b State Key Laboratory of NBC Protection for Civilian, Beijing 102205, China ;
c Polymer Research Center, Korea Institute of Science and Technology, Seoul 136-791, Republic of Korea
Microorganisms are prevalent on surfaces that we touch everyday. Infections caused by these small organisms result in significant morbidity and, in some cases, mortality [1]. Recently, there has been much effort to overcome such infections by preparing bactericidal materials based on antibiotics [2, 3], silver nanoparticles [4], phenols [5] and peptides [6]. However, such approach has some fundamental flaws such as loss of antibacterial chemicals and possibility of drug resistance in pathogens through mutation [7]. Therefore, it is desirable to design new antibacterial materials and methods to eliminate the issues associated with uncontrolled release of biocides. Chemical binding of antibacterial monomers to polymer chains has been carried out to prevent antimicrobial ingredients from leaching [8, 9, 10, 11, 12, 13]. However, the preparation of these polymers require complicated design and cumbersome procedures. There is an urgent need for a more convenient method to prepare antibacterial polymers.
UV photopolymerization is a feasible industrial process with applications ranging widely from polymeric coatings to inks and adhesives [14, 15]. Recently, thiol-ene photopolymerizable systems are gaining popularity in both academic research and industrial applications, because they possess all the desirable traits of a "click" reaction with short reaction times, wide functional group and solvent tolerance, few to no byproducts [16]. The thiol-ene UV curable coatings are based on a mixture of multifunctional thiols and multifunctional enes. This polymerization process occurs through well-known radical intermediates that involve a free-radical addition followed by a chain transfer reaction [17]. Furthermore, these cross-linked networks are characterized by advantages such as lower film stress, relatively narrow glass transitions, low shrinkage, reduced oxygen inhibition and relatively high functional group conversion compared with those made by acrylic cross-linkers [18].
Poly[(mercaptopropyl)methylsiloxane] (PMMS) with multifunctional enes is suitable for the thiol-ene systems [19]. As for antibacterial agents, some imidazolium salts with long alkyl chains are known to have high antimicrobial properties and are widely used as disinfectants [20]. In this work, we present a facile protocol to prepare PMMS-based antibacterial polymer coatings via a twostep sequential thiol-ene click chemistry. As shown in Scheme 1, by controlling the molar ratio of PMMS and antibacterial monomer, PMMS-based antibacterial polymers can be partially functionalized in the first-step thiol-ene click process. After then the leaving spare mercapto groups were used as cross-linking sites to form networks in the second-step thiol-ene click process.

|
Download:
|
| Scheme. 1. Polysiloxane-based antibacterial polymer coatings prepared by a twostep sequential thiol-ene click chemistry protocol. | |
2. Experimental
Unless otherwise noted, ACS reagent grade chemicals and solvents were purchased from commercial vendors and used without further purification. Poly(mercaptomethyl)siloxane (PMMS, SMS-992) was purchased from Gelest Inc. Triallyl cyanurate (TAC, 99%), deuterated solvents for NMR and 2, 2- dimethoxy-2-phenylacetophenone (DMPA, 99%) were purchased from (J&K). 1H NMR spectra were recorded in DMSO-d6 (J&K) using TMS as internal standard on a Agilent 400 MHz. The NMR spectra were analyzed and processed using MestReNova-6.1.1-6384 software. Thermogravimetric analysis (TGA) was performed under a controlled atmosphere of N2 with a Mettler TGA/DSC 1 instrument between 25 ℃ and 600 ℃ at a heating rate of 10 ℃/min. Dynamic mechanical analysis (DMA) was performed using a TA Instruments Q800 dynamic mechanical analyzer in tension film mode equipped with a gas cooling accessory. Samples were heated from -80 ℃ to 140 ℃ for the samples at a ramp rate of 3 ℃/min. Pencil hardness was evaluated on photocured films according to the standard test method GB/T 6739-2006. Contact angles (θ) were obtained with a VCA Optima contact angle measuring instrument (AST Products, Inc.) with a drop size of 1.0 μL of deionized water. The θ values were determined five times for a reliable average value.
