Metal-organic frameworks (MOFs) are highly ordered crystalline materials prepared by self-assembly of metal ions with organic linkers to yield low density network structures of diverse topology [1]. MOFs,with their fascinating diverse structures,permanent nanoscale porosity,high surface area and uniform structured cavities [2, 3, 4],have become a rapidly developing research area and have attracted much attention in the last few years [5, 6]. They have been considered as alternative precursors to construct nanoporous carbons since the use of MOFs as hard templates to synthesize nanoporous carbon materials through high temperature carbonization process can offer the resulting nanoporous carbons with ordered micro-,meso-,and macropores depending on the original hard templates. Also,their synthesis process is easy and favorable for large scale production. So far,MOF-5 [7],ZIF-8 [8],ZIF-67 [9], Ni-MOF [10],and Al-MOF [11] have been demonstrated as promising precursors for yielding highly nanoporous carbons. MOFs derived nanoporous carbons (MOF-C) have shown good thermal and chemical stability,highly ordered mesoporous structure and high surface area [12, 13, 14]. They have been reported for the use in adsorption [15, 16, 17],catalysis [18],and electrochemical capacitance [19]. It has been reported that MOF-C materials were promising and efficient adsorbents with fast adsorption rate and high adsorption capacities [15, 20]. However,their nanoscale sizes and light weight can make them difficult to separate and recover from sample solution. On the other hand,the incorporation of magnetic property into the MOF-C materials can make the separation easier and convenient by using magnetic solid-phase extraction (MSPE).
MSPE is a mode of solid-phase extraction based on the use of magnetic adsorbents. In MSPE,a magnetic adsorbent is first dispersed in a sample solution. After adsorption for an appropriate time,the powdery magnetic material can be easily separated from the solution by using an external magnet without the need of additional filtration or centrifugation procedures,which makes the separation rapid and effective. When the external magnetic field is removed,the separated powdery magnetic material can be redispersed in a solution,which makes the following washing and desorption operations convenient [21, 22]. Since the magnetic adsorbent can be suspended in sample solution,the contact area between the analytes and the adsorbent is large,which is favorable for fast mass transfer and rapid reach of extraction equilibrium [23]. For the above reasons,MSPE has been increasingly explored in the field of sample preparation.
The MOF-5 framework is one of the most representative MOFs, in which oxocentered Zn4 tetrahedra are connected through linear terephthalic acid to give a cubic network structure and a threedimensional channel system [4]. The terephthalic acid in MOF-5 is rich in carbon and therefore it can serve as both the carbon precursor and as the template for the synthesis of nanoporous carbon materials.
In this work,a MOF-5-derived porous carbon MOF-5-C was synthesized by the direct carbonization of MOF-5 as both the precursor and template. Then,the magnetic MOF-5-derived porous carbon (Fe3O4@MOF-5-C) was fabricated by the chemical coprecipitation method. The prepared Fe3O4@MOF-5-C showed a high specific surface area and also strong magnetism due to the presence of iron oxide nanoparticles. Chlorophenols (CPs) are widely used as herbicides,insecticides,wood preservatives and disinfectants [24] and they are also often used in mushroom compost,although they are toxic and carcinogenic for human beings [25]. With the growth of mushrooms,the chlorophenols could remain in them. Considering that the Fe3O4@MOF-5-C could have a good adsorption capacity towards the benzene-ring structured compounds such as CPs via van der Waals forces,p- p stacking and hydrophobic interactions,some CPs were selected as the model analytes to evaluate the extraction performance of the Fe3O4@MOF-5-C. The relevant experimental parameters were optimized,and as a result,a simple and sensitive analytical method for the determination of CPs in mushrooms by Fe3O4@MOF-5-C based MSPE followed by HPLC was established.
