Methyldopa,a catechol derivative,is widely used as an antihypertensive agent. It is a centrally acting α2-adrenoreceptor agonist that reduces sympathetic tone and produces a fall in blood pressure [1]. The quantification of methyldopa is of importance for both pharmaceutical and clinical reasons [1, 2, 3]. A number of techniques have been reported for the quantification of methyldopa including chromatography [4, 5],kinetic methods [3, 6],spectrophotometry [1, 7],1H NMR [8] and electrochemical methods [9, 10, 11]. Spectrophotometry and chromatography are the two most widely utilized techniques for the quantification of methyldopa. However,they require a time-consuming derivatization step and selectivity makes these techniques unsuitable for a routine quantification. On the other hand,voltammetry enables high sensitivity and rapidness to the analysis of a wide range of drugs [12, 13, 14, 15, 16]. However,the selectivity of voltammetry is sometimes restricted due to the interference from the other redox active molecules [17, 18, 19, 20]. For example,molecules such as AA and UA display oxidation behaviour at potentials close to those required for the oxidation of the hypertensive drug,methyldopa. An important strategy to overcome such problems is to modify the electrodes by chemically modifying the electrodes [21, 22, 23, 24, 25, 26]. Carbon nanotubes (CNTs) are attractive modifying materials for the fabrication of nanosensors because of their capability to provide strong catalytic activity and minimize surface fouling [27]. The modification of electrodes with CNTs for use in analytical sensing has also been documented to result in low detection limits,high sensitivities and reduction of overpotentials [28].
In this paper,we report experiments to confirm the high electrocatalytic activity of a glassy carbon electrode modified with MWCNTs for the detection of methyldopa in the presence of AA and UA. The experimental results showed that the surface modification of electrodes with MWCNTs could shift the voltammetric response of methyldopa in analytically useful manners and provide lower detection limits.
2. Experimental 2.1. Chemical reagentsMethyldopa,AA and UA obtained from Sigma-Aldrich were used as received. Chloroform was obtained from Merck. MWCNTs were purchased from NanoLab,USA. Methyldopa solutions were prepared using 0.1 mol/L phosphate buffer at pH 4.0. Ultra pure water was used for the preparation of the solutions. Oxygen-free nitrogen was bubbled through the cells prior to each experiment. All experiments were carried out at room temperature.
2.2. InstrumentationAn EcoChemie Autolab PGSTAT 12 potentiostat/galvanostat (The Netherlands) with the electrochemical software package 4.9 was used for the voltammetric experiments. A three-electrode system was used: a GCE as working electrode (WE) [3 mm in diameter (Bioanalytical Systems,Lafayette,USA)],a Pt wire as counter electrode (CE) and an Ag/AgCl as reference electrode (RE) (Metrohm,Switzerland). Prior to the modification with MWCNTs,the GCE was polished with 1 μm and 0.3 μm alumina suspensions. The electrode was then sonicated for 5 min in ethanol. All experiments were carried out at room temperature.
2.3. Preparation of the modified electrode1 μg of MWCNTs was dispersed in 5 mL of chloroform. Then it was placed into an ultrasonic bath for 10 min,after which an aliquot of known volume was cast onto the electrode surface and the solvent was allowed to be evaporated.
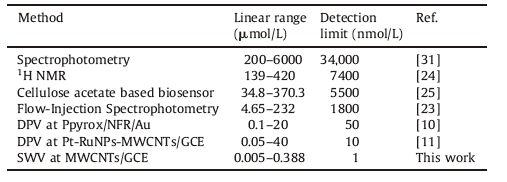
3. Results and discussion 3.1. Cyclic voltammetry of methyldopaFig. 1a shows a cyclic voltammogram of 4.5 μmol/L methyldopa recorded at a bare glassy carbon electrode. Methyldopa exhibits an oxidation peak at 676 mV and a corresponding reduction peak at 170 mV. The separation between the anodic and cathodic peak potentials (ΔEp) was 506 mV. Fig. 1b shows the voltammograms of methyldopa at various scan rates. The peak currents were proportional to the square root of the scan rates,suggesting that the voltammetric response of methyldopa was controlled by diffusion. However,methyldopa shows an oxidation peak at 425 mVand a reduction peak at 403 mV. The separation of the peak potential (ΔEp) was 22 mV at a GCE modified with MWCNTs as given in Fig. 2a. Methyldopa undergoes a reversible two-electron oxidation at MWCNTs/GCE. It is clearly seen that the voltammetric response of methyldopa was improved at GCE modified with MWCNTs. Cyclic voltammograms of methyldopa at a GCE modified with MWCNTs at different scan rates are shown in Fig. 2b. The peak currents were directly proportional to the scan rates for an electrode modified with MWCNTs. It was shown that the response of anodic peak current of methyldopa has changed from a square root to a linear dependence on scan rate. This has been attributed to the transition from planar diffusion to a thin layer character as the signals were seen immediately on exposure of the MWCNTs/ GCE to solution,and changed only slightly over a period of ca. 10 min [29].

