Quantum dots (QDs) after excitation are prone to electron or energy transfer with their donor or receptor,which quenches their photoluminescence [1, 2]. This property of QDs can be taken advantage of in molecular detection,so that the concentration signal of an analyte can be transformed into an optical signal [2, 3]. Testing systems based on the principle of QDs-fluorescence resonance energy transfer (FRET) have been used in the detection of protein-small molecules or protein-protein combination [4, 5, 6], immunoassay [7],DNA hybridization probe [8, 9],enzyme activity measurement [10],and other fields. On the contrary,sensors based on the principle of QDs-PIET were subsequently studied and applied to very few fields [3, 11, 12, 13, 14]. A PIET-based receptorsubstrate working mechanism was proposed recently: the quencher is adsorbed electrostatically onto the surface of QDs, thus quenching the phosphorescence of QDs; when a receptor that may combine with the quencher is added,and the quencher desorbs from the surface of the QDs,which recovers the phosphorescence of QDs [3]. The above studies are based on the fluorescence of QDs,but there is little research about PIET sensors based on the room temperature phosphorescence (RTP) QDs.
As the research on QDs has intensified,the RTP of QDs and its application in detection became two hotspots. RTP-based methods outperform fluorescence-based methods in many ways. For instance,phosphorescence is rare and more sensitive compared with fluorescence [15]. The longer lifetime of phosphorescence helps to avoid interference from the background fluorescence and scattering light in biofluids [16, 17]. RTP methods also avoid the superposition between the excitation spectrum and the emission spectrum [17],and do not need the addition of any deoxidant or revulsive [18]. Thus,RTP-based detection can be widely applied into many fields [18, 19, 20, 21].
Methylene blue (MB) is an alkaline biological dye with a flat structure that has long been applied in clinical diagnosis and chemical analysis. MB can be used to detoxify the poisoning by nitrite,sulfonamide salt,prussiate or CO and can also be used to screen out anti-cancer drugs in vitro.Ononehand,MBcanbe inserted intothedouble chains ofDNAmolecules toa formnon-electric,active substance with DNA. On the other hand,the molecular structure of MB contains N+(CH3)2,which can electrostatically interact with the negatively-charged PO43- outside the double chain of DNA [22].
DNA as the major carrier of genetic information is the crucial factor for species continuation and evolution. Even slight structural change in DNA will lead to changes in genetic traits or the occurrence of various diseases. Thus,DNA has become a research hotspot in many fields. Fluorescence-based methods with high sensitivity,selectivity,and multiple parameters play key roles in quantitative DNA detection [23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33]. However,the weak endogenous fluorescence of DNA cannot be directly utilized into structural or quantitative analysis [30, 34]. Recently,some nano-sensors including QDs have been successfully applied into DNA detection and analysis [30, 32, 35, 36].
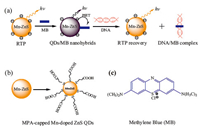
The objectives of this study are to prepare a nanohybrid formed from MPA-coated Mn-doped ZnS QDs andMBthrough electrostatic interaction,and to explore the feasibility whether the RTP sensor based on this nanohybrid can be used in DNA detection. Through PIET,MB will quench the RTP of Mn-doped ZnS QDs. With the addition of DNA,MB binds with DNA through inserting and static electricity,and then desorbs from the surfaces of QDs,which recovers the RTP of the Mn-doped ZnS QDs (Fig. 1). On this basis,a DNA detection method based on the properties of RTP was set up. Since this method avoids the interference from the background fluorescence or scattering light in vivo,and thus,avoids complex sample pretreatment. Thus,this method is very feasible for detection of trace DNA in biofluids.
|
Download:
|
| Fig. 1. (a) Schematic illustration of fabricating Mn-doped ZnS QDs/MB nanohybrids for DNA detection; (b) The structure of MPA-capped Mn-doped ZnS QDs; (c) The structure of MB. | |
2. Experimental 2.1. Materials and chemicals
MPA(J & KScientific,Beijing,China),Zn(Ac)2•2H2O,Mn(Ac)2•4H2O, and Na2S•9H2O (Tianjing Kermel Chemical Reagent Co.,China) were used to prepare Mn-doped ZnS QDs. Methylene blue (MB) was purchased in Beijing chemical plant. Salmon Testes DNA was provided by Sigma. Ultrapure water (18.2MVcm) was obtained from aWater Pro water purification system (Labconco Corporation, Kansas City,MO).
