b Department of Chemistry, Graduate University of Advanced Technology, Kerman, Iran
Electrochemical sensors satisfy many of the requirements in analysis,such as high selectivity,easy preparation,good selectivity and sensitivity,and fast response [1, 2, 3, 4, 5, 6]. Furthermore,the limited amount of electrode materials makes only a restricted number of electroactive materials suitable for electrochemical detection with high sensitivity and selectivity [7, 8, 9, 10]. So,efforts have been made to modify the electrode surfaces for the purpose of lowering the overpotential,improving the sensitivity with high current density for effective enrichment of the desired substance and/or restraining the effect of interferences [11, 12, 13, 14].
Promazine is a medicine that belongs to the phenothiazine class of antipsychotics. An old drug used to treat schizophrenia, promazine is still being prescribed alongside newer agents such as olanzapine and quetiapine. It has predominantly anticholinergic side effects,though extra pyramidal side effects are not uncommon either [15]. The monitoring of promazine is significant for quality assurance in the pharmaceutical industry and for obtaining optimum therapeutic concentrations in body fluids to minimize toxicity [16]. Therefore,the detection of this drug is important in biological samples such as urine and drug tablets. In comparison to other methods for determination of promazine,the proposed electrochemical sensor has attracted more interest due to its sensitivity,low cost,accuracy,high dynamic range and simplicity.
To the best of our knowledge,there are no reports for the application of ZnO/NPs-modified carbon ionic liquid paste electrode for the determination of promazine. The electrochemical behaviors of promazine at ZnO/NPs/ILs/CPE,carbon paste electrode modified with ionic liquid (IL/CPE),ZnO/NPs carbon paste electrode (ZnO/NPs/CPE),and carbon paste electrode (CPE) were investigated. The results showed the superiority of ZnO/NPs/ILs/ CPE to the other electrodes in terms of both reversibility and sensitivity. We also evaluate the analytical performance of ZnO/ NPs/ILs/CPE for the voltammetric determination of promazine in real samples such as drug tablets,urine and serum.
2. Experimental 2.1. Chemicals and apparatusIn this study we used a Potentiostat/Galvanostat (m-Autolab with PGSTAT 302 N (Eco Chemie,the Netherlands) coupled with a Pentium Ⅳ personal computer connected to an HP laser jet 6L printer,and experiments were performed in a three-compartment cell. A conventional three-electrode cell assembly consisting of a platinum wire as an auxiliary electrode and an Ag/AgCl/KClsat electrode as a reference electrode was used. The working electrode was a ZnO/NPs/ILs/CPE. All chemicals were of A.R. grade and were used as received without any further purification. Promazine was purchased from Sigma. Mineral oil was obtained from Fluka. Graphite powder (< 50 mm) was purchased from Fluka. All solutions were prepared using double distilled water having a specific conductivity of 0.4-0.9 mS. ZnO nanoparticles were synthesized according to our previous report [17].
2.2. Preparation of the electrodeZnO/NPs/ILs/CPE was prepared by mixing 0.2 g of ionic liquids, 0.8 g of the liquid paraffin,0.1 g of ZnO/NPs,and 0.90 g of graphite powder. Then the mixture was well mixed for 60 min until a uniformly wetted paste was obtained. A portion of the paste was filled firmly into one glass tube as described above to prepare ZnO/ NPs/ILs/CPE. ZnO/NPs/CPE was prepared by hand-mixing of 0.90 g of graphite powder and 0.10 g ZnO/NPs plus paraffin and mixed well for 60 min until a uniformly wetted paste was obtained.
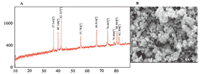
3. Results and discussionZnO/NPs were analyzed by XRD analyses. The XRD pattern of ZnO/NPs,in the 2θ range of 10-90°,is shown in Fig. 1A. The average grain size of the sampleswas estimated with the help of the Scherrer equation using the diffraction intensity of (1 0 1; 2θ = 42.327° ) peak. The mean grain size (D= 38 nm) of the particles was determined from the XRD line broadening measurement using the Scherrer equation. The morphology of the as-grown nanostructures was characterized by SEM. Typical SEM micrograph of the ZnO/NPs is shown in Fig. 1B. The presence of dark points in this figure in nanoscale size confirms that this nanoparticle was synthesized.