|
Download:
|
| Fig. 1.(a) A cyclic voltammogram of 4.5 μmol/L methyldopa at bare GCE in 0.1 mol/L PBS at pH: 4.0,scan rate: 50 mV/s,and (b) cyclic voltammograms of 4.5 μmol/L methyldopa at bare GCE in 0.1 mol/L PBS at pH: 4.0. Scan rates: from 25 mV/s to 125 mV/s. | |

|
Download:
|
| Fig. 2.(a) A cyclic voltommogram of 4.5 μmol/L methyldopa at a GCE modified with 0.15 μg MWCNT in 0.1 mol/L PBS at pH: 4.0,scan rate: 50 mV/s,and (b) cyclic voltommograms of 4.5 μmol/L methyldopa at a GCE modified with 0.15 μg in 0.1 mol/L PBS at pH: 4.0. Scan rates: from 10 mV/s to 100 mV/s. | |
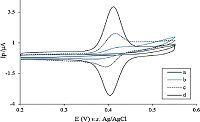
The effect of the surface film on the electrode surface was examined. Fig. 3a exhibits the cyclic voltammograms of methyldopa at a GCE modified with various amounts of MWCNTs. The separation of anodic and cathodic peak potentials for the redox couple of methyldopa was reduced and the cyclic voltammetric behaviour of methyldopa was seen to be more reversible with the increase of the thickness of MWCNTs. The experimental data also showed that an increase of thickness on the surface had promoted the catalytic activity towards the oxidation of methyldopa.
|
Download:
|
| Fig. 3.Cyclic voltammograms of 10 nmol/L methyldopa at a GC electrode modified with 0.01 μg (a),0.05 μg (b),0.10 μg (c) and 0.15 μg (d) MWCNTs in 0.1 mol/L PBS at pH: 4.0. Scan rate: 50 mV/s. | |
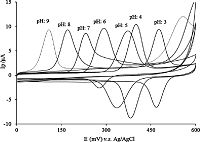
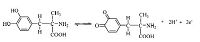
The effect of pH on anodic peak potential of methyldopa using GCEs modified with 0.15 μg MWCNTs is shown in Fig. 4. The results showed that the oxidation peak potential shifted towards negative potential with increased pH values,indicating that the oxidation of methyldopa includes transfer of protons. The slope of anodic peak potential (Epa) vs. pH is 59.9 mV/pH. This indicated that the proportion of electrons and protons involved in the reaction is 1:1. Since the number of electrons and protons are equal in the electrode reaction,the number of protons involved in the whole electrode reaction is 2. The proposed electrode reaction of methyldopa at MWCNTs/GCE is given in Scheme 1.
|
Download:
|
| Fig. 4.Cyclic voltammograms of methyldopa at MWCNTs/GCE in 0.1 mol/L PBS at various pH values. Scan rate: 50 mV/s. | |
|
Download:
|
| Scheme 1.The electrode reaction of methyldopa at MWCNTs/GCE. | |
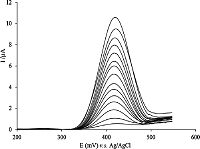
Fig. 5 shows the square wave voltammetric determination of methyldopa at MWCNTs/GCE. The peak currents were linear with the concentrations of methyldopa over two intervals in the range of 0.005-0.068 μmol/L and 0.068-0.388 μmol/L. The first linear regression equation was Ipa(mA) = 6.5201 + 9.0041C μmol/L with a correlation coefficient of 0.9970. The second was Ipa(mA) = 0.1165 + 109.0807C μmol/L with a correlation coefficient of 0.9989. The detection limit was 1.0 nmol/L (S/N = 3). Also,Table 1 shows a comparison of the analytical parameters obtained by the proposedmethodwith severalmethods for the determination ofmethyldopa. As shown in Table 1,a lower concentration range and the lowest detection limit for methyldopa were obtained by the proposed method. The parameters obtained in this study clearly indicate that the proposed electrode is more sensitive for the determination of methyldopa.