2.2. ApparatusThe morphology and microstructure of QDs were characterized by a JEM-2100 Transmission Electron Microscope (TEM,Japan). Phosphorescence was measured by a Cary Eclipse fluorescence spectrophotometer (Varian American Pty Ltd.,USA),equipped with a plotter unit and a quartz cell (1 cm × 1 cm) in the phosphorescence mode. The pH was measured with a pH meter (Jinpeng Analytical Instruments Co.,Ltd.,China). Absorption spectra were measured using a Shimadzu UV-29100 UV/vis Spectrophotometer. The resonance light scattering (RLS) spectra were recorded in the same spectrofluorometer by scanning both the excitation and emission monochromators (Δλ = 0) from 200 nm to 700 nm.
2.3. Synthesis of Mn-doped ZnS QDsMn-doped ZnS QDs were synthesized in aqueous solution as per a published method [32, 37] with minor modification. The specific steps are as follows: 5 mL of 0.1 mol/L Zn(Ac)2,2 mL of 0.01 mol/L Mn(Ac)2,and 50 mL of 0.04 mol/L MPA were added to a three-neck flask. The mixture was adjusted to pH 11 with 1 mol/L NaOH. At r.t. under argon conditions,and after ventilation of nitrogen for 30 min to ensure the MPA fully with Zn2+ and Mn2+ complex,5 mL of 0.1 mol/L Na2S was then injected into the mixture. After stirring for 20 min,the solution was aged at 50 8C in open air for 2 h. The QDs were purified by precipitation with ethanol,centrifugation, washing with ethanol,and vacuum drying. The obtained QDs powder is highly soluble in water.
2.4. Assay condition and RTP measurementTo study the MB effect of the MPA-capped Mn-doped ZnS QDs on the RTP intensity,we prepared a 2.0 × 10-5 mol/L mother liquor from MB; then added different volumes of the mother liquor into the phosphate-buffered saline solution (PBS,pH 7.4,20 mmol/L). MPA-capped Mn-doped ZnS QDs were dissolved in water to form a 2.0 mg/mL solution,which then (100 mL) was added to each of the above MB solutions. After 5 min,the phosphorescence at an excitation wavelength of 295 nm was measured. For DNA detection,the Salmon Testes DNA was made into a 100 mg/L mother liquor. The assay solutions containing MPA-capped Mndoped ZnS QDs (100 mL),MB (600 mL),and varying concentrations of DNA (0-20 mg/L) were prepared in 5 mL of PBS (20 mmol/L,pH 7.4). Reactions proceeded for 5 min before spectrophotometry.
2.5. Sample pretreatmentUrine was sampled from healthy volunteer. The urine samples were diluted 100 times with secondary water before analysis without any other pretreatment.
2.6. Measurement proceduresPBS (0.2 mol/L,500 mL),MB (2.0 × 10-5 mol/L,600 mL),MPAcapped Mn-doped ZnS QDs (2.0 mg/mL,100 mL),and urine (50 mL) were sequentially added to a 5 mL calibrated test tube. A recovery study was carried out in the samples spiked with 2.0,3.0 and 4.0 mg/L DNA. The mixtures were diluted to 5 mL with ultrapure water,mixed thoroughly,and aged for 5 min. Then the phosphorescence of the mixtures was measured at an excitation wavelength of 295 nm. Experiments were repeated three times.
