b Department of Chemistry, School of Sciences, Gujarat University, Ahmedabad 380009, India
The emergence and recent advance of nanoscience and nanotechnology open new opportunities for the application of nanomaterials in various fields. There has been an great increase in interest in the use of nanoparticles for the development of novel analytical tools [1]. Metal nanoparticles have a surface plasmon resonance absorption in the UV-visible region and it arises from the coherent existence of free electrons in the conduction band due to the small particle size [2]. Silver nanoparticles (AgNps) have strong surface plasmon resonance and therefore used as colorimetric sensors [3, 4]. As a result,the efforts are on for the synthesis of stable water dispersible AgNps because of its various advantages like simplicity,rapidity and benefits of cost-effectiveness. It has been found that the colorimetric assay worked well not only in test tubes,but also it could couple with micro fluidic device for rapid,portable,field detection of real environmental samples [5, 6]. As silver nanoparticles are also found to be fluorescent in nature,therefore this property has been exploited for the development of analytical techniques for detection of various analytesbased on fluorescence measurements [7, 8, 9, 10].
Heavy metal ions are of great concern to the scientific community,especially chemists,biologists,and environmentalists,because of certain disadvantages associated with them. In spite of the fact that some heavy metal ions play important roles in living systems,they are very toxic above a certain level and causes serious environmental and health problems [11].
Among all heavy metal ions,mercury is highly toxic. Mercury [12] contamination occurs through variety of natural and anthropogenic sources including oceanic and volcanic emissions,gold mining,combustion of fossil fuels [13, 14],long-term damage to biological systems and bring oxidative damage to human body [15].
Several techniques for the determination of mercury ions have been reported over the past few years. These include,inductively coupled plasma mass spectroscopy [16, 17, 18],spectrophotometry [19],neutron activation analysis [20],anodic stripping voltammetry [21],X-ray fluorescence spectrometry [22],electrothermal atomic absorption spectrometry [23],atomic fluorescence spectrometry [24],atomic absorption spectrometry [25, 26, 27] and potentiometric ion-selective electrodes [28]. Among these much attention has been given to spectrofluorimetry because the fluorometric methods can reach a much lower detection limit and the instrumentation involved is widely available. Hg(Ⅱ) responsive chemosensors with fluorescence response offer a promisingly simple and rapid approach in monitoring the aqueous Hg(Ⅱ) in biological and environmental samples. As far as sensitivity is considered,fluoroionophores showing fluorescence enhancement upon metal ion binding are favoured over those exhibiting fluorescence quenching [29, 30, 31, 32]. Therefore important practical challenge is to have a water soluble sensors which can sense Hg(Ⅱ) ions via turn-on mechanism and appropriately meet the criteria of selectivity over other competing metal ion contaminants and of optical sensitivity in aqueous solution.
Herein,we report the synthesis of stable water dispersible silver nanoparticles (AgNps) using calix[4]pyrrole octahydrazide (CPOH) as a reducing as well as capping agent. CPOH-AgNps acts as chemosensor which possess excellent selectivity and sensitivity for the detection Hg(Ⅱ) ions over other metal ions by colorimetry and spectrofluorimetry. CPOH-AgNps have also been used successfully for the determination of Hg(Ⅱ) ions in ground water and industrial effluent water samples.
2. Experimental 2.1. Chemicals and instrumentsAll metal salts including silver nitrate were purchased from Sigma-Aldrich. Other reagents and solvents of analytical grade were purchased from commercial sources and used without further purification. All aqueous solutions were prepared from Millipore water (resistivity,18 MV cm @25 ℃; Millipore Systems). TLC plates (F-2009) fluorescence active were procured from the Merck.
The melting points (uncorrected) were recorded from a VEEGO (Model; VMP-DS) melting point apparatus. FT-IR spectra were recorded on Bruker,tensor 27 Infrared spectrometer with samples prepared as KBr pellets. 1H NMR spectra were recorded on a FTNMR model Bruker,Advance Ⅱ (500 MHz) at 298 K with TMS as the internal reference. Mass spectra were recorded on JEOL SX 102/ DA 6000 mass spectrometer using Xenon/Argon (6 kV,10 mA) as the FAB-gas and ESI-MS were recorded on micromass Quarter2 mass spectrometer. Absorption spectra were recorded on a Jasco V-570 UV-visible spectrophotometer. pH of the solutions was measured using pH analyzer LI 614-Elico. The Malvern Zetasizer (Model; ZEN3600) was used for particle size (e.g.,hydrodynamic diameter) and the zeta potential measurements by Laser Doppler Electrophoresis (LDE). TEM images were recorded in MACK/model JEOL,JEM 2100 at an accelerated voltage of 200 kV. Fluorescence spectra were recorded on Jasco FP-6500 spectrofluorimeter.
