI:10.1039/C4NR03322A |
| [26] | Q.Q. Zhou, Y.R. Li, L. Huang, C. Li, G.Q. Shi. Three-dimensional porous graphene/ polyaniline composites for high-rate electrochemical capacitors. J. Mater. Chem. A 2 (2014) 17489–17494. DOI:10.1039/C4TA03639E |
| [27] | A.R. Liu, C. Li, H. Bai, G.Q. Shi. Electrochemical deposition of polypyrrole/sulfonated graphene composite films. J. Phys. Chem. 114 (2010) 22783–22789. |
| [28] | W. Fan, C. Zhang, W.W. Tjiu, et al. Graphene-wrapped polyaniline hollow spheres as novel hybrid electrode materials for supercapacitor applications. ACS Appl. Mater. Interfaces 5 (2013) 3382–3391. DOI:10.1021/am4003827 |
| [29] | N.B. Trung, T.V. Tam, H.R. Kim, et al. Three-dimensional hollow balls of graphene-polyaniline hybrids for supercapacitor applications. Chem. Eng. J. 255 (2014) 89–96. DOI:10.1016/j.cej.2014.06.028 |
| [30] | J. Luo, Q. Ma, H.H. Gu, Y. Zheng, X.Y. Liu. Three-dimensional graphene-polyaniline hybrid hollow spheres by layer-by-layer assembly for application in supercapacitor. Electrochim. Acta 173 (2015) 184–192. DOI:10.1016/j.electacta.2015.05.053 |
 2016, Vol. 27
2016, Vol. 27 


