b Key Laboratory of Synthetic and Self-Assembly Chemistry for Organic Functional Molecules, Shanghai Institute of Organic Chemistry, Chinese Academy of Sciences, Shanghai 200032, China
Organic field-effect transistors (OFETs),one of the most significant branches of modern organic electronics,have earned keen interest in recent decades for their potential applications in flexible large-area display,smart tags and cards,and physical/ chemical sensors [1-4]. On the one hand,as more and more innovative p-conjugation organic semiconductors are designed and synthesized,carrier mobility,the major electrical characteristic of OFET,has surpassed that of amorphous silicon (0.1-1.0 cm2 V-1 s-1) and reached 5 cm2 V-1 s-1 or even higher for both small molecules and polymer,p-type and n-type organic semiconductors [5-13]. In addition,solution-processed OFETs with high performance (high charge mobility,low threshold voltage and high current on/off ratio) and stability have experienced remarkable improvements [14-17] .However,the commercial application of OFETs is still limited due to the relatively lower device performance,not ideal device stability and the challenging device integration technology. One the other hand,great efforts have been devoted to the design and fabrication of multi-functional OFETs. As a result,combining traditional OFETs with functional molecular building blocks to install multi-functional OFETs has been the latest research tendency. Considerable novel multi-functional devices based on OFET such as phototransistors [18-20],light-emitting transistors [21, 22],sensors [23, 24] and memories [25, 26],have been achieved by molecular tailoring,modification of dielectric layer and electrode layer,construction of active layer,etc. Some reviews on device fabrication strategies and challenges have been previously published [27, 28]. Smart use of these organic electronic devices by the introduction of stimuli responsive materials is one of the common ways [29-32]. Organic functional materials that are sensitive to gas,light,pressure,temperature and pH have already been employed to OFETs,affording multifunctional devices such as sensors and switches [24, 33-38]. Phototransistors particularly the devices that are electrically and optically responsive due to the integration with photochromic molecules are attracting more and more attention [39-42]. Photochromic molecules can reversibly switch between two isomers result in the reversible color change simultaneously,when exposed to particular wavelength of light. Nonionic-ionic spiropyran,open ring-closed ring diarylethene and trans-cis azobenzene are three main sorts of photochromic systems frequently used in photoresponsive OFETs [43]. Bi-functional OFETs combined with these photo-switchable molecules are promising candidates for new applications in light sensors and optical memory where the photostimulation can be precise controlled on direction,intensity and wavelength [44].
A typical OFET is composed of an active layer,a dielectric layer,a gate electrode and source/drain electrode as well as their corresponding interfaces including electrode/active layer interface,dielectric/active layer interface,and organic/organic layer interface [45]. Both the functional layers and interfaces have key influences on the device performance. In this contribution,we attempt to present a brief overview on recent advances in the study of photoactive OFETs involving photochromic molecules in active layers,in dielectric layers,and on the electrode/active layer interface.
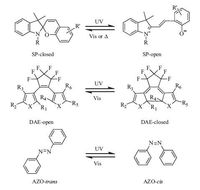
2. Photochromic moleculesPhotochromic molecules can reversibly switch between two isomers induced by light or heat,where the two forms have different absorption spectra and cause the discoloration intuitively. Generally,the variation from colorless isomer to colored isomer is induced by ultraviolet (UV) irradiation,and the restoring course to the initial state can be performed by visible (Vis) light or thermal relaxation. During the isomerization,the physical and chemical properties of these photochromic compounds can also be changed,including refractive indices,dielectric constants,geometrical structures,energy levels,polarity,magnetic properties and oxidation/reduction potentials [43, 46]. These controllable property tunings have inspired people to apply photochromic system in photoelectric field such as photo-switches [47-49],sensors [50, 51],molecular machine [52-54],data storage [55-57],liquid crystal [58-60],and so on. Spiropyran,diarylethene and azobenzene are three most widely studied organic photochromic molecules,and their structures and the photochromism process of these molecules are presented in Fig. 1.
Spiropyran (SP) is one of the oldest families of photochromic molecules. As shown in Fig. 1,the cleavage of the Cspiro-O bond in the parent colorless SP-isomer was triggered by UV light,making the spiro structure transform into the merocyanine form. The recovery process can easily occur either by exposure to visible light or by thermal relaxation. Besides the color variance,the open and closed SP forms differ a lot in electric dipole moment,structure,emission behavior,protonation and chemical affinity,thus make the SP responsive to multiple stimuli such as light,pH [61],metal ions [62, 63] and heat. Based on this unique characteristic,many investigations on the study of SP-based dynamic materials have been reported such as SP-functional polymers [64],light-controlled surface wettability [65],photo-control inorganic nanoparticles and so on [66].
It is generally accepted that diarylethene (DAE) derivatives bearing two thiophene-derived groups is the most suitable photochromic compounds for practical application. The most outstanding features of DAE compounds are their relatively high light-sensitivity,fatigue resistance and thermal stability [46, 67]. Photochromism mechanism of DAE is shown in Fig. 1. With the irradiation of UV/vis light,DAE switches between an open-ring form and closed-ring form. Highlighted for the thermal stability of both isomers,DAE molecules have become the hottest system for light-triggered photo-switches,like fluorescent switch,supramo- lecular switch,liquid crystal switch,crystalline and amorphous switch. These DAE-based photo-switches have been summarized in Tian’s excellent review [49].
|
Download:
|
| Figure 1. Photochromic mechanism of three typical photochromic molecules. top,spiropyran from SP-closed to SP-open; middle,diarylethene from an open-ring form to a closed-ring form; bottom,azobenzene from a trans-isomer to a cis-isomer. | |
Discovered in the mid-18th century,azobenzene (AZO) was once regarded as a coloring agent used for the synthesis of desired dyes until its cis-form was firstly found by Hartley in 1937 [68]. Composed of two phenyl rings linked by an azo linkage (-N=N-),AZO can switch between two cis-trans isomers induced by particular wavelengths of light. It is demonstrated that the cis form can convert back to the trans isomer either by vis light or by thermal relaxation in the dark. This steady transformation rate depends on the light intensity and wavelength,ambient temperature,and the host matrix (be in solution,liquid crystal,monolayer,or polymer) [69]. A large number of expending researches on photobiology [70, 71] and photoelectrochemical information storage [59, 72-74] have emerged according to the facile geometric isomerization of AZO.
3. Photochromic molecule in OFET active layerThe performance of OFETs mainly depend on the properties of the device active layer where free charge carriers transport between the source and drain electrodes and come into being of source/drain current (IDS) under a gate voltage. Bifunctional OFETs with photochromic molecules and organic semiconductors are modulated by light and voltage simultaneously. The most direct way to realize this attempt is by blending photochromic molecules with organic semiconductors in the active layer. In this way,different photochromic isomers would have different doping effects on the organic semiconductors and reversibly modulate the output signal.
3.1. Photochromic molecule doped OFET active layer 3.1.1. Diarylethene doped OFET active layerAs mentioned above,diarylethene (DAE) is a potential photochromic system for photo-triggered OFETs thanks to its thermal and energy levels stabilities in both isomers. In 2012,Samori et al. [39] fabricated bi-component OFETs based on DAE derivative as photo-tunable component and poly(3-hexylthio- phene) (P3HT) as semiconductor matrix. A couple of DAE molecules with different ionization energies (IE): DAE_1 and DAE_2 were designed and synthesized (Fig. 2a and b). As illustrated in Fig. 2c,the drain source current declined spontaneously under the irradiation of UV and OFETs doped with DAE_1 and DAE_2 both showed reversible transistor characteristics switch,while DAE_1 performed better to light stimulate,as large as 80% drain current modulation was observed. Since the hole transport is primary occurs in the HOMO levels of organic semiconductor,the result may be caused by the better HOMO levels match between P3HT and DAE-closed than DAE-open. Similarly,the HOMO energy gap between P3HT and DAE-1c (0.3 eV) is narrower than that between P3HT and DAE-2c (0.4 eV),leading to a greater modulation degree in DAE_1 doped active layer as illustrated in Fig. 2c.

