b College of Chemistry and Chemical Engineering, Xianyang Normal University, Xianyang 712000, China
Organic field-effect transistors (OFETs) have gained great attention over the last years because of their potential applications in low-cost, flexible, and large-area electronic products, such as digital displays, electronic paper, radio frequency identification tags, and label-free sensors [1-3].
Due to their unique merits such as bioadhension, biocompatibility, biodegradability and no need for chemical synthesis, natural biological materials, namely, DNA [4], carbohydrates [5], peptides and proteins [6, 7] etc., have been extensively integrated in OFETs devices fabrication as gate substrate, dielectrics and organic semiconductor (OSC). If used properly, these natural materials can actually simplify the OFET fabrication process, reduce the costs, and enhance the performance of the devices. So, the incorporation of natural biological materials in OFETs unfolds new perspectives for the exploitation of biosysteμS and holds much promise in bioelectronics application [8].
In this mini-review, we summarize and discuss the recent advances of proteins as functional interlayers in OFETs. This review is mainly restricted to the discussion ofback-gate OFETs which are made from a gate electrode, a dielectric layer, an OSC, a source and a drain electrode from bottom to top in turn. Specific focus is given
on the proteins integrated into the device structure, either to act as dielectric materials or to perform as the functional interlayer between the dielectric and the OSC. At last, the possibility of amyloid, a special type of protein structures with multiple excellent properties, serving as useful building blocks for OFET is also discussed.
2. Proteins as dielectric layersAmong the structural layers in OFET architectures, proteins can serve as dielectric layers for the majority of naturally occurring proteins have insulating properties. Furthermore, proteins are biodegradable and often have unique properties that cannot be easily achieved by conventional organic or inorganic insulating materials. In the subsequent sections, the development of OFETs based on protein dielectrics is discussed. Among the most used proteins employed directly as dielectrics are silk fibroin, bovine serum albumin (BSA), collagen, and gelatin. Special attention is paid to the significant enhancement of electric properties of the devices. Meanwhile, the remaining challenges are also outlined.
2.1. Silk fibroinSilk fibroin (SF) is one of the silk proteins spun by silkworμS. It is a natural biopolymer consisting of the repeated amino acids of glycine (Gly) and alanine (Ala) in alternating sequence. The structure consists of extended polypeptide chains bonded together by lateral N—H ⋅⋅⋅〇 hydrogen bonds to form antiparallel-chain pleated sheets [9]. The merits of SF, such as low cost, lightweight, and large area, make it promising potential for a wide variety of applications in organic electronic devices. To the best of our knowledge, SF was the first and one of the most reported proteins in OFETs so far.
Capelli et al. [10] integrated SF into electronic and optoelectronic devices, in which SF was used as a thin film dielectric in an OFET and an organic light emitting transistor (OLET) device. The comparisons of the mobility, threshold values and on/off ratio for silk p and n-type OFETs and respective standard poly(methyl methacrylate) (PMMA) and SiO2 devices were as shown in Table 1. The results demonstrated natural SF can be successfully used as a dielectric material for fabricating high-performance n- and p-type organic transistors and light emitting transistors.
Taking SF as the gate dielectric and a flexible poly(ethylene terephthalate) (PET) as plastic substrate. Wang et al. [11] developed a pentacene organic thin-film transistors (OTFT) with a very high field-effect mobility (μFE) value of 23.2 cm2 V-1 s-1 in the saturation regime and a low operating voltage of -3 V. Based on the detection results by atomic force microscopy (AFM) and grazing incidence X-ray diffraction (GIXRD), the authors considered that the primary roles of the SF dielectric is to increase the pentacene orthorhombic phase and to reduce the amorphous phase in the pentacene layer prepared by thermal evaporation. In a word.the operating speed of pentacene based OTFT was greatly improved by choosing SF as the gate dielectric material.
Solution-processed OFETs with SF as gating material and poly(3-hexylthiophene) (P3HT) as the semiconducting layer has been reported by Shi et al. [12]. The distinctive characteristic of such OFETs can be represented from the following aspects: low threshold of -0.77 V, low-operating voltage (0--3 V) and high carrier mobility of 0.21 cm2 V-1 s-1. The enhancement of the performance is attributed to an array of highly ordered fibers structure originated from the high content of b strands in SF dielectric, which leads to reduce the trapping sites at the semiconductor/dielectric interface.
