b Department of Pharmaceutics, School of Pharmacy, East China University of Science and Technology, Shanghai 200237, China;
c Shanghai Key Laboratory of New Drug Design, East China University of Science and Technology, Shanghai 200237, China;
d Shanghai Institute of Health Sciences, Shanghai 201318, China
Nanoparticle (NP) drug delivery systems (NDDSs) have been widely investigated as new drug delivery vehicles in the pharmaceutical research area. They are expected to change the landscape of healthcare and pharmaceutical industries in the foreseeable future [1, 2]. The use of NDDSs enables to achieve: (i) Improved solubilization and delivery of poorly water-soluble drugs; (ii) protection of labile drugs from degradation; (iii) modified tissue distribution profile of drugs; (iv) targeted delivery of drugs into cells via endocytosis rather than non-specific diffusion; (v) co-delivery of two or more drugs for combination therapy; and (vi) visualization of drug delivery sites by inclusion of imaging modalities [3, 4, 5, 6, 7].
Biodegradable and biocompatible natural polymers,such as chitosan (CS) and β-cyclodextrin ( β-CD),have been extensively used to prepare NDDSs in the last decade. CS is an attractive nontoxic,biodegradable and biocompatible linear polymer which has been employed in the preparation of NPs. The polymer features the cationic nature which allows for fabricating NPs by ionic cross-linking method with anions,leading to the controlled release of loaded agents. Moreover,the abundant amino groups of CS are readily available for functionalization [8]. A number of research groups have reported the use of CS-based NPs for drug delivery [9, 10, 11, 12]. However,the ability of CS alone to directly encapsulate water insoluble drugs is poor as the loading efficiency is usually rather low. Different from linear polymeric CS,β-CD has a truncated cone structure with a hydrophilic outer surface and a hydrophobic interior cavity. As a result of hydrophobic interactions,a variety of lipophilic drugs can be loaded into the hydrophobic cavity of β-CD,leading to the improved solubility, loading efficiency,and stability of lipophilic drugs. Thus,β-CD and its derivatives such as hydroxypropyl- β-cyclodextrin (HP- β-CD),sulphobutylether - β-cyclodextrin (SB- β-CD) and 2,6-di-O-methyβ-cyclodextrin (DM- β-CD) are often used for drug formulation [13, 14, 15]. These β-CD derivatives with different physicochemical properties behave differently for various drugs in the formation of stable inclusion complexes. For example,DM- β-CD is the most effective inclusion host for paclitaxel as it can achieve the best paclitaxel solubility among all CD derivatives studied [16]. Another example is albendazole,the ability of β-CD citrate derivative to form inclusion complex is better than that of β-CD and HP- β-CD [17]. Different CD derivatives even affect the way to prepare inclusion complex as in the case of celecoxib. DM- β-CD is able to include the drug both in the solid and solution state while HP- β-CD can only form inclusion complex with celecoxibin a solid state [18]. Therefore,there is a recent research trends to incorporate different β-CDs into CS NPs (CD/CS NPs) to further improve its versatility and loading and controlled-releasing profiles for various poorly water-soluble drugs [19, 20, 21, 22, 23].
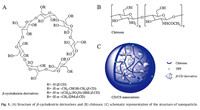
Herein,the incorporation of various β-CD and its derivatives (shown in Fig. 1A) with CS (shown in Fig. 1B) to form a series of drug nanocarriers (shown in Fig. 1C) by ionic gelation method using tripolyphosphate (TPP) as the crosslinker were systematically studied. Three representative β-CD derivatives with different physicochemical properties,which are HP- β-CD,SB- β-CDand DMβ-CD,were selected achieving loading versatility for different poorly water-soluble drugs. The size,zeta potential,and morphology of the resulting NPs were characterized. Compositions of NPs were investigated with elemental analysis. The in vitro stability of NPs was also investigated under pH 6.8 in phosphate buffer solution (PBS).
|
Download:
|
| Fig. 1. (A) Structure of β-cyclodextrin derivatives and (B) chitosan; (C) schematic representation of the structure of nanoparticle. | |
NPs were prepared using the mild ionic gelation method previously developed by our lab [12]. NPs were spontaneously obtained via ionic gelation between the positively charged amino groups of CS and negatively charged TPP.