2.2. Synthesis of compounds (
|
Download:
|
| Scheme. 1. Synthesis of compound CP, ECP and CPOH. | |
Synthesis of meso-tetra(methyl) meso-tetra(3,5-dihydroxyphenyl) calix[4]pyrrole (CP): A mixture of 3,5-dihydroxyacetophenone (2.28 g 0.015 mol) and freshly distilled pyrrole (1.0 mL,0.015 mol) were dissolved in 30 mL of ethanol. To the above solution of BF3(OEt)2 1.0 mL was added dropwise. The reaction mixture was subjected to microwave irradiation for approximately 10-12 min with a break after every 2 min,for the purpose of stirring. After 10-12 min,the dark brown mixture was quenched in 200 mL of distilled water with constant stirring. Triethylamine was added dropwise to neutralize the acid. Light pink coloured precipitate was filtered quickly and made soluble in diethyl ether,dried over MgSO4 and concentrated under reduced pressure to get solid residue (enriched α,α,α,α-isomer) which was further separated and purified by column chromatography to get white crystalline solid of compound CP [33]. White solid (70% yield),m.p. 225 ℃,FT-IR (KBr disk,cm-1): 3415,1645 (Fig. S1 in Supporting information). 1H NMR (500 MHz,DMSO-d6): δ 1.73 (s,12H),5.35 (d,4H),5.86 (d,4H),7.25-7.84 (m,24H),8.60 (m,4H) (Fig. S2 in Supporting information). ESI-MS m/z: 806 [M + 1]+ (Fig. S3 in Supporting information). 13C NMR (500 MHz,DMSO-d6): δ 158.6,157.2,138.1,106.74,104.5,100.3,44.5,28.6 (Fig. S4 in Supporting information).
Synthesis of meso-tetra(methyl) meso-tetra(3,5-diphenoxy acetate) calix[4]pyrrole (ECP): Meso-tetra(methyl) meso-tetra(3,5-dihydroxyphenyl) calix[4]pyrrole (CP) (2.00 g,2.5 μmol) and K2CO3 (2.76 g,8 μmol) were suspended in dry acetone (100 mL) and stirred for 3 h. Ethyl bromoacetate (3.3 g,8 μmol) was added and the suspension was refluxed for 72 h. After cooling,the solution was filtered off to remove K2CO3,and the solvent removed in vacuum. A brown oil was obtained which was dissolved in dichloromethane (40 mL) and washed with water (4 × 15 mL). The organic phase was separated and then dried with MgSO4. The solvent was removed in vacuum affording an oil which was triturated with alcohol (50 mL) affording the product as a brownish powder which was collected by filtration and dried under high vacuum. The product was dried,recrystallized in chloroform to give compound ECP. Light brownish solid (66% yield),m.p. 85 ℃. FT-IR (KBr disk,cm-1): 2982,1770 (Fig. S5 in Supporting information). 1H NMR (500 MHz,CDCl3): δ 1.90 (t,12H,J = 7.0 Hz,CH3),2.56 (t,12H,CCH3),3.81 (q,12H,J = 7.0 Hz,OCH2),4.65 (s,16H,OCH2CO),6.16 (d,8H,Py,CH),6.35 (d,8H,J = 8.5 Hz,ArH),6.92 (s,4H,NH) (Fig. S6 in Supporting information). ESI-MS m/z: 1509 [M + H2O]+ (Fig. S7 in Supporting information). 13C NMR (500 MHz in CDCl3): δ 169.3,160.5,142.1,130.3,107.2,104.5,97.6,65.4,61.2,53.2,14.2 (Fig. S8 in Supporting information).