Different from the above documents using SF as gate dielectric independently, high sensitivity OFET based NO2 gas sensors with silk fibroin (SF) and PMMA bilayer dielectric was reported by Li et al. [13]. The results revealed that the sensing properties of the OFET with PMMA/SF bilayer dielectric was significantly enhanced compared to that with the PMMA dielectric. The authors suggested that an increased saturation current and charge mobility can be caused by the interaction between the NO2 and a great quantity of hydroxyl groups (-OH) of serine and the amidogen of SF molecules, thus enhancing the performance of this kind of OFET.
2.2. Chicken albumenChicken albumen is more easily obtained, processable, and inexpensive than other biomaterials for OFET devices. Chang et al. [14] used chicken albumen without any purification as a gate dielectric in pentacene- and C60-based OFETs. The schematic of the structure of an OFET fabricated with albumen dielectrics was shown in Fig. 1. According to the method presented by the authors, a high-quality albumen dielectric layer can be prepared via spincoating and subsequent thermal treatments. The output current of these OFETs reached 1.7-5×10-6 A without obvious hysteresis and the gate leakage currents were roughly 10-10 A. Compared with common polymeric dielectrics, such as PMMA and polystyrene (PS) dielectrics, the output currents of the OFETs with albumen dielectrics were double those of ordinary polymer-based OFETs. At last, the authors suggested that the intrinsic properties of chicken albumen, such as the stability, aging effect, tolerance to temperature and moisture, loss factor under different frequency regime, etc., should be further investigated for the practical application of chicken albumen dielectrics.

|
Download:
|
| Figure 1. (a) Sketch of the OFET structure fabricated with albumen dielectrics. (b) Schematic diagram of denaturation and the cross-linking reaction of albumen protein under heat treatments. (c) Scheme for the formation of a disulfide bond between two cysteine groups on different protein chains. Reprinted with permission from Ref. [14]. © 2011 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim. | |
2.3. Bovine serum albumin (BSA)
Bovine serum albumin (BSA) is a natural protein, in which the percent of acidic and basic amino acid residues reach ca. 34% in total [15]. So BSA is known for its good hydration ability. The effect of humidity on OFETs fabricated with BSA as gate dielectric was investigated by Lee et al. [16]. The researchers found that pentacene OFETs with BSA as the gate dielectric exhibited a field-effect mobility value (μFE, sat, ) of 0.3 cm2 V-1 s-1 in the saturation regime and a threshold voltage (VTH) of ca.-16V in the VTH reduced to -0.7 V in a relative humidity of 47%. In fact, hydrated BSA can act as a natural polyelectrolyte, in which mobile ions can be generated by the dissociation of absorbed water molecules. The formation of electric double-layer capacitors (EDLCs) works well in explaining the high capacitance and low threshold voltage. Although hydrated BSA is more attractive than dry BSA in terμS of device performance, but for practical applications, the OFET device should be well packaged to avoid the influence of humidity variation.
|
|
Table 1 The parameters comparison of silk p and n-type OFETs with respective standard PMMA and SiO2 devices [10]. |
2.4. Collagen
Similar to the OFETs fabricated with BSA as gate dielectric, the pentacene organic thin film transistors (OTFTs) with collagen hydrolysate as the gate dielectric exhibited improved performance by water absorption [17]. In vacuum, the device had a field-effect mobility (μFE) value of 0.8 cm2 V-1 s-1 and an on/off ratio of 105. When the device was exposed to air, the drain current increased rapidly with time from 10-9 to 10-5 A and the μFE value increased to ca. 15.5 cm2 V-1 s-1. The enhancement of μFE was attributed to the interaction of water in air ambient and OH-groups in collagen hydrolysate. However, the increase of leakage current and accompanied reduction of on/off ratio to 5×103 was also discovered. Therefore, it is important and necessary to find an effective method able to control the amount of moisture in the collagen hydrolysate accurately before this kind of OTFT was put into practical application.