2. Experimental 2.1. Reagents and chemicalsHPLC-grade methanol was purchased from Sinopharm Chemical Reagent Co.,(Beijing,China). Zn(NO3)2•6H2O,terephthalic acid (benzene-1,4-dicaboxylic acid),N,N-dimethylformamide (DMF), acetonitrile,acetone,hydrochloric acid,and sodium hydroxide were purchased from Beijing Chemical Reagents Company. The water used throughout the work was double-distilled on an SZ-93 automatic double-distiller (Shanghai Yarong Biochemistry Instrumental Factory). 2-Chlorophenol (2-CP),3-chlorophenol (3-CP), 2,3-dichlorophenol (2,3-CP),and 2,4-dichlorophenol (2,4-CP) were purchased from Agricultural Environmental Protection Institution (Tianjin,China). A mixture stock solution containing 2-CP,3-CP, 2,3-CP,and 2,4-CP each at 40.0 μg mL-1 was prepared in methanol. A series of standard solutions was prepared by mixing an appropriate amount of the stock solution with methanol in a 10 mL volumetric flask. All the standard solutions were stored at 4 8C in the dark.
2.2. InstrumentsAn Agilent Technologies 1260 Infinity LC/G1314F with 1260 VWD detector was used for all the experiments. A Centurysil C18 column (250 mm × 4.6 mm id,5.0 mm) from Dalian Johnsson Separation Science Technology Corporation (Dalian,China) was used for separations. The mobile phase was a mixture of methanol-water (60:40 v/v) at a flow rate of 1.0 mL min-1. The UV monitoring wavelength was chosen at 280 nm.
Scanning electron microscopy (SEM) images were observed on an S-4300 SEM instrument (HITACHI,Japan). Transmission electron microscopy (TEM) was conducted on a JEOL model JEM-2011(HR) (Tokyo,Japan). X-ray diffraction (XRD) (Brooker D8 ADVANC,Germany) measurements were made with Cu Ka radiation. The Brunauer-Emmett-Teller (BET) surface areas were determined from the N2 adsorption at 300 K using V-Sorb 2800P (China). The magnetic property was analyzed using an MPMS-XL-7 vibrating sample magnetometer (Quantum Design,USA) at room temperature.
2.3. Preparation of Fe3O4@MOF-5-CMOF-5 was prepared according to the reported method [26]. The carbonization of the MOF-5 nanoparticles was performed at 900 8C for 6 h under Ar flow to get the nanoporous carbon MOF- 5-C. For the preparation of the magnetic composite Fe3O4@MOF-5- C,0.2 g MOF-5-C was first suspended in 150 mL water at 50 8C under N2 atmosphere,and then,0.46 g (1.724 mmol) FeCl2•4H2O and 0.24 g (0.86 mmol) FeCl3•6H2O were added. After the solution was sonicated (200 W,40 kHz) for 10 min,the pH of the solution was adjusted to 11 with 25% NH3•H2O to precipitate the iron oxides. Then,the reaction was carried out at 50 8C for 1 h under constant mechanical stirring. The resulting powdery magnetic material was separated from the aqueous dispersion by an external magnet. Then,the precipitate was washed with double-distilled water until the pH reached 7. Finally,the Fe3O4@MOF-5-C composite was dried under vacuum. The schematic illustration for the synthesis of the Fe3O4@MOF-5-C is shown in Fig. 1. The reproducibility of the formation of Fe3O4@MOF-5-C from five different batches of Fe3O4@MOF-5-C was investigated. The experimental results showed that the adsorption capacity of the adsorbent for the analytes has no obvious changes with a relative standard deviation (RSD) of 6.1% from batch to batch.

|
Download:
|
| Fig. 1. Schematic illustration of the synthetic process of Fe3O4@MOF-5-C. | |
2.4. Sample preparation
Fresh mushroom samples were homogenized in a laboratory homogenizer. A 200.0 g portion of the homogenized mushroom sample was accurately weighed,and then placed in four 50 mL screw capped PTFE centrifugal tubes with a conical bottom. The samples were centrifuged at 9000 rpm for 20 min. Then,the sedimented phase in each tube was washed with 25 mL of doubledistilled water twice. All the supernatants were combined together in a 500 mL volumetric flask,to which double-distilled water was added to complete the volume. The obtained sample solution was stored at 4 8C for the following MSPE extractions.
