b Department of Pharmacy, Fujian University of Traditional Chinese Medicine, Fuzhou 350122, China ;
c Department of Radiology, Shanghai Eastern Hepatobiliary Surgery Hospital, Second Military Medical University, Shanghai 200438, China
Photodynamic therapy (PDT) now is a well recognized approach to cancer therapy for the selective destruction of tumors by visible light in presence of a photosensitizer (PS) and cell oxygen [1]. It is based on the principle that the interaction between light and PS in tumor tissues generates cytotoxic reactive oxygen species (ROS) created through either electron transfer (type Ⅰ) or energy transfer (type Ⅱ) reactions to inactivate the tumor cells [2].
Porfimer sodium, the first clinically approved porphyrin-type PS for the treatment of bladder cancer in the world, has suffered some drawbacks such as its complex component, inefficient absorption (ε = 1170 L mol-1 cm-1) at long wavelength (λmax = 630 nm), prolonged cutaneous phototoxicity up to 4-6 weeks due to its slow clearance in skin tissues [3].
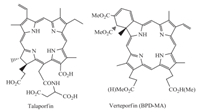
In the recent decade, the so-called second generation chlorintype PSs such as natural chlorophyll-a derivatives have generated interest due to its low skin phototoxicity, rapid clearance from tissues and strong absorption at long wavelengths (λmax > 650 nm) to take full advantage of greater tissue penetration [4, 5, 6]. Among them, talaporfin [7] and verteporfin (BPD-MA) [8] were approved for PDT applications (Fig. 1).
|
Download:
|
| Fig. 1. Two clinical available semisynthetic chlorin-type photosensitizers. | |
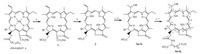
Pyropheophorbid-a (2), the one of chlorophyll-a derivative as chlorin-type PS, has poor water solubility to hamper its clinical development. Introducing amino acid at 173-position and alkoxy at 31-position was reported to individually improve the water solubility and the biological activity of chlorin-based derivatives [9, 10, 11, 12]. In this regard, a series of novel water-soluble amino acid conjugates of pyropheophorbide-a ethers 4a-4j were synthesized (Scheme 1) and investigated their in vitro photodynamic antitumor activity against three kinds of tumor cell lines.
|
Download:
|
| Scheme. 1. Synthetic route for the titled compounds 4a–4j. Reagents and conditions: (a) cond. aqueous HCl–Et2O, 0–5 ℃, 30 min; (b) HOAc, reflux, 4 h; (c) 33% HBr–HOAc, 24 h; (d) alcohol, CH2Cl2, K2CO3, 2 h; Alcohol donors: R = CH3 (3a), n-C3H7 (3b), n-C5H11 (3c), n-C6H13 (3d), n-C8H17 (3e); (e) L-Asp(OBut)2 or L-Glu(OBut)2, EDCI, HOBt, DIPEA, CH2Cl2, r.t., 12 h; (f) CH2Cl2–TFA (1:1), r.t., 2 h. Alcohol donors and amino acid residues: m = 1 and R = CH3 (4a), n-C3H7 (4b), n-C5H11 (4c), n-C6H13 (4d), n-C8H17 (4e); m = 2 and R = CH3 (4f), n-C3H7 (4g), n-C5H11 (4h), n-C6H13 (4i), n-C8H17 (4j). | |
2. Experimental 2.1. Chemicals and instruments
Melting points were measured on a XRD micro melting point apparatus and uncorrected. 1H NMR spectra were recorded on Bruker MSL-300 or MSL-600 using TMS as the internal reference. Mass spectra were collected on an API-3000 LC-MS spectrometer. UV absorption spectra were measured on an Agilent UV 8453 spectrophotometers. Elemental analysis was carried out using a PE2400 Ⅱ instrument. Column chromatography was performed on silica gel (size 10-40 μm, Qingdao Haiyang Chemical, China). All reagents and solvents purchased from commercial vendors and were used without further purifications. The key intermediate pheophorbide-a (1) was obtained via cond. aqueous HCl degradation of chlorophyll-a in Et2O (Scheme 1) by the methodology developed in our laboratory using crude chlorophyll extracts in silkworm excrements [13].