|
Download:
|
| Figure 2. Devices components, geometry, energetics and reversible transistor characteristics. Schematic representation of the investigated bottom-gate bottom-contact OFET devices (a). Energy level diagram that depicts theHOMOlevels of the employed components of P3HT and DAE derivative and highlights the mechanism of photomodulation in the OFET determined by CV and UPS (b). Static photoswitching of the OFET devices that contained DAE_1o or DAE_2o at fixed time intervals (steps) of ultraviolet and white light irradiation indicated by the violet and grey shaded areas, respectively (c), at the end of each step the light was turned off to measure a transfer curve, ID is the drain current, and normalizing factor I0 is the first drain-current value measured in step number 0 (in the dark) for each compound. (a–c) Reproduced with permission [39], Copyright (2012) NPG. | |
Several years later,Samori and coworkers extended their work by using DAE with different alkyl substituents (Fig. 3c and d) as photochromic molecules and small molecule 2,7-dialkyl-ben- zothieno(3,2-b)benzothiophene (BTBT) decorated with C12H25 alkyl chains (C12-BTBT) as the semiconductor matrix material (Fig. 3a and b) [75]. The reversible characteristics were compared to those based on P3HT. As is shown in Fig. 3e,under short irradiation time,the OFETs based on BTBT performed better than those based on P3HT,although the small molecules may underwent phase separation in the active layer. According to their report,as high as 0.21 cm2 V-1 s-1 hole-mobility was achieved and the optimized photomodulation (ID/I0) exceeded 90% (Fig. 3e) [75].

|
Download:
|
| Figure 3. Chemical structures of organic semiconductor BTBT (a),photo-switchable DAE derivatives (b) and phototransistor characteristics of pure BTBT and the DAE- doped BTBT (c),where the violet and green shaded areas correspond to the irradiations with the lights at 365 and 546 nm,respectively. (a-c) Reproduced with permission [75],Copyright (2015) NPG. | |
Most recently,n-type organic semiconductors,[6,6]-phenyl- C61-butyric acid methyl ester (PCBM) and indene-C60 bis-adduct (ICBA),are used as matrix in those binary photochromic systems for the first time (Fig. 4a) [76]. Meanwhile,DAE molecules are functionalized with electron-withdrawing substituents to lower their LUMO energy levels (Fig. 4b) [76]. It was discovered that blending n-type semiconductors with photochromic molecules was an efficient method to prepare photo-modulated transistors.

|
Download:
|
| Figure 4. Chemical structures of n-type organic semiconductors (PCBM and ICBA) and photo-switchable DAE derivatives (DAE-FN and DAE-F) (a), and their respective LUMO levels determined by cyclic voltammetry with fullerene derivatives in black color and the DAE derivatives in blue (open form) and red (closed form) colors (b). (a and b) Reproduced with permission [76], Copyright (2015) RSC. | |
In summary,these OFETs doped with DAE derivatives in active layers exhibit both voltage- and light-modulation on current. More importantly,high current switch efficiency,fatigue resistance and thermal stability were achieved in these studies [39, 75, 76]. Polymeric,small molecular,p-type and n-type organic semiconductors with DAE dopants are well demonstrated for phototransistors. However,the key influence factors of the current switch efficiency and the mechanism of the modulation still remain to be analyzed in depth.
3.1.2. Spiropyran doped OFET active layerAnother regular used photochromic molecule with outstanding properties is spiropyran (SP) which exhibits a large different in electric dipole moment before and after isomerization. The dipole moment of ionic polarized SP-open is 13.9 D while the SP-closed form is 6.4 D [77, 78].
Guo et al. [40] blended P3HT with spiropyran to form a hybrid active layer of a bottom-gate,top-contact OFET (Fig. 5a). They observed that the device mobility decreased gradually with the increase of the SP concentration. For the balance of carrier mobility and switch efficiency,the SP concentration was fixed at 10 wt%. As illustrated in Fig. 5b,the absolute value of drain source current (IDS) increased when UV light turned on and restored slowly under the irradiation of visible light,affording a switch cycle. Some obvious cycles with full recovery of IDS were also acquired during a period of 40 min (Fig. 5c) while OFETs based on pristine P3HT only showed gradual current decrease no matter irradiated with which kind of light (Fig. 5d insert). The authors pointed out that this result may be caused by two reasons. One is that the SP-open with larger dipole moment formed electrostatic environment and thus lead to a more conductive blended active layer. The changed molecular orbital energy structure may also support an explanation for the increased IDS since the HOMO-LUMO gap of SP was narrowed from 3.03 eV into 2.66 eV during the isomerization when the SP-closed changed to the SP- open. Another possible reason is that the HOMO level value of SP changed from -5.57 eV to -5.74 eV when SP-closed form transformed to SP-open isomer. As a result,the increased energy gap between the HOMO levels of P3HT and SP-open drove the carrier transport (Fig. 5e).

|
Download:
|
| Figure 5. Device structure and photochromic characteristics. Schematic representation of the device structure with P3HT-SP blends as an active semiconducting layer and SP molecules switching back-and-forth between SP-closed and SP-open triggered by light with different wavelengths (a). One full switching cycle of the time-dependent behavior of the same device,VD = -30 V,VG = -10 V (b). Time trace of ID for the same device over a period of 〜40 min,showing the reversible photoswitching events under irradiation of UV light and visible light (c),the inset shows the photo response of control thin films formed from a pure P3HT solution in CHCl3 (10 mg mL1) without SP. VD = -30 V,VG= -10 V. Energy level diagram and working model of P3HT-SP hybrid systems (d). (a-d) Reproduced with permission [40],Copyright (2012) RSC. | |
Later on,Wakayama et al.proposed a similar work [79],where the P3HT was replaced by poly(triarylamine) (PTAA) (Fig. 6),but the switch behavior is opposite to Guo’s report. The IDS reduced when SP-closed transformed to SP-open and reversed under the irradiation of visible light. In their opinion,a possible mechanism for this phenomenon is that the ionic polarized SP-open works as acarrier scattering site and trap the carriers in blend film thus leading to the IDS decrease under UV light [79].