2.5. MSPE proceduresFor the MSPE,8.0 mg of Fe3O4@MOF-5-C was added to 50 mL sample solution in a conical flask. The mixture was shaken on a slow-moving platform shaker for 10 min. Subsequently,the Fe3O4@MOF-5-C was separated from the sample solution by putting an external magnet at the bottom of the conical flask and the resulting supernatant was discarded. Next,the Fe3O4@MOF-5- C and the residual solution were all transferred to a 10 mL centrifuge tube. A magnet was put outside of the tube wall to aggregate the Fe3O4@MOF-5-C,and the residual supernatant was completely removed by a pipette. Finally,0.4 mL (0.2 mL × 2) of alkaline methanol (1% 1 mol L-1 [8TD$DIF] NaOH) as the desorption solution was added to the tube,and the analytes were desorbed from the Fe3O4@MOF-5-C by vortexing the tube for 1 min. After magnetic separation,the desorption solution was transferred to a 2 mL microcentrifuge tube and 4 μL of 1 mol L-1 HCl aqueous solution was added into the solution to neutralize its pH. Finally,20 mL of the resulting desorption solution was injected into the HPLC system for analysis. After being washed three times with 1 mL of alkaline methanol and then with 1 mL of water by vortexing for 1 min,the Fe3O4@MOF-5-C was reused for the next extraction.
3. Results and discussion 3.1. Characterization of Fe3O4@MOF-5-CThe morphology of the Fe3O4@MOF-5-C was investigated by both SEM and TEM. The SEM image in Fig. 2a shows that the Fe3O4@MOF-5-C possesses a nanoporous structure. A comparison between Fig. 2a and b revealed that the Fe3O4 nanoparticles were successfully coated on the surface of MOF-5-C. The distribution of the Fe3O4 particles on the MOF-5-C was also observed from the TEM image of the Fe3O4@MOF-5-C (Fig. 2c).

|
Download:
|
| Fig. 2. The SEM images of the Fe3O4@MOF-5-C (a) and MOF-5-C (b), and the TEM image of the Fe3O4@MOF-5-C (c). | |
The XRD patterns of the MOF-5,MOF-5-C and Fe3O4@MOF-5-C are shown in Fig. S1 in Supporting information. The X-ray diffraction graph of MOF-5 shows that all the main peaks are attributed to the Zn. As shown in the XRD pattern of the MOF-5-C, the main peaks attributed to Zn disappeared,indicating that when the MOF-5 was carbonized at 900 8C,the Zn metal was vaporized away along with the Ar flow. In the XRD pattern of the Fe3O4@MOF-5-C,the diffraction peaks at 2u = 30.128,35.548, 43.228,53.528,57.148,and 62.688 correspond to the crystal indices of (2 2 0),(3 1 1),(4 0 0),(4 2 2),(5 1 1),and (4 4 0) of crystalline magnetite (Fe3O4),respectively. The above data matched well with the ones from the JCPDS card (19-0629) for Fe3O4.
Nitrogen adsorption-desorption isotherms were determined to evaluate the surface area and porous structure of the Fe3O4@MOF- 5-C. Fig. 3 shows that the nitrogen adsorption-desorption isotherms of the Fe3O4@MOF-5-C are typical type IV curves with hysteresis loops,which are characteristic of mesoporous materials. The specific surface area,total pore volume,and average pore diameter of the Fe3O4@MOF-5-C were determined to be 1058 m2 g-1,1.37 cm3 g-1,and 2.04 nm,respectively.

|
Download:
|
| Fig. 3. N2 adsorption-desorption isotherms of the Fe3O4@MOF-5-C. | |
The magnetization curve of the Fe3O4@MOF-5-C was measured at room temperature using a superconducting quantum interference device. As observed in Fig. S2 in Supporting information,the magnetic hysteresis loop was an S-shaped curve,suggesting that the Fe3O4@MOF-5-C had a super paramagnetic behavior. The saturation magnetization value of the Fe3O4@MOF-5-C was 32.8 emu g-1.