2.2. Chemical synthesisPyropheophorbide-a (2): A suspension of 1 (2.0 g, 3.38 mmol) in HOAc (100 mL) was refluxing under an atmosphere of N2 for 4 h. The reaction mixture was poured into H2O (1 L) and then extracted with CH2Cl2. The organic layer was washed with water, dried over anhydrous Na2SO4, and evaporated. The residue was purified on a silica gel column (CH2Cl2:CH3COCH3:CH3OH:HCO2H = 60:1:1:0.1 as eluent) to obtain 2 (1.5 g, 83.1%) as bright black solid. Physical and spectroscopic characterization data of compound 2 was given in Supporting information.
Pyropheophorbide-a ether derivatives (3a-3e): A suspension of 2 (0.5 g, 0.94 mmol) in 33% HBr-HOAc (50 mL) was stirred overnight at room temperature and then evaporated. The residue was added dissolved in CH2Cl2 (50 mL). K2CO3 (1.0 g) and the appropriate alcohol donors (10 mL) were then added and allowed to stir at room temperature for 2 h. The reaction mixture was added H2O (300 mL) and then extracted with CH2Cl2. The organic layer was washed with water, dried over anhydrous Na2SO4, and evaporated. The residue was purified on a silica gel column (CH2Cl2:CH3COCH3:CH3OH:HCO2H = 80:1:1:0.1 as eluent) to give compounds 3a-3e as bright black solid in yield of 42.3%-69.0%. Physical and spectroscopic characterization data of compounds 3a-3e were given in Supporting Information.
Amino acid conjugates of Pyropheophorbide-a ethers (4a-4j): To a solution of compounds 3a-3e (0.40 mmol) in anhydrous CH2Cl2 (100 mL) was individually added 1-ethyl-3-(3-dimethyla-minopropyl)-carbodiimide hydrochlorate (EDCI) (0.48 mmol, 1.2 equiv.) and 1-hydroxybenzotriazole (HOBt) (0.48 mmol l, 1.2 equiv.) to stir until completely dissolved under in ice-salt bath. After 30 min, L-Asp(OBut)2·HCl or L-Glu(OBut)2·HCl (0.48 mmol 1.2 equiv.) and N, N-diisopropylethylamine (DIPEA) (0.48 mmol l, 1.2 equiv.) were mixed in CH2Cl2 (30 mL) and poured into above reaction mixture. The mixture was allowed to stir at room temperature overnight under nitrogen. It was diluted with CH2Cl2 (150 mL) and then washed with 5% aqueous citric acid, brine and water, respectively. The organic layer was dried over anhydrous Na2SO4 and evaporated. The residue was dissolved in dry CH2Cl2/TFA (1:1, 30 mL) and stirred at room temperature for 2 h. The resulting mixture was diluted with CH2Cl2 and adjusted to pH 5-6 with 10% NaHCO3 and purified on a silica gel column (CH2Cl2:CH3COCH3:CH3OH:HCO2H = 80:1:1:0.1 as eluent) to give the target compounds 4a-4j.
