b Department of Chemistry, Shahid Beheshti University, G. C., Evin, Tehran 1983963113, Iran
Multicomponent reactions (MCRs), particularly one-pot processes, are of current interest to organic chemists because of being rapid, their convergence, productivity, facile execution [1] cost-effectiveness such as atom economy [2] and lower the costs of reagents and solvents [3], protection-deprotection steps, less tedious work-up and purification [4].Multicomponent reactions provide unmatched opportunities for the expeditious increase of complexity and diversity in synthetic outcomes. Isocyanide-based multicomponent reactions (IMCRs) have emerged as an efficient and powerful tool in synthetic organic chemistry [5]. One class of these reactions is the generation of zwitterionic intermediates with the nucleophilic addition of isocyanides to activate acetylene [6].
Compounds containing furan rings are extensively found in many biologically active natural products. They are used in preparation of many pharmaceutical products such as ascorbic acid [7], ranitidine [8], phomactin A [9], azimilide [10], dantrolene [11], nitrofurazone [12], perillene [13], teubrevin G and teulepicin [14]. Moreover, in commerce and business, furans are important intermediates in the preparation of dyes, essential oils, agrochemical bioregulators, cosmetics and photosensitizers [15, 16].
Much attention has been devoted to a large variety of nitrogencontaining heterocyclics and heterocyclic quinoxalines because of their pharmacological properties and clinical applications [17]. The quinoxaline derivatives are an important group of azapolycyclics [18], while indenoquinoxaline derivatives are important classes of N-heterocycles since both are useful intermediates for spiroindeno synthesis. The main structure of many spiro compounds exhibit valuable (advantageous) pharmacological properties such anti-tumor agents [19, 20], anti-cancer [21], natural alkaloids [22], and also biological properties like antibacterial, anti-microbial [23], with an inhibitor growth factor receptor [24] of particular interest. Spiroheterocycles are also of considerable interest because the presence of a spirocarbon provides a strengthening of the structure [22, 25] and together with a variety of furanes are the main important core of many pharmacological agents [15].
Existing furan andquinoxalinemoieties in one spiromolecule can be attractive to organic and biological chemists due to the incorporation ofmore than one heterocyclic scaffold in one structure causes interesting biological properties.We therefore sought toMW irradiation in good yields [26].
In continuation of this work, develop a simple and versatile procedure for the combinatorial synthesis of a spiro-substituted furan-indenoquinoxaline library for biological screening.
In 2004 Azizian et al., reported the synthesis of spirofuranindenoquinoxalines via a three-component condensation reaction in DMF using herein, we report a one-pot, four-component procedure for synthesis of (Z)-dialkyl-5-(alkylimino)-5H-spiro[furan-2, 11'- indeno[1, 2-b]quinoxaline]-3, 4-dicarboxylates in excellent yields, via four component reaction ninhydrin, benzene-1, 2-diamines, and zwitterionic made up of dialkylacetylenedicarboxylates and alkylisocyanide in the CH2Cl2 at room temperature (Scheme 1).
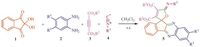
|
Download:
|
| Scheme. 1. Preparation of spirofuran-indenoquinoxalines derivatives. | |
2. Experimental
Melting points and IR spectra of all compounds were measured with an Electrothermal 9200 apparatus and a Perkin-Elmer 783 FTIR spectrometer, respectively. Also, the 1H NMR and 13C NMR spectrum were recorded on a Bruker Avance DPX-250 instrument using CDCl3 and DMSO-d6 as internal standard at 250 and 62.5 MHz, respectively. All of the compounds were purchased from Fluka, Merck, and Aldrich companies, and used without further purification.
2.1. General procedure for the preparation of compounds 5Ninhydrin 1 (1 mmol), benzene-1, 2-diamine 2 (1 mmol) were added at r.t. to CH2Cl2 (10 mL) while stirring. After ca. 10 min, the appropriate acetylenedicarboxylate 3 (1 mmol) in dichloromethane (5 mL) and the appropriate isocyanide 4 (1 mmol) in dichloromethane (5 mL) simultaneously were added dropwise over 20 min and the reaction mixture was stirred for 8 h. After completion of the reaction, the solvent was removed under vacuum and the product 5 was crystallized out from an EtOH-H2O and washed with Et2O (4mL × 2) to give a white crystalline solid.