|
Download:
|
| Figure 6. Schematic illustrations of the transistor involving SP and PTAA,together with the chemical structures of PTAA and SP. Open-ring isomers of SP are reversibly transformed into closed-ring isomers and vice versa by alternating UV and VIS light irradiation. Reproduced with permission [79],Copyright (2013) RSC. | |
The extension work of PTAA/SP hybrid active layer was applied on dual-gate transistors [80]. Wakayama et al.fabricated a dualgate transistor with two channels,a PTAA/SP hybrid layer and a PTAA pristine layer were modulated by top gate and bottom gate independently (Fig. 7a). The calculated carrier mobility from saturation regime of two channels was 1.8×10-4 cm2 V-1 s-1 (top channel) and 2.3×10-4 cm2 V-1 s-1 (bottom channel). In the irradiation of UV or vis light,the IDS through the bottom channel did not change,while the IDS through the top channel had a 6% reduction induced by UV light,and was restored by the irradiation of VIS light (Fig. 7b). The result of the reversible IDS modulation was consistent with their previous report [79].

|
Download:
|
| Figure 7. Device configuration and circuit diagram of photochromism-based dual-gate transistor (a). Changes in transfer characteristics of the top and bottom channels at VDS of -50 V under UV and VIS irradiation for 30 min (b). (a and b) Reproduced with permission [80],Copyright (2013) ACS. | |
Recently,a one-step spin-coated dual-gate OFET was demonstrated by Wakayama and coworkers [81]. As shown in Fig. 8a,the mixed P3HT/SP solution is spin-coated on the Si/SiO2 substrate,and the SP molecules were mainly distributed in the bottom layer around the substrate,thus forming a top SP-free layer and a bottom SP-rich layer. As shown in Fig. 8b,this phase separation was proved by the transfer characteristics since the top channel was photoinactive and the bottom channel was photo-active.

|
Download:
|
| Figure 8. Schematic illustrations of the dual-gate transistor and the chemical structures of P3HT and spiropyran (a),the open-ring isomer of spiropyran reversibly transforms into the closed-ring isomer and vice versa by alternating UV - VIS irradiation. Transfer characteristics of the top and bottom channels at a VDS of -50 V under UV or visible irradiation for 10 min (b). (a and b) Reproduced with permission [81],Copyright (2014) ACS. | |
Doping active layer with photochromic molecules is an effective way to form multifunctional transistors modulated by light. High switching efficiency and low fatigue are key properties of these phototransistors as well as their phase separation degree. Although some possible theories have emerged for explaining the results,the precise mechanism of the modulation process remains unclear.
3.2. Pristine photochromic molecule as OFET active layerWith the goal of realizing better mixture and higher concentration of photochromic system in semiconductors,some published studies focused on combining these two parts (photochromic molecule and organic semiconductor) by covalent bond,then the extended pristine photochromic materials can be deposited as semiconductors layer directly [82, 83]. Similar methods were often used and proved efficient in other research areas by connecting different molecular blockings to get the expected properties in a molecular system. Several important parameters should be taken into account for,such as energy level and intermolecular interaction.
Fuhrmann-Lieker et al. synthesized an AZO glass with two AZO chromophores based on two triarylamine cores (AZOPD) (Fig. 9) [82]. This dimer was proved to be a bipolar semiconductor. Thehole and electron mobilities in linear regimes are 1.9 ×10-6 cm2 V-1 s-1 and 5×10-7 cm2 V-1 s-1,respectively. The p-type bottom-gate top-contact transistor based on AZOPD presented switchable behavior in the irradiation of 436 nm blue light. The hole mobility decreased from 9.8×10-6 cm2 V-1 s-1 to 0.7× 10-6 cm2 V-1 s-1,and the threshold voltage shifted to a more negative value. However,after thermal relaxing in dark for 30 min,the transistors didn’t behave full recovery with the mobility only restored about 30% and the threshold remained pinned.

|
Download:
|
| Figure 9. Chemical structure of AZOPD. | |
DAE derivative 1,2-bis(2-methyl-5-(4-biphenyl)-3-thienyl) hexafluorocyclopentene was synthesized by Chikyow and coworkers as a photochromic semiconductor [83] (Fig. 10a). The drain current of this derivative was demonstrated optically and electrically switchable. Irradiated with UV light,the open-isomer transformed to closed form. As a result,the energy level changed. As illustrated in Fig. 10b,the HOMO level of the open-ring is -6.31 eV and that of the closed form is -5.70 eV,and the latter is much closer to the work function of Au electrode (-5.1 eV). In other words,the closed-isomer was easier for carrier injection due to the narrowing of injection barrier from Au electrode to active layer. What’s more,during the photochromism,the closing of the DAE ring can be regarded as an extension of p-conjugation system. Such kind of transformation may have an effect on the molecule aggregation. As expected,a great reversible change of drain current (VD = -100 V,VG = -100 V) induced by UV and vis light was demonstrated in Fig. 10c and 10d. The optical current on/off ratio was as high as 102,which is larger than most reported corresponding values for DAE derivatives. Another highlight of this work was the long retention time of both ON and OFF state,and the modulated ON and OFF states can be maintained in dark for as long as one week. Nevertheless,the programming cycle endurance was not satisfactory enough as the current on/off ratio decreased gradually during the back and forth processes.

|
Download:
|
| Figure 10. Device structure and photochromic characteristics. Schematic illustration of an optically and electrically driven DAE transistor (a). Energy levels of DAE molecules with open- and closed-ring configurations and the work function of Au (B),the HOMO and LUMO energy levels of the respective isomers were estimated by cyclic voltammetry and absorption spectroscopy. Optically modulation of the drain current (Vd =-100 V,Vg =-100 V) against a sequence of photoirradiation (c). Retention properties of ON and OFF states induced by light irradiation ( Vd = -100 V,Vg = -100 V) (d). (a-d) Reproduced with permission [83],Copyright (2013) ACS. | |
4. Photochromic molecules in OFET dielectric layer or on its surface
It is confirmed that carrier transport occurs on a few semiconductor molecule layers close to the dielectric layer [84],thus the active layer/dielectric layer interface become a significant focus in the studies of OFETs. The interface trap density affects the device mobility and threshold voltage dramatically. Besides,the
morphology of organic layer and the aggregation of organic grains are also influenced by the interface [45]. Therefore,several attempts such as blend photochromic molecules with the dielectrics or modify dielectric layer surface by photochromic molecules have been demonstrated for photoresponsive OFETs.
4.1. Hybrid dielectric layernovel method has been proposed by blending photochromic molecule with dielectric polymer to form photo-active OFET hybrid gate dielectric layer. It is demonstrated that the device stability can get an improvement compared with that of devices doped photochromic molecule in active layers,because in this method the photochromic component is not mixed with active layer and thus phase separation or illumination would not do much damage to the semiconductor layer.
Shen et al. used polymethylmethacrylate (PMMA) and spir- opyran to form a PMMA/SP hybrid dielectric layer in a bottom-gate,top-contact OFET based on pentacene semiconductor (Fig. 11a) [85]. As buffering matrix,pristine PMMA does not have photo- switchable characteristic,while SP molecules can isomerize flexibly between SP-closed and SP-open. In this manner,recoverable current increase induced by UV light was observed (Fig. 11c). As shown in Fig. 11c,the perfect switching circles could last for 6 h and the mobility and Ion/off ratio did not diminish. During the course of this experiment,the capacitance of hybrid dielectric film changed reversibly between 2.10 μ/cm2 and 2.14 μ/cm2 corresponding to drain current circulation (Fig. 11b). Therefore,the change of dielectric capacitance may be the prior reason for the photoresponsive results.