N-(3-Devinyl-3-(1-methoxy)ethylPyropheophorbide-a-173-acyl)-L-aspartic acid (4a): Yield 54.2%, mp 168-169 ℃; 1H NMR (300 MHz, DMSO-d6): δ 12.10 (s, 2H, 2× CO2H), 9.65 (s, 1H, 10-H), 9.38 (s, 1H, 5-H), 8.88 (s, 1H, 20-H), 8.20 (m, 1H, CONH), 6.02 (q, 1H, J = 6.8 Hz, 31-H), 5.22 (d, 1H, J = 21.0 Hz, 132-Hb), 5.10 (d, 1H, J = 21.0 Hz, 132-Ha), 4.56-4.59 (m, 1H, 17-H), 4.28-4.32 (m, 1H, 18- H), 3.82 (m, 1H, CONHCHCO), 3.69 (q, 2H, J = 7.2 Hz, 81-CH2), 3.62 (s, 3H, 12-CH3), 3.42 (s, 3H, 31-OCH3), 3.35 (s, 3H, 7-CH3), 3.17 (s, 3H, 2-CH3), 2.22-2.03 (m, 6H, 172-CH2 + 171-CH2 + CONHCHCH2), 2.02 (d, 3H, J = 6.8 Hz, 32-CH3), 1.76 (d, 3H, J = 7.2 Hz, 18-CH3), 1.59 (t, 3H, J = 7.2 Hz, 82-CH3), -2.04 (s, 1H, NH); MS (ESI+) m/z: 682.57 [M+H]+ (100%); Anal. Calcd. for C38H43N5O7:C 66.94, H 6.36, N 10.27; Found: C 67.14, H 6.31, N 10.33.
N-(3-Devinyl-3-(1-propoxy)ethylPyropheophorbide-a-173-acyl)-L-aspartic acid (4b): Yield 55.8%, mp > 300 ℃; 1H NMR (300 MHz, DMSO-d6): δ 9.80 (1H, splitted s, 10-H), 9.76 (s, 1H, 5-H), 8.83 (s, 1H, 20-H), 7.95 (m, 1H, CONH), 6.0 (q, 1H, J = 6.8 Hz, 31-H), 5.23 (d, 1H, J = 21.0 Hz, 132-Hb), 5.09 (d, 1H, J = 21.0 Hz, 132- Ha), 4.51-4.58 (m, 1H, 17-H), 4.28 (m, 1H, 18-H), 3.74 (m, 1H, CONHCHCO), 3.70 (q, 2H, J = 7.3 Hz, 81-CH2), 3.63 (s, 3H, 12-CH3), 3.49 (m, 2H, 31-OCH2), 3.39 (s, 3H, 7-CH3), 3.22 (s, 3H, 2-CH3), 2.12-2.30 (m, 6H, 172-CH2 + 171-CH2 + CONHCHCH2), 2.02 (d, 3H, J = 6.8 Hz, 32-CH3), 1.76 (d, 3H, J = 7.6 Hz, 18-CH3), 1.63 (t, 3H, J = 7.3 Hz, 82-CH3), 0.82-0.93 (m, 5H, 31-OCH2CH2CH3), -1.98 (s, 1H, NH); MS (ESI+) m/z: 710.56 [M+H]+ (100%); Anal. Calcd. for C40H47N5O7: C 67.68, H 6.67, N 9.87; Found: C 67.89, H 6.61, N 9.92.
N-(3-Devinyl-3-(1-pentyloxy)ethylPyropheophorbide-a-173- acyl)-L-aspartic acid (4c): Yield 60.1%, mp > 300 ℃; 1H NMR (300 MHz, DMSO-d6): δ 9.80 (splitted s, 1H, 10-H), 9.75 (s, 1H, 5-H), 8.83 (s, 1H, 20-H), 7.95 (m, 1H, CONH), 5.98 (q, 1H, J = 6.8 Hz, 31-H), 5.23 (d, 1H, J = 18.0 Hz, 132-Hb), 5.09 (d, 1H, J = 18.0 Hz, 132-Ha), 4.59-4.56 (m, 1H, 17-H), 4.31-4.28 (m, 1H, 18-H), 3.74 (m, 1H, CONHCHCO), 3.71 (q, 2H, J = 7.7 Hz, 81-CH2), 3.62 (s, 3H, 12-CH3), 3.46-3.51 (m, 2H, 31-OCH2), 3.38 (s, 3H, 7-CH3), 3.22 (s, 3H, 2-CH3), 2.33-2.12 (m, 6H, 172-CH2 + 171-CH2 + CONHCHCH2), 2.02 (d, 3H, J = 6.8 Hz, 32-CH3), 1.76 (d, 3H, J = 6.6 Hz, 18-CH3), 1.63 (t, 3H, J = 7.7 Hz, 82-CH3), 1.25 (m, 4H, 31-OCH2(CH2)2C2H5), 0.72-0.86 (m, 5H, O(CH2)3CH2CH3), -1.98 (s, 1H, NH); MS (ESI+) m/z: 738.51 [M+H]+ (100%); Anal. Calcd. for C42H51N5O7:C 68.36, H 6.97, N 9.49; Found: C 68.58, H 6.92, N 9.56.