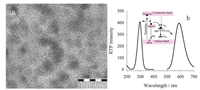
3. Results and discussion 3.1. Characterization of the MPA-capped Mn-doped ZnS QDsThe size of MPA-capped Mn-doped ZnS QDs was determined by TEM to be about 3.5 nm (Fig. 2a). The maximum excitation peak occurred at 295 nm and a narrow emission band was centered at 590 nm: hv1 is the fluorescence occurring from the surface defect of ZnS QDs; hv2 is the phosphorescence attributed to the transition of Mn2+ from the triplet state (4T1) to the ground state (6A1) (Fig. 2b). As reported,Mn-doped ZnS QDs exhibit an orange phosphorescence emission (about 590 nm) [20],which is attributed to the energy transfer from the band gap of ZnS to Mn2+ dopant and the subsequent 4T1 to 6A1 transition of the Mn2+ incorporated into the ZnS host lattice [38].
|
Download:
|
| Fig. 2. (a) TEM image of MPA-capped Mn-doped ZnS QDs; (b) The excitation and RTP emission spectra of Mn-doped ZnS QDs (40 mg/L). Solutions were prepared in PBS buffer (20 mmol/L, pH 7.4). | |
3.2. Synthesis of MPA-coated Mn-doped ZnS QDs/MB nanohybrids
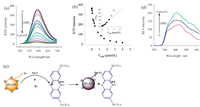
A nanohybrid was prepared by the electrostatic interaction between MPA-coated Mn-doped ZnS QDs and MB. As the MB dosage increased,the RTP intensity of Mn-doped ZnS QDs at 590 nm was gradually weakened (Fig. 3a). The RTP intensity was gradually stabilized at the MB concentration of 2.4 mmol/L and unchanged with further addition ofMB(Fig. 3b). The inset in Fig. 3b indicates high linearity within the range 0.02-2.4 mmol/L MB,the RTP quenching of the QDs by MB fitted the following equation: P0/P = 3.8334CMB + 0.9299 (R = 0.986),where P0 and P are the phosphorescence intensity of Mn-doped ZnS QDs without and at a given MB concentration.
|
Download:
|
| Fig. 3. (a) MB concentration-dependent RTP emission of the MPA-capped Mn-doped ZnS QDs. The concentration of the MPA-capped Mn-doped ZnS QDs is 40 mg/L; (b) The change of the RTP intensity with the increase of the concentration of MB. The inset shows the linear fitted result of P0/P; (c) Illustration of the PIET of QDs after the addition of MB; (d) MB concentration dependent RLS of Mn-doped ZnS QDs. | |
The coating of MPA onto the surfaces of Mn-doped ZnS QDs not only enhanced the water-solubility of QDs,but also negatively charged the surfaces of QDs. Then the negatively-charged QDs electrostatically interacted with the positively-charged MBto form a Mn-doped ZnS QDs/MB nanohybrid. Moreover,through PIET,MB as an effective electron receptor can quench the RTP of Mn-doped ZnS QDs (Fig. 3c).
As showed in the RLS spectra (Fig. 3d),after the addition of MB, the MPA-coated Mn-doped ZnS QDs polymerized with MB,and then,the hydrophobic interaction was gradually enhanced to form larger aggregate particles. The RLS intensity of MPA-coated Mndoped ZnS QDs is very low. After their interaction,with the gradual increase of MB concentration,the RLS intensity is gradually enhanced. These results indicate that the electrostatic interaction between QDs and MB forms a polymer,and their polymerization degree is improved through hydrophobic interaction.