|
Download:
|
| Figure 11. Schematic representation of the device structure showing how photochromic SP alter the dielectric capacitance for reversibly controlling the device performance under UV and visible light illumination (a). The representative four full switching cycles of the gradual transition of the capacitance for the same device by taking the data at 1 MHz frequency (b). Time trace of the drain current for the same device over a period of 6 h with the first 10 cycles expanded for clarity,showing the reversible photoswitching events under irradiation of UV and visible lights (VD = -50 V,and VG =-30 V) (c). (a)-(c) Reproduced with permission [85],Copyright (2010) Wiley. | |
Similar results were achieved by Lutskyk et al. [86]. They dissolved spiropyran into polymer insulator PMMA,and used a perylene bisimide derivative as n-type active layer (Fig. 12). The increased IDS was detected under the illumination of UV light and the current could be restored to the original state by thermal relaxation or by irradiation with visible light. A small threshold voltage back and forth shift was also observed during the experiment. In addition,it was found that the current on/off ratio depend on the gate voltage,and the response current on/off ratio get to the highest values (102-103) when the gate voltage was larger than the threshold voltage. More recently,a kinetic study of light-driven current switch processes was carried out by Lutsyk and coworkers [87],which confirmed the circulation of the dielectric capacitance during the photochromic process.

|
Download:
|
| Figure 12. Schematic architecture of an OFET. The acronyms in the parentheses refer to the names of materials used in the OFET. The chemical formulas of the materials employed in the study and the scheme of the SP-MR photochromic reaction are shown on the right side of the figure. Reproduced with permission [86],Copyright (2011) ACS. | |
4.2. Pristine photochromic molecule in dielectric layer
In consideration of the limitation of SP’s low solubility in PMMA [85],Guo and coworkers recently incorporated photochromic SP units to the main chain of PMMA,affording new copolymers SP-co- MMA with different SP and MMA ratios (Fig. 13) [88]. The bottom- gate,top-contact OFETs based on pentacene with this copolymer as dielectric layer was fabricated. As expected,the IDS gradually increased to its maximum value after the UV light irradiation for 20 min,which was attributed to the high conductance state caused by the SP-open forms. Subsequent current restoration lasted for 1 h. It is worth mentioning that the photosensitivity in this case was higher than that of SP/PMMA hybrid dielectric system [85]. This exciting phenomenon may be explained by the high content of SP units in the copolymer (as high as 95% contribution) compared with small SP molecules dissolved hybrid dielectric systems [85, 86]. Similar to SP/PMMA hybrid dielectric [85, 86],two switching mechanisms were analyzed and demonstrated. One is the dipole moment shift of SP which leads to the capacitance change in the dielectric/active layer. The other is the scattering effect of the ionic polarized SP-open. These two mechanisms may complement with each other to explain the photo-modulation of OFETs. Therefore,building multi-functional copolymers is a promising strategy for preparing perfect photosensitive devices.

|
Download:
|
| Figure 13. Photochromism of SP-co-MMA between SP-closed and SP-open triggered by UV/vis light. Reproduced with permission [88],Copyright (2015) Wiley. | |
4.3. Photochromic SAM at dielectric layer/active layer interface
To realize an effective integration of photochromic molecules and dielectric,the modification of dielectric/active interface by using SP self-assembled monolayer (SAM) bonded on the dielectric surface was carried out by Guo and coworkers in 2011 [89]. As illustrated in Fig. 14,carboxyl acids of SPs are tethered to the surface-bonded amino groups by amide linkages,and make the SAM stably stand on the SiO2 surface with an angle of inclination of 47.2°. A series of characterizations including IR spectrum,X-ray photoelectron spectroscopy and low-angle X-ray revealed the successful attachment of SP molecules on the SiO2 surface. The OFETs’ mobility and threshold presented reversible response towards UV/vis light,which was caused by the channel conductance switch,where there was an increase in the local electric field induced by the ionic SP-open alignment on the dielectric/active layer interface.

|
Download:
|
| Figure 14. The structure of OFET and SP-SAM on the SiO2 surface. The SP-SAM on the dielectric/active layer interface of a bottom-gate top-contact OFET can undergo a photochromism process between SP-closed and SP-open triggered by light at specific wavelengths (a). Model structure of SP SAM on the SiO2 surface with an angle of inclination of 47.2° (b). (a and b) Reproduced with permission [89],Copyright (2011) ACS. | |
Recently,Guo and coworkers extended their research by designing and synthesizing a DAE derivative terminated by a phosphonic acid group,and these DAE molecules can react with the HfO2 bottom dielectric at surface to form SAM as illustrated in and in the insert figure [42]. The distinct variation in HOMO and LUMO levels of DAE-open and DAE-closed lead to the photoresponsive behavior. These photo-switchable devices could be applied for OFET based nonvolatile memories. The signal writing was done by the illumination of 633 nm light with different intensities,and the stored data was erased by applying a -3 V gate pulse. Organic memory transistors with low operation voltage (≤3V),long retention time (half a year),high fatigue resistance ( <104 memory circles) and good nondestructive signal process ability were achieved and demonstrated in detail [42]. Further application of these organic memory transistors arrays was carried out on scanning photocurrent mapping precisely and reversibly. Such kind of novel and effectual interfacial technology has provided a potential route for realizing the practical application of photoactive transistors as multi-functional devices.
Tao and coworkers synthesized a series of AZO derivatives with different substitutions (Fig. 15b) and attached them on the dielectric layer (Fig. 15a) [90]. Bottom-gate top-contact OFET was fabricated,where pentence was used as semiconductor channel and Au electrodes were deposited through a shadow mask. As shown in Fig. 15c,OFET based on 1-CF3 SAM-modified dielectric layer achieved a positive shift of IDS and Vth by UV irradiation,and returned to its origin state after being kept in the dark for 16 h (Fig. 15d). However,the shift direction of devices based on 1-H and 1-CH3 was opposite (Fig. 15e and f. The electron- donating or electron-accepting substitutions of AZO derivatives may be the main reason for these difference results [90].