For CS NPs without CD,different concentrations (0.05%-0.20%, w/w) of CS solutions were prepared with the addition of glacial acetic acid (1.75 times of CS weight). Then the pH of CS solutions was adjusted to 4.0-6.0 using 0.1 mol/L NaOH. The cross-linking agent TPP (0.08%-0.20%,w/w) was added dropwise to 5 mL CS solution with magnetic stirring at 800 rpm and continued stirring for a certain period.
CD/CS NPs were prepared by dissolving various amounts of β-CD/HP- β-CD/SB- β-CD/DM- β-CD in TPP solution (0.1%,w/v) described for the preparation of CS NPs without CD under the optimal condition. The concentration of CS solution was 1.0 mg/mL. The mass ratio of CS to TPP was kept at 5:1,and the pH was adjusted to 4.5. NPs were spontaneously formed upon mixing of these polycationic and polyanionic phases under stirring. The purified NPs were freeze dried to obtain dry NPs with 3% mannitol as lyophilized protective agent. Every sample was prepared in triplicate and the results represented the average value.
The particle size and zeta potential of all the NPs in aqueous solution were determined by dynamic light scattering (DLS, NanoZS4700 nanoseries,Malvern Instruments,UK) equipped with 4 MW He-Ne laser at a wave length of 633 nm at 25 ℃.
Morphological evaluations of CS NPs,SB- β-CD/CS NPs,DM- β-CD/CS NPs and HP- β-CD/CSNPs were performed with transmission electron microscopy (TEM). For TEM images,the particle morphology of polymers was observed on JEM-2010 JEOL from Japan. The sample was stained with 2% (w/v) phosphotungstic acid and then dropped on a copper grid.
The measured C,H and N mass fraction of the CD/CS NPs sample was used to determine the mass ratio of CD to CS in the sample. The N mass fraction of NPs was used to determine the CS content. After the CS content was determined,the contribution of C mass fraction arising from CS could be calculated,and the CD’s contribution to the C mass fraction could be deduced by subtraction this calculated figure from the measured total C mass fraction value,thus the content of CD could be calculated. The remaining fraction of the NP composition was attributed to counterions TPP. Incorporation efficiency is a parameter showing the percentage of the starting materials which were actually incorporated into the NP. Incorporation efficiency of CD can be calculated as follows:
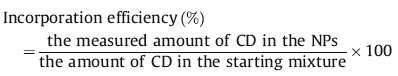
For the stability experiment,the NPs were freshly prepared, isolated,and resuspended in PBS (pH 6.8) at 0.1% (w/v). Then,the NPs were incubated at 25 ℃ under agitation (100 rpm). At time points of 0,0.5,1,2,4,6,12,24,48 and 72 h,samples were collected and their size distribution was measured by DLS. Each experiment was performed in triplicate.
All data were presented as a mean value with its standard deviation (mean ± SD). Multiple group comparison was conducted by one-way analysis of variance (ANOVA) and then by LSD using statistical software (SPSS,Chicago). Differences were considered to be significant when p < 0.05 using a two-tailed paired t-test. 3. Results and discussion
First,we prepared CS NPs using a technique based on the ionic gelation between CS and TPP. The results suggested that size and zeta potential of CS NPs depended largely on concentration of CS and TPP,pH value,and the mass ratio of CS to TPP (CS/TPP).
