b College of Chemistry and Chemical Engineering, University of Jinan, Jinan 250022, China;
c Graduate University of Chinese Academy of Sciences, Beijing 100080, China
Supercapacitors are gaining increased attention owing to the diminishing supplies of fossil fuel and the rapid growing demand for renewable sources [1]. It is well known that the most frequently used materials for supercapacitors are mainly carboN-based materials [2, 3, 4],conducting polymers [5] and transition metal oxides [6, 7]. CarboN-based materials have advantages with regard to their high specific surface area,high power density and excellent electric conductivity. However,carboN-based materials are known to have low specific capacitance and are prone,in general,to aggregation [8, 9, 10, 11, 12, 13]. Additionally,the transition metal oxides,with charge-discharge capability based on their redox reactions,often suffer from inferior rate performance,low utilization rate and poor cycle stability even though possessing an extremely attractive theoretical capacitance value [14, 15, 16, 17]. Among the numerous electrode materials,nickel-based ones with the advantages of natural abundance and low cost,combined with their energy storage performance in basic electrolytes and environmental compatibility,have made them one of the most promising for pseudocapacitors [18, 19]. In particular,the transmission rates of ions and electrons during the charge-discharge process,which determine the specific capacitance and the rate performance of the supercapacitors,need to be mentioned. It is generally accepted that the redox reaction between the active materials and the electrolyte ions occurs only in a thin layer on the surface of the active materials [20]. The preparation of active pseudocapacitance materials with large specific surface area,porosity and good electronic conductivity is therefore highly preferred in order to enhance the performance of the supercapacitors [21, 22].
In order to meet the above mentioned requirements,an effective approach may be to construct hierarchical porous structured,carbon-transition metal composites with high electrochemical performance [23]. The hierarchical porous structure generally consists of one/two dimensional nanostructures featuring a large specific surface area to provide more effective contact between the electrolyte ions and the active materials,so as to raise their utilization rate and enhance the electrochemical performance of the supercapacitors [24]. Owing to the large specific surface area,outstanding electronic conductivity and a two-dimensional lamellar structure [25, 26],graphene sheets (GS) are usually incorporated into pseudocapacitance materials to enhance their electrochemical performance [27, 28, 29, 30].
In this work,a series of composites based on nickel compound- graphene sheets (NiBC-GS) are prepared by flocculation of intercalated nickel dichloride-graphene oxide from a solution using hydrazine hydrate as flocculant and reductant,followed by a subsequent annealing process. The NiBC-GS designed as a loosely packed structure with GS sheets uniformly distributed in the NiCl2-hydrazine complex (Ni(N2H4)xCl2) matrix can facilitate the efficient transport/migration of ions and are expected to possess excellent specific capacitances. More importantly,the application of the prepared Ni(N2H4)xCl2-GS,as an electrode composite for use in supercapacitors,is a new attempt and has not been reported by others. The texture and morphology of the obtained composites were characterized by powder X-ray diffraction (XRD),thermogravimetric analysis (TGA) and field-emission scanning electron microscopy (FESEM). In addition,the variation of specific capacitances of NiBC-GS with the annealing temperature was also studied in detail. 2. Experimental
All chemicals are analytical grade,purchased from J&K China Chemical Ltd and used as received without further purification. Graphene oxide (GO) suspension (0.1 mg mL-1) was prepared by a modified Hummer’s method [31, 32].