|
Download:
|
| Fig. 1. (A) XRD patterns of as-synthesized ZnO/NPs. (B) SEM images of ZnO/NPs. | |
The electrochemical oxidation of the promazine occurs at the nitrogen atom in a potential range of (0.0-0.8 V). This signal is independent of pH value [18]. So,we select pH 7.0 to mimic biological conditions for the analysis. The microscopic areas were calculated from the slope of the Ip - n1/2 relation (taking concentration of K4Fe(CN)6 as 1.0 mmol/L,concentration of KCl electrolyte as 0.10 mol/L,n = 1,DR = 7.6 × 10-6 cm/s) for all of the electrodes. It is 0.25 cm2 (average of five measurements) for the (ZnO/NPs/ILs/CPE),0.19 cm2 for ILs/CPE,0.14 cm2 for ZnO/NPs/CPE and 0.09 cm2 for the CPE. Fig. 2A (inset) shows the current density derived from the cyclic voltammograms of 400 mmol/L promazine (pH 7.0) at the surface of different electrodes with a scan rate of 50 mV/s. The results show that the presence of both ZnO/NPs and ILs causes the good conductivity of the electrode. Fig. 2A shows cyclic voltammograms of 400 mmol/L promazine at pH 7.0 at the surface of different electrodes with a scan rate of 50 mV/s. ZnO/ NPs/ILs/CPE exhibited a significant oxidation peak current around 685 mV with the peak current of 41.6 mA (Fig. 2A,curve d). In contrast,low redox activity peak was observed at ZnO/NPs/CPE (Fig. 2A,curve b) and at unmodified CPE (Fig. 2 curve a) under the same conditions. The promazine oxidation peak potential at ZnO/ NPs/CPE and at CPE observed around 740 and 750 mV vs. the reference electrode with the oxidation peak current of 19.2 and 10.1 mA,respectively. In addition,at the surface of bare ILs/CPE,the oxidation peak appeared at 690 mV with a peak current of 27.5 mA (Fig. 2A,curve c),which indicated the presence of ILs in CPE could enhance the peak currents and decrease the oxidation potential. The ZnO/NPs with suitable electronic properties together with an ionic liquid promoted charge transfer reactions,good anti-fouling properties,especially when mixed with more conductive compounds such as ILs when used as an electrode.

|
Download:
|
| Fig. 2. (A) Cyclic voltammograms of (a) CPE, (b) ZnO/NPs/CPE, (c) ILs/CPE and (d) ZnO/NPs/ILs/CPE in the presence of 400 mmol/L promazine at pH 7.0, respectively. Inset: the current density derived from cyclic voltammogram responses of 400 mmol/L promazine at pH 7.0 at the surface of different electrodes. (B) Plot of Ipa versus n1/2 for the oxidation of promazine at ZnO/NPs/ILs/CPE. Inset shows cyclic voltammograms of promazine at ZnO/NPs/ILs/CPE at different scan rates (a–h) of 5, 15, 20, 50, 70, 100.0, 150.0 and 200 mV/s in 0.1 mol/L phosphate buffer, pH 7.0. | |
In scan rate investigation,we observed a linear variation of the peak current with the square root of scan rate (v1/2 (r2 = 0.9921 (Fig. 2B). This result clearly indicates a diffusion-controlled electrooxidative process. To obtain information about the ratedetermining step,the Tafel plot was drawn,as derived frompoints in the Tafel region of the cyclic voltammogram. The slope of the Tafel plot was equal to 2.3RT/n(1 - a)F,which arrived 0.2172 and 0.2240 for scan rates of 50 and 100 mV/s,respectively. Therefore, weobtained amean value of 0.74 (Fig. S1 in Supporting informationinformation).
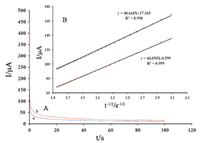
Chronoamperometry was used with ZnO/NPs/ILs/CPE to determine the diffusion coefficient of promazine (Fig. 3). We have determined the diffusion coefficient,D,of promazine using the cottrell equation:
|
Download:
|
| Fig. 3. (A) Chronoamperograms obtained at ZnO/NPs/ILs/CPE in the presence of (a) 300 and (b) 400 mmol/L promazine in the buffer solution (pH 7.0). (B) Cottrell’splot for the data from the chronoamperograms | |
| $I = nFA{D^{1/2}}{C_b}{\pi ^{ - 1/2}}{t^{ - 1/2}}$ | (1) |
According to the above equation,we calculated a diffusion coefficient of 9.5 × 10-5 cm2/s for promazine.