(Z)-Dimethyl-5-(tert-butylimino)-5H-spiro[furan-2, 11'- indeno[1, 2-b]quinoxaline]-3, 4-dicarboxylate (5a): White crystal, mp: 277-279 ℃; IR (KBr): vmax 1750, 1725, 1685; 1 H NMR (250 MHz, CDCl3): δ 1.56 (s, 9H, 3Me), 3.39 (s, 3H, OMe), 3.96 (s, 3H, OMe), 7.44-7.48 (m, 1H, Ar), 7.50-7.64 (m, 3H, Ar), 7.66-7.79 (m, 3H, Ar), 8.12-8.20 (m, 1H, Ar); 13C NMR (62.5 MHz, CDCl3): δ 30.70, 31.40, 54.09, 54.30, 61.12, 112.24, 113.31, 124.55, 126.87, 130.59, 130.98, 131.13, 132.22, 132.74, 133.24, 139.62, 141.92, 142.39, 144.44, 154.17, 157.32, 162.05.
(Z)-Dimethyl-5-(cyclohexylimino)-5H-spiro[furan-2, 11'- indeno[1, 2-b]quinoxaline]-3, 4-dicarboxylate (5b): White crystal; mp 233-235 ℃; IR (KBr): vmax 1751, 1728, 1681, 1439; 1H NMR (250 MHz, DMSO-d6): δ 1.18-1.89 (m, 10H, 5CH2 of cyclohexyl), 4.09 (q, 1H, J = 18.5 Hz, CH-N of cyclohexyl), 3.31 (s, 3H, OMe), 3.91 (s, 3H, OMe), 7.53-7.56 (m, 1H, Ar), 7.65-7.77 (m, 3H, Ar), 7.80- 7.95 (m, 3H, Ar), 8.16-8.21 (m, 1H, Ar); 13CNMR(62.5 MHz, DMSOd6): d 23.67, 25.20, 32.21, 32.31, 33.12, 38.13, 52.96, 53.24, 64.16, 88.12, 119.71, 123.34, 125.67, 125.94, 129.40, 130.05, 131.4, 131.61, 133.00, 137.06, 141.16, 141.5, 143.12, 162.22, 166.19.
(Z)-Diethyl-5-(cyclohexylimino)-5H-spiro[furan-2, 11'- indeno[1, 2-b]quinoxaline]-3, 4-dicarboxylate (5c): White crystal; mp 220-222 ℃; IR (KBr): vmax 2940, 1743, 1720, 1685, 1651; 1H NMR (250 MHz, CDCl3): δ 0.84 (m, 3H, Me), 1.45 (m, 3H, Me), 1.20- 1.82 (m, 10H, 5CH2 of cyclohexyl), 3.61 (m, 1H, CH-N of cyclohexyl), 3.85 (dq, 2H, 2J = 15.7 Hz, 3J = 6.5 Hz, OCH2), 4.51 (m, 2H, OCH2), 7.28-8.23 (m, 8H, Arom.); 13C NMR (62.5 MHz, CDCl3): δ 13.78, 14.53, 25.07, 25.16, 2609, 33.58, 33.63, 57.14, 62.08, 62.95, 90.26, 123.15, 124.91, 129.65, 129.80, 130.45, 130.96, 131.86, 132.62, 138.36, 139.27, 142.12, 142.14, 143.36, 143.42, 154.46, 155.14, 157.68, 159.88, 162.16.
(Z)-Dimethyl-5-(tert-butylimino)-8'-methyl-5H-spiro[furan- 2, 11'-indeno[1, 2-b]quinoxaline]-3, 4-dicarboxylate (5d): White crystal; mp 222-225 ℃; IR (KBr): vmax 1752, 1731, 1666, 1649; 1H NMR (250 MHz, CDCl3): δ 1.25 (s, 9H, 3Me), 2.53 (s, 3H, Me), 3.44 (s, 3H, OMe), 4.03 (s, 3H, OMe), 7.49-8.20 (m, 7H, Ar.); 13C NMR (62.5 MHz, CDCl3): δ 29.95, 33.64, 53.15, 53.64, 55.68, 90.76, 123.24, 124.72, 129.66, 129.78, 130.47, 130.96, 131.86, 132.62, 138.30, 140.60, 141.02, 142.17, 143.29, 143.44, 153.06, 154.37, 157.58, 160.48, 162.86.