3.3. Factors affecting stability of Mn-doped ZnS QDs/MB nanohybridsFactors affecting the RTP intensity of Mn-doped ZnS QDs/MB nanohybrids include pH,time and NaCl. In particular,pH is the major influence factor. As pH rises within 4.5-9.0,the RTP intensity first increases and then decreases. Within pH 6.8-7.8,the RTP intensity gradually stabilizes and maximizes (Fig. 4a). Thus,human physical pH 7.4 is selected as the optimal level. At pH 7.4,the RTP of the Mn-doped ZnS QDs/MB nanohybrids is unchanged within 30 min (Fig. 4b). The RTP of the nanohybrids is unchanged with presence of 0-0.02 mol/L NaCl (Fig. 4c). However,with further addition of NaCl,the RTP intensity is enhanced,mainly because the high salinity solution contained high content of Na+,which inhibited the polymerization between MB and the Mn-doped ZnS QDs,thereby interfered with the quenching of phosphorescence.

|
Download:
|
| Fig. 4. (a) Effect of pH on the RTP emission of the MPA-capped Mn-doped ZnS QDs, and their nanohybrids with MB; (b) Time-dependent RTP emission of the Mn-doped ZnS QDs/MB nanohybrids; (c) Effect of NaCl concentration on the RTP emission of the Mn-doped ZnS QDs/MB nanohybrids. The concentration of Mn-doped ZnS QDs and MB are 40 mg/L and 2.4 mmol/L, respectively. | |
3.4. Mn-doped ZnS QDs/MB nanohybrids used as a DNA RTP sensor
Fig. 5A shows the ultraviolet spectra of MB (curve a),DNA (curve b),and MPA-coated Mn-doped ZnS QDs (curve c). The addition of MB led to the enhancement of ultraviolet spectra and occurrence of red shift (curve d),indicating the occurrence of interaction between Mn-doped ZnS QDs and MB. With the addition of DNA,the ultraviolet intensity of the Mn-doped ZnS QDs/MB nanohybrids is further enhanced (curve e) and further red-shifts. The reason is that DNA competes with QDs for MB,and thus,MB is desorbed from the surfaces of the QDs and forms a new hybrid through embedding and electrostatic interaction. These phenomena indicate the occurrence of interaction between Mn-doped ZnS QDs and MB,and between DNA and the Mn-doped ZnS QDs/MB nanohybrids.

|
Download:
|
| Fig. 5. (A) UV–vis absorption spectra of a) MB, b) DNA, c) the MPA-capped Mn-doped ZnS QDs, d) the MPA-capped Mn-doped ZnS QDs/MB nanohybrids, e) the MPA-capped Mn-doped ZnS QDs/MB + DNA; DNA concentration-dependent RTP emission of (B) the MPA-capped Mn-doped ZnS QDs/MB hybrids; (C) the MPA-capped Mn-doped ZnS QDs without MB; (D) DNA concentration dependent RLS of the MB. | |
With further addition of DNA,the RTP intensity of the nanohybrids is gradually enhanced (Fig. 5B),indicating that this hybrid is applicable for use as an RTP sensor in DNA detection. After different concentrations of DNA (0-40 mg/L) were added into the Mn-doped ZnS QDs,the RTP intensity of the QDs was basically unchanged (Fig. 5C). The reason is that the MPA-coated Mn-doped ZnS QDs and the DNA are both negatively charged and thus mutually repel in weak alkaline solutions. As showed in the RLS spectra of MB-DNA interaction (Fig. 5D),with the presence of certain concentrations of MB,the addition of DNA led to gradual enhancement of RLS intensity,indicating the occurrence of interaction and polymerization between MB and DNA.
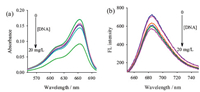
3.5. Action mechanism between MB and DNAAs showed in the absorption spectra between MB and DNA (Fig. 6a),the addition of DNA resulted in the significantly reduced intensity and red-shift in the absorption peak of MB,indicating the occurrence of intercalation between MB and DNA [39, 40, 41, 42].
|
Download:
|
| Fig. 6. | |
Because of strong fluorescence in MB,the addition of DNA led to the reduced fluorescence intensity of MB,but no shift in the peak (Fig. 6b). The quenching of fluorescence indicates that the very strong inter-electronic action of the MB/DNA hybrid leads to the structural changes in excited-state electrons [43].