Squarewavevoltammetry (withanamplitudepotential of50mV and a frequency of 12 Hz) was used to determine promazine. SWVs clearly show that the plot of the peak current vs. promazine concentration is linear for 0.08-450.0 mmol/L of promazine (Fig. 4). The detection limit (LOD) was 0.04 mmol/L according to the definition of LOD = 3sb/m (where sb is the standard deviation of the blank signal (n = 8) and m is the slope of the calibration).

|
Download:
|
| Fig. 4. The plots of the electrocatalytic peak current as a function of promazine concentration. Inset shows the SWVs of ZnO/NPs/ILs/CPE in 0.1 mol/L PBS (pH 7.0) containing different concentrations of promazine. From inner to outer (a–m) correspond to 0.08, 8.0, 20.0, 50.0, 100.0, 150.0, 200.0, 250.0, 300.0, 350.0, 400.0, 420.0 and 450.0 mmol/L of promazine. | |
The repeatability and stability of ZnO/NPs/ILs/CPE was investigated by the SWV measurements of 10.0 mmol/L promazine. The relative standard deviation (RSD%) for seven successive assays was 1.5%. When using seven different electrodes,the RSD% for nine measurements was 2.6%. When the electrode was stored in the laboratory,the modified electrode retains 95% of its initial response after two weeks and 92% after 50 days. These results indicate that ZnO/NPs/ILs/CPE has good stability and reproducibility, and could be used for promazine.
After the above experimental investigation,we found that 1000-fold glucose,sucrose,lactose,fructose,Mg+2,Ca+2,Br-,K+,and glycine; 700-fold valine and methionine did not interfere with the determination of promazine. The result shows that only ascorbic acid could be an interference for the determination of promazine using this modified electrode. The interference of ascorbic acid can be minimized using ascorbic oxidase enzyme,which exhibits high selectivity for the oxidation of ascorbic acid,if necessary.
Determination of promazine in tablets,urine and pharmaceutical serum samples was examined to demonstrate the ability of the modified electrode to the determination of promazine in real samples. Standard addition method was used to calculate the promazine concentrations. The results given in Table 1 indicate that the modified electrode retained its efficiency for the determination of promazine in real samples.
|
|
Table 1 Determination of promazine in pharmaceutical and urine samples (n = 3). |
4. Conclusion
In this research,we reported the synthesis of ZnO/NPs nanocomposite as a highly sensitive sensor for the voltammetric determination of promazine using a carbon paste electrode in the presence of 1-methyl-3-butylimidazolium bromide as a binder for the first time. Compared with traditional CPEs,a decrease of overpotential of oxidation of promazine was about 65 mV with a 4.0-fold increment in the peak current when using ZnO/NPs/ILs/ CPE. Under the best conditions,the oxidation peak current was proportional to the promazine concentration in the range of 0.08 to 450 mmol/L with the detection limit of 0.04 mmol/L. The proposed method was successfully applied to the promazine detection in real samples such as drug tablets and urine.
Appendix A. Supplementary dataSupplementary data associated with this article can be found,in the online version,at http://dx.doi.org/10.1016/j.cclet.2016.01.026.