Synthesis of CPOH: Octaacetatemeso-tetra(methyl) meso-tetra(3,5-diphenoxy acetate) calix[4]pyrrole(ECP) (2 g (w/v)),1.3 μmol) and hydrazine hydrate (1.3 g (w/v),20.8 μmol) were suspended in 120 mL of fresh distilled methanol: toluene (50:50) and stirred reflux for 72 h. The solvent was removed in vacuum and the white solid was suspended in dichloromethane (50 mL). The suspension was filtered and washed with DCM (3 × 20 mL). The crude compound was recrystallized in hot water to give pure white solid compound CPOH [34]. White crystal (46% yield),m.p. 155 ℃. FT-IR (KBr disk,cm-1): 3221,1571 (Fig. S9 in Supporting information). 1H NMR (500 MHz,DMSO-d6): δ 1.83 (t,12H,J = 7.0 Hz,CH3),4.32 (t,16H,OCH2),4.38 (q,16H,NH2),6.19 (d,8H,Py,CH),6.36 (d,8H,J = 8.5 Hz,ArH),8.49-8.99 (s,4H,NH). 9.37 (s,8H,CONH) (Fig. S10 in Supporting information). ESI-MS m/z: 1378.9 [M-H]- (Fig. S11 in Supporting information). 13C NMR (500 MHz,DMSO-d6): δ 166.3,160.5,142.3,130.2,107.2,104.2,97.6,66.4,53.1,14.1.
2-3. Procedure for the synthesis of calix[Silver nanoparticles were prepared by reduction of silver nitrate in presence of calix[4]pyrrole octahydrazide. A series of experiments were performed,varying the concentration of silver nitrate and calix[4]pyrrole octahydrazide to obtain highly stable silver nanoparticles. In a typical procedure,25 mL of 1.0 μmol/L solution of calix[4]pyrrole octahydrazide was rapidly added into the 25 mL of 1.0 μmol/L boiling solution of AgNO3. The heating was continued only for an hour and the solution was stirred further at room temperature for about 2 h. The colour of the solution changed from colourless to pale yellow,indicates the successful formation of calix[4]pyrrole octahydrazide capped silver nanoparticles. The resulting colloidal solution (0.0053%) was then subjected to centrifugation at REMI (R-℃) 14000RPM. The centrifugate was checked for absence of Ag ions and CPOH by atomic absorption spectrophotometry and UV-visible spectrophotometry,respectively. The residue was washed with copious amount of deionized water to remove any unbound molecules and again redispersed in deionized water to get 0.0053% solution of CPOH-AgNps for further studies (Scheme 2).
|
Download:
|
| Scheme. 2. Schematic representation showing the preparation of silver nanoparticles (CPOH-AgNps). | |
2.4. General procedure for the UV-visible and fluorescence measurements
Stock solution of the CPOH-AgNps (0.0053%) and of nitrate salts (2 μmol/L) of various cations Pb(Ⅱ),Cd(Ⅱ),Mn(Ⅱ),Fe(Ⅲ),Ni(Ⅱ),Zn(Ⅱ),Hg(Ⅱ),Co(Ⅱ),Cu(Ⅱ)were prepared in deionized water. 2.5 mL of CPOH-AgNps and 2.5 mL of each cation were taken in a 5 mL volumetric flask,so that the effective concentration of the metal ions is 1 μmol/L. UV-visible spectra and fluorescence spectra of the resulting solution was compared with surface plasmon resonance band (428 nm) and emission spectra (580 nm) of CPOH-AgNps.
3. Results and discussion 3.1. Synthesis and mechanism of formation of silver nanoparticles (CPOH-AgNps)In this study,water soluble calix[4]pyrrole octahydrazide (CPOH) which has all the power and properties of hydrazide to act as reducing agent and its web type structure with an inherent hollow cavity making it a very effective stabilizing agent have been used for the formation of water dispersible stable nanoparticles (CPOH-AgNps). Reduction of AgNO3 by calix[4]pyrrole octahydrazide occurs via oxidation of the amino group i.e.,transfer of electrons from the amine of calix[4]pyrrole octahydrazideto the Ag+ ions. CPOH can passivate the surface of silver nanoparticles and stabilize nanoparticles owing to the coordination of nitrogen atoms of hydrazide (-NH-NH2) group with Ag atoms at the surface of silver nanoparticles. The resulting metallic silver nucleates to form silver nanoparticles and further stabilize it electrostatically as follows [35, 36, 37].