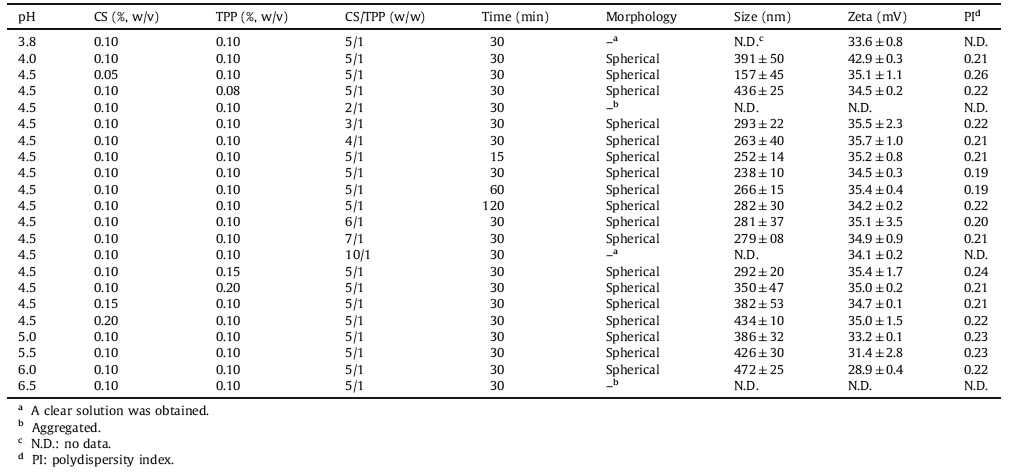
Since CS is a weak basic polysaccharide with an average amino group density of 0.837 per disaccharide unit,it is insoluble at neutral and alkaline environment. While in acidic medium,these amine groups are positively charged,thus conferring to a highly charged polysaccharide chain. Therefore,the surface charge density of CS molecule is strongly dependent on solution pH. The phenomenon is crucial in the pH-responsive formationprocess of CS NPs via ionic cross-linking. To study the effects of pH values on the formation of CS NPs,a fixed CS and TPP concentration (0.10%,w/v),and CS/TPP weight ratio (5:1) were used in the NP preparation with variable solution pH values. Our results showed that CS NPs could only be formed in pH ranging 4.0-6.0 (Table 1). The aggregation at pH 6.5 was likely due to decreased electrostatic repulsion between NPs related to the reduced protonation level on the particle surface at a less acidic solution. This idea of deprotonation was also supported by the results shown in Table 1,which showed that zeta potential of NPs decreased with increasing pH value.
| Table 1 Physicochemical properties of CS NPs. |
Given that CS is a polycationic electrolyte,our approach to the formation of NPs is based on the induced gelation process from the addition of the counterion TPP. It is known that both the inter- and intra-molecular linkages between TPP and the positively charged amino groups of CS are responsible for the success of the gelation process. Our results showed that the formation of NPs was only successful for a certain mass ratio range of CS to TPP (Table 1). Whenthe massratio of CS toTPP was toohigh( ≥10),theamount of TPP was not enough for crosslinking to form nanoparticles. When the ratio was too low ( ≤2),the excess negative-charged TPP might disrupt the positive particle-particle repulsion barrier that defined their stability as individual colloids,or/and act as cross-linking agent between formed NPs,thus leading to particle aggregation.
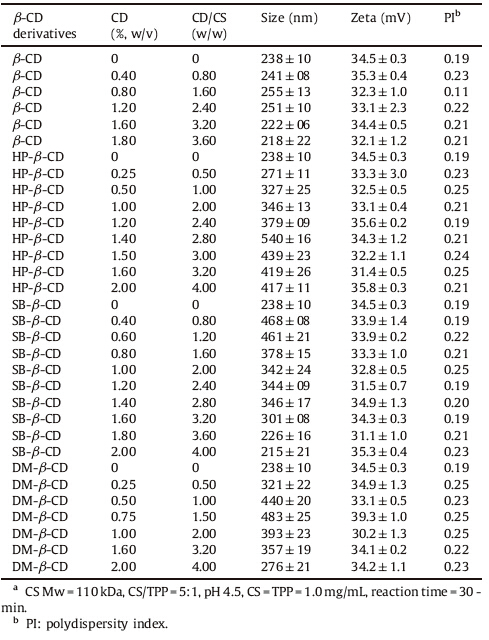
When it comes to CD/CS NPs,we hypothesized that physicochemical properties of CD may be critical for its interaction with CS and therefore,we selected a group of different CDs: β-CD,HPβ-CD,SB- β-CD and DM- β-CD. Among them,β-CD,HP- β-CD and DM- β-CD are neutral molecules,whereas SB- β-CD is polyanionic. Our results suggested that all these CDs could form spherical NPs with CS (Table 2).