|
Download:
|
| Figure 15. Polarization switch with photochromic isomerization of AZO moieties incorporated in the active channel (a). Structure of compounds used (b). Transfer characteristics of devices with the monolayer of 1-CF3 (c),1-H (e),and 1-CH3 (f) as a function of UV irradiation (356 nm),and 1-CF3 device stored in dark after UV irradiation (d). Insert in (c) shows the curves for mixed monolayer of 1-CF3. (a-f) Reproduced with permission [90],Copyright (2012) ACS. | |
5. Photochromic SAM on OFET electrode/active layer interface
The electrode/active layer interface is also a very important interface in OFETs,where the carriers inject from the source electrode to the semiconductor cannel. Suitable work function of electrode is the key for high-mobility environment-stable OFETs. Modification of electrode/active layer interface by photochromic monolayer is a strategy for realizing photo-triggered OFETs. Except for organic electronics,photochromic SAMs have been widely adopted for applications in other research fields,including surface wettability [91],DNA delivery [92],and single-cell adhesion [93].
In 2011,Samori et al. exploited a thiol-terminated biphenyl AZO (Fig. 16,insert) to form SAM on the electrode/active layer interface of a bottom-gate,bottom-contact OFET based on n-type organic semiconductor PDIF-CN2 (Fig. 16) as channel layer (Fig. 16) [41]. Unexpectedly,the flexible photo-isomerization of AZO was not restricted by its high orientation and tight packing,the device presented reversible field-effect photoresponsive. During the in situ experiment,the IDS increased when the device was exposed to the UV light and restored to initial state by 24 h relaxation in the dark,accompanied by a tiny threshold voltage switch. It was demonstrated the thinner cis-isomer AZO monolayer decreased the carrier injection barrier at the interface,and thus contributed to the enhanced IDS current. In addition to the resistance switch,a change of interface film morphology,modulation of work function and formation of an empty cavity induced by the isomerization of AZO are likely to have an impact on the modulation process. The theoretical study of the work function change in organic multifunctional OFETs was recently reported [94]. Four different techniques,Kelvin probe,Kelvin probe force microscopy and ultraviolet photoelectron spectroscopy were used to demonstrate that the work function of gold electrode was modulated during the AZO light-triggered isomerization [94]. Most recently,Samori and coworkers [95] designed and fabricated a photoresponsive top-gate bottom-contact OFET based on n-type polymer P(NDI2OD-T2) with thiolated-DAE-functionalized drain and source electrodes. The maximum switching ratio ever for OFETs with DAE-functionalized electrodes without detectable fatigue during the course of the performed switching cycles was obtained. More importantly,both states,i.e. o-DAE and c-DAE,are stable for at least over five months.