| $AgN{{O}_{3}}+RNHN{{H}_{2}}\to A{{g}^{0}}+RNHN{{H}_{2}}^{+}+NO_{3}^{-}$ |
A drop of dilute solution of aqueous nanoparticles was placed on carbon coated copper grids and was dried in vacuum and directly observed in the TEM. The morphology and particles size of CPOH-AgNps as shown in Fig. 1a revealed that the particles are roughly spherical in shape and uniform in size,as well as,well dispersed with a narrow size distribution with an average particles size of 18 ± 2 nm (Fig. 1b). The size distribution of the CPOH-AgNps was also determined using particle size analyzer where the average hydrodynamic diameter was found to be 26 ± 3 nm. These higher values were due to the light scattered by the core particle and the layers formed on the surface of the particles. Energy-dispersive X-ray (EDX) analysis spectrum recorded in the spot-profile mode from one of the densely populated CPOH-AgNps regions on the surface of film. Strong signals from Ag atoms while weaker signals from C,O,Si,Cu and Ca atoms were observed (Fig. 1c). The overall particles charge in a particular medium is denoted as their zeta potential value which is also responsible for deciding the fate of stability. Here,synthesized silver nanoparticles (CPOH-AgNps) had a 13 ± 2 MeV zeta potential value,which is sufficient to keep the particles away from aggregation and maintained the stability,moreover positive value also suggests that hydrazide groups were successfully introduced onto the surface of nanoparticles.

|
Download:
|
| Fig. 1. (a) TEM image of silver nanoparticles (CPOH-AgNps); (b) Particle size distribution graph and (c) Energy-dispersive X-ray (EDX). | |
3.3. Effect of pH and time on stability of CPOH-AgNps
Stability of silver nano particles has been investigated by change in their SPR band and fluorescence intensity at different pH. SPR band of CPOH-AgNps shows negligible change at pH other than 7.0 for a day or two but CPOH-AgNps tend to agglomerate after that. It is noteworthy that when their agglomerated form is sonicated for 15-20 min they retain their originality with a negligible compromise in their SPR band and thereby size (Fig. 2a). No change in SPR band and absorbance of the CPOH-AgNps at pH 7.0 was recorded up to 120 days (Fig. 2b). Likewise,fluorescence intensity of silver nanoparticles CPOH-AgNps decreases slightly at pH other than 7.0 and the no change in wavelength (Fig. 2c). Therefore,pH 7.0 was selected to carry out all experiments on CPOH-AgNps and concluded that CPOH-AgNps shows maximum stability and fluorescence intensity at pH 7.0.

|
Download:
|
| Fig. 2. (a) Effect of pH on surface plasmon resonance of CPOH-AgNps; (b) Stability of silver nanoparticles (CPOH-AgNps) with respect to time on wavelength/absorbance; (c) Effect of pH on fluorescence intensity/wavelength. | |
3.4. Colorimetric detection of Hg(Ⅱ) using CPOH-AgNps
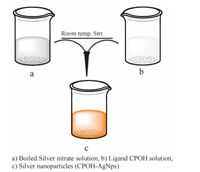
Although studies using silver nanoparticles have been reported for the colorimetric detection of Hg(Ⅱ) ions [10, 38] but CPOH- AgNPs have been found to be unique in their performance for selective and sensitive naked eye detection of Hg(Ⅱ) ions in aqueous samples for the following reasons: (i) Very low concentration of nanoparticles (0.00066%) is needed for detection of Hg(Ⅱ) ions; (Ⅱ) High stability of water dispersible nanoparticles; (Ⅱi) Selectivity among various metal ions. When 2 mL of CPOHAgNps (0.00066%) which is yellowish brown in colour are mixed with 2 mL (2 μmol/L) of each metal ion (Pb(Ⅱ),Cd(Ⅱ),Mn(Ⅱ),Fe(Ⅲ),Ni(Ⅱ),Zn(Ⅱ),Hg(Ⅱ),Co(Ⅱ) and Cu(Ⅱ) separately. There was no visible change in colour with all the metal ions except Hg where the colour changed instantly from yellowish brown to colourless (Fig. 3). The repeated observation confirms that method is simple,sensitive and selective for colorimetric detection of Hg(Ⅱ) ions in aqueous media.