2.5. GelatinAlthough gelatin thin film is considered hydrophobic [18], a small amount of adsorbed water molecules in gelatin dielectric is enough to change the electrical performance of the OFETs dramatically. The research by Mao et al. [19] shown that gelatin can work effectively as the gate dielectric for n-type N, N-dioctyl- 3, 4, 9, 10-perylene tetracarboxylic diimide (PTCDI-C8)-based OFETs. When the operation environment was switched from vacuum to air ambient, the field-effect mobility value in the saturation regime and the threshold voltage value of the device were enhanced respectively from (0.22 cm2 V-1 s-1, 55 V) to (0.74 cm2 V-1 s-1, 2.6 V). The authors considered that various amino acid residues of gelatin may be surrounded by water locally at the molecular level; furthermore, the water molecules may interact with acidic and basic amino acid residues to form positive and negative charged ions in gelatin. The increase of the quasi-static capacitance (QSC) value of the Au/gelatin/Au structure from 5 nF cm-2 in vacuum to 9 nF cm-2 in air ambient with a relative humidity of 57% was an obvious proof of the generation of charged ions in gelatin.
3. Protein interlayer between the dielectric and the OSCProteins not only can serve as dielectric but they can also been integrated in OFETs as biointerlayer between the gate dielectric and the OSC. The main function of this biointerlayer is acting as recognition elements of OFET sensing platforμS.
3.1. StreptavidinStreptavidin (SA) is a hydrophilic tetrameric protein. It is well known that SA has an extraordinarily high affinity for biotin, and the tight binding between SA and biotin offers useful bioanalytical advantages as well as an attractive model for studying protein- ligand interactions [20].
As shown in Fig. 2A. Angione et al. [21] integrated streptavidin (SA) into an OFET structure by spin coating SA layers between the SiO2 dielectric and the poly-3-hexylthiophene (P3HT) OSC. The capability of SA layer to bind biotin molecules was completely not affected even after chloroform spin spreading. The data indicated that biotin concentration of 10 part per trillion (ppt) can cause a drastic reduction of the current flowing in the 2D transport region. The SA functional biological interlayer (FBI)-OFET response to the biotin concentration as low as 10 ppt was about nine times higher than the response to pure water. To prove that biotin-induced electronic response is due actually to the sole SA-biotin complex formation, the control experiments have been designed with bare P3HT OFET, bovine serum albumin (BSA, a protein not able to bind biotin)-OFETs as negative-blank controls. In fact.there was no response to biotin exposure be recorded in all cases (Fig. 2B). All these experiments made clear that SA FBI-OFETs is a successful device with super high sensitivity and selectivity.

|
Download:
|
| Figure 2. (A) Sketch of SA FEI-OFET structure and IDS - VG curves at VDS = -80 V measured in pure water and at different biotin concentrations. (B) The biotin calibration curve of the SA FBI-OFET (blue circles), P3HT OFET (black squares) and the BSA FBI-OFET (red diamonds). Reprinted with permission from Ref. [21]. © 2012 National Academy of Sciences. | |
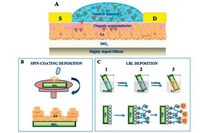
The further work about SA FBI-OFETs by Magliulo et al. [22] confirmed that the device fabricated by them is ultrasensitive label-free biosensors capable to detect biotin at 3 ppt. Both spincoating deposition process and controllable layer-by-layer (LbL) assembly had been employed to deposit the SA on the SiO2 dielectric surface (Fig. 3). The structural and morphological of two kinds of SA FBI-OFETs were characterized by X-ray specular reflectivity (XSR) and grazing incidence small angle scattering (GISAXS) [23]. The results indicated that a higher percentage of surface covering with an irregular morphology and layer discontinuity was obtained in the case of spin-deposited SA, whereas a more homogeneous coating was obtained by an LbL deposition. It was as expected that the optimization of the FBI analytical performances had been achieved through a controllable LbL deposition. But, as a kind of more simple and easy processable method, spin-coating deposition was an advisable alternative for potential low cost production of equally highly performing sensors.