|
Download:
|
| Figure 16. Schematic representation of the device structure showing the reversible isomerization reaction (trans-cis) that takes place at the interface between the semiconductor and AZO-functionalized electrodes. Reproduced with permission [41],Copyright (2011) Wiley. | |
6. Conclusion and outlook
In recent years,multi-functional organic electronic devices have emerged and become the center of attention due to their potential applications for recording and storage,sensors and monitors. Various strategies have been demonstrated by using photochromic molecules for fabricating photoresponsive OFETs,including doping the semiconductor layer,self-assembling on the device interface of dielectric layer/active layer or electrode/active layer and blending with the polymer matrix. Therefore,great progress have been made in photo-modulation transistors,for example,as high as 102-103 photo-modulated current on/off ratios have been achieved by a hybrid SP/PMMA dielectric layer [87],and several photochromic systems are proved suitable for reversible transformations with high fatigue resistance and stability.
However,opportunities and challenges co-exist in the fundamental study and applications of photoresponsive OFETs. There are several key points to be noted,(i) since the low photo-modulated current on/off ratio and less device stability,the device performance should be much improved,together with the fatigue resistance; (ii) it is very important for design and synthesis of new photochromic molecules with excellent reversibility and stability for both isomers,low fatigue,rapid response,nondestructive readout capability,as well as the excellent blending ability with general organic semiconductors; (iii) design and synthesis of novel organic semiconductors with covalently linked photochromic units should be highlighted for their directly applications in phototransistors; (iv) systematic and thorough study on photo- modulated mechanism of phototransistors involving organic semiconductors and photochromic molecules; (v) new applications of photoresponsive OFETs as novel multi-functional electronic devices.
Acknowledgments This study was supported financially by the National Natural Science Foundation of China (Nos. 21302212 and 21522209) and the "Strategic Priority Research Program" (No. XDB12010100)| [1] | B. Crone, A. Dodabalapur, A. Gelperin, et al. Electronic sensing of vapors with organic transistors. Appl. Phys. Lett. 78 (2001) 2229–2231. DOI:10.1063/1.1360785 |
| [2] | C.D. Dimitrakopoulos, P.R. Malenfant. Organic thin film transistors for large area electronics. Adv. Mater. 14 (2002) 99–117. DOI:10.1002/(ISSN)1521-4095 |
| [3] | A. Kraft. Organic field-effect transistors-the breakthrough at last. ChemPhysChem 2 (2001) 163–165. DOI:10.1002/(ISSN)1439-7641 |
| [4] | F. Wü rthner. Kunststoff-Transistoren werden reif für Massenanwendungen in der Mikroelektronik. Angew. Chem. 113 (2001) 1069–1071. DOI:10.1002/(ISSN)1521-3757 |
| [5] | H. Minemawari, T. Yamada, H. Matsui, et al. Inkjet printing of single-crystal films. Nature 475 (2011) 364–367. DOI:10.1038/nature10313 |
| [6] | Y. Yuan, G. Giri, A.L. Ayzner, et al., Ultra-high mobility transparent organic thin film transistors grown by an off-centre spin-coating method, Nat. Commun. 5(2014). |
| [7] | D. Shukla, S.F. Nelson, D.C. Freeman, et al. Thin-film morphology control in naphthalene-diimide-based semiconductors: high mobility n-type semiconductor for organic thin-film transistors. Chem. Mater. 20 (2008) 7486–7491. DOI:10.1021/cm802071w |
| [8] | L. Ying, B.B. Hsu, H. Zhan, et al. Regioregular pyridal[1-3] thiadiazole π-conjugated copolymers. J. Am. Chem. Soc. 133 (2011) 18538–18541. DOI:10.1021/ja207543g |
| [9] | J.H. Oh, S. Liu, Z. Bao, R. Schmidt, F. Wü rthner. Air-stable n-channel organic thinfilm transistors with high field-effect mobility based on N N'-bis (heptafluorobutyl)-3,4:9,10-perylene diimide. Appl. Phys. Lett. 91 (2007) 212107. DOI:10.1063/1.2803073 |
| [10] | H.R. Tseng, L. Ying, B.B. Hsu, et al. High mobility field effect transistors based on macroscopically oriented regioregular copolymers. Nano Lett. 12 (2012) 6353–6357. DOI:10.1021/nl303612z |
| [11] | H.R. Tseng, H. Phan, C. Luo, et al. High-mobility field-effect transistors fabricated with macroscopic aligned semiconducting polymers. Adv. Mater. 26 (2014) 2993–2998. DOI:10.1002/adma.201305084 |
| [12] | X. Gao, Z. Zhao. High mobility organic semiconductors for field-effect transistors. Sci. China Chem. 58 (2015) 947–968. DOI:10.1007/s11426-015-5399-5 |
| [13] | X. Gao, Y. Hu. Development of n-type organic semiconductors for thin film transistors: a viewpoint of molecular design. J. Mater. Chem. C 2 (2014) 3099–3117. DOI:10.1039/c3tc32046d |
| [14] | S. Allard, M. Forster, B. Souharce, H. Thiem, U. Scherf. Organic semiconductors for solution-processable field-effect transistors (OFETs). Angew. Chem. Int. Ed. 47 (2008) 4070–4098. DOI:10.1002/(ISSN)1521-3773 |
| [15] | M.M. Payne, S.R. Parkin, J.E. Anthony, C.C. Kuo, T.N. Jackson. Organic field-effect transistors from solution-deposited functionalized acenes with mobilities as high as 1 cm2/Vs. J. Am. Chem. Soc. 127 (2005) 4986–4987. DOI:10.1021/ja042353u |
| [16] | H. Sirringhaus. Device physics of solution-processed organic field-effect transistors. Adv. Mater. 17 (2005) 2411–2425. DOI:10.1002/(ISSN)1521-4095 |
| [17] | F. Zhang, Y. Hu, T. Schuettfort, et al. Critical role of alkyl chain branching of organic semiconductors in enabling solution-processed n-channel organic thinfilm transistors with mobility of up to 3.50 cm2 V-1 s-1. J. Am. Chem. Soc. 135 (2013) 2338–2349. DOI:10.1021/ja311469y |
| [18] | J. Labram, P. Wöbkenberg, D. Bradley, T. Anthopoulos. Low-voltage ambipolar phototransistors based on a pentacene/PC 61 BM heterostructure and a selfassembled nano-dielectric. Org. Electron. 11 (2010) 1250–1254. DOI:10.1016/j.orgel.2010.04.024 |
| [19] | B. Mukherjee, M. Mukherjee, Y. Choi, S. Pyo. Control over multifunctionality in optoelectronic device based on organic phototransistor. ACS Appl. Mater. Interfaces 2 (2010) 1614–1620. DOI:10.1021/am100127q |
| [20] | Y.Y. Noh, D.Y. Kim, K. Yase. Highly sensitive thin-film organic phototransistors: Effect of wavelength of light source on device performance. J. Appl. Phys. 98 (2005) 074505. DOI:10.1063/1.2061892 |
| [21] | R. Capelli, S. Toffanin, G. Generali, et al. Organic light-emitting transistors with an efficiency that outperforms the equivalent light-emitting diodes. Nat. Mater. 9 (2010) 496–503. DOI:10.1038/nmat2751 |
| [22] | A. Hepp, H. Heil, W. Weise, et al. Light-emitting field-effect transistor based on a tetracene thin film. Phys. Rev. Lett. 91 (2003) 157406. DOI:10.1103/PhysRevLett.91.157406 |
| [23] | R.S. Dudhe, J. Sinha, A. Kumar, V.R. Rao. Polymer composite-based OFET sensor with improved sensitivity towards nitro based explosive vapors. Sens. Actuators B 148 (2010) 158–165. DOI:10.1016/j.snb.2010.04.022 |
| [24] | S.C. Mannsfeld, B.C. Tee, R.M. Stoltenberg, et al. Highly sensitive flexible pressure sensors with microstructured rubber dielectric layers. Nat. Mater. 9 (2010) 859–864. DOI:10.1038/nmat2834 |