|
Download:
|
| Fig. 3. Visual colour change of silver nanoparticles (CPOH-AgNps) upon addition of different metal ions (1 μmol/L) at pH 7.0. | |
3.5. Interaction of Hg(Ⅱ) with CPOH-AgNps by UV-visible and fluorescence spectroscopy measurements
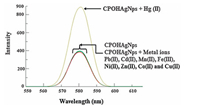
Interaction of different cations with CPOH-AgNps concluded that except Hg(Ⅱ),no other metal ion i.e. Pb(Ⅱ),Cd(Ⅱ),Mn(Ⅱ),Fe(ⅡI),Ni(Ⅱ),Zn(Ⅱ),Co(Ⅱ) and Cu(Ⅱ) showed any noticeable change in the SPR band of CPOH-AgNps (Fig. 4). To further study the interaction behaviour of Hg(Ⅱ) with CPOH-AgNps,different concentrations of aqueous solutions of Hg(Ⅱ) ranging from 1 nmol/L to 1 μmol/L were mixed with nanoparticles and absorption spectra were recorded. Interestingly,with the increasing concentration of Hg(Ⅱ) ions SPR band shifted 21 nm towards the lower wavelength region (i.e. from 428 nm to 407 nm,blue shift). Morris et al. [39] have also demonstrated that the adsorption of Hg(Ⅱ) results in a blue shift of the surface plasmon mode of Ag nanoparticles. The authors believe that the shift to lower wavelength is related to a change in the polarization of the near surface region (Hg is known to lower the work function of Ag surfaces by 0.3 eV) [40].

|
Download:
|
| Fig. 4. UV–visible spectra of CPOH-AgNps after the addition of various metal ions. | |
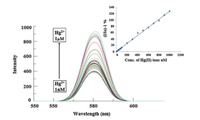
The emission spectra of the CPOH-AgNps were recorded at 580 nm over a wide range of pH (3.0 to 11.0) (Fig. 2c) and the maximum emission intensity was observed at pH 7.0. Therefore emission spectra of CPOH-AgNPs with various metal ions i.e. Pb(Ⅱ),Cd(Ⅱ),Mn(Ⅱ),Fe(ⅡI),Ni(Ⅱ),Zn(Ⅱ),Hg(Ⅱ),Co(Ⅱ),Cu(Ⅱ) and Hg(Ⅱ) ions were recorded at pH 7.0 (Fig. 5). It was observed that none other than Hg(Ⅱ) showed an increase in fluorescence intensity. The linear range of minimum and maximum detection of Hg(Ⅱ) ions was determined by means of fluorescence titration of CPOH-AgNPs (0.0053%) with increasing concentration of Hg(Ⅱ) ions. It was noted that fluorescence intensity,at 580 nm,gradually increases with an increase in the concentration of Hg(Ⅱ) ions (inset Fig. 6). The minimum and maximum fluorescence enhancement was observed at 1 nmol/L and 1 μmol/L concentration of Hg(Ⅱ) ions,respectively (Fig. 6),which is considered as the minimum and maximum detection limit CPOH-AgNps. This observation is in contrast with what has been observed by John et al. [41] where functionalized mercaptothiadiazole capped silver nanoparticles (AgNps) have been used for determination of Hg(Ⅱ) ions in aqueous solution using by spectrofluorimetry and decrease in emission intensity was ascribed to the aggregation of AgNps. In our case there is no aggregation of nanoparticles on addition of Hg(Ⅱ) ions,SPR bands shows blue shift,the emission intensity is increased,therefore it is deduced that CPOH-AgNps can be used as selective and sensitive sensor for Hg(Ⅱ) ions. The fluorescence property of the sensor is due to the silver metal nanoparticle. In the unbound state CPOH was not found to be fluorescent. As indicated in the graphical abstract,the sensing mechanism occurs through the internal charge transfer from Hg(Ⅱ) to CPOH. After addition of Hg(Ⅱ),the electrons are transferred from CPOH to Hg(Ⅱ) resulting in ICT between Hg(Ⅱ) and CPOH. This type of interaction causes enhancement in the fluroscence of nanoparticles [42, 43].