|
Download:
|
| Figure 3. (A) Schematic view of the FBI-OFET biosensor comprising a functional streptavidin (SA) interlayer deposited on the SiO2 dielectric surface. (B) Illustration of the spincoating procedure for the deposition of the SA layer. (C) Illustration of the LbL deposition of the SA layer. Reprinted with permission from Ref. [23]. © 2014American Chemical Society. | |
3.2. Ferritin protein
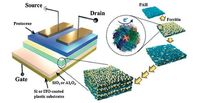
A ferritin nanoparticle (NP) is a highly stable iron-storing protein [24]. Thanks to the presence of redox sites, it can be effectively employed as charge-storing gate dielectrics in place of metal NPs for use in transistor memory devices. A novel type of OFET memory device based on ferritin NPs multilayers that displays nonvolatile memory properties was reported by Kim et al. [25]. As shown in Fig. 4, ferritin multilayer filμS were prepared through the successive layer-by-layer (LbL) deposition of cationic poly(allylamine hydrochloride) (PAH) and anionic ferritin NPs onto SiO2 -coated or Al2O3-coated plastic substrates. The good programmable memory properties had been conferred this protein NP-based OFET memory devices, namely, large memory window (ΔVT > 20 V), a fast switching speed (10 μS), high ON/OFF current ratio (> 104). When ferritin NP multilayers were deposited onto plastic substrates, and high-k Al2O3 gate dielectrics was used in place of SiO2, the device could be effectively extended to flexible OFET memory devices with the low operating voltages ( <10V) and reliable mechanical bending stability.
|
Download:
|
| Figure 4. Schematic representation ofthe OFET memory device with LbL-assembled (PAH/ferritin NP)n multilayered gate dielectrics. Reprinted with permission from Ref. [25]. © 2013 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim. | |
4. Protein as top gate
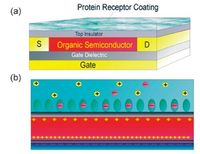
Maddalena et al. [26] have proposed an OFET with integrated sulfate binding protein (SBP) for sensing of sulfate ions. As shown in Fig. 5, the protein receptor layer was covalently coupled to a maleimide-functionalized polystyrene layer, and could be regarded as a dual-gate field-effect transistor. The only difference was that the top gate had been replaced by the bioreceptors. As depicted in Fig. 5b, when a negative bias is applied on the bottom gate, the transistor accumulates positive charge at the bottom insulator-semiconductor interface. So, positive counter charges environment, especially in different humidity or different ionic concentration. Even so, these OFETs with naturally proteins as functional biointerlayers are no doubt an emerging but really exciting novel research field, and worth of making greater efforts. will accumulate rapidly in the semiconductor with protein receptors capture negative sulfate ions. Furthermore, the authors found that the protein remained active even after being dried and the sulfate ions could be detected by a shift in the threshold voltage.
|
Download:
|
| Figure 5. (a) Schematic view of the bio-FET device. (b) Illustration of binding of sulfate ions to the receptor and charge compensation in the semiconductor. Reprinted with permission from Ref. [26]. © 2010 American Institute of Physics. | |
5. Potential application of amyloid in the OFETs
Amyloid fibrils are a common form of protein nanostructure resulting from the aggregation of soluble proteins into β-sheet rich supramolecular polymers [27]. It is previously best known by people for their association with neurodegenerative diseases including Alzheimer’s disease and Parkinson’s disease [28]. However, as a biomaterial with many excellent properties, namely, precisely assembled fibrillar structures, tunable functionalities, excellent mechanical properties, and ultra stability over a wide range of harsh conditions, amyloids have the potential to serve as useful building blocks for functional materials and bionanoelectronic devices [29]. By now, many naturally occurring proteins (e.g.., silk fibroin, bovine serum albumin, collagen and gelatin) have been integrated in the structure of OFETs, but comparable amyloid-based devices have not yet appeared. In view of the unique properties of amyloids, advanced OFETs based on amyloids would also be expected in the future.