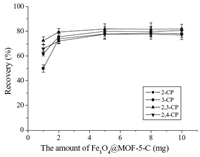
3.2. Effect of the amount of Fe3O4@MOF-5-CThe effect of the amount of Fe3O4@MOF-5-C on the MSPE of the CPs was investigated with 50 mL of water solution containing 80.0 ng mL-1 each of the CPs. Fig. 4 shows that the extraction recoveries increased when the amount of Fe3O4@MOF-5-C was increased from 1 mg to 5 mg,and then reached a plateau when the amounts of the magnetic sorbent exceeded 5 mg. The adsorption capacities of Fe3O4@MOF-5-C for 2-CP,3-CP,2,3-CP and 2,4-CP were measured to be 3.94,4.09,4.12 and 3.86 μg mg-1, respectively. To ensure that the adsorbent was sufficient for the extraction,8 mg of the Fe3O4@MOF-5-C was chosen for the following experiments.
|
Download:
|
| Fig. 4. Effect of the amount of the Fe3O4@MOF-5-C on the extraction recovery of CPs. Extraction conditions: sample volume, 50 mL; extraction time, 30 min; desorption solvent, 0.4 mL (0.2 mL × 2) alkaline methanol; concentration of each of the analytes, 80.0 ng mL-1. | |
3.3. Effect of extraction time and ionic strength
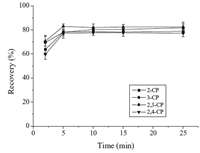
The extraction time profiles from 2 min to 30 min for the MSPE of the CPs are shown in Fig. 5. The results indicated that all of the CPs reached extraction equilibrium when the extraction time was increased to about 10 min. Therefore,10 min was chosen as the extraction time.
|
Download:
|
| Fig. 5. Effect of extraction time on the extraction recovery of CPs. Extraction conditions: amount of the sorbents, 8 mg; other conditions are the same as in Fig. 4. | |
Ionic strength is another parameter affecting the extraction efficiency. Adding salt to the aqueous sample solution can decrease the solubility of the organic analytes in aqueous phase due to the salting-out effect,which is often favorable for the extraction. However,on the other hand,the salt addition can also increase the viscosity of the solution,which is unfavorable for the extraction [27]. The effect of ionic strength was studied by changing the NaCl concentrations in the sample from 0% to 20%. The results indicated that the extraction recoveries for the CPs had a negligible change in the investigated NaCl concentration range. Therefore,no NaCl was added to the samples for the subsequent experiments.
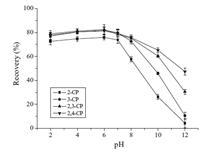
3.4. Effect of the pH of sample solutionThe CPs are acidic compounds with pKa values between 6.44 and 8.85 [25]. Their existing forms will be influenced by the pH of the sample solution. Therefore,the interactions between the CPs and the adsorbent will be affected by the sample solution pH. The effect of the sample solution pH on the extraction of the CPs was investigated in the pH range from 2 to 12. The result (Fig. 6) shows that the extraction recoveries for the CPs had no significant changes when the sample solution pH was changed from 2.0 to 6.0, and then a big decline of the extraction recoveries for all the CPs was observed when the sample solution pH was increased over 7.0. The reason for this may be that the CPs existed in a neutral molecular form in acidic media. When the pH was increased over 7.0,the CPs were ionized and their affinity toward the Fe3O4@MOF- 5-C became weakened. On the other hand,the zeta potential of the Fe3O4@MOF-5-C was measured to be about -5.0 mV at the pH value of 6,suggesting that the adsorbent was nearly neutral under the experimental conditions. Therefore,under the experimental conditions,the Fe3O4@MOF-5-C can interact with the analytes via non-covalent forces,such as van der Waals forces,π-π stacking, and hydrophobic interactions. Considering that the pH of the sample solution from the mushroom samples fell in the range of 5- 6,for the convenience of the experiment,the sample solution pH was not adjusted.