These results show that the interaction between MB and DNA involves two aspects: the reductive MB can be inserted into the DNA; and the oxidative MB can electrostatically bind to the phosphoric acid skeleton of DNA. Though the action between Mndoped ZnS QDs and MB is also electrostatic interaction,it is inferior to the action between DNA and MB. Thus,DNA can desorb MB from the Mn-doped ZnS QDs/MB nanohybrids,and thus recover the RTP of the Mn-doped ZnS QDs.
3.6. Characterization of Mn-doped ZnS QDs/MB nanohybrids as an RTP sensorBased on these results,a quantitative method for DNA detection by Mn-doped ZnS QDs/MB nanohybrids was designed. The linearity between the RTP recovering value of Mn-doped ZnS QDs/MB nanohybrids (DRTP) and the DNA concentration under the optimal conditions was obtained (Fig. 7).

|
Download:
|
| Fig. 7. Plots of ΔRTP as a function of DNA concentration show one linear ranges. Buffer, 20 mmol/L PBS (pH 7.4); MPA-capped Mn-doped ZnS QDs, 40 mg/L; MB, 2.4 mmol/L. | |
The linear ranges of DNA are 0.2-20 mg/L,and the linear equations are ΔRTP = 5.757CDNA + 3.328 (R = 0.995). This method has a detection limit (3σ) of 0.113 mg/L (where σ is the standard deviation from 11 continuous parallel detections of 0.2 mg/L DNA). For systems without DNA and with 0.2 mg/L DNA,the 11 continuous parallel detections have a relative standard deviation of 2.6%. The detection limit of this method is higher to that of other nanoparticle,nonspecific DNA quantitative determination methods (Table 1). But the system has a wider linear range and the RTP suffers from less background interference from biological fluids,so this system can detect DNA content in complex biological fluids without complex pretreatments.
|
|
Table 1 Comparison of the proposed method with different analytical techniques reported for determination of DNA. |
3.7. Selectivity of Mn-doped ZnS QDs/MB nanohybrids as an RTP sensor
Some common metal ions and biomolecules in biological fluids were used to investigate their interferences on detection of DNA by Mn-doped ZnS QDs/MB nanohybrids RTP sensor (Table 2). With 2 mg/L DNA,the RTP change of MPA-capped Mn-doped ZnS QDs was not affected by a 1000-fold of Na+,250-fold of K+,10-fold of Ca2+,5-fold of Mg2+,50-fold Glucose,25-fold of L-Cys,25-fold of L-Gly or 50-fold of L-His. Physiologically relevant Na+ ions caused interferences for DNA analysis. However,the detection limit is only 0.113 mg/L. Thus,the designed biosensor can work with small amounts of urine samples,Na+ and most potential interferences can be largely eliminated by simple dilution.
|
|
Table 2 Effect of co-existing substance on the RTP intensity of 2 mg/L DNA |
3.8. Sample analysis
Further experimentation was performed to validate whether the Mn-doped ZnS QDs/MB nanohybrids can be used in determination of DNA content. The standard addition recoveries on urine samples spiked with DNA are 96%-102% (Table 3). Pretreatment was not needed in all samples.
|
|
Table 3 Recovery for the determination of DNA in urine samples (mean±s; n = 3). |
4. Conclusions
Based on the PIET between Mn-doped ZnS QDs and MB,an accurate and quantitative method for the quantitative detection of DNA in biological fluids was proposed. This method effectively utilizes the unique RTP property of QDs. MB binds with Mn-doped ZnS QDs via electrostatic interaction,and then quenches the RTP of the QDs through PIET; [18TD$DIF]With the addition of DNA,DNA combines with MB,and MB will be desorbed from the surface of Mn-doped ZnS QDs,thereby releasing the RTP of Mn-doped ZnS QDs. The present QDs-based RTP method does not need deoxidants or other inducers as required by conventional RTP detection methods,and avoids interference from background fluorescence or scattering light in vivo. Therefore,this method is very feasible for detection of trace DNA in biofluids.