2.1. Synthesis of 1-allyl-3-decylimidazolium bromide1-Decylimidazole was synthesized according to previous literature [21]. 1-Decylimidazole (5.49 g, 26.3 mmol) and 3- bromopropene (3.1 g, 26.3 mmol) were added to a 100 μL round-bottom flask containing 30 μL ethanol with condenser. The reaction mixture was stirred for 24 h. Solvent was then removed under reduced pressure. The crude product was washed three times with ether and dried in a vacuum oven at 50 ℃ for 24 h to give the 1-allyl-3-decylimidazolium bromide (ADIm).
1-Allyl-3-decylimidazolium bromide: This compound was obtained in 97% as a yellow viscous oil. 1H NMR (DMSO-d6, 400 MHz): δ 9.30 (s, 1H, -N-CH-N-), 7.87 (s, 1H, -N-CH-CH-N-), 7.78 (s, 1H, -N-CH-CH-N-), 6.07 (m, 1H, -CH=CH2), 5.32 (dd, 2H, =CH2), 4.87 (d, 2H, -CH2-CH=CH2), 4.19 (t, 2H, -N-CH2-), 1.79 (m, 2H, -CH2-CH2-), 1.24 (broad, 14H, -CH2-CH3), 0.85 (t, 3H, -CH3).
2.2. Synthesis of polymersTypical procedure was used to prepare PMMSx-g-ADIm: ADIm (613 mg, 1.86 mmol), PMMS (500 mg, 3.72 mmol-SH) and DMPA (0.05 equiv. to each-ene moiety, 24 mg, 0.094 mmol) dissolving in DMF (50 μL) were added into a Schlenk-type flask. The flask was purged with dry argon for about 3 min and then exposed to the hand-held UV-lamp (λexc = 365 nm) for 30 min. These reactions were monitored by 1H NMR spectroscopy and were used without further purification for the preparation of crosslinked films. Relative content of PMMS and ADIm were listed in Table 1.
PMMS10-g-ADIm: 1H NMR (DMSO-d6, 400 MHz): δ 9.30 (s, 1H, - N-CH-N-), 7.80 (s, 2H, -N-CH-CH-N-), 4.23 (d, 2H, -N-CH2-), 4.14 (d, 2H, -N-CH2-CH2), 2.44 (m, 22H, -CH2-S-CH2-, HS-CH2-), 2.13 (s, 8H, -SH), 2.04 (m, 2H, -N-CH2-), 1.76 (m, 2H, -N-CH2- CH2-), 1.55 (s, 20H, ≡Si-CH2-CH2-), 1.20 (s, 14H, -CH2-CH3), 0.82 (t, 3H, -CH3), 0.58 (s, 20H, ≡Si-CH2-), 0.05 (s, 30H, ≡Si-CH3).
PMMS25-g-ADIm: 1H NMR (DMSO-d6, 400 MHz): δ 9.28 (s, 1H, - N-CH-N-), 7.80 (s, 2H, -N-CH-CH-N-), 4.22 (d, 2H, -N-CH2-), 4.13 (d, 2H, -N-CH2-CH2), 2.45 (m, 10H, -CH2-S-CH2-, HS-CH2-), 2.17 (s, 3H, -SH), 2.03 (m, 2H, -N-CH2-), 1.76 (m, 2H, -N-CH2- CH2-), 1.55 (s, 8H, ≡Si-CH2-CH2-), 1.20 (s, 14H, -CH2-CH3), 0.82 (t, 3H, -CH3), 0.57 (s, 8H, ≡Si-CH2-), 0.05 (s, 12H, ≡Si-CH3).
PMMS50-g-ADIm: 1H NMR (DMSO-d6, 400 MHz): δ 9.31 (s, 1H, - N-CH-N-), 7.81 (s, 2H, -N-CH-CH-N-), 4.23 (d, 2H, -N-CH2-), 4.15 (d, 2H, -N-CH2-CH2), 2.45 (m, 6H, -CH2-S-CH2-, HS-CH2-), 2.19 (s, 1H, -SH), 2.03 (m, 2H, -N-CH2-), 1.76 (m, 2H, -N-CH2- CH2-), 1.52 (s, 4H, ≡Si-CH2-CH2-), 1.20 (s, 14H, -CH2-CH3), 0.82 (t, 3H, -CH3), 0.57 (s, 4H, ≡Si-CH2-), 0.04 (s, 6H, ≡Si-CH3).