6. ConclusionProtein-based OFETs, either baring n-type or p-type OSCs, proved to be excellent dielectrics or functional biointerlayers, resulting in devices with excellent electronic performance or recognition performance. Although the humidity affected greatly the electrical performance of the devices in some cases, the advantages of these protein integrated OFETs were also obvious. The future directions of these devices mainly lie in some aspects as follows: Firstly, it is necessary to exploit new proteins with superior stability or excellent electrochemical properties for developing the performance of OFETs. Secondly, the operation mechanisμS of protein biointerlayers are still not fully discovered, and further efforts should be made to find out the truth behind the observed phenomena. Lastly, some specific technique probleμS for practical applications are far from completely solved, for example, the function decline of OFETs with degradation of protein interlayers, the data consistency of the devices in different work
Acknowledgment We thank the funding from the National Natural Science Foundation of China (NSFC. Nos. 21374057, 51303100), the Fundamental Research Funds for the Central Universities (No. GK201502001, GK201301006), the 111 Project (No. B14041) and Program for Changjiang Scholars and Innovative Research Team in University (No. IRT-14R33), Natural Science Basic Research Plan in Shaanxi Province of China (No. 2015JM2048) as well as Open Project of State Key Laboratory of Supramolecular Structure and Materials (No. sklssm201626). W. Z. thanks the support of the Project funded by China Postdoctoral Science Foundation (No. 2014M560747).Natural Science Basic Research Plan in Shaanxi Province of China (No. 2016JM5024) and the Scientific Research Program funded by Shaanxi Provincial Education Department (No. 14JK1801).| [1] | X.K. Gao, Z. Zhao. High mobility organic semiconductors for field-effect transistors. Sci. China Chem. 58 (2015) 947–968. DOI:10.1007/s11426-015-5399-5 |
| [2] | Y.G. Wen, Y.Q. Liu, Y.L. Guo, G. Yu, W.P. Hu. Experimental techniques for the fabrication and characterization of organic thin films for field-effect transistors. Chem. Rev. 111 (2011) 3358–3406. DOI:10.1021/cr1001904 |
| [3] | S.H. Kim, K. Hong, W. Xie, et al. Electrolyte-gated transistors for organic and printed electronics. Adv. Mater. 25 (2013) 1822–1846. DOI:10.1002/adma.v25.13 |
| [4] | B. Singh, N.S. Sariciftci, J.G. Grote, F.K. Hopkins. Bio-organic-semiconductor-fieldeffect-transistor based on deoxyribonucleic acid gate dielectric. J. Appl. Phys. 100 (2006) 024514. DOI:10.1063/1.2220488 |
| [5] | A. Petritz, A. Wolfberger, A. Fian, et al. Cellulose as biodegradable high-k dielectric layer in organic complementary inverters. Appl. Phys. Lett. 103 (2013) 153303. DOI:10.1063/1.4824701 |
| [6] | A. Dezieck, O. Acton, K. Leong, et al. Threshold voltage control in organic thin film transistors with dielectric layer modified by a genetically engineered polypeptide. Appl. Phys. Lett. 97 (2010) 013307. DOI:10.1063/1.3459978 |
| [7] | M. Irimia-Vladu, E.D. Głowacki, G. Voss, S. Bauer, N.S. Sariciftci. Green and biodegradable electronics. Mater. Today 15 (2012) 340–346. DOI:10.1016/S1369-7021(12)70139-6 |
| [8] | M. Magliulo, K. Manoli, E. Macchia, G. Palazzo, L. Torsi. Tailoring functional interlayers in organic field-effect transistor biosensors. Adv. Mater. 27 (2015) 7528–7551. DOI:10.1002/adma.v27.46 |
| [9] | R.E. Marsh, R.B. Corey, L. Pauling. An investigation of the structure of silk fibroin. Biochim. Biophys. Acta 16 (1955) 1–34. DOI:10.1016/0006-3002(55)90178-5 |
| [10] | R. Capelli, J.J. Amsden, G. Generali, et al. Integration of silk protein in organic and light-emitting transistors. Org. Electron. 12 (2011) 1146–1151. DOI:10.1016/j.orgel.2011.04.005 |
| [11] | C.H. Wang, C.Y. Hsieh, J.C. Hwang. Flexible organic thin-film transistors with silk fibroin as the gate dielectric. Adv. Mater. 23 (2011) 1630–1634. DOI:10.1002/adma.201004071 |