| [1] | C. Burda, X.B. Chen, R. Narayanan, M.A. El-Sayed. Chemistry and properties of nanocrystals of different shapes. Chem. Rev. 105 (2005) 1025–1102 |
| [2] | F.M. Raymo, I. Yildiz. Luminescent chemosensors based on semiconductor quantum dots. Phys. Chem. Chem. Phys. 9 (2007) 2036–2043 |
| [3] | I. Yildiz, M. Tomasulo, F.M. Raymo. A mechanism to signal receptor-substrate interactions with luminescent quantum dots. Proc. Natl. Acad. Sci. U. S. A. 103 (2006) 11457–11460 |
| [4] | I.L. Medintz, A.R. Clapp, H. Mattoussi, et al. Self-assembled nanoscale biosensors based on quantum dot FRET donors. Nat. Mater. 2 (2003) 630–638 |
| [5] | A.R. Clapp, I.L. Medintz, J.M. Mauro, et al. Fluorescence resonance energy transfer between quantum dot donors and dye-labeled protein acceptors. J. Am. Chem. Soc. 126 (2004) 301–310 |
| [6] | I.L. Medintz, J.H. Konnert, A.R. Clapp, et al. A fluorescence resonance energy transfer-derived structure of a quantum dot-protein bioconjugate nanoassembly. Proc. Natl. Acad. Sci. U. S. A. 101 (2004) 9612–9617 |
| [7] | S.P. Wang, N. Mamedova, N.A. Kotov, W. Chen, J. Studer. Antigen/antibody immunocomplex from CdTe nanoparticle bioconjugates. Nano Lett. 2 (2002) 817–822 |
| [8] | D.J. Zhou, L.M. Ying, X. Hong, et al. A compact functional quantum dot-DNA conjugate:preparation, hybridization, and specific label-free DNA detection. Langmuir 24 (2008) 1659–1664 |
| [9] | H. Peng, L.J. Zhang, T.H.M. Kjällman, C. Soeller, J. Travas-Sejdic. DNA hybridization detection with blue luminescent quantum dots and dye-labeled single-stranded DNA. J. Am. Chem. Soc. 129 (2007) 3048–3049 |
| [10] | L.F. Shi, N. Rosenzweig, Z. Rosenzweig. Luminescent quantum dots fluorescence resonance energy transfer-based probes for enzymatic activity and enzyme inhibitors. Anal. Chem. 79 (2007) 208–214 |
| [11] | S.R. Guo, D.D. Bao, S. Upadhyayula, et al. Photoinduced electron transfer between pyridine coated cadmium selenide quantum dots and single sheet graphene. Adv. Funct. Mater. 23 (2013) 5199–5211 |
| [12] | D.B. Cordes, S. Gamsey, B. Singaram. Fluorescent quantum dots with boronic acid substituted viologens to sense glucose in aqueous solution. Angew. Chem. Int. Ed. Engl. 45 (2006) 3829–3832 |
| [13] | J.F. Callan, R.C. Mulrooney, S.K. Kamila, B. McCaughan. Anion sensing with luminescent quantum dots-a modular approach based on the photoinduced electron transfer (PET) mechanism. J. Fluoresc. 18 (2008) 527–532 |
| [14] | S. Basili, T.D. Giacco, F. Elisei, R. Germani. An acridinium-based sensor as a fluorescent photoinduced electron transfer probe for proton detection modulated by anionic micelles. Org. Biomol. Chem. 12 (2014) 6677–6683 |
| [15] | J.M. Traviesa-Alvarez, I. Sánchez-Barragán, J.M. Costa-Fernández, R. Pereiro, A. Sanz-Medel. Room temperature phosphorescence optosensing of benzo. Analyst 132 (2007) 218–223 |