| [25] | T.B. Singh, N. Marjanovic, G. Matt, et al. Nonvolatile organic field-effect transistor memory element with a polymeric gate electret. Appl. Phys. Lett. 85 (2004) 5409–5411. DOI:10.1063/1.1828236 |
| [26] | W. Wu, H. Zhang, Y. Wang, et al. High-performance organic transistor memory elements with steep flanks of hysteresis. Adv. Funct. Mater. 18 (2008) 2593–2601. DOI:10.1002/adfm.v18:17 |
| [27] | C.a. Di, F. Zhang, D. Zhu. Multi-functional integration of organic field-effect transistors (OFETs): advances and perspectives. Adv. Mater. 25 (2013) 313–330. DOI:10.1002/adma.201201502 |
| [28] | Y. Guo, G. Yu, Y. Liu. Functional organic field-effect transistors. Adv. Mater. 22 (2010) 4427–4447. DOI:10.1002/adma.v22:40 |
| [29] | R. Yerushalmi, A. Scherz, M.E. van der Boom, H.B. Kraatz. Stimuli responsive materials: new avenues toward smart organic devices. J. Mater. Chem. 15 (2005) 4480–4487. DOI:10.1039/b505212b |
| [30] | E.S. Matsuo, T. Tanaka. Patterns in shrinking gels. Nature 358 (1992) 482–485. DOI:10.1038/358482a0 |
| [31] | W. Frey, D.E. Meyer, A. Chilkoti. Dynamic addressing of a surface pattern by a stimuli-responsive fusion protein. Adv. Mater. 15 (2003) 248–251. DOI:10.1002/adma.200390058 |
| [32] | S. Mendez, L.K. Ista, G.P. López. Use of stimuli responsive polymers grafted on mixed self-assembled monolayers to tune transitions in surface energy. Langmuir 19 (2003) 8115–8116. DOI:10.1021/la034632l |
| [33] | R.C. Naber, C. Tanase, P.W. Blom, et al. High-performance solution-processed polymer ferroelectric field-effect transistors. Nat. Mater. 4 (2005) 243–248. DOI:10.1038/nmat1329 |
| [34] | J. Covington, J. Gardner, D. Briand, N. De Rooij. A polymer gate FET sensor array for detecting organic vapours. Sens. Actuators B 77 (2001) 155–162. DOI:10.1016/S0925-4005(01)00687-6 |
| [35] | A.N. Sokolov, B.C. Tee, C.J. Bettinger, J.B.H. Tok, Z. Bao. Chemical and engineering approaches to enable organic field-effect transistors for electronic skin applications. Acc. Chem. Res. 45 (2011) 361–371. |
| [36] | V. Podzorov, V. Pudalov, M. Gershenson. Light-induced switching in back-gated organic transistors with built-in conduction channel. Appl. Phys. Lett. 85 (2004) 6039–6041. DOI:10.1063/1.1836877 |
| [37] | F. Zhang, C. Di, N. Berdunov, et al. Ultrathin film organic transistors: precise control of semiconductor thickness via spin-coating. Adv. Mater. 25 (2013) 1401–1407. DOI:10.1002/adma.v25.10 |
| [38] | J.T. Mabeck, G.G. Malliaras. Chemical and biological sensors based on organic thin-film transistors. Anal. Bioanal. Chem. 384 (2006) 343–353. |
| [39] | E. Orgiu, N. Crivillers, M. Herder, et al. Optically switchable transistor via energy-level phototuning in a bicomponent organic semiconductor. Nat. Chem. 4 (2012) 675–679. DOI:10.1038/nchem.1384 |
| [40] | Y. Li, H. Zhang, C. Qi, X. Guo. Light-driven photochromism-induced reversible switching in P3HT-spiropyran hybrid transistors. J. Mater. Chem. 22 (2012) 4261–4265. DOI:10.1039/C1JM14872A |
| [41] | N. Crivillers, E. Orgiu, F. Reinders, M. Mayor, P. Samorì. Optical modulation of the charge injection in an organic field-effect transistor based on photochromic self-assembled-monolayer-functionalized electrodes. Adv. Mater. 23 (2011) 1447–1452. DOI:10.1002/adma.201003736 |
| [42] | H. Chen, N. Cheng, W. Ma, et al., Design of a photoactive hybrid bilayer dielectric for flexible nonvolatile organic memory transistors, ACS nano (2015). |
| [43] | Y. Wakayama, R. Hayakawa, H.S. Seo. Recent progress in photoactive organic field-effect transistors. Sci. Technol. Adv. Mater. 15 (2014) 024202. DOI:10.1088/1468-6996/15/2/024202 |
| [44] | M. Yoshida, K. Suemori, S. Uemura, et al. Development of field-effect transistortype photorewritable memory using photochromic interface layer. Jpn. J. Appl. Phys. 49 (2010) 04D. |
| [45] | C.A. Di, Y. Liu, G. Yu, D. Zhu. Interface engineering: an effective approach toward high-performance organic field-effect transistors. Acc. Chem. Res. 42 (2009) 1573–1583. DOI:10.1021/ar9000873 |
| [46] | M. Irie. Diarylethenes for memories and switches. Chem. Rev. 100 (2000) 1685–1716. DOI:10.1021/cr980069d |
| [47] | L. Hou, X. Zhang, T.C. Pijper, W.R. Browne, B.L. Feringa. Reversible photochemical control of singlet oxygen generation using diarylethene photochromic switches. J. Am. Chem. Soc. 136 (2014) 910–913. DOI:10.1021/ja4122473 |
| [48] | F.M. Raymo, M. Tomasulo. Optical processing with photochromic switches. Chem. Eur. J. 12 (2006) 3186–3193. DOI:10.1002/(ISSN)1521-3765 |
| [49] | H. Tian, S. Yang. Recent progresses on diarylethene based photochromic switches. Chem. Soc. Rev. 33 (2004) 85–97. DOI:10.1039/b302356g |
| [50] | V.A. Bren. Fluorescent and photochromic chemosensors. Russ. Chem. Rev. 70 (2001) 1017–1036. DOI:10.1070/RC2001v070n12ABEH000667 |
| [51] | K. Fries, S. Samanta, S. Orski, J. Locklin, Reversible colorimetric ion sensors based on surface initiated polymerization of photochromic polymers, Chem. Commun. (2008) 6288-6290. |
| [52] | M. Alonso, V. Reboto, L. Guiscardo, A. San Martin, J. Rodriguez-Cabello. Spiropyran derivative of an elastin-like bioelastic polymer: photoresponsive molecular machine to convert sunlight into mechanical work. Macromolecules 33 (2000) 9480–9482. DOI:10.1021/ma001348h |
| [53] | V. Balzani, A. Credi, M. Venturi. Light powered molecular machines. Chem. Soc. Rev. 38 (2009) 1542–1550. DOI:10.1039/b806328c |
| [54] | Y. Norikane, N. Tamaoki. Light-driven molecular hinge: a new molecular machine showing a light-intensity-dependent photoresponse that utilizes the trans-cis isomerization of azobenzene. Org. Lett. 6 (2004) 2595–2598. DOI:10.1021/ol049082c |
| [55] | C.C. Corredor, Z.L. Huang, K.D. Belfield. Two-photon 3D optical data storage via fluorescence modulation of an efficient fluorene dye by a photochromic diarylethene. Adv. Mater. 18 (2006) 2910–2914. DOI:10.1002/(ISSN)1521-4095 |
| [56] | S. Kawata, Y. Kawata. Three-dimensional optical data storage using photochromic materials. Chem. Rev. 100 (2000) 1777–1788. DOI:10.1021/cr980073p |
| [57] | S. Luo, K. Chen, L. Cao, et al. Photochromic diarylethene for rewritable holographic data storage. Opt. Express 13 (2005) 3123–3128. DOI:10.1364/OPEX.13.003123 |
| [58] | T. Ikeda, T. Sasaki, K. Ichimura. Photochemical switching of polarization in ferroelectric liquid-crystal films. Nature 361 (1993) 428–430. DOI:10.1038/361428a0 |
| [59] | T. Ikeda, O. Tsutsumi. Optical switching and image storage by means of azobenzene liquid-crystal films. Science 268 (1995) 1873–1875. DOI:10.1126/science.268.5219.1873 |
| [60] | Y. Yokoyama, T. Sagisaka, Reversible control of pitch of induced cholesteric liquid crystal by optically active photochromic fulgide derivatives, Chem. Lett. (1997) 687-688. |
| [61] | F.M. Raymo, S. Giordani. Signal processing at the molecular level. J. Am. Chem. Soc. 123 (2001) 4651–4652. DOI:10.1021/ja005699n |