2.3. Preparation of antibacterial polymer coatings via thiol-ene click photopolymerizationFor practical photocuring, triallyl cyanurate (TAC) was added to the above PMMSx-g-ADIm while maintaining a one-to-one ratio of thiol to alkene. Relative content of PMMSx-g-ADIm and TAC were listed in Table 1. DMPA was incorporated at 0.1 wt% as a photoinitiator. DMPA was dissolved using a small amount (no more than 200 μL) of N, N'-dimethyl formamide (DMF) and then added into the thiol-ene formulations. After mixing thoroughly, the resins were coated onto a round glass pane by means of a film applicator and then irradiated under a 365 nm lamp (intensity, 160 mW/cm2) for 3 min in ambient conditions to get transparent photocured films with a usual size of 22 mm in diameter and ~100 μm in thickness. The cross-linked samples were soaked in deionized water for 24 h to remove residual DMF and dried in vacuum at 50 ℃ for 24 h.
|
|
Table 1 Antibacterial films prepared from different proportion of components. |
3. Results and discussion
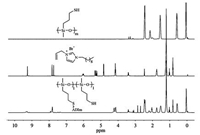
The desired antibacterial component, 1-allyl-3-decylimidazolium bromide (ADIm), was synthesized by a quaternization reaction of 1-decylimidazole with 3-bromopropene. The corresponding polymers, PMMSx-g-ADIm, were prepared by a radicalinitiated thiol-ene addition reaction of PMMS and ADIm. 1H NMR spectra of the corresponding peak assignments of the commercially available polymer PMMS, a antibacterial monomer ADIm and a corresponding polymer PMMS50-g-ADIm are shown in Fig. 1. In comparison, the terminal olefin protons of ADIm originally located at 5.2-5.4 ppm disappeared, which proved a successful grafting reaction. The leaving methine proton of S-H bond of PMMS appearing at ~2.2 ppm was used as cross-linking sites to form networks in the second-step thiol-ene click process.
|
Download:
|
| Fig. 1. 1H NMR spectra of PMMS, ADIm, and PMMS50-g-ADIm. | |
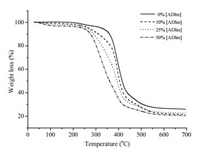
The thermal stability of the hybrid ternary networks was investigated by thermogravimetric analysis. Fig. 2 shows the TGA thermograms of the ADIm-PMMS-TAC system as a function of ADIm concentration. The initial onset of degradation was observed around 250 ℃ and was not obviously affected by the composition of the ternary networks. However, thermal stability of the crosslinked networks significantly decrease with the increase of content of ADIm in the films. Decrease in thermal stability could be attributed to the decrease of cross-linked degree of polymer.
|
Download:
|
| Fig. 2. Degradation profiles from TGA of antimicrobial polymer films with varying concentration of ADIm. | |
Mechanical and physical properties of the thiol-ene networks were investigated by dynamic mechanical analysis (DMA). DMA was used to obtain storage (E') and loss moduli (E") as a function of temperature. The glass transition temperature (Tg) was determined from the peak maximum of tan δ curve. Fig. 3a shows tan d curves for the ADIm-PMMS-TAC ternary system with increasing concentration of ADIm. A shift of tan δ peak to lower temperature was observed with increasing concentration of ADIm, indicating an decrease of Tg for the thiol-ene networks. Fig. 3b shows a marked decrease in the rubbery storage modulus of the ADIm-PMMS-TAC ternary system with increasing concentration of ADIm. The observed decrease in Tg and rubbery modulus can be attributed to an decrease in the relative crosslink density. The surface hardness properties of antibacterial films were also investigated. A decrease in pencil hardness was abserved by increasing ADIm in the photocurable formulation (Table 2). These results are consistent with the glass transition temperature reduced.