| [1] | M.R. Ganjali, P. Norouzi, M. Ghorbani, A. Sepehri. Fourier transform cyclic voltammetric technique for monitoring ultratrace amounts of salbutamol at gold ultra microelectrode in flowing solutions. Talanta 66 (2005) 1225–1233 |
| [2] | P. Norouzi, M.R. Ganjali, T. Alizadeh, P. Daneshgar. Fast Fourier continuous cyclic voltammetry at gold ultramicroelectrode in flowing solution for determination of ultra trace amounts of penicillin G. Electroanalysis 18 (2006) 947–954 |
| [3] | T. Alizadeh, M.R. Ganjali, M. Zare, P. Norouzi. Development of a voltammetric sensor based on a molecularly imprinted polymer (MIP) for caffeine measurement. Electrochim. Acta 55 (2010) 1568–1574 |
| [4] | P. Norouzi, M.R. Ganjali, A. Sepehri, M. Ghorbani. Novel method for fast determination of ultra trace amounts of timolol maleate by continuous cyclic voltammetry at Au microelectrode in flowing injection systems. Sens. Actuators B Chem. 110 (2005) 239–245 |
| [5] | A. Taherkhani, H. Karimi-Maleh, A.A. Ensafi, et al. Simultaneous determination of cysteamine and folic acid in pharmaceutical and biological samples using modified multiwall carbon nanotube paste electrode. Chin. Chem. Lett. 23 (2012) 237–240 |
| [6] | M.L. Yola, N. Atar. A novel voltammetric sensor based on gold nanoparticles involved in p-aminothiophenol functionalized multi-walled carbon nanotubes:application to the simultaneous determination of quercetin and rutin. Electrochim. Acta 119 (2014) 24–31 |
| [7] | V.K. Gupta, M.L. Yola, N. Atar, et al. Electrochem-ically modified sulfisoxazole nanofilm on glassy carbon for determination of cadmium (ò) in water samples. Electrochim. Acta 105 (2013) 149–156 |
| [8] | Z.F. Wang, H. Zhang, Z.P. Wang, et al. Trace analysis of Ponceau 4R in soft drinks using differential pulse stripping voltammetry at SWCNTs composite electrodes based on PEDOT:PSS derivatives. Food Chem. 180 (2015) 186–193 |
| [9] | H. Karimi-Maleh, P. Biparva, M. Hatami. A novel modified carbon paste electrode based on NiO/CNTs nanocomposite and (9,10-dihydro-9,10-ethanoanthracene-11, 12-dicarboximido)-4-ethylbenzene-1,2-diol as a mediator for simultaneous determination of cysteamine, nicotinamide adenine dinucleotide and folic acid. Biosens. Bioelectron. 48 (2013) 270–275 |
| [10] | P.K. Brahman, R.A. Dar, K.S. Pitre. DNA-functionalized electrochemical biosensor for detection of vitamin B1 using electrochemically treated multiwalled carbon nanotube paste electrode by voltammetric methods. Sens. Actuators B Chem. 177 (2013) 807–812 |
| [11] | P.K. Brahman, R.A. Dar, S. Tiwari, K.S. Pitre. Electrochemical behavior of gatifloxacin at multi-walled carbon nanotube paste electrode and its interaction with DNA. Rev. Anal. Chem. 31 (2012) 83–92 |
| [12] | Z.F. Wang, Z.P. Wang, H. Zhang, et al. Electrochemical sensing application of poly (acrylic acid modified EDOT-co-EDOT):PSS and its inorganic nanocomposite with high soaking stability, adhesion ability and flexibility. RSC Adv. 5 (2015) 12237–12247 |
| [13] | P. Norouzi, F. Faridbod, B. Larijani, M.R. Ganjali. Glucose biosensor based on MWCNTs-gold nanoparticles in a nafion film on the glassy carbon electrode using flow injection FFT continuous cyclic voltammetry. Int. J. Electrochem. Sci. 5 (2010) 1213–1224 |
| [14] | H. Karimi-Maleh, F. Tahernejad-Javazmi, N. Atar, et al. A novel DNA biosensor based on a pencil graphite electrode modified with polypyrrole/functionalized multiwalled carbon nanotubes for determination of 6-mercaptopurine anticancer drug. Ind. Eng. Chem. Res. 54 (2015) 3634–3639 |
| [15] | http://en.wikipedia.org/wiki/Promazine21 November 2014. |
| [16] | B. Rezaei, A. Mokhtari. Chemiluminescence determination of promazine in human serum and drug formulations using Ru (phen)32+-Ce (IV) system and a chemometrical optimization approach. Luminescence 24 (2009) 183–188 |
| [17] | H. Karimi-Maleh, S. Rostami, V.K. Gupta, M. Fouladgar. Evaluation of ZnO nanoparticle ionic liquid composite as a voltammetric sensing of isoprenaline in the presence of aspirin for liquid phase determination. J. Mol. Liq. 201 (2015) 102–107 |
| [18] | A.A. Ensafi, M. Taei, T. Khayamian, H. Karimi-Maleh, F. Hasanpour. Voltammetric measurement of trace amount of glutathione using multiwall carbon nanotubes as a sensor and chlorpromazine as a mediator. J. Solid State Electrochem. 14 (2010) 1415–1423 |
 2016, Vol. 27
2016, Vol. 27 