(Z)-Dimethyl-5-(cyclohexylimino)-8'-methyl-5H-spiro[furan- 2, 11'-indeno[1, 2-b]quinoxaline]-3, 4-dicarboxylate (5e): White crystal; mp 237-238 ℃; IR (KBr): vmax 1752, 1729, 1675, 1646; 1H NMR (250 MHz, CDCl3): δ 1.18-1.84 (m, 10H, 5CH2 of cyclohexyl), 2.51 (s, 3H, Me), 3.44 (s, 3H, OMe), 3.59 (m, 1H, CH-N of cyclohexyl), 4.03 (s, 3H, OMe), 7.38-8.25 (m, 7H, Ar); 13C NMR (62.9 MHz, CDCl3): δ 20.65, 20.84, 25.12, 26.06, 33.57, 53.11, 53.70, 57.24, 90.78, 122.96, 124.74, 128.95, 129.66, 131.84, 132.18, 138.65, 139.13, 140.35, 140.98, 141.56, 142.24, 142.33, 142.86, 153.51, 156.40, 160.45, 162.65.
(Z)-Dimethyl-5-(tert-butylimino)-7', 8'-dichloro-5H-spiro[- furan-2, 11'-indeno[1, 2-b]quinoxaline]-3, 4-dicarboxylate (5f): White crystal; mp: 219-222 ℃; IR (KBr): vmax 1758, 1764, 1671, 1653; 1H NMR (250 MHz, CDCl3): δ 1.35 (s, 9H, Me), 3.35 (s, 3H, OMe), 3.84 (s, 3H, OMe), 7.17-8.22 (m, 6H, Ar); 13C NMR (62.9 MHz, CDCl3): δ 29.7, 52.78 53.3, 56.9, 78.49, 118.59, 123.06, 123.14, 124.50, 129.86, 130.57, 131.08, 131.68, 132.2, 132.81, 143.14, 153.74, 161.43, 163.64.
(Z)-Dimethyl-7', 8'-dichloro-5-(cyclohexylimino)-5H-spiro[- furan-2, 11'-indeno[1, 2-b]quinoxaline]-3, 4-dicarboxylate (5g): White crystal; mp: 247-249 ℃; IR (KBr): vmax 1750, 1726, 1684, 1440; 1H NMR (250 MHz, CDCl3): δ 1.11-1.92 (m, 10H, 5CH2 of cyclohexyl), 3.42 (s, 3H, OMe), 3.99 (s, 3H, OMe), 7.25-8.30 (m, 6H, Ar); 13C NMR (62.5 MHz, CDCl3): δ 25.6, 29.7, 33.16, 52.78 53.3, 56.9, 78.49, 118.59, 123.06, 123.14, 124.50, 129.86, 130.57, 131.08, 131.68, 132.2, 132.81, 143.14, 153.74.
3. Results and discussionRecent success in conjunction with isocyanide-based, multicomponent reactions provided a means to constructing novel heterocycles systems [15]. The polycyclic heterocycles, like indenoquinoxalines, leads to forming the rings of spiroindenoquinoxalines. In our initial test reaction, the three components, between 11H-indeno[1, 2-b]quinoxalin-11-one 6a, tert-butyl isocyanide 4 and dimethylacetylenedicaboxylate (DMAD) 3 without any catalyst in dry CH2Cl2 at r.t for 10 h, gave (Z)-dimethyl-5-(tertbutylimino)- 5H-spiro[furan-2, 11'-indeno[1, 2-b]quinoxaline]-3, 4- dicarboxylate 5a in excellent yield (Scheme 2).

|
Download:
|
| Scheme. 2. The synthesis of spirofuran-indenoquinoxalines via a three-component condensation reaction. | |
To optimize the conditions, various aprotic and protic solvents were investigated at reflux and r.t. conditions. In protic solvents the desired product was not obtained. However, the desired product was formed in aprotic solvents and was produced under the. Best the conditions in dry CH2Cl2 at r.t. with excellent yield. The results are summarized in Table 1.
|
|
Table 1 Optimization of conditions for the formation of spirofuran-indenoquinoxaline 5a. |
To minimize the steps required for the formation of the product and to simplify the procedure, a one-pot, four-component reaction was planned (Scheme 1) understanding the necessity to separate intermediate 6 as well as the control of reaction time. Our studies for this model reaction revealed that 5a could be synthesized in a one-pot reaction if 6a was prepared in situ from condensation of ninhydrin 1 with benzene-1, 2-diamine 2a at r.t. within 10 min., then treated with equimolar amounts of DMAD and tert-butyl isocyanide within 8 h at r.t (Scheme 3). Therefore, the implication of 11H-indeno[1, 2-b]quinoxalin-11-one 6a in the initial reaction is confirmed.