|
Download:
|
| Fig. 6. Effect of sample solution pH on the extraction recovery of the CPs. Extraction conditions: extraction time, 10 min; other conditions are the same as in Fig. 5. | |
3.5. Desorption conditions
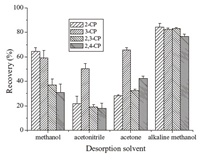
The desorption solvent was optimized to achieve a good desorption of the analytes from the Fe3O4@MOF-5-C. The most commonly used solvents,including methanol,alkaline methanol, acetonitrile,and acetone were tested for the desorption of the CPs from Fe3O4@MOF-5-C. The desorption capabilities of those solvents are given in Fig. 7. Under the same extraction and elution conditions,alkaline methanol provided the best desorption efficiency. The reason for this may be explained as that the alkaline condition was favorable for the ionization of the CPs,thus reducing their affinity to the adsorbent and facilitating the elution. Therefore,alkaline methanol was chosen as the desorption solvent.
|
Download:
|
| Fig. 7. Effect of the desorption solvents on the extraction recovery of the CPs. | |
The effect of the volume of alkaline methanol on desorption was also investigated by varying its volumes from 200 mL to 600 mL (0.2 mL × 1,0.2 mL × 2,0.2 mL × 3). The results showed that the extraction recoveries for all the CPs increased with increased volume of the alkaline methanol from 200 mL to 400 mL and then the extraction recoveries were almost constant. On the other hand, further increasing the elution volume was found to result in a decline in analytical signal due to the dilution effect. Therefore, 0.4 mL (0.2 mL × 2) was chosen.
3.6. Reusability of the Fe3O4@MOF-5-CThe reusability of the adsorbent was investigated in this study. After the desorption of the analytes from the adsorbent,the used Fe3O4@MOF-5-C was washed three times with 1 mL alkaline methanol and then with 1 mL water by vortexing for 1 min for the next extraction. Fig. S3 shows that the adsorbent can be reused 13 times without a significant loss of its extraction efficiency.
3.7. Analytical performance of the methodThe figures of merit for the developed MSPE method with Fe3O4@MOF-5-C as the adsorbent for the extraction of CPs followed by HPLC-UV detection are summarized in Table 1. The linear response for all the analytes was ranged from 0.8 ng g-1 to 100.0 ng g-1 with the correlation coefficients (r2) of 0.9923- 0.9963,which were evaluated using 50 mL of sample solution spiked with the CPs each at the concentrations of 1.0,5.0,10.0, 20.0,40.0,60.0,80.0,and 100.0 ng g-1,respectively. The limits of detection (LODs) at S/N = 3 were estimated to be from 0.25 ng g-1 [10TD$DIF] to 0.30 ng g-1 for the CPs. Five replicate analyses for the mushroom sample containing 40 ng mL-1 of each of the CPs were performed under the optimal conditions,and the resulting relative standard deviations (RSDs) were between 3.5% and 6.8%.
|
Download:
|
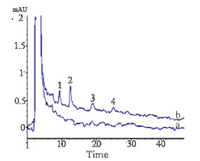
| Fig. 8. The typical chromatograms of mushroom (a) and the mushroom spiked with the CPs at each concentration of 5.0 ng g-1 (b). Peak identifications: (1). 2-CP, (2). 3- CP, (3). 2,3-CP, and (4). 2,4-CP. | |
|
|
Table 1 Analytical performance data for the CPs by the MSPE method |
3.8. Analysis of real samples
The method was applied to analyze mushroom samples bought from a local market in Baoding (China). The relevant data are listed in Table 2. The mushroom sample 2 was found to contain 0.89 ng g-1 of 2,3-CP. To test the accuracy of the method,the samples spiked with the CPs each at 5.0 and 10.0 ng g-1 were used to determine the recoveries of the analytes for the method. The resulting determined recoveries fell in the range from 85.4% to 97.5% with RSDs between 4.2% and 7.5%. Fig. 8 shows the typical chromatograms of the mushroom samples before and after being spiked with the CPs.
|
|
Table 2 Analytical results for the determination of CPs in mushroom samples. |
3.9. Comparison of the Fe3O4@MOF-5-C with other reported adsorbents
Some relevant data for the comparison between the Fe3O4@- MOF-5-C and other reported adsorbents in the literature [28, 29, 30, 31] for the extraction of CPs are listed in Table S1 in Supporting information. The data show that the current Fe3O4@MOF-5-C based method had comparable or even better performance compared to the other reported [1TD$DIF]relevant methods in terms of linearity,LODs,and RSDs.