N-(3-Devinyl-3-(1-hexyloxy)ethylPyropheophorbide-a-173- acyl)-L-aspartic acid (4d): Yield 51.6%, mp > 300 ℃; 1H NMR (300 MHz, DMSO-d6): δ 9.80 (s, 1H, 10-H), 9.75 (s, 1H, 5-H), 8.81 (s, 1H, 20-H), 8.05 (m, 1H, CONH), 6.21 (q, 1H, J = 6.8 Hz, 31-H), 5.20 (d, 1H, J = 18.0 Hz, 132-Hb), 5.05 (d, 1H, J = 18.0 Hz, 132-Ha), 4.55 (m, 1H, 17-H), 4.26 (m, 1H, 18-H), 4.12 (m, 1H, CONHCHCO), 3.71 (q, 2H, J = 7.5 Hz, 81-CH2), 3.62 (s, 3H, 12-CH3), 3.49 (m, 2H, 31-OCH2), 3.39 (s, 3H, 7-CH3), 3.22 (s, 3H, 2-CH3), 2.12-2.33 (m, 6H, 172- CH2 + 171-CH2 + CH2 + CONHCHCH2), 2.02 (d, 3H, J = 6.8 Hz, 32-CH3), 1.76 (d, 3H, J = 6.6 Hz 18-CH3), 1.63 (t, 3H, J = 7.5 Hz, 82-CH3), 1.27 (m, 6H, 31-OCH2(CH2)3C2H5), 0.80-0.90 (m, 5H, 31-O(CH2)4CH2CH3); MS (ESI+) m/z: 752.53 [M+H]+ (100%). Anal. Calcd. for C43H53N5O7:C 68.69, H 7.10, N 9.31; Found: C 68.89, H 7.05, N 9.38.
N-(3-Devinyl-3-(1-octyloxy)ethylPyropheophorbide-a-173-acyl)-L-aspartic acid (4e): Yield 53.0%, mp > 300 ℃; 1H NMR (300 MHz, (CD3)2CO): δ 9.87 (s, 1H, 10-H), 9.71 (s, 1H, 5-H), 8.78 (s, 1H, 20-H), 7.45 (m, 1H, CONH), 6.02 (q, 1H, J = 6.8 Hz, 31-H), 5.21 (d, 1H, J = 18.0 Hz, 132-Hb), 5.04 (d, 1H, J = 18.0 Hz, 132-Ha), 4.77 (m, 1H, 17-H), 4.60 (m, 1H, 18-H), 4.38 (m, 1H, CONHCHCO), 3.69 (q, 2H, J = 7.5 Hz 81-CH2), 3.58 (s, 3H, 12-CH3), 3.55 (m, 2H, 31-OCH2), 3.39 (s, 3H, 7-CH3), 3.22 (s, 3H, 2-CH3), 2.31-2.20 (m, 6H, 172-CH2 + 171-CH2 + CH2 + CONHCHCH2CO2H), 2.06 (d, 3H, J = 6.8 Hz, 32-CH3), 1.78 (d, 3H, J = 6.6 Hz, 18-CH3), 1.64 (t, 3H, J = 7.5 Hz, 82- CH3), 1.20 (m, 4H, 31-OCH2(CH2)2C5H11), 0.81-0.82 (m, 8H, 31- O(CH2)3(CH2)4CH3), 0.63 (t, 3H, J = 7.5 Hz, 31-O(CH2)7CH3), -1.84 (s, 2H, NH × 2); MS (ESI+) m/z: 780.62 [M+H]+ (100%). Anal. Calcd. for C45H57N5O7:C 69.30, H 7.37, N 8.98; Found: C 69.49, H 7.29, N 9.05.