| Table 2 Physicochemical properties of CD/CS NPs.a |
To probe the effects of process parameters on NPs’ size and zeta potential,the physicochemical properties of CS NPs prepared under different conditions were characterized. As seen from Table 1,the sizes of CS NPs were ranged from 157 nm to 472 nm. The zeta potential was from 28.88 mV to 42.90 mV,which indicated the good stability of NPs. The size,zeta potential,and PI of CS NPs largely depended on the concentration of CS and TPP, the pH value,and the weight ratio of CS to TPP. It was found out that NPs formed at pH 4.5 had a relatively smaller size. The zeta potential of NPs decreased with increasing solution pH value, which was ascribed to the reduced degree of protonation at the surface of CS NPs. The effect of CS concentration on particle size was also very prominent. It was demonstrated that the size of CS NPs became larger with the increasing CS concentration. As for TPP concentration,stable NPs could be formed from 1.0 mg/mL to 2.0 mg/mL. Additionally,the mass ratio of CS to TPP also played an important role in the distribution of particle size. The smallest particle size was obtained at the mass ratio of 5 (i.e. mole ratio of CS unit to TPP was 9.9). Generally speaking,one molecule of TPP could make five ionic cross-linking interactions with amino groups of CS, which indicates that two CS units could bind to one TPP molecule. Addition of TPP to CS in 1/5 weight ratio presumably led to the most efficient cross-linking of amino groups. Almost all the amino groupsof CS were involved in cross-linking,resulting in a relatively stable CS NPs. In conclusion,this study demonstrated that rational design and modulation of the particle size and surface charge of CS NPs could be achieved by the right control of these critical process parameters.
When it comes to the preparation of CD/CS NPs,we adopted the optimized process parameters from the above CS NPs (CS/ TPP = 5:1,pH 4.5,CS = TPP = 1.0 mg/mL,reaction time = 30 min). The results demonstrated that the size of CD/CS NPs mostly depended on the types and concentration of CDs (Table 2). In addition,the incorporation of β-CD did not affect the size and zeta potential of NPs significantly,whereas the incorporation of HP- β-CD,SB- β-CD and DM- β-CD led to an increase in the particle size at low concentrations. When the concentration of β-CD,HP- β-CD and DM- β-CD reached a certain number,the particle size started to reduce. The reason might be that at low concentration,CDs could facilitate the incorporation process into CS to form larger particles, whereas at higher concentration,CDs might lead to a more compact nanostructure due to a series of weak intermolecular forces such as hydrogen bond/hydrophobic interactions between CDs and CS. In the case of SB- β-CD/CS NPs,the size decreased from 346 nm to 226 nm as the concentration of SB- β-CD increased from 14.0 mg/mL to 18.0 mg/mL. The reduction in size could be explained by the increased ionic interactions between cationic CS and anionic SB- β-CD. The zeta potential of four series of NPs was positive and the values were close to 33 mV. It suggested that neutral or anionic CDs were mainly inside the NPs and did not mask the positive charge of CS on the surface.
The morphology of CS NPs,SB- β-CD/CS NPs DM- β-CD/CS NPs and HP- β-CD/CS NPs was investigated by TEM. It was observed that NPs had spherical morphology with smooth surface (Fig. 2). Because of the presence of SB- β-CD,DM- β-CD and HP- β-CD,the size of NPs had a minor increase compared with that of CS NPs. However,no noticeable change in the appearance of NPs was observed. The value of particle size was smaller than the data determined by DLS since the TEM measured the particle size in dry state,while DLS measured the particle size in solution. The size determined by DLS included hydrated layers surrounding the particles,thus the value was larger than that in dry state.

|
Download:
|
| Fig. 2. TEM photographs of (A) CS NP, (B) SB- β-CD/CS NP, (C) DM- β-CD/CS NP and (D) HP- β-CD/CS NP. | |
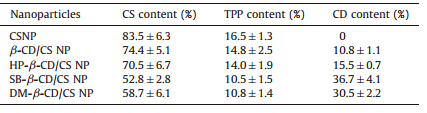
In this work,composition of the CD/CS NPs prepared with the same mass ratio of CDs to CS and the incorporation efficiency of CDs were determined by elemental analysis. Using this technique, the content of CS and CD of the NPs could be determined easily by comparing the C-N mass ratios. As shown in Table 3,CS NPs were analyzed and taken as a reference for the CD-containing formulations. The content of CS was 83.5% and the content of counterions was 16.5%,values which were very close to the theoretical ratio (CS/TPP = 5/1). When the weight ratio of CDs to CS was 3.2,the TPP content of all CD/CS NPs decreased compared with that of CS NPs. This was because the incorporation of CDs substituted some of the TPP molecules inside CS NPs. The incorporation efficiency of β-CD,HP- β-CD,SB- β-CD and DM- β-CD in NPs were 10.8%,15.5%,36.7% and 30.5%,respectively. For neutral β-CD,HP- β-CD and DM- β-CD,the force between CDs and CS was a series of weak intermolecular forces such as hydrogen bond/hydrophobic interactions,which was not quite strong. However,for anionic SB- β-CD,the relatively high incorporation efficiency was justified by the presence of negatively charged groups in SB- β-CD and their ionic interactions with the positively charged CS molecules.