|
Download:
|
| Scheme. 3. A synthesis of spirofuran-indenoquinoxalines via isocyanid-based one-pot four-component reaction. | |
Having optimized the conditions, various isocyanide 4 and acetylenedicarboxylate 3 were condensed with ninhydrin 1 and benzene-1, 2-diamines 2 to afford the corresponding products 5a- 5g (Scheme 1). The results presented in Table 2 show that the various benzene-1, 2-diamine and the methyl and ethyl acetylenedicarboxylates and isocyanides were successfully applied in this process to afford the corresponding (Z)-dialkyl-5-(alkylimino)-5Hspiro[furan-2, 11'-indeno[1, 2-b]quinoxaline]-3, 4-dicarboxylates in excellent yields.
|
|
Table 2 One-pot, four-component synthesis of spirofuran-indenoquinoxalines 5a–5g. |
The plausible mechanism for the synthesis of (Z)-dialkyl-5- (alkylimino)-5H-spiro[furan-2, 11'-indeno[1, 2-b]quinoxaline]-3, 4- dicarboxylate compounds 5 is shown in Scheme 4. We envisioned that this reaction could be realized in a one-pot, two-step manner. Initially, ninhydrin 1 and the benzene-1, 2-diamine 2 react to form the corresponding indenoquinoxalinone A, the zwitterionic intermediate B, formed by the 1:1 interaction between the isocyanide and acetylenedicarboxylate, which attacks preferentially the carbonyl group of indeno-quinoxalines 6, leading to dipolar intermediate C, and subsequently leading to ring fivemembered and the spirocyclic product 5a.

|
Download:
|
| Scheme. 4. Proposed mechanism for the synthesis of (Z)-dimethyl-5-(tert-butylimino)-5H-Spiro[furan-2, 11'-indeno[1, 2-b]quinoxaline]-3, 4-dicarboxylate. | |
In order to elucidate the structure of these compounds, we determined the structure of compound 5a by X-ray crystallography (X-STEP32 Version1.07b, X-ray structure evaluation package, 2000, Stoe&Cie, Darmstadt, Germany). The ORTEP diagram of 5a is presented in Fig. 1. X-Ray data for 5a: C26H23N3O5, M = 457.47, orthorhombic system, space group P212121, a = 11.053(2) , b = 12.311(3) , c = 16.914(3) Å , V = 2301.6(8) Å 3, Z = 4, Dcalc = 1.320 g cm-3, μ(Mo-Ka) = 0.093mm-1 , T = 120(2) K, crystal size of 0.50 × 0.38 × 0.10mm3. The X-ray diffraction measurement was made on a STOE IPDS-2T diffractometer with graphite monochromatedMo- Ka radiation. The structurewas solved using SHELXS. The structure refinementand data reductionwas carried outwithSHELXL using the X-STEP32 suite of programs. The non-hydrogen atomswere refined anisotropically by full matrix least-squares on F2 values to final R1 = 0.0901, wR2 = 0.1158 and S = 1.069 with 312 parameters using 6009 independent reflections. Hydrogen atoms were added in idealized positions. The crystallographic information file has been deposited with the Cambridge Data Centre, CCDC 1032226.

|
Download:
|
| Figure 1. ORTEP diagram of 5a. Thermal ellipsoids are at 30% probability level. | |
4. Conclusion
In conclusion, we have succeeded in developing a novel approach and a clean, convenient, simple and inexpensive method for the synthesis of spiroindenoquinoxalines via a one-pot reaction. This procedure offers significant advantages because of the minimization of labor, time, and cost. Noteworthy, our work presents a very easy and simple reaction, at r.t conditions, without any catalyst and in a short time frame. Additionally, the products obtained can be easily isolated via crystallization.