| [16] | J. Kuijt, F. Ariese, U.A.T. Brinkman, C. Gooijer. Room temperature phosphorescence in the liquid state as a tool in analytical chemistry. Anal. Chim. Acta 488 (2003) 135–171 |
| [17] | I. Sánchez-Barragán, J.M. Costa-Fernández, A. Sanz-Medel, M. Valledor, J.C. Campo. Room-temperature phosphorescence (RTP) for optical sensing. Trends Anal. Chem. 25 (2006) 958–967 |
| [18] | Y. He, H.F. Wang, X.P. Yan. Exploring Mn-doped ZnS quantum dots for the roomtemperature phosphorescence detection of enoxacin in biological fluids. Anal. Chem. 80 (2008) 3832–3837 |
| [19] | Y. Li, X.Y. Liu, H.Y. Yuan, D. Xiao. Glucose biosensor based on the room-temperature phosphorescence of TiO2/SiO2 nanocomposite. Biosens. Bioelectron. 24 (2009) 3706–3710 |
| [20] | Z.F. Zhang, Y.M. Miao, Q.D. Zhang, L.W. Lian, G.Q. Yan. Selective room temperature phosphorescence detection of heparin based on manganese-doped zinc sulfide quantum dots/polybrene self-assembled nanosensor. Biosens. Bioelectron. 68 (2015) 556–562 |
| [21] | H.F. Wang, Y. He, T.R. Ji, X.P. Yan. Surface molecular imprinting on Mn-doped ZnS quantum dots for room-temperature phosphorescence optosensing of pentachlorophenol in water. Anal. Chem. 81 (2009) 1615–1621 |
| [22] | M.T. Carter, M. Rodriguez, A.J. Bard, Voltammetric studies of the interaction of metal chelates with DNA. 2. Tris-chelated complexes of cobalt (Ⅲ) and iron (Ⅱ) with 1. 10-phenanthroline and 2,2'-bipyridine. J. Am. Chem. Soc. 111 (1989) 8901–8911 |
| [23] | G. Song, L. Li, L. Liu, et al. Fluorometric determination of DNA using a new ruthenium complex Ru(bpy)2PIP(V) as a nucleic acid probe. Anal. Sci. 18 (2002) 757–759 |
| [24] | Q. Liu, Z.H. Pu, A.M. Asiri, X.P. Sun. Bamboo-like nitrogen-doped carbon nanotubes toward fluorescence recovery assay for DNA detection. Sens. Actuators B:Chem. 206 (2015) 37–42 |
| [25] | W. Wei, C.Y. Gao, Y.X. Xiong, et al. A fluorescence method for detection of DNA and DNA methylation based on graphene oxide and restriction endonuclease hpaⅡ. Talanta 131 (2015) 342–347 |
| [26] | M.A. Breimer, Y. Gelfand, O.A. Sadik. Integrated capillary fluorescence DNA biosensor. Biosens. Bioelectron. 18 (2003) 1135–1147 |
| [27] | Y. Ye, J.T. Stivers. Fluorescence-based high-throughput assay for human DNA (cytosine-5)-methyltransferase 1. Anal. Biochem. 401 (2010) 168–172 |
| [28] | B. Liu, G.C. Bazan. Homogeneous fluorescence-based DNA detection with watersoluble conjugated polymers. Chem. Mater. 16 (2004) 4467–4476 |
| [29] | C.Q. Zhu, S.J. Zhuo, H. Zheng, et al. Fluorescence enhancement method for the determination of nucleic acids using cationic cyanine as a fluorescence probe. Analyst 129 (2004) 254–258 |
| [30] | L.L. Zhao, X. Wu, H.H. Ding, J.H. Yang. Fluorescence enhancement effect of morinnucleic acid-L-cysteine-capped nano-ZnS system and the determination of nucleic acid. Analyst 133 (2008) 896–902 |