| [12] | L.L. Shi, X.J. Xu, M.C. Ma, L.D. Li. High-performance, low-operating voltage, and solution-processable organic field-effect transistor with silk fibroin as the gate dielectric. Appl. Phys. Lett. 104 (2014) 023302. DOI:10.1063/1.4862198 |
| [13] | X.L. Li, W. Shi, X.G. Yu, J.S. Yu. Performance improvement of organic field-effect transistor based nitrogen dioxide gas sensor using biocompatible PMMA/silk fibroin bilayer dielectric. J. Mater. Sci. Mater. Electron. 26 (2015) 7948–7954. DOI:10.1007/s10854-015-3448-7 |
| [14] | J.W. Chang, C.G. Wang, C.Y. Huang, et al. Chicken albumen dielectrics in organic field-effect transistors. Adv. Mater. 23 (2011) 4077–4081. DOI:10.1002/adma.v23.35 |
| [15] | J.E. Nielsen, G. Vriend. Optimizing the hydrogen-bond network in Poisson-Boltzmann equation-based pKa calculations. Proteins: Struct. Funct. Genet. 43 (2001) 403–412. DOI:10.1002/(ISSN)1097-0134 |
| [16] | C.Y. Lee, J.C. Hwang, Y.L. Chueh, et al. Hydrated bovine serum albumin as the gate dielectric material for organic field-effect transistors. Org. Electron. 14 (2013) 2645–2651. DOI:10.1016/j.orgel.2013.07.004 |
| [17] | C.Y. Hsieh, J.C. Hwang, T.H. Chang, et al. Enhanced mobility of organic thin film transistors by water absorption of collagen hydrolysate gate dielectric. Appl. Phys. Lett. 103 (2013) 023303. DOI:10.1063/1.4813075 |
| [18] | P. Guerrero, P.M. Stefani, R.A. Ruseckaite, K. de la Caba. Functional properties of films based on soy protein isolate and gelatin processed by compression molding. J. Food Eng. 105 (2011) 65–72. DOI:10.1016/j.jfoodeng.2011.02.003 |
| [19] | L.K. Mao, J.Y. Gan, J.C. Hwang, T.H. Chang, Y.L. Chueh. The role of water in the device performance of n-type PTCDI-C8 organic field-effect transistors with solution-based gelatin dielectric. Org. Electron. 15 (2014) 920–925. DOI:10.1016/j.orgel.2014.01.023 |
| [20] | M. González, L.A. Bagatolli, I. Echabe, et al. Interaction of biotin with streptavidin. J. Biol. Chem. 272 (1997) 11288–11294. DOI:10.1074/jbc.272.17.11288 |
| [21] | M.D. Angione, S. Cotrone, M. Magliulo, et al. Interfacial electronic effects in functional biolayers integrated into organic field-effect transistors. Proc. Natl. Acad. Sci. USA 109 (2012) 6429–6434. DOI:10.1073/pnas.1200549109 |
| [22] | M. Magliulo, A. Mallardi, R. Gristina, et al. Part per trillion label-free electronic bioanalytical detection. Anal. Chem. 85 (2013) 3849–3857. DOI:10.1021/ac302702n |
| [23] | M. Magliulo, D. Altamura, C. Di Franco, et al. Structural and morphological study of a poly(3-hexylthiophene)/streptavidin multilayer structure serving as active layer in ultra-sensitive OFET biosensors. J. Phys. Chem. C 118 (2014) 15853–15862. DOI:10.1021/jp504652u |
| [24] | X. Lin, J. Xie, L. Zhu, et al. Hybrid ferritin nanoparticles as activatable probes for tumor imaging. Angew. Chem. Int. Ed. 50 (2011) 1569–1572. DOI:10.1002/anie.201006757 |
| [25] | B.J. Kim, Y. Ko, J.H. Cho, J. Cho. Organic field-effect transistor memory devices using discrete ferritin nanoparticle-based gate dielectrics. Small 9 (2013) 3784–3791. DOI:10.1002/smll.v9.22 |
| [26] | F. Maddalena, M.J. Kuiper, B. Poolman, et al. Organic field-effect transistor-based biosensors functionalized with protein receptors. J. Appl. Phys. 108 (2010) 124501. DOI:10.1063/1.3518681 |
| [27] | T.P.J. Knowles, M.J. Buehler. Nanomechanics of functional and pathological amyloid materials. Nat. Nanotechnol. 6 (2011) 469–479. DOI:10.1038/nnano.2011.102 |
| [28] | R. Paparcone, S. Keten, M.J. Buehler. Atomistic simulation of nanomechanical properties of Alzheimer's Aβ(1-40) amyloid fibrils under compressive and tensile loading. J. Biomech. 43 (2010) 1196–1201. DOI:10.1016/j.jbiomech.2009.11.026 |
| [29] | X.Y. Wang, Y.F. Li, C. Zhong. Amyloid-directed assembly of nanostructures and functional devices for bionanoelectronics. J. Mater. Chem. B 3 (2015) 4953–4958. DOI:10.1039/C5TB00374A |
 2016, Vol. 27
2016, Vol. 27 