| [1] | G.H. Mahdavinia, M. Mirzazadeh, B. Notash, A rapid and simple diversity-oriented synthesis of novel 3-amino-2'-oxospiro [benzo[c]pyrano[3, 2-a]phenazine-1, 3'-indoline]-2-carbonitrile/carboxylate derivatives via a one-pot, four-component domino reaction, Tetrahedron Lett. 54 (2013) 3487-3492. |
| [2] | M. Ghandi, A. Tabatabaei Ghomi, M.J. Kubicki. Synthesis of cyclopentadiene-fused chromanones via one-pot multicomponent reactions. J. Org. Chem. 78 (2013) 2611–2616. DOI:10.1021/jo302790y |
| [3] | H.Y. Zhou, W. Zhang, B. Yan. Use of Cyclohexylisocyanide and methyl, 2-isocyanoacetate as convertible isocyanides for microwave-assisted fluorous synthesis of 1, 4-benzodiazepine-2, 5-dione library. J. Comb. Chem. 12 (2010) 206–214. DOI:10.1021/cc900157w |
| [4] | B. Jiang, S.J. Tu, P. Kaur, W.G. Wever Li. Four-component domino reaction leading to multifunctionalized quinazolines. J. Am. Chem. Soc. 131 (2009) 11660–11661. DOI:10.1021/ja904011s |
| [5] | E. Soleimani, M. Zainali, N. Ghasemi, B. Notash, Isocyanide-based multicomponent reactions: synthesis of 2-(1-(alkylcarbamoyl)-2, 2dicyanoethyl)-N-alkylbenzamide and 1, 7-diazaspiro[4, 4]nonane-2, 4-dione derivatives, Tetrahedron 69 (2013) 9832-9838. |
| [6] | (a) A.A. Esmaeili, H. Zendegani, Three-component reactions involving zwitterionic intermediates for the construction of heterocyclic systems: one pot synthesis of highly functionalized γ-iminolactones, Tetrahedron 61 (2005) 4031-4034; (b) I. Ugi, From isocyanides via four-component condensations to antibiotic syntheses, Angew. Chem. Int. Ed. Engl. 21 (1982) 810-819. |
| [7] | M. Ghandi, N. Zarezadeh. Three-component one-pot synthesis of quinoline-furan conjugates from acetylenedicarboxylate, isocyanide, and, 2-chloroquinoline-3-carbaldehyde. . |
| [8] | J.L. Roux, H. Gallard, J.P. Croué, S. Papot, M. Deborde. NDMA formation by chloramination of ranitidine: kinetics and mechanism. Environ. Sci. Technol. 46 (2012) 11095–11103. DOI:10.1021/es3023094 |
| [9] | P.J. Mohr, R.L. Halcomb. Total synthesis of (+)-phomactin a using a B-alkyl Suzuki macrocyclization. J. Am. Chem. Soc. 125 (2003) 1712–1713. DOI:10.1021/ja0296531 |
| [10] | K.E. Miller, J.F. Carpenter, R.R. Brooks. Inhibition of isoproterenol-induced tachycardia by azimilide in the isolated perfused guinea pig heart. Cardiovasc. Drug Ther. 12 (1998) 83–91. DOI:10.1023/A:1007766220822 |
| [11] | T. Hosoya, H. Aoyama, T. Ikemoto, et al. Dantrolene analogues revisited: general synthesis and specific functions capable of discriminating two kinds of Ca2+ release from sarcoplasmic reticulum of mouse skeletal muscle. Bioorg. Med. Chem. 11 (2003) 663–673. DOI:10.1016/S0968-0896(02) 00600-4 |
| [12] | C. Van Poucke, C. Detavernier, M. Wille, et al. Investigation into the possible natural occurence of semicarbazide in macrobrachium rosenbergii Prawns. J. Agric. Food Chem. 59 (2011) 2107–2112. DOI:10.1021/jf103282g |
| [13] | D.K. Barma, A. Kundu, R. Baati, C. Mioskowski, J.R. Falck. A convenient preparation of, 3-substituted furans: synthesis of perillene and dendrolasin. Org. Lett. 4 (2002) 1387–1389. DOI:10.1021/ol025708s |
| [14] | D.G. Velazquez, R. Luque. Efficient and straightforward preparation of a building block for ( )-teubrevin G synthesis via chemically diversed oriented synthesis. Tetrahedron Lett. 52 (2011) 7004–7007. DOI:10.1016/j.tetlet.2011.10.112 |