4. ConclusionA magnetic nanoporous carbon Fe3O4@MOF-5-C was prepared as the MSPE adsorbent for the extraction of CPs from mushroom samples. The synthesized nanocomposite showed a high surface area,superparamagnetism,and good adsorption performance. The MSPE coupled with HPLC-UV detection for the CPs provides an alternative approach for a simple,fast and sensitive determination of the analytes.
Appendix A. Supplementary dataSupplementary data associated with this article can be found,in the online version,athttp://dx.doi.org/10.1016/j.cclet.2016.01.021.
| [1] | Y.L. Hu, Z.L. Huang, J. Liao, G.K. Li. Chemical bonding approach for fabrication of hybrid magnetic metal-organic framework-5:high efficient adsorbents for magnetic enrichment of trace analytes. Anal. Chem. 85 (2013) 6885–6893 |
| [2] | Q.W. Li, W.Y. Zhang, O.Š. Miljanić, et al. Docking in metal-organic frameworks. Science 325 (2009) 855–859 |
| [3] | H.C. Zhou, J.R. Long, O.M. Yaghi. Introduction to metal-organic frameworks. Chem. Rev. 112 (2012) 673–674 |
| [4] | S.S. Kaye, A.D. Dailly, O.M. Yaghi, J.R. Long. Impact of preparation and handling on the hydrogen storage properties of Zn4O (1,4-benzenedicarboxylate)3(MOF-5). J. Am. Chem. Soc. 129 (2007) 14176–14177 |
| [5] | L.E. Kreno, K. Leong, O.K. Farha, et al. Metal-organic framework materials as chemical sensors. Chem. Rev. 112 (2012) 1105–1125 |
| [6] | J.R. Li, J. Sculley, H.C. Zhou. Metal-organic frameworks for separations. Chem. Rev. 112 (2012) 869–932 |
| [7] | B. Liu, H. Shioyama, H.L. Jiang, X.B. Zhang, Q. Xu. Metal-organic framework (MOF) as a template for syntheses of nanoporous carbons as electrode materials for supercapacitor. Carbon 48 (2010) 456–463 |
| [8] | H.L. Jiang, B. Liu, Y.Q. Lan, et al. From metal-organic framework to nanoporous carbon:toward a very high surface area and hydrogen uptake. J. Am. Chem. Soc. 133 (2011) 11854–11857 |
| [9] | L. Hao, C. Wang, Q.H. Wu, et al. Metal-organic framework derived magnetic nanoporous carbon:novel adsorbent for magnetic solid-phase extraction. Anal. Chem. 86 (2014) 12199–12205 |
| [10] | L. Chen, J. Bai, C. Wang, et al. One-step solid-state thermolysis of a metal-organic framework:a simple and facile route to large-scale of multiwalled carbon nanotubes. Chem. Commun. 44 (2008) 1581–1583 |
| [11] | M. Hu, J. Reboul, S. Furukawa, et al. Direct carbonization of Al-based porous coordination polymer for synthesis of nanoporous carbon. J. Am. Chem. Soc. 134 (2012) 2864–2867 |
| [12] | W. Chaikittisilp, K. Ariga, Y. Yamauchi. A new family of carbon materials:synthesis of MOF-derived nanoporous carbons and their promising applications. J. Mater. Chem. A 1 (2013) 14–19 |
| [13] | J.K. Sun, Q. Xu. Functional materials derived from open framework templates/precursors:synthesis and applications. Energy Environ. Sci. 7 (2014) 2071–2100 |
| [14] | S.J. Yang, T. Kim, J.H. Im, et al. MOF-derived hierarchically porous carbon with exceptional porosity and hydrogen storage capacity. Chem. Mater. 24 (2012) 464–470 |
| [15] | A. Banerjee, R. Gokhale, S. Bhatnagar, et al. MOF derived porous carbon-Fe3O4 nanocomposite as a high performance, recyclable environmental superadsorbent. J. Mater. Chem. 22 (2012) 19694–19699 |