|
Download:
|
| Fig. 5.SWVs of various concentrations of methyldopa at MWCNTs/GCE in 0.1 mol/L PBS at pH 4.0. Methyldopa concentrations: 0.002 μmol/L; 0.004 μmol/L; 0.012 μmol/L; 0.016 μmol/L; 0.022 μmol/L; 0.028 μmol/L; 0.036 μmol/L; 0.044 μmol/L; 0.054 μmol/L; 0.068 μmol/L; 0.086 μmol/L; 0.110 μmol/L; 0.188 μmol/L; 0.228 μmol/L; 0.388 μmol/L. Frequency: 20 Hz. Step potential: 100 mV/s. Amplitude: 50 mV/s. | |
|
|
Table 1 The application results of various methods for the determination of methyldopa. |
The relative standard deviation (RSD) of 10 successive scans was 2.3% for 10 nmol/L methyldopa. This indicated that the reproducibility of the electrode was good. Also,the results obtained are comparable with the results obtained from a cellulose acetate modified ionic liquid sensor with an RSD of 4.3% [9],a DPV at Ppyrox/NFR/Au with an RSD of 2.6% [10] and a DPV at Pt-RuNPs- MWCNTs/GCE with an RSD of 3.05% [11]. Also,20 cycles of scanning in 0.1 mol/L PBS in the potential range 0.0-0.8 V could regenerate clean background CV curves for the next study. The peak current of methyldopa has decreased by 5% over 15 days of storage in 0.1 mol/L PBS.
3.5. Detection of methyldopa in the presence of AA and UAVoltammetry provides a sensitive approach to the quantification of many drugs [12, 13, 14]. However,the interference from the other electrochemically active molecules can restrict this approach as shown in Fig. 6a where only one broad oxidation peak is observed for a mixture of AA,methyldopa and UA at a bare GCE. Fig. 6b exhibits the cyclic voltammograms of increasing concentrations of methyldopa in the presence of both AA and UA. The current response increased linearly with the concentration of methyldopa,indicating that AA and UA did not interfere with its determination. It is clearly demonstrated that the modification of a GCE with MWCNTs improved peak separation from interfering compounds such as AA and UA,and thus enabled the detection of methyldopa in the presence of AA and UA.

|
Download:
|
| Fig. 6.(a) Cyclic voltammogram of the mixture 50 μmol/L AA,0.05 μmol/L methyldopa and 50 μmol/L UA at bare GCE,and (b) cyclic voltammograms of the mixture of 50 μmol/L AA,50 μmol/L UA and increasing concentrations of methyldopa at MWCNTs/GCE. Methyldopa concentrations: 0.05 μmol/L; 0.1 μmol/L; 0.15 μmol/L; 0.2 μmol/L; 0.25 μmol/L; 0.3 μmol/L; 0.35 μmol/L. Supporting electrolyte: 0.1 mol/L PBS at pH 4.0. Scan rate: 50 mV/s. | |
Square wave voltammetric (SWV) determination of methyldopa in tablets was referred to the regression equation. The analysis of tablets using the proposed procedure is summarized in Table 2. A mean recovery of 98.2% with an RSD of 1.9% was obtained by the proposed electrode. The results of the drug analysis obtained from the proposed method were in close agreement with the claimed value. The results obtained by the proposed method were also comparable with the standard USP method with a mean recovery of 101.7% [30]. Also,the results obtained were comparable with the results obtained from a DPV at Pt-RuNPs-MWCNTs/GCE with a recovery of 95.5% to 105.5% [11],a cellulose acetate modified ionic liquid sensor with a recovery of 94.1% to 105.9% [9] and a spectrophotometric method with a recovery of 98.8% [31]. However,the results obtained from the proposed method indicated that this method could be used for the determination of methyldopa in drug samples.
|
|
Table 2 The analysis of methyldopa tablets using the proposed procedure. |
The oxidation of methyldopa exhibited a quasi-reversible response on a bare electrode. It was clearly shown that the voltammetric response of methyldopa was improved at MWCNTs/ GCE. Also,the separation in the anodic and cathodic peak potentials for the redox couple of methyldopa was reduced and the voltammetric behaviour of methyldopa was seen to be more reversible with the increase of the amount of MWCNTs on the electrode surface. The results showed that the peak currents were proportional to the concentrations of methyldopa over two intervals in the range of 0.005-0.068 μmol/L and 0.068- 0.388 μmol/L with a detection limit of 1.0 nmol/L. It was shown that the proposed procedure improved peak separation from interfering compounds such as AA and UA,reduced the potential required to oxidizemethyldopa and reduced the detection limit. The results indicate that the proposed method is accurate and can be applied for the determination of methyldopa in pharmaceuticals.