| [62] | C. Lenoble, R.S. Becker. Photophysics, photochemistry, kinetics, and mechanism of the photochromism of 60-nitroindolinospiropyran. J. Phys. Chem. 90 (1986) 62–65. DOI:10.1021/j100273a015 |
| [63] | D.A. Parthenopoulos, P.M. Rentzepis. Three-dimensional optical storage memory. Science 245 (1989) 843–845. DOI:10.1126/science.245.4920.843 |
| [64] | R. Klajn. Spiropyran-based dynamic materials. Chem. Soc. Rev. 43 (2014) 148–184. DOI:10.1039/C3CS60181A |
| [65] | I. Vlassiouk, C.-D. Park, S.A. Vail, D. Gust, S. Smirnov. Control of nanopore wetting by a photochromic spiropyran: a light-controlled valve and electrical switch. Nano Lett. 6 (2006) 1013–1017. DOI:10.1021/nl060313d |
| [66] | B.I. Ipe, S. Mahima, K.G. Thomas. Light-induced modulation of self-assembly on spiropyran-capped gold nanoparticles: a potential system for the controlled release of amino acid derivatives. J. Am. Chem. Soc. 125 (2003) 7174–7175. DOI:10.1021/ja0341182 |
| [67] | S. Abe, K. Uchida, I. Yamazaki, M. Irie. Fatigue-resistance property of diarylethene LB films in repeating photochromic reaction. Langmuir 13 (1997) 5504–5506. DOI:10.1021/la970555g |
| [68] | G. Hartley. The cis-form of azobenzene. Nature 140 (1937) 281. |
| [69] | K.G. Yager, C.J. Barrett. Novel photo-switching using azobenzene functional materials. J. Photochem. Photobiol. A 182 (2006) 250–261. DOI:10.1016/j.jphotochem.2006.04.021 |
| [70] | A.J. Harvey, A.D. Abell. Azobenzene-containing, peptidyl α-ketoesters as photobiological switches of α-chymotrypsin. Tetrahedron 56 (2000) 9763–9771. DOI:10.1016/S0040-4020(00)00883-8 |
| [71] | O. Sadovski, A.A. Beharry, F. Zhang, G.A. Woolley. Spectral tuning of azobenzene photoswitches for biological applications. Angew. Chem. Int. Ed. 48 (2009) 1484–1486. DOI:10.1002/anie.v48:8 |
| [72] | M.-S. Ho, A. Natansohn, P. Rochon. Azo polymers for reversible optical storage. 9. Copolymers containing two types of azobenzene side groups. Macromolecules 29 (1996) 44–49. DOI:10.1021/ma950485p |
| [73] | Z.F. Liu, K. Hashimoto, A. Fujishima, Photoelectrochemical Information Storage Using an Azobenzene Derivative, 1990. |
| [74] | P. H. Rasmussen, P. Ramanujam, S. Hvilsted, R.H Berg, et al. A remarkably efficient azobenzene peptide for holographic information storage. J. Am. Chem. Soc. 121 (1999) 4156–4161. |
| [75] | M. El Gemayel, K. Börjesson, M. Herder, et al. Optically switchable transistors by simple incorporation of photochromic systems into small-molecule semiconducting matrices. Nat. Commun. 6 (2015) . |
| [76] | K. Börjesson, M. Herder, L. Grubert, et al. Optically switchable transistors comprising a hybrid photochromic molecule/n-type organic active layer. J. Mater. Chem. C 3 (2015) 4156–4161. DOI:10.1039/C5TC00401B |
| [77] | Q. Shen, Y. Cao, S. Liu, M.L. Steigerwald, X. Guo. Conformation-induced electrostatic gating of the conduction of spiropyran-coated organic thin-film transistors. J. Phys. Chem. C 113 (2009) 10807–10812. DOI:10.1021/jp9026817 |
| [78] | B. Bunker, B. Kim, J. Houston, et al. Direct observation of photo switching in tethered spiropyrans using the interfacial force microscope. Nano Lett. 3 (2003) 1723–1727. DOI:10.1021/nl034759v |
| [79] | Y. Ishiguro, R. Hayakawa, T. Chikyow, Y. Wakayama. Optical switching of carrier transport in polymeric transistors with photochromic spiropyran molecules. J. Mater. Chem. C 1 (2013) 3012–3016. DOI:10.1039/c3tc30130c |
| [80] | Y. Ishiguro, R. Hayakawa, T. Yasuda, T. Chikyow, Y. Wakayama. Unique device operations by combining optical-memory effect and electrical-gate modulation in a photochromism-based dual-gate transistor. ACS Appl. Mater. Interfaces 5 (2013) 9726–9731. DOI:10.1021/am402833k |
| [81] | Y. Ishiguro, R. Hayakawa, T. Chikyow, Y. Wakayama. Optically controllable dualgate organic transistor produced via phase separation between polymer semiconductor and photochromic spiropyran molecules. ACS Appl. Mater. Interfaces 6 (2014) 10415–10420. DOI:10.1021/am501884q |
| [82] | M. Arlt, A. Scheffler, I. Suske, et al. Bipolar redox behaviour, field-effect mobility and transistor switching of the low-molecular azo glass AZOPD. Phys Chem. Chem. Phys. 12 (2010) 13828–13834. DOI:10.1039/c0cp00643b |
| [83] | R. Hayakawa, K. Higashiguchi, K. Matsuda, T. Chikyow, Y. Wakayama. Optically and electrically driven organic thin film transistors with diarylethene photochromic channel layers. ACS Appl. Mater. Interfaces 5 (2013) 3625–3630. DOI:10.1021/am400030z |
| [84] | R. Ruiz, A. Papadimitratos, A.C. Mayer, G.G. Malliaras. Thickness dependence of mobility in pentacene thin-film transistors. Adv. Mater. 17 (2005) 1795–1798. DOI:10.1002/(ISSN)1521-4095 |
| [85] | Q. Shen, L. Wang, S. Liu, et al. Photoactive gate dielectrics. Adv. Mater. 22 (2010) 3282–3287. DOI:10.1002/adma.201000471 |
| [86] | P. Lutsyk, K. Janus, J. Sworakowski, et al. Photoswitching of an n-type organic field effect transistor by a reversible photochromic reaction in the dielectric film. J. Phys. Chem. C 115 (2011) 3106–3114. |
| [87] | P. Lutsyk, K. Janus, J. Sworakowski, A. Kochalska, S. Nešpůrek. Kinetic study of light-driven processes in photochromic dye-doped polymers used as gate insulators in photoswitchable organic field effect transistors. Chem. Phys. 404 (2012) 22–27. DOI:10.1016/j.chemphys.2012.01.020 |
| [88] | H. Zhang, J. Hui, H. Chen, et al. Synergistic photomodulation of capacitive coupling and charge separation toward functional organic field-effect transistors with high responsivity. Adv. Electron. Mater. 1 (2015) 1500159. DOI:10.1002/aelm.201500159 |
| [89] | H. Zhang, X. Guo, J. Hui, et al. Interface engineering of semiconductor/dielectric heterojunctions toward functional organic thin-film transistors. Nano Lett. 11 (2011) 4939–4946. DOI:10.1021/nl2028798 |
| [90] | C.W. Tseng, D.C. Huang, Y.T. Tao. Electric bistability induced by incorporating self-assembled monolayers/aggregated clusters of azobenzene derivatives in pentacene-based thin-film transistors. ACS Appl. Mater. Interfaces 4 (2012) 5483–5491. DOI:10.1021/am3013906 |
| [91] | H.S. Lim, J.T. Han, D. Kwak, M. Jin, K. Cho. Photoreversibly switchable superhydrophobic surface with erasable and rewritable pattern. J. Am. Chem. Soc. 128 (2006) 14458–14459. DOI:10.1021/ja0655901 |
| [92] | F.L. Callari, S. Petralia, S. Conoci, S. Sortino. Light-triggered DNA release by dynamic monolayer films. N. J. Chem. 32 (2008) 1899–1903. DOI:10.1039/b808118b |
| [93] | M. Wirkner, J.M. Alonso, V. Maus, et al. Triggered cell release from materials using bioadhesive photocleavable linkers. Adv. Mater. 23 (2011) 3907–3910. DOI:10.1002/adma.v23.34 |
| [94] | N. Crivillers, A. Liscio, F. Di Stasio, et al. Photoinduced work function changes by isomerization of a densely packed azobenzene-based SAM on Au: a joint experimental and theoretical study. Phys. Chem. Chem. Phys. 13 (2011) 14302–14310. DOI:10.1039/c1cp20851a |
| [95] | T. Mosciatti, M.G. del Rosso, M. Herder, et al. Light-modulation of the charge injection in a polymer thin-film transistor by functionalizing the electrodes with bistable photochromic self-assembled monolayers. Adv. Mater. 31 (2016) 6606–6611. |
 2016, Vol. 27
2016, Vol. 27 