| [31] | H.T. Li, L.M. Ying, J.J. Green, S. Balasubramanian, D. Klenerman. Ultrasensitive coincidence fluorescence detection of single DNA molecules. Anal. Chem. 75 (2003) 1664–1670 |
| [32] | L.Y. Wang, L. Wang, F. Gao, Z.Y. Yu, Z.M. Wu. Application of functionalized CdS nanoparticles as fluorescence probe in the determination of nucleic acids. Analyst 127 (2002) 977–980 |
| [33] | J.Q. Zhuang, X.D. Zhang, G. Wang, et al. Synthesis of water-soluble ZnS:Mn2+ nanocrystals by using mercaptopropionic acid as stabilizer. J. Mater. Chem. 13 (2003) 1853–1857 |
| [34] | S. Udenfriend, P. Zaltzman. Fluorescence characteristics of purines, pyrimidines, and their derivatives:measurement of guanine in nucleic acid hydrolyzates. Anal. Biochem. 3 (1962) 49–59 |
| [35] | Y.M. Miao, Y.T. Li, Z.F. Zhang, G.Q. Yan, Y. Bi. "Turn off-on" phosphorescent biosensors for detection of DNA based on quantum dots/acridine orange. Anal. Biochem. 475 (2015) 32–39 |
| [36] | Y.M. Miao, Z.F. Zhang, Y. Gong, G.Q. Yan. Phosphorescent quantum dots/doxorubicin nanohybrids based on photoinduced electron transfer for detection of DNA. Biosens. Bioelectron. 59 (2014) 300–306 |
| [37] | P. Wu, Y. He, H.F. Wang, X.P. Yan. Conjugation of glucose oxidase onto Mn-doped ZnS quantum dots for phosphorescent sensing of glucose in biological fluids. Anal. Chem. 82 (2010) 1427–1433 |
| [38] | J.H. Chung, C.S. Ah, D.J. Jang. Formation and distinctive decay times of surface- and lattice-bound Mn2+ impurity luminescence in ZnS nanoparticles. J. Phys. Chem. B 105 (2001) 4128–4132 |
| [39] | S.A. Tysoe, R.J. Morgan, A.D. Baker, T.C. Strekas. Spectroscopic investigation of differential binding modes of Δ- and Λ-Ru(bpy)2(ppz)2+ with calf thymus DNA. J. Phys. Chem. 97 (1993) 1707–1711 |
| [40] | E.C. Long, J.K. Barton. On demonstrating DNA intercalation. Acc. Chem. Res. 23 (1990) 271–273 |
| [41] | J. Liu, T.B. Lu, H. Li, et al. DNA-binding and cleavage studies of a dinuclear copper (Ⅱ) complex with a 26-membered hexaazamacrocycle. Transit. Metal Chem. 27 (2002) 686–690 |
| [42] | L.S. Lerman. Structural considerations in the interaction of DNA and acridines. J. Mol. Biol. 3 (1961) 18–30 |
| [43] | Y.X. Li, J.L. Chen, C.Q. Zhu, et al. Preparation and application of cysteine-capped ZnS nanoparticles as fluorescence probe in the determination of nucleic acids. Spectrochim. Acta A:Mol. Biomol. Spectrosc. 60 (2004) 1719–1724 |
| [44] | Z.S. Yu, X.Y. Ma, Q. Zhang, B. Yu. Application of ZnS:Mn/ZnS quantum dots in quantitative analysis of DNA. J. Instr. Anal. 30 (2011) 789–794 |
| [45] | L.L. Wang, S.P. Liu, W.J. Liang, et al. Detection of DNA utilizing a fluorescent reversible change of a biosensor based on the electron transfer from quantum dots to polymyxin B sulfate. J. Colloid Interface Sci. 448 (2015) 257–264 |
 2016, Vol. 27
2016, Vol. 27 