| Table 3 Composition of different series of CD/CS NPs. |
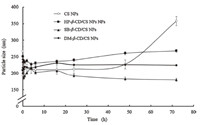
Time-dependent size change of different CD/CS NPs in PBS is shown in Fig. 3. Initially,the particle size reduced rapidly due to the change of environmental conditions. This stage of rapid particle size reduction is designated as the instantaneous stage. The time to finish the instantaneous stage depends on the initial size of the NPs. The following stage is called the aging stage. The insignificant size change during this period is due to the balance between intermolecular entanglement,syneresis,inter/intra-molecular hydrogen bonding formation and new crystalline regions formation. The third stage is swelling/aggregation of the NPs. Swelling is possibly due to the inflow of water into the NPs by osmosis. Swelling of NPs will lead to the fracture of polymer matrix. Aggregation might be due to the collision and adhesion of NPs during storage [24].
|
Download:
|
| Fig. 3. Particle size change of different nanoparticles following incubation in PBS (pH 6.8) for 72 h at 25 ℃. Data were expressed as mean ± SD (n = 3). | |
For CS NPs withoutCD,a significant swelling uponincubation in PBS after 48 h was found. However,the other groups were still stable after incubated in PBS for up to 72 h. This result was consistent with our previous conclusion that CD/CS NPs presented a more compact inner structure due to a series of weak intermolecular forces or ionic interactions between CDs and CS. 4. Conclusion
This study reports the development of a series of novel drug nanocarrier through entrapping β-CD and its derivatives within CS through a mild ionic gelation technique. The influences of process parameters including solution pH value,concentration of CS and TPP,mass ratio of CS to TPP,gelation time,and the concentration of CD on physicochemical properties of the NPs are investigated systematically. The optimal CD/CS NPs are obtained with uniform size and good stability in PBS (pH 6.8). The size of CD/CS NPs is greatly affected by the types of CDs incorporated and their concentrations. This current research provides a versatile CD/CS NPs-based drug delivery platform for incorporating poorly watersoluble drugs with different physicochemical properties which constitutes our future endeavor.
Acknowledgments
This work was financially supported by Postdoctoral Science Foundation of China (No. 2014M550222),Shanghai Postdoctoral Sustentation Fund (No. 14R21410500). The authors also acknowledge the support from School of Pharmacy,Fudan University & the Open Project Program of Key Lab of Smart Drug Delivery (Fudan University),Ministry of Education (No. SDD2014-2),State Key Laboratory of Molecular Engineering of Polymers (Fudan University,No. K2015-15),and the Fundamental Research Funds for the Central Universities (Nos. 22A201514055 and WY1213013 ECUST).
| [1] | L. Zhang, F.X. Gu, J.M. Chan, et al., Nanoparticles in medicine: therapeutic applications and developments, Clin. Pharmacol. Ther. 83 (2008) 761–769. |
| [2] | D. Brambilla, B. Le Droumaguet, J. Nicolas, et al., Nanotechnologies for Alzheimer's disease: diagnosis, therapy, and safety issues, Nanomedicine 7 (2011) 521–540. |
| [3] | A.O. Elzoghby, W.M. Samy, N.A. Elgindy, Albumin-based nanoparticles as potential controlled release drug delivery systems, J. Control. Release 157 (2012) 168–182. |
| [4] | R. Singh, J.W. Lillard Jr., Nanoparticle-based targeted drug delivery, Exp. Mol. Pathol. 86 (2009) 215–223. |
| [5] | M. Ferrari, Cancer nanotechnology: opportunities and challenges, Nat. Rev. Cancer 5 (2005) 161–171. |