| [16] | R. Li, Y.P. Yuan, L.G. Qiu, W. Zhang, J.F. Zhu. A rational self-sacrificing template route to metal-organic framework nanotubes and reversible vapor-phase detection of nitroaromatic explosives. Small 8 (2012) 225–230 |
| [17] | R. Li, X.Q. Ren, X. Feng, et al. A highly stable metal- and nitrogen-doped nanocomposite derived from Zn/Ni-ZIF-8 capable of CO2 capture and separation. Chem. Commun. 50 (2014) 6894–6897 |
| [18] | J. Kim, N.D. McNamara, T.H. Her, J.C. Hicks. Carbothermal reduction of Ti-modified IRMOF-3:an adaptable synthetic method to support catalytic nanoparticles on carbon. ACS Appl. Mater. Interfaces 5 (2013) 11479–11487 |
| [19] | J. Liu, H. Wang, C. Wu, et al. Preparation and characterization of nanoporous carbon-supported platinum as anode electrocatalyst for direct borohydride fuel cell. Int. J. Hydrogen Energy 39 (2014) 6729–6736 |
| [20] | N.L. Torad, M. Hu, S. Ishihara, et al. Direct synthesis of MOF-derived nanoporous carbon with magnetic Co nanoparticles toward efficient water treatment. Small 10 (2014) 2096–2107 |
| [21] | X.S. Li, G.T. Zhu, Y.B. Luo, B.F. Yuan, Y.Q. Feng. Synthesis and applications of functionalized magnetic materials in sample preparation. TrAC Trends Anal. Chem. 45 (2013) 233–247 |
| [22] | A. Ríos, M. Zougagh, M. Bouri. Magnetic (nano)materials as an useful tool for sample preparation in analytical methods. A review. Anal. Methods 5 (2013) 4558–4573 |
| [23] | C. Wang, R.Y. Ma, Q.H. Wu, M. Sun, Z. Wang. Magnetic porous carbon as an adsorbent for the enrichment of chlorophenols from water and peach juice samples. J. Chromatogr. A 1361 (2014) 60–66 |
| [24] | A. Ghaffari, M.S. Tehrani, S.W. Husain, M. Anbia, P.A. Azar. Adsorption of chlorophenols from aqueous solution over amino-modified ordered nanoporous silica materials. J. Nanostruct. Chem. 4 (2014) 114–123 |
| [25] | M. Sun, P.L. Cui, S.J. Ji, et al. Octadecyl-modified graphene as an adsorbent for hollow fiber liquid phase microextraction of chlorophenols from honey. Bull. Korean Chem. Soc. 35 (2014) 1011–1015 |
| [26] | C. Petit, T.J. Bandosz. MOF-graphite oxide composites:combining the uniqueness of graphene layers and metal-organic frameworks. Adv.Mater. 21 (2009) 4753–4757 |
| [27] | L. Wang, X.H. Zang, C. Wang, Z. Wang. Graphene oxide as a micro-solid-phase extraction sorbent for the enrichment of parabens from water and vinegar samples. J. Sep. Sci. 37 (2014) 1656–1662 |
| [28] | J.F. Liu, X. Liang, Y.G. Chi, et al. High performance liquid chromatography determination of chlorophenols in water samples after preconcentration by continuous flow liquid membrane extraction on-line coupled with a precolumn. Anal. Chim. Acta 487 (2003) 129–135 |
| [29] | Y.Q. Cai, Y.E. Cai, S.F. Mou, Y.Q. Lu. Multi-walled carbon nanotubes as a solid-phase extraction adsorbent for the determination of chlorophenols in environmental water samples. J. Chromatogr. A 1081 (2005) 245–247 |
| [30] | J.T. Wang, W.N. Wang, Q.H. Wu, C. Wang, Z. Wang. Extraction of some chlorophenols from environmental waters using a novel graphene-based magnetic nanocomposite followed by hplc determination. J. Liq. Chromatogr. Relat. Technol. 37 (2014) 2349–2362 |
| [31] | Q. Liu, J.B. Shi, L.X. Zeng, et al. Evaluation of graphene as an advantageous adsorbent for solid-phase extraction with chlorophenols as model analytes. J. Chromatogr. A 1218 (2011) 197–204 |
 2016, Vol. 27
2016, Vol. 27 