|
Download:
|
| Fig. 3. Plots of (a) tan δ vs. temperature and (b) storage modulus vs. temperature for different loading percentages of ADIm in networks. | |
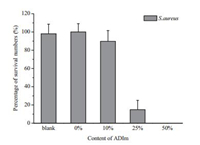
Antimicrobial properties of the prepared films were evaluated by dynamic shake method [22]. The films on the circular glass (diameter: 22 mm) were shaken with 10 μL bacterial suspension containing Staphylococcus aureus (ATCC 292130) (107 cells) in a 50 μL conical tube for 90 min at 37 ℃. The bacterial suspension was then aliquoted and diluted appropriately, and colony forming units (CFUs) were determined. Fig. 4 shows the number of surviving cells after contact with the samples with different content of ADIm. Film-4 containing 50% ADIm showed an antibacterial efficiency of 100%, whereas Film-3 containing 25% ADIm showed a reduced efficiency of 85.4%. Film-2 containing 10% ADIm exhibited much weaker biocidal ability (<14%). The high antibacterial efficiency of the film containing 50% ADIm was mainly attributed to the presence of a large portion of ADIm in the film, which is in accordance with the previous observations that the high cationic surface charge density is critical for a high killing efficiency of Escherichia coli [23]. The water contact angles decreased gradually with the decrease of cross-link density for the crosslinked polymeric films, which were measured to be 75.48, 72.78, 69.58, 36.98 for antibacterial polymer films with 0%, 10%, 25%, 50% ADIm, respectively (Table 2). This indicated that with higher content of the more hydrophilic monomer of ADIm, the hydrophilicity of the film was increased.
|
Download:
|
| Fig. 4. The antimicrobial activity of films with different content of ADIm of 0%, 10%, 25%, 50% with respect to the control tests without films. | |
|
|
Table 2 Water contact angle and pencil hardness of the films containing increasing antibacterial monomer content measured by the contact angle meter and pencil hardness test (GB/T 6739-2006), respectively. |
4. Conclusion
In summary, PMMS-based antibacterial polymer coatings were prepared through a two-step sequential thiol-ene click chemistry. These films with different content of ADIm displayed different properties. With the decrease of crosslinking degree, these films showed lower thermal stability, Tg, rubbery modulus and higher hydrophilicity. Film-4 bearing 50% imidazolium bromide groups was tested as a nonleaching antimicrobial material and displayed excellent antimicrobial activity against S. aureus with 100% killing efficiency. Such antibacterial materials are expected to find applications in the fields of water purification systems, medical devices and health care products.
| [1] | C.C. Zhou, P. Li, X.B. Qi, et al. A photopolymerized antimicrobial hydrogel coating derived from epsilon-poly-L-lysine. Biomaterials 32 (2011) 2704–2712 |
| [2] | M.A. Fischbach, C.T. Walsh. Antibiotics for emerging pathogens. Science 325 (2009) 1089–1093 |
| [3] | J. Wang, X.L. Du, W.S. Pan, X.J. Wang, W.J. Wu. Photoactivation of the cryptochrome/photolyase superfamily. J. Photochem. Photobiol. 22 (2015) 84–102 |
| [4] | S. Shankar, J.W. Rhim. Amino acid mediated synthesis of silver nanoparticles and preparation of antimicrobial agar/silver nanoparticles composite films. Carbohydr. Polym. 130 (2015) 353–363 |
| [5] | D. James Bound, P.S. Murthy, P. Srinivas. Synthesis and antibacterial properties of 2,3-dideoxyglucosides of terpene alcohols and phenols. Food Chem. 185 (2015) 192–199 |