N-(3-Devinyl-3-(1-methoxy)ethylPyropheophorbide-a-173-acyl)-L-glutamic acid (4f): Yield 73.3%, mp > 300 ℃; 1H NMR (300 MHz, DMSO-d6): δ 9.70 (s, 2H, 10- and 5-H), 8.85 (s, 1H, 20-H), 8.15 (m, 1H, CONH), 5.95 (q, 1H, J = 6.6 Hz, 31-H), 5.20 (d, 1H, J = 18.0 Hz, 132-Hb), 5.07 (d, 1H, J = 18.0 Hz, 132-Ha), 4.56 (m, 1H, 17- H), 4.30 (m, 1H, 18-H), 4.10 (m, 1H, CONHCHCO), 3.68 (q, 2H, J = 7.4 Hz, 81-CH2), 3.58(s, 3H, 12-CH3), 3.47 (s, 3H, 31-OCH3), 3.40(s, 3H, 7-CH3), 3.21 (s, 3H, 2-CH3), 2.25-2.03 (m, 6H, 172-CH2 + 171- CH2 + CONHCH(CO2H)CH2CH2CO2H), 2.02(d, 3H, J = 6.6 Hz, 32-CH3), 1.77 (d, 3H, J = 6.9 Hz, 18-CH3), 1.61 (t, 3H, J = 7.4 Hz, 82-CH3), 1.20-1.40 (m, 2H, CONHCH(CO2H)CH2CH2CO2H), -1.99 (s, 1H, NH); MS (ESI+) m/z: 696.55 [M+H]+ (100%). Anal. Calcd. for C39H45N5O7:C 67.32, H 6.52, N 10.07; Found: C 67.51, H 6.48, N 10.12.
N-(3-Devinyl-3-(1-propoxy)ethylPyropheophorbide-a-173-acyl)-L-glutamic acid (4g): Yield 59.5%, mp > 300 ℃; 1H NMR (300 MHz, DMSO-d6): δ 12.38 (s, 2H, 2× CO2H), 9.80 (splitted s, 1H, 10-H), 9.74 (s, 1H, 5-H), 8.84 (s, 1H, 20-H), 8.16 (m, 1H, CONH), 6.0 (q, 1H, J = 6.3 Hz, 31-H), 5.21 (d, 1H, J = 18.0 Hz, 132-Hb), 5.09 (d, 1H, J = 18.0 Hz, 132-Ha), 4.55 (m, 1H, 17-CH), 4.31 (m, 1H, 18-CH), 4.20 (m, 1H, CONHCHCO), 3.71 (q, 2H, J = 7.7 Hz, 81-CH2), 3.62 (s, 3H, 12-CH3), 3. 49 (m, 2H, 31-OCH2), 3.43 (s, 3H, 7-CH3), 3.21 (s, 3H, 2-CH3), 2.25-2.07 (m, 6H, 172-CH2 + 171-CH2 + CONHCH (CO2H)CH2CH2CO2H), 2.02 (d, 3H, J = 6.3 Hz, 32-CH3), 1.76 (d, 3H, J = 6.8 Hz, 18-CH3), 1.63 (t, 3H, J = 7.7 Hz, 82-CH3), 1.13-1.38 (m, 4H, CONHCH(CO2H)CH2CH2CO2H + 31-OCH2CH2CH3), 0.90 (t, 3H, J = 7.5 Hz, 31-O(CH2)2CH3), -1.98 (s, 1H, NH); MS (ESI+) m/z: 724.54 [M+H]+ (40%); MS (ESI-) m/z: 722.55 [M-H]+ (70%), 1445.46 [2M-H]+ (100%). Anal. Calcd. for C41H49N5O7:C 68.03, H 6.82, N 9.68; Found: C 68.23, H 6.77, N 9.73.