| [6] | S. Naahidi, M. Jafari, F. Edalat, K. Raymond, A. Khademhosseini, Biocompatibility of engineered nanoparticles for drug delivery, J. Control. Release 166 (2013) 182–194. |
| [7] | J.D. Byrne, T. Betancourt, L. Brannon-Peppas, Active targeting schemes for nanoparticle systems in cancer therapeutics, Adv. Drug Del. Rev. 60 (2008) 1615–1626. |
| [8] | S.A. Agnihotri, N.N. Mallikarjuna, T.M. Aminabhavi, Recent advances on chitosanbasedmicro- andnanoparticles in drug delivery, J. Control. Release 100 (2004) 5–28. |
| [9] | S.R. Jameela, P.G. Latha, A. Subramoniam, A. Jayakrishnan, Antitumour activity of mitoxantrone-loaded chitosan microspheres against Ehrlich ascites carcinoma, J. Pharm. Pharmacol. 48 (1996) 685–688. |
| [10] | S. Mitra, U. Gaur, P.C. Ghosh, A.N. Maitra, Tumour targeted delivery of encapsulated dextran–doxorubicin conjugate using chitosan nanoparticles as carrier, J. Control. Release 74 (2001) 317–323. |
| [11] | P. Yousefpour, F. Atyabi, E. Vasheghani-Farahani, A.A. Movahedi, R. Dinarvand, Targeted delivery of doxorubicin-utilizing chitosan nanoparticles surfacefunctionalized with anti-Her2 trastuzumab, Int. J. Nanomed. 6 (2011) 1977–1990. |
| [12] | Z.T. Yuan, Y.J. Ye, F. Gao, et al., Chitosan-graft-β-cyclodextrin nanoparticles as a carrier for controlled drug release, Int. J. Pharm. 446 (2013) 191–198. |
| [13] | S.S. Gao, J. Sun, D.J. Fu, et al., Preparation, characterization and pharmacokinetic studies of tacrolimus-dimethyl-β-cyclodextrin inclusion complex-loaded albumin nanoparticles, Int. J. Pharm. 427 (2012) 410–416. |
| [14] | Y.J. Ye, Y. Sun, H.L. Zhao, et al., A novel lactoferrin-modified β-cyclodextrinnanocarrier for brain-targeting drug delivery, Int. J. Pharm. 458 (2013) 110–117. |
| [15] | C. Yang, Recent progress in supramolecular chiral photochemistry, Chin. Chem. Lett. 24 (2013) 437–441. |
| [16] | H. Hamada, K. Ishihara, N. Masuoka, K. Mikuni, N. Nakajima, Enhancement of water-solubility and bioactivity of paclitaxel using modified cyclodextrins, J. Biosci. Bioeng. 102 (2006) 369–371. |
| [17] | A. García, D. Leonardi, M.O. Salazar, M.C. Lamas, Modified β-cyclodextrin inclusion complex to improve the physicochemical properties of albendazole. Complete in vitro evaluation and characterization, PLOS ONE 9 (2014) e88234. |
| [18] | C.A. Ventura, S. Tommasini, A. Falcone, et al., Influence of modified cyclodextrins on solubility and percutaneous absorption of celecoxib through human skin, Int. J. Pharm. 314 (2006) 37–45. |
| [19] | J.G. Ji, S.L. Hao, W.Q. Liu, et al., Preparation and evaluation of O-carboxymethylchitosan/cyclodextrin nanoparticles as hydrophobic drug delivery carriers, Polym. Bull. 67 (2011) 1201–1213. |
| [20] | A. Vyas, S. Saraf, S. Saraf, Encapsulation of cyclodextrincomplexed simvastatin in chitosan nanocarriers: a novel technique for oral delivery, J. Incl. Phenom. Macrocycl. Chem. 66 (2010) 251–259. |
| [21] | A.A. Mahmoud, G.S. El-Feky, R. Kamel, G.E.A. Awad, Chitosan/sulfobutyletherb- cyclodextrin nanoparticles as a potential approach for ocular drug delivery, Int. J. Pharm. 413 (2011) 229–236. |
| [22] | Y. Yang, Y.M. Zhang, Y. Chen, J.T. Chen, Y. Liu, Targeted polysaccharide nanoparticle for adamplatin prodrug delivery, J. Med. Chem. 56 (2013) 9725–9736. |
| [23] | D. Zhao, Y. Chen, Y. Liu, Comparative studies on molecular induced aggregation of hepta-imidazoliumyl-β-cyclodextrin towards anionic surfactants, Chin. Chem. Lett. (2014), http://dx.doi.org/10.1016/j.cclet.2014.11.028. |
| [24] | M.L. Tsai, R.H. Chen, S.W. Bai, W.Y. Chen, The storage stability of chitosan/tripolyphosphate nanoparticles in a phosphate buffer, Carbohydr. Polym. 84 (2011) 756–761. |