|
Download:
|
| Fig. 5. Fluorescence spectra of CPOH-AgNps on addition various metal ions(1 μmol/L). | |
|
Download:
|
| Fig. 6. Fluorescence response of CPOH-AgNps on addition of Hg(Ⅱ) solution (1 nmol/L to 1 μmol/L). The inset shows a Stern Volmer plot of relative intensity versus concentration of Hg(Ⅱ). | |
3.6. Application
Herein,we report the selective detection of Hg(Ⅱ) ions in industrial waste water (Vatva,GIDC,Ahmedabad) and tap water of University area which was found to be 9.53 nmol/L and 4.41 nmol/L,respectively. To further confirm,standard addition method was adopted. To the 0.9 mL CPOH-AgNps solution,0.05 mL of standard Hg(Ⅱ) ions of different strengths (25,50,75 and 100 nmol/L) was spiked,followed by the addition of 0.05 mL sample solution. Since increase in relative fluorescence intensity is linear with successive addition of nmol/L concentration of Hg(Ⅱ) ions. The fluorescence intensity of the resulting solution of four different sets was measured and graph was plotted between the percent intensity increment (I/I0) - 1 and concentration of Hg(Ⅱ) ions,where (I) and (I0) are the fluorescence intensity of CPOHAgNps in presence of Hg(Ⅱ) ions and absence of Hg(Ⅱ) ions. The reference concentration (inset Fig. 7) is subtracted from the computed Hg(Ⅱ) concentrations to obtain the Hg(Ⅱ) ions concentration in the test solution (Fig. 7 and Table 1).

|
Download:
|
| Fig. 7. Graph shows the concentration of Hg(Ⅱ) ions found in water samples; Inset shows linearity of standard solution of Hg(Ⅱ) ions up to 100 nmol/L. | |
|
|
Table 1 Determination of Hg(II) ions in different water samples. |
4. Conclusion
Stable water dispersible silver nanoparticles (AgNps) have been synthesized using calix[4]pyrrole octahydrazide (CPOH) which act as both reducing as well as stabilizing agent. CPOH-AgNps were found to be stable over 120 days at varied pH. CPOH-AgNps can be used as a selective and sensitive sensor for colorimetric and spectrofluorimetric determination of Hg(Ⅱ) ions in natural aqueous samples. Micromolar concentration of Hg(Ⅱ) ions can be detected by naked eye as the colour of CPOH-AgNps changes from yellow to colourless. Nanomolar concentration of Hg(Ⅱ) ions can be determined by spectrofluorimetry via a turn on mechanism. Minimum and maximum linear range of detection of Hg(Ⅱ) ions by spectrofluorimetry is 1 nmol/L to 1 μmol/L. The method has been successfully applied for determination of Hg(Ⅱ) in tap water and industrial effluent water samples. The CPOH-AgNps can also be explored for their use in the detection of Hg(Ⅱ) ions in environmental and biological samples.
Appendix A. Supplementary dataSupplementary data associated with this article can be found,in the online version,at http://dx.doi.org/10.1016/j.cclet.2016.01.012.
| [1] | K. Omidfar, F. Khorsand, M.D. Azizi. New analytical applications of gold nanoparticles as label in antibody based sensors. Biosens. Bioelectron. 43 (2013) 336–347 |
| [2] | C. Burda, X.B. Chen, R. Narayanan, M.A. El-Sayed. Chemistry and properties of nanocrystals of different shapes. Chem. Rev. 105 (2005) 1025–1102 |
| [3] | Y.H. Lin, C.E. Chen, C.Y. Wang, et al. Silver nanoprobe for sensitive and selective colorimetric detection of dopamine via robust Ag-catechol interaction. Chem. Commun. 47 (2011) 1181–1183 |
| [4] | Y. Wang, F. Yang, X.R. Yang. Colorimetric detection of mercury(Ⅱ) ion using unmodified silver nanoparticles and mercury-specific oligonucleotides. ACS Appl. Mater. Interfaces 2 (2010) 339–342 |
| [5] | S. He, D. Li, C. Zhu, et al. Design of a gold nanoprobe for rapid and portable mercury detection with the naked eye. Chem. Commun. 40 (2008) 4885–4887 |