| [6] | W. Yuan, H.T. Jin, Z.H. Ren, et al. Effects of antibacterial peptide on humoral immunity in weaned piglets. Food Agric. Immunol. 26 (2015) 682–689 |
| [7] | A.C. Engler, N. Wiradharma, Z.Y. Ong, D.J. Coady, J.L. Hedrick. Emerging trends in macromolecular antimicrobials to fight multi-drug-resistant infections. Nano Today 7 (2012) 201–222 |
| [8] | U. Mizerska, W. Fortuniak, J. Chojnowski, et al. Polysiloxane cationic biocides with imidazolium salt (Ims) groups, synthesis and antibacterial properties. Eur. Polym. J. 45 (2009) 779–787 |
| [9] | V. Sambhy, B.R. Peterson, A. Sen. Antibacterial and hemolytic activities of pyridinium polymers as a function of the spatial relationship between the positive charge and the pendant alkyl tail. Angew. Chem. Int. Ed. 47 (2008) 1250–1254 |
| [10] | S.C.M. Fernandes, P. Sadocco, A. Aonso-Varona, et al. Bioinspired antimicrobial and biocompatible bacterial cellulose membranes obtained by surface functionalization with aminoalkyl groups. ACS Appl. Mater. Interfaces 5 (2013) 3290–3297 |
| [11] | S.C.M. Fernandes, P. Sadocco, J. Causio, et al. Antimicrobial pullulan derivative prepared by grafting with 3-aminopropyltrimethoxysilane:Characterization and ability to form transparent films. Food Hydrocolloids 35 (2014) 247–252 |
| [12] | G.Y. Li, X.D. Li, Z.D. Chen, et al. Large areas of centimeters-long sic nanowires synthesized by pyrolysis of a polymer precursor by a CVD route. J. Phys. Chem., C 113 (2009) 17655–17660 |
| [13] | Z.Y. Chu, R.A. He, X.B. Zhang, X.D. Li. The production of polycarbosilane-derived porous carbon fibers. Carbon 50 (2012) 2669–2672 |
| [14] | B.J. Sparks, T.J. Kuchera, M.J. Jungman, et al. Cyclic tetravinylsiloxanetetraols as hybrid inorganic-organic thiol-ene networks. J. Mater. Chem. 22 (2012) 3817–3824 |
| [15] | Y.H. Li, K.D. Ahn, D.P. Kim. Synthesis and properties of UV curable polyvinylsilazane as a precursor for micro-structuring. Polym. Adv. Technol. 26 (2015) 245–249 |
| [16] | G. Colucci, S. Mana, L. Conzatti, M. Sangermano. Hybrid organic-inorganic silicate/thiol-ene photocured coatings. Surf. Coat. Technol. 206 (2012) 2719–2724 |
| [17] | M. Sangermano, G. Colucci, M. Fragale, G. Rizza. Hybrid organic-inorganic coatings based on thiol-ene systems. React. Funct. Polym. 69 (2009) 719–723 |
| [18] | Q. Fu, J.H. Liu, W.F. Shi. Preparation and photopolymerization behavior of multifunctional thiol-ene systems based on hyperbranched aliphatic polyesters. Prog. Org. Coat. 63 (2008) 100–109 |
| [19] | J. Heo, T. Kang, S.G. Jang, et al. Improved performance of protected catecholic polysiloxanes for bioinspired wet adhesion to surface oxides. J. Am. Chem. Soc. 134 (2012) 20139–20145 |
| [20] | L. Carson, P.K.W. Chau, M.J. Earle, et al. Antibiofilm activities of 1-alkyl-3-methylimidazolium chloride ionic liquids. Green Chem. 11 (2009) 492–497 |
| [21] | S. Khabnadideh, Z. Rezaei, A. Khalafi-Nezhad, et al. Synthesis of n-alkylated derivatives of imidazole as antibacterial agents. Bioorg. Med. Chem. Lett. 13 (2003) 2863–2865 |
| [22] | H.K. He, B. Adzima, M.Q. Zhong, et al. Multifunctional photo-crosslinked polymeric ionic hydrogel films. Polym. Chem. 5 (2014) 2824–2835 |
| [23] | H. Murata, R.R. Koepsel, K. Matyjaszewski, A.J. Russell. Permanent, non-leaching antibacterial surfaces-2:how high density cationic surfaces kill bacterial cells. Biomaterials 28 (2007) 4870–4879 |
 2016, Vol. 27
2016, Vol. 27 