| [15] | (a) M. Adib, E. Sheikhi, A. Kavoosi, H.R. Bijanzadeh, Synthesis of 2-(alkylamino)-5-{alkyl[(2-oxo-2H-chromen-3-yl)carbonyl]amino}-3, 4-furandicarboxylates using a multi-component reaction in water, Tetrahedron 66 (2010) 9263-9269; (b) A. Dö mling, Recent developments in isocyanide based multicomponent reactions in applied chemistry, Chem. Rev. 106 (2006) 17-89. |
| [16] | H.N.C. Wong, Y. Yang, Regiospecific synthesis of 3, 4-disubstituted furans and 3-substituted furans using 3, 4-bis(tri-n-butylstannyl)furan and 3-(tri-n-butylstannyl) f, Tetrahedron 50 (1994) 9583-9608. |
| [17] | (a) F. Chen, J. Zheng, M. Huang, Y. Li, One-pot three-component synthesis of novel spiroindenoquinoxalines, Res. Chem. Intermed. 41 (2015) 5545-5554; (b) G.C.R. Rezanejade Bardajee, ZrOCl2.8H2O in water: an efficient catalyst for rapid one-pot synthesis of pyridopyrazines, pyrazines and 2, 3-disubstituted quinoxalines, Chimie 16 (2013) 872-877. |
| [18] | (a) A. Hasaninejad, N. Golzar, A. zare, One-pot, four-component synthesis of novel spiro[indeno[2, 1-b]quinoxaline-11, 4'-pyran]-2'-amines, J. Heterocycl. Chem. 50 (2013) 608-614; (b) A. Hasaninejad, N. Golzar, M. Shekouhy, A. Zare, Diversity-oriented synthesis of novel 2'-aminospiro[11H-indeno[1, 2-b]quinoxaline-11, 4'-[4H]pyran] derivatives via a one-pot four-component reaction, Helv. Chim. Acta 94 (2011) 2289-2294. |
| [19] | (a) A. Hasaninejad, S. Firoozi, One-pot, sequential four-component synthesis of benzo[c]pyrano[3, 2-a]phenazine, bis-benzo[c]pyrano[3, 2-a]phenazine and oxospiro benzo[c]pyrano[3, 2-a]phenazine derivatives using 1, 4-diazabicyclo[2.2.2]octane (DABCO) as an efficient and reusable solid base catalyst, Mol. Diversity 17 (2013) 499-513; (b) R. Ghadari, F. Hajishaabanha, M. Mahyari, A. Shaabani, H.R. Khavasi, An unexpected route toward the synthesis of spiro-benzo[b]acridine-furan derivatives, Tetrahedron. Lett. 53 (2012) 4018-4021. |
| [20] | A. Hasaninejad, S. Firoozi, F. Mandegani, An efficient synthesis of novel spiro[benzo[ c]pyrano[3, 2-a]phenazines] via domino multi-component reactions using lproline as a bifunctional organocatalyst, Tetrahedron Lett. 54 (2013) 2791-2794. |
| [21] | N. Tanaka, Y. Kashiwada, S.Y. Kim, et al., Acylphloroglucinol, biyouyanagiol, biyouyanagin b, and related spiro-lactones from Hypericum chinense, J. Nat. Prod. 72 (2009) 1447-1452. |
| [22] | D.M. Pore, P.B. Patil, D.S. Gaikwad, et al. Green access to novel spiro pyranopyrazole derivatives. Tetrahedron Lett. 54 (2013) 5876–5878. DOI:10.1016/j.tetlet.2013.08.084 |
| [23] | M.T. Maghsoodlou, S.M. Habibi-Khorassani, A. Moradi, et al. One-pot threecomponent synthesis of functionalized spirolactones by means of reaction between aromatic ketones, dimethyl acetylenedicarboxylate, and N-heterocycles. Tetrahedron 67 (2011) 8492–8495. DOI:10.1016/j.tet.2011.09.017 |
| [24] | T.A. Shoker, K.I. Ghattass, J.C. Fettinger, M.J. Kurth, M.J. Haddadin, Unusual Friedlander reactions: a route to novel quinoxaline-based heterocycles, Org. Lett. 14 (2012) 3704-3707. |
| [25] | (a) N. Zohreh, A. Alizadeh, Uncatalyzed one-pot synthesis of highly substituted pyridazines and pyrazoline-spirooxindoles via domino SN/condensation/Aza-ene addition cyclization reaction sequence, ACS Comb. Sci. 15 (2013) 278-286; (b) H. Chen, D. Shi, Efficient one-pot synthesis of novel spirooxindole derivatives via three-component reaction in aqueous medium, J. Comb. Chem. 12 (2010) 571-576. |
| [26] | J. Azizian, A.R. Karimi, A.A. Mohammadi, M.R. Mohammadizadeh. Three component synthesis of some g-spiroiminolactones under microwave-assisted solventfree conditions. Heterocycles 63 (2004) 2225–2229. DOI:10.3987/COM-04-10125 |
 2016, Vol. 27
2016, Vol. 27 