N-(3-Devinyl-3-(1-pentyloxy)ethylPyropheophorbide-a-173- acyl)-L-glutamic acid (4h): Yield 53.5%, mp 180-182 ℃; 1H NMR (300 MHz, DMSO-d6): δ 12.53 (s, 2H, 2× CO2H), 9.81 (s, 1H, 10-H), 9.75 (s, 1H, 5-H), 8.84 (s, 1H, 20-H), 8.10 (m, 1H, CONH), 6.00 (q, 1H, J = 6.0 Hz, 31-H), 5.22 (d, 1H, J = 18.0 Hz, 132-Hb), 5.09 (d, 1H, J = 18.0 Hz, 132-Ha), 4.56 (m, 1H, 17-H), 4.32 (m, 1H, 18-H), 4.18-4.20 (m, 1H, CONHCHCO), 3.71 (q, 2H, J = 8.3 Hz, 81-CH2), 3.62 (s, 3H, 12-CH3), 3.48-3.50 (m, 2H, 31-OCH2), 3.39 (s, 3H, 7- CH3), 3.21 (s, 3H, 2-CH3), 2.33-2.10 (m, 6H, 172-CH2 + 171- CH2 + CH2CO2H), 2.01 (d, 3H, J = 6.0 Hz, 32-CH3), 1.76 (d, 3H, J = 6.8 Hz, 18-CH3), 1.63 (t, 3H, J = 8.3 Hz, 82-CH3), 1.20 (m, 8H, CONHCH(CO2H)CH2CH2CO2H + 31-OCH2(CH2)3CH3), 0.90 (t, 3H, J = 6.8 Hz, O(CH2)4CH3), -1.98 (s, 1H, NH); MS (ESI+) m/z: 752.99 [M+H]+ (100%). Anal. Calcd. for C43H53N5O7:C 68.69, H 7.10, N 9.31; Found: C 69.81, H 7.05, N 9.37.
N-(3-Devinyl-3-(1-hexyloxy)ethylPyropheophorbide-a-173- acyl)-L-glutamic acid (4i): Yield 69.3%, mp 184-185 ℃; 1H NMR (300 MHz, DMSO-d6): δ 12.29 (s, 2H, 2× CO2H), 9.79 (s, 1H, 10-H), 9.71 (s, 1H, 5-H), 8.83 (splitted s, 1H, 20-H), 8.14 (m, 1H, CONH), 5.95 (q, 1H, J = 5.7 Hz, 31-H), 5.21 (d, 1H, J = 18.0 Hz, 132-Hb), 5.09 (d, 1H, J = 18.0 Hz, 132-Ha), 4.56 (m, 1H, 17-H), 4.32 (m, 1H, 18-H), 4.22 (m, 1H, CONHCHCO), 3.69 (q, 2H, J = 7.3 Hz, 81-CH2), 3.60 (s, 3H, 12-CH3), 3.42 (m, 2H, 31-OCH2), 3.35 (s, 3H, 7-CH3), 3.20 (s, 3H, 2-CH3), 2.23-2.11 (m, 6H, 172-CH2 + 171-CH2 + CH2CO2H), 2.02 (d, 3H, J = 5.7 Hz, 32-CH3), 1.76 (d, 3H, J = 6.5 Hz, 18-CH3), 1.61 (t, 3H, J = 7.3 Hz, 82-CH3), 1.11-1.28 (m, 10H, CONHCH(CO2H)CH2CH2- CO2H + 31-OCH2(CH2)4CH3), 0.81 (t, 3H, J = 5.4 Hz, 31-O(CH2)5CH3), -1.97 (1H, s, NH); MS (ESI+) m/z: 766.26 [M+H]+ (100%). Anal. Calcd. for C44H55N5O7:C 69.00, H 7.24, N 9.14; Found: C 69.23, H 7.17, N 9.20.