AcknowledgmentThe authors greatly appreciate the financial support from the University of Harran (HÜ BAK).
| [1] | P.R.S. Ribeiro, J.A. Gomes Neto, L. Pezza, H.R. Pezza, Flow-injection spectrophotometric determination of methyldopa in pharmaceutical formulations, Talanta 67(2005) 240-244. |
| [2] | E.A. Gadkariem, K.E.E. Ibrahim, N.A.A. Kamil, M.E.M. Haga, H.A. El-Obeid, A new spectrophotometric method for the determination of methyldopa, Saudi Pharm. J. 17(2009) 289-293. |
| [3] | M. Tubino, D.C.D.V. Batista, J.A.R. Rodrigues, Kinetic method for the determination of alpha-methyldopa in pharmaceutical preparations:analytical procedure and reaction mechanism considerations, Anal. Lett. 39(2006) 327-339. |
| [4] | S. Li, H. Wu, Y. Yu, et al., Quantitative analysis of levodopa, carbidopa and methyldopa in human plasma samples using HPLC-DAD combined with second-order calibration based on alternating trilinear decomposition algorithm, Talanta 81(2010) 805-812. |
| [5] | C. Muzzi, E. Bertocci, L. Terzuoli, et al., Simultaneous determination of serum concentrations of levodopa, dopamine, 3-O-methyldopa and alpha-methyldopa by HPLC, Biomed. Pharmacother. 62(2008) 253-258. |
| [6] | T.S. James, C.L.O. Hoo, D.M. William, Determination of methyldopa in pharmaceutical dosage forms and biological fluids based on oxidation at the tubular carbon electrode, J. Pharm. Sci. 63(2006) 954-955. |
| [7] | L.K. Abdulrahman, A.M. Al-Abachi, M.H. Al-Qaissy, Flow injection-spectrophotometeric determination of some catecholamine drugs in pharmaceutical preparations via oxidative coupling reaction with p-toluidine and sodium periodate, Anal. Chim. Acta 538(2005) 331-335. |
| [8] | Z. Talebpour, S. Haghgoo, M. Shamsipur, 1H nuclear magnetic resonance spectroscopy analysis for simultaneous determination of levodopa, carbidopa and methyldopa in human serum and pharmaceutical formulations, Anal. Chim. Acta 506(2004) 97-104. |
| [9] | S.K. Moccelini, A.C. Franzoi, I.C. Vieira, J. Dupont, C.W. Scheeren, A novel support for laccase immobilization:cellulose acetate modified with ionic liquid and application in biosensor for methyldopa detection, Biosens. Bioelectron. 26(2011) 3549-3554. |
| [10] | M.B. Gholivand, M. Amiri, Preparation of polypyrrole/nuclear fast red films on gold electrode and its application on the electrocatalytic determination of methyl-dopa and ascorbic acid, Electroanalysis 21(2009) 2461-2467. |
| [11] | S. Shahrokhian, S. Rastgar, Electrodeposition of Pt-Ru nanoparticles on multiwalled carbon nanotubes:application in sensitive voltammetric determination of methyldopa, Electrochim. Acta 58(2011) 125-133. |
| [12] | A. Kutluay, M. Aslanoglu, Nickel nanoparticles functionalized multi-walled carbon nanotubes at platinum electrodes for the detection of bromhexine, Sens. Actuators B 192(2014) 720-724. |
| [13] | Xi. Yang, B. Feng, P. Yang, Y. Ding, Y. Chen, J. Fei, Electrochemical determination of toxic ractopamine at an ordered mesoporous carbon modified electrode, Food Chem. 145(2014) 619-624. |
| [14] | H.R. Zare, F. Chatraei, Preparation and electrochemical characteristics of electrodeposited acetaminophen on ruthenium oxide nanoparticles and its role as a sensor for simultaneous determination of ascorbic acid, dopamine and N-acetylL-cysteine, Sens. Actuators B 160(2011) 1450-1457. |
| [15] | A. Salimi, S. Lashgari, A. Noorbakhsh, Carbon nanotubes-ionic liquid and chloropromazine modified electrode for determination of NADH and fabrication of ethanol biosensor, Electroanalysis 22(2010) 1707-1716. |
| [16] | X.H. Liu, L. Li, X.P. Zhao, X.Q. Lu, Electrochemical behavior of rutin on a multiwalled carbon nanotube and ionic liquid composite film modified electrode, Colloids Surf. B 81(2010) 344-349. |