| [6] | G.L. Wang, X.Y. Zhu, H.J. Jiao, Y.M. Dong, Z.J. Li. Ultrasensitive and dual functional colorimetric sensors for mercury(Ⅱ) ions and hydrogen peroxide based on catalytic reduction property of silver nanoparticles. Biosens. Bioelectron. 31 (2012) 337–342 |
| [7] | L. Shang, S.J. Dong. Silver nanocluster-based fluorescent sensors for sensitive detection of Cu(Ⅱ). J. Mater. Chem. 18 (2008) 4636–4640 |
| [8] | Y.W. Zhang, H.L. Li, X.P. Sun. Silver nanoparticles as a fluorescent sensing platform for nucleic acid detection. Chin. J. Anal. Chem. 39 (2011) 998–1002 |
| [9] | L. Shang, S.J. Dong. Sensitive detection of cysteine based on fluorescent silver clusters. Biosens. Bioelectron. 24 (2009) 1569–1573 |
| [10] | B. Roy, P. Bairi, A.K. Nandi. Selective colorimetric sensing of mercury(Ⅱ) using turn off-turn on mechanism from riboflavin stabilized silver nanoparticles in aqueous medium. Analyst 136 (2011) 3605–3607 |
| [11] | D.T. Quang, J.S. Kim. Fluoro- and chromogenic chemodosimeters for heavy metal ion detection in solution and biospecimens. Chem. Rev. 110 (2010) 6280–6301 |
| [12] | J. Dessingou, K. Tabbasum, A. Mitra, V.K. Hinge, C.P. Rao. Lower rim 1,3-di{4-antipyrine}amide conjugate of calix. J. Org. Chem. 77 (2012) 1406–1413 |
| [13] | O. Malm. Gold mining as a source of mercury exposure in the Brazilian Amazon. Environ. Res. 77 (1998) 73–78 |
| [14] | J.M. Benoit, W.F. Fitzgerald, A.W.H. Damman. The biogeochemistry of an ombrotrophic bog:evaluation of use as an archive of atmospheric mercury deposition. Environ. Res. 78 (1998) 118–133 |
| [15] | M.R. Knecht, M. Sethi. Bio-inspired colorimetric detection of Hg2+ and Pb2+ heavy metal ions using Au nanoparticles. Anal. Bioanal. Chem. 394 (2009) 33–46 |
| [16] | D. Karunasagar, J. Arunachalam, S. Gangadharan. Development of a 'collect and punch' cold vapour inductively coupled plasma mass spectrometric method for the direct determination of mercury at nanograms per litre levels. J. Anal. At. Spectrom. 13 (1998) 679–682 |
| [17] | A.N. Anthemidis, G.A. Zachariadis, C.E. Michos, J.A. Stratis. Time-based on-line preconcentration cold vapour generation procedure for ultra-trace mercury determination with inductively coupled plasma atomic emission spectrometry. Anal. Bioanal. Chem. 379 (2004) 764–796 |
| [18] | C.F. Harrington, S.A. Merson, T.M. D'Silva. Method to reduce the memory effect of mercury in the analysis of fish tissue using inductively coupled plasma mass spectrometry. Anal. Chim. Acta 505 (2004) 247–254 |
| [19] | Z. Marczenko. Separation and Spectrophotometric Determination of Elements, John Wiley and Sons. New York, NY (1986) |
| [20] | J.C. Yu, J.M. Lo, C.M. Wai. Extraction of gold and mercury from sea water with bismuth diethyldithiocarbamate prior to neutron activation-γ-spectrometry. Anal. Chim. Acta 154 (1983) 307–312 |
| [21] | P. Ugo, L.M. Moretto, P. Bertoncello, J. Wang. Determination of trace mercury in salt water at screen-printed electrodes modified with sumichelate Q10R. Electroanalysis 10 (1998) 1017–1021 |
| [22] | L. Bennun, J. Gomez. Determination of mercury by total-reflection X-ray fluorescence using amalgamation with gold. Spectrochim. Acta, B At. Spectrosc. 52 (1997) 1195–1200 |
| [23] | C. Burrini, A. Cagnini. Determination of mercury in urine by ET-AAS using complexation with dithizone and extraction with cyclohexane. Talanta 44 (1997) 1219–1223 |
| [24] | A. Shafawi, L. Ebdon, M. Foulkes, P. Stockwell, W. Corns. Determination of total mercury in hydrocarbons and natural gas condensate by atomic fluorescence spectrometry. Analyst 124 (1999) 185–189 |