N-(3-Devinyl-3-(1-octyloxy)ethylPyropheophorbide-a-173-acyl)-L-glutamic acid (4j): Yield 79.3%, mp 180-182 ℃; 1H NMR (300 MHz, DMSO-d6): δ 12.58 (s, 2H, 2× CO2H), 9.79 (s, 1H, 10-H), 9.72 (s, 1H, 5-H), 8.82 (s, 1H, 20-H), 7.90 (m, 1H, CONH), 5.95 (q, 1H, J = 6.8 Hz, 31-H), 5.21 (d, 1H, J = 18.0 Hz, 132-Hb), 5.08 (d, 1H, J = 18.0 Hz, 132-Ha), 4.56 (m, 1H, 17-H), 4.30 (m, 1H, 18-H), 4.16 (m, 1H, CONHCHCO), 3.69 (q, 2H, J = 7.8 Hz, 81-CH2), 3.60 (s, 3H, 12-CH3), 3.45 (m, 2H, 31-OCH2), 3.35 (s, 3H, 7-CH3), 3.20 (s, 3H, 2-CH3), 2.20-2.13 (m, 6H, 172-CH2 + 171-CH2 + CH2CO2H), 2.02 (d, 3H, J = 6.8 Hz, 32-CH3), 1.75 (d, 3H, J = 6.8 Hz, 18-CH3), 1.62 (t, 3H, J = 7.8 Hz, 82-CH3), 0.82-1.37 (m, 17H, CONHCH(CO2H)CH2CH2- CO2H + 31-OCH2(CH2)6CH3), -1.98 (s, 1H, NH); MS (ESI-) m/z: 792.63 [M-H]+ (100%). Anal. Calcd. for C46H59N5O7:C 69.58, H 7.49, N 8.82; Found: C 69.76, H 7.41, N 8.90.
The determination method of in vitro dark toxicity and phototoxicity for target compounds 4a-4j was given in Supporting information.
3. Results and discussion 3.1. SynthesisIt was designed that Pyropheophorbide-a ethers (3a-3e), which was generated via the addition of Pyropheophorbide-a (2) with hydrobromic acid followed by substitution with alcohol donors (ROH), coupled with carboxyl-protected amino acids in the presence of condensation agent and catalyst followed by removal of protective group with trifluoroacetic acid (TFA) to yield the target compounds (4a-4j).
Compound 2 was prepared by acid degradation of pheophorbide-a (1) in refluxing HOAc. Compound 1 was synthesized according to our reported procedure [13]. All of the target amino acid conjugates of Pyropheophorbide-a ethers 4a-4j were new compounds and their structures were confirmed by 1H NMR, MS and elemental analysis. The UV-vis spectral data showed that they possessed more efficient absorption (ε = 1.7 × 104 to 9.8 × 104 L mol-1 cm-1) at longer maximum absorption wavelength ranged from 651 nm to 666 nm than porfimer sodium (Table 1), indicating their greater tissue penetration [4, 5, 6].
|
|
Table 1 UV–vis data of the titled compounds 4a–4j. |
3.2. In vitro dark cytotoxicity and photodynamic efficacy
The dark toxicity and phototoxicity of the titled compounds were screened with the MTT assay against HCT116, MDA-MB-231 and MKN45 cells using BPD-MA as control (Table 2). In order to realize parallel control to eliminate the experimental error caused by the solvent, the tested compounds 4a-4j and BPD-MA were both prepared into the DMSO-mediated 10% PBS solution. The results displayed that all compounds exhibited better phototoxicity and less dark toxicity against three kinds of tumor cell lines than BPD-MA. A structure-activity relationship analysis revealed that their PDT antitumor activity, particularly against HCT116 cells, enhanced with ether carbon chain growth, the one with 6-8 Carbon atoms as the best. In addition, the type of water-soluble amino acid such as L-aspartic acid or L-glutamic acid had little effect on the activity. Among them, compound 4d and 4j exhibited the best in vitro PDT antitumor efficacy and their IC50 values for HCT116 cell were individually 41 nmol/L and 33 nmol/L, which represented 7.8- and 9.7-fold increase of antitumor potency compared to BPD-MA, respectively. In the next in vivo animal antitumor trials, they could be made into water-soluble sodium salt for intravenous administration.