| [17] | A. Kutluay, M. Aslanoglu, Selective detection of albuterol in the presence of uric acid using a glassy carbon electrode modified with multi-walled carbon nanotubes and poly(pivalic acid), Sens. Actuators B 177(2013) 703-709. |
| [18] | A. Babaei, M. Zendehdel, B. Khalilzadeh, A. Taheri, Simultaneous determination of tryptophan, uric acid and ascorbic acid at iron(ⅡI) doped zeolite modified carbon paste electrode, Colloids Surf. B 66(2006) 226-232. |
| [19] | H. Beitollahi, I. Sheikhshoaie, Electrocatalytic and simultaneous determination of isoproterenol, uric acid and folic acid at molybdenum(VI) complex-carbon nanotube paste electrode, Electrochim. Acta 56(2011) 10259-10263. |
| [20] | R. Hosseinzadeh, R.E. Sabzi, K. Ghasemlu, Effect of cetyltrimethyl ammonium bromide(CTAB) in determination of dopamine and ascorbic acid using carbon paste electrode modified with tin hexacyanoferrate, Colloids Surf. B 68(2009) 213-217. |
| [21] | J.B. Raoof, R. Ojani, H. Beitollahi, L-Cysteine voltammetry at a carbon paste electrode bulk-modified with ferrocenedicarboxylic acid, Electroanalysis 19(2007) 1822-1830. |
| [22] | H.F. Zhang, Z.C. Meng, Q. Wang, J.B. Zheng, A novel glucose biosensor based on direct electrochemistry of glucose oxidase incorporated in biomediated gold nanoparticles-carbon nanotubes composite film, Sens. Actuators B 158(2011) 23-27. |
| [23] | H.R. Zare, N. Nasirizadeh, H. Ajamain, A. Sahragard, Preparation, electrochemical behavior and electrocatalytic activity of chlorogenic acid multi-wall carbon nanotubes as a hydroxylamine sensor, Mater. Sci. Eng. C 31(2011) 975-982. |
| [24] | A. Kutluay, M. Aslanoglu, Multi-walled carbon nanotubes/electro-copolymerized cobalt nanoparticles-poly(pivalic acid) composite film coated glassy carbon electrode for the determination of methimazole, Sens. Actuators B 171-172(2012) 1216-1221. |
| [25] | M.M. Rahman, M.J.A. Shiddiky, M.D.A. Rahman, Y.D. Shim, A lactate biosensor based on lactate dehydrogenase/nictotinamide adenine dinucleotide(oxidized form) immobilized on a conducting polymer/multiwall carbon nanotube composite film, Anal. Biochem. 384(2009) 159-165. |
| [26] | M. Keyvanfard, R. Shakeri, H. Karimi-Maleh, K. Alizad, Highly selective and sensitive voltammetric sensor based on modified multiwall carbon nanotube paste electrode for simultaneous determination of ascorbic acid, acetaminophen and tryptophan, Mater. Sci. Eng. C 33(2013) 811-816. |
| [27] | M. Keyvanfard, M. Ahmadi, F. Karimi, K. Alizad, Voltammetric determination of cysteamine at multiwalled carbon nanotubes paste electrode in the presence of isoproterenol as a mediator, Chin. Chem. Lett. 25(2014) 1244-1246. |
| [28] | T.L. Lu, Y.C. Tsai, Sensitive electrochemical determination of acetaminophen in pharmaceutical formulations at multiwalled carbon nanotube-alumina-coated silica nanocomposite modified electrode, Sens. Actuators B 153(2011) 439-444. |
| [29] | M.C. Henstridge, E.J.F. Dickinson, M. Aslanoglu, C. Batchelor-McAuley, R.G. Compton, Voltammetric selectivity conferred by the modification of electrodes using conductive porous layers or films:The oxidation of dopamine on glassy carbon electrodes modified with multiwalled carbon nanotubes, Sens. Actuators B 145(2010) 417-427. |
| [30] | United States Pharmacopeia National Formulary 23rd Revision, US Pharmacopeial Convention, Rockville, MD, 1995. |
| [31] | I. da Cruz Vieira, O. Fatibello-Filho, Spectrophotometric determination of methyldopa and dopamine in pharmaceutical formulations using a crude extract of sweet potato root(Ipomoea batatas(L.) Lam.) as enzymatic source, Talanta 46(1998) 559-564. |
 2016, Vol.27
2016, Vol.27