| [25] | J.V. Cizdziel, S. Gerstenberger. Determination of total mercury in human hair and animal fur by combustion atomic absorption spectrometry. Talanta 64 (2004) 918–921 |
| [26] | E. Kopysc, K. Pyrzynska, S. Garbos, E. Bulska. Determination of mercury by coldvapor atomic absorption spectrometry with preconcentration on a gold-trap. Anal. Sci. 16 (2000) 1309–1312 |
| [27] | Y. Yamini, N. Alizadeh, M. Shamsipur. Solid phase extraction and determination of ultra trace amounts of mercury(Ⅱ) using octadecyl silica membrane disks modified by hexathia-18-crown-6-tetraone and cold vapour atomic absorption spectrometry. Anal. Chim. Acta 355 (1997) 69–74 |
| [28] | P. Bühlmann, E. Pretsch, E. Bakker. Carrier-based ion-selective electrodes and bulk optodes. 2. Ionophores for potentiometric and optical sensors. Chem. Rev. (1998) 1593–1688 |
| [29] | H. Zhang, Q.L. Wang, Y.B. Jiang. 8-Methoxyquinoline based turn-on metal fluoroionophores. Tetrahedron Lett. 48 (2007) 3959–3962 |
| [30] | D.Y. Wu, W. Huang, Z.H. Lin, et al. Highly sensitive multiresponsive chemosensor for selective detection of Hg2+ in natural water and different monitoring environments. Inorg. Chem. 47 (2008) 7190–7201 |
| [31] | R. Martínez, F. Zapata, A. Caballero, et al. 2-Aza-1,3-butadiene derivatives featuring an anthracene or pyrene unit:highly selective colorimetric and fluorescent signaling of Cu2+cation. Org. Lett. 8 (2006) 3235–3238 |
| [32] | K. Rurack, M. Kollmannsberger, U. Resch-Genger, J. Daub. A selective and sensitive fluoroionophore for HgⅡ, AgⅠ, and CuⅡ with virtually decoupled fluorophore and receptor units. J. Am. Chem. Soc. 122 (2000) 968–969 |
| [33] | G. Gil-Ramírez, J. Benet-Buchholz, E.C. Escudero-Adán, P. Ballester. Solid-state self-assembly of a calix. J. Am. Chem. Soc. 129 (2007) 3820–3821 |
| [34] | K.D. Bhatt, D.J. Vyas, B.A. Makwana, S.M. Darjee, V.K. Jain. Highly stable water dispersible calix. Spectrochim. Acta, A:Mol. Biomol. Spectrosc. 121 (2014) 94–100 |
| [35] | U. Nickel, A. zu Castell, K. Pöppl, S. Schneider. A silver colloid produced by reduction with hydrazine as support for highly sensitive surface-enhanced raman spectroscopy. Langmuir 16 (2000) 9087–9091 |
| [36] | M. Chen, Y.G. Feng, X. Wang, et al. Silver nanoparticles capped by oleylamine:formation, growth, and self-organization. Langmuir 23 (2007) 5296–5304 |
| [37] | J.D.S. Newman, G.J. Blanchard. Formation of gold nanoparticles using amine reducing agents. Langmuir 22 (2006) 5882–5887 |
| [38] | V. Bhalla, R. Tejpal, M. Kumar, A. Sethi. Terphenyl derivatives as "turn on" fluorescent sensors for mercury. Inorg. Chem. 48 (2009) 11677–11684 |
| [39] | T. Morris, H. Copeland, E. McLinden, S. Wilson, G. Szulczewski. The effects of mercury adsorption on the optical response of size-selected gold and silver nanoparticles. Langmuir 18 (2002) 7261–7264 |
| [40] | C. Radhakumary, K. Sreenivasan. Gold nanoparticles generated through "green route" bind Hg2+ with a concomitant blue shift in plasmon absorption peak. Analyst 136 (2011) 2959–2962 |
| [41] | N. Vasimalai, G. Sheeba, S.A. John. Ultrasensitive fluorescence-quenched chemosensor for Hg(Ⅱ) in aqueous solution based on mercaptothiadiazole capped silver nanoparticles. J. Hazard. Mater. 213- 214 (2012) 193–199 |
| [42] | H. Liu, X. Hao, C.H. Duan, et al. Al3+-induced far-red fluorescence enhancement of conjugated polymer nanoparticles and its application in live cell imaging. Nanoscale 5 (2013) 9340–9347 |
| [43] | Y.F. Xu, Y.H. Liu, X.H. Qian. Novel cyanine dyes as fluorescent pH sensors:PET. ICT mechanism or resonance effect? J. Photochem. Photobiol., A Chem. 190 (2007) 1–8 |
 2016, Vol. 27
2016, Vol. 27 