Two series of samples,nickel chloride-based compounds without GS incorporation (NiBC) and with GS incorporation (NiBC-GS),wererespectively prepared. In a typical procedure for preparation of NiBC-GS,950 mg (0.004 mol) of nickel dichloride (NiCl2·6H2O) was dissolved in 150 mL of N,N-dimethylformamide (DMF) and mixed with 5.2 mL of GO (0.52 mg GO powders) suspension in DMF under stirring and ultrasonic at room temperature,followed by drop-wise addition of 2 mL of aqueous solution (80 wt%) of hydrazine hydrate (N2H4·H2O,about 0.02 mol, making N2H4/Ni2+ at 5.0 by mole) under stirring to induce the flocculation reaction. The reaction was allowed to proceed for 30 min upon completion of the N2H4·H2O addition. The temperature was then increased to 80 ℃ and held for 30 min in order for GO in the flocculate to be reduced to GS by N2H4·H2O. The product was rinsed twice with 100 mL of ethanol,and dried in air flow at 50 ℃ for 12 h to obtain NiBC-GS composite,denoted as NiBC-GS50, with the ending number indicating drying temperature. The samples denoted as NiBC-GS250 or NiBC-GS450 were obtained the same way as for NiBC-GS50,except that they were treated further after ethanol rinsing and drying at 50 ℃,followed by an annealing process at 250 ℃ and 450 ℃,respectively. This annealing was conducted by an increase in temperature at a rate of 5 ℃ min-1, and the sample held at the target temperature for 4 h. For preparation of NiBC samples,NiBC-250 for example,the same procedure was followed without addition of GO suspension.
The structure and morphology of the composites were characterized by powder X-ray diffraction (XRD,Rigaku D/Max 2400,Cu-Kα radiation),thermogravimetric analysis (TGA,STA 449C,air flow) and field-emission scanning electron microscopy (FESEM,JEOL JSM-6701F). Cyclic voltammetry (CV) and galvanostatic charge/discharge experiments were carried out using a CHI 660C electrochemical workstation (Shanghai CH Instrument Company,China) in an electrolyte solution of 2.0 mol L-1 KOH in a three-electrode system. The working electrode was fabricated by pasting a homogeneous slurry of active material (4 mg),carbon black and poly(tetrafluoroethylene) in a mass ratio of 80:15:5 into a porous nickel foam. A platinum (Pt) sheet and an Ag/AgCl (KClsaturated) electrode served as the counter electrode and the reference electrode,respectively. The specific capacitance was calculated according to the following equation:

A color change was observed with the addition of N2H4·H2O solution to the NiCl2 solution in DMF for preparation of NiBC samples. This color change was dependent on the added amount of N2H4·H2O solution. The NiCl2 solution,initially green,became purple along with an obvious generation of flocculation or precipitation when 1 mL of N2H4·H2O solution (80 wt%) was added to 150 mL of the NiCl2 solution (containing 950 mg,i.e., 0.004 mol of NiCl2·6H2O,giving N2H4/Ni2+ molar ratio of 2.5). With further addition of another 1 mL of N2H4·H2O solution, achieving a N2H4/Ni2+ molar ratio of 5.0,the reaction medium turned pink. Such color evolutionwas also previously reported for the reaction system conducted in a full aqueous medium [33, 34]. The flocculate thus prepared was filtered and dried at 50 ℃ for 12 h to obtain a powder sample of NiBC50 with pink color. During the preparation of GS-containing samples,the same color evolution was observed,except the final flocculation composites turned black with the reduction of GO to GS by N2H4·H2O after raising the temperature to 80 ℃ and maintaining this temperature for 30 min.
Generally,different complexes can be formed by the reaction of NiCl2 and N2H4·H2O solutions,depending on their relative amounts [34, 35, 36, 37]. At a low N2H4/Ni2+ ratio of 2.5,Ni(N2H4)2Cl2 is formed with a purple color; while a pink crystal form of Ni(N2H4)3Cl2 can be produced at a higher N2H4/Ni2+ ratio of 5.0. It is conceivable that initially formed Ni(N2H4)2Cl2 may remain at the end of the reaction,although part of it is certainly transformed into the Ni(N2H4)3Cl2 complex. It is to point out that there is no scientific support indicating that the presence of GO or GS would interfere with the reaction between NiCl2 and N2H4·H2O; namely, the same nickel-hydrazine complexes were formed in GS incorporated NiBC. With the presence of black-colored GS, however,the color of the nickel-hydrazine complexes is masked.
To observe the