|
|
Table 2 Cytotoxicity (IC50, μmol/L) for the titled compounds against tumor cells. |
4. Conclusion
In summary, 10 new water-soluble amino acid conjugates of Pyropheophorbide-a ethers (4a-4j) were synthesized and evaluated for their in vitro photodynamic antitumor activity against HCT116, MDA-MB-231 and MKN45 cells. All compounds exhibited more potent phototoxicity and lower dark toxicity against all tested tumor cell lines than BPD-MA. In particular, compounds 4d and 4j were the two most effective PSs, which showed 7.8- and 9.7- fold more potent antitumor activity against HCT116 cells compared to BPD-MA. These results demonstrated that compounds 4d and 4j could be promising PSs for PDT applications because of their strong absorption at long wavelength, high phototoxicity, low dark cytotoxicity and good water-solubility, and worthy of further study.
Appendix A. Supplementary dataSupplementary data associated with this article can be found, in the online version, at http://dx.doi.org/10.1016/j.cclet.2016.03.019.
| [1] | P. Agostinis, K. Berg, K.A. Cengel, et al. Photodynamic therapy of cancer:an update. Cancer J. Clin. 61 (2011) 250–281 |
| [2] | R.W. Redmond, I.E. Kochevar. Symposium-in-print:singlet oxygen invited review. Photochem. Photobiol. 82 (2006) 1178–1186 |
| [3] | N. Drogat, C. Gady, R. Granet, V. Sol. Design and synthesis of water-soluble polyaminated chlorins and bacteriochlorins-with near-infrared absorption. Dyes Pigments 98 (2013) 609–614 |
| [4] | K.T. Oliveira, P.B. Momo, F.F. Assis, et al. Chlorins:natural sources, synthetic developments and main applications. Curr. Org. Synth. 11 (2014) 42–58 |
| [5] | J.Z. Yao, W.N. Zhang, C.Q. Sheng, et al. Design, synthesis, and in vitro photodynamic activities of benzochloroporphyrin derivatives as tumor photosensitizers. Bioorg. Med. Chem. Lett. 18 (2008) 293–297 |
| [6] | J.L. Zhang, L. Deng, J.Z. Yao, et al. Synthesis and photobiological study of novel chlorin photosensitizer BCPD-18Ma for photodynamic therapy. Bioorg. Med. Chem. 19 (2011) 5520–5528 |
| [7] | D. Kessel. Determinants of photosensitization by mono-L-aspartylchlorin e6. Photochem. Photobiol. 49 (2008) 447–452 |
| [8] | R.R. Allison, G.H. Downie, R. Cuenca, et al. Photosensitizers in clinical PDT. Photodiagn. Photodyn. 1 (2004) 27–42 |
| [9] | T. Hitoshi, I. Yasuaki, T. Takuya, et al. Synthesis of chlorophyll-amino acid conjugates as models for modification of proteins with chromo/fluorophores. Bioorg. Med. Chem. 22 (2014) 1421–1428 |
| [10] | W.G. Roberts, F.Y. Shiau, J.S. Nelson, et al. In vitro characterization of monoaspartyl chlorin e6 and diaspartyl chlorin e6 for photodynamic therapy. J. Natl. Cancer Inst. 80 (1998) 330–336 |
| [11] | J.Z. Yao, W.D. Shen, J.F. Liu, et al. Photochemotherapeutic effects of 2-(1-hydroxy1) ethyl chlorin f and its ether derivatives on sarcoma 180 transplanted in mice. Chin. J. Laser Med. Surg 9 (2001) |
| [12] | J.Z. Yao, W.H. Chen, X. He, et al. Synthesis and photosensitizing abilities as tumorphotobiological activities of chlorine f methyl ether. Acta Pharm. Sin. 35 (2000) 63–66 |
| [13] | J.Z. Yao, X. He, W.D. Shen, et al. Synthesis of tumor-photobiological activity of chlorine f. Chin. Pharm. J. 34 (1999) 846–848 |
 2016, Vol. 27
2016, Vol. 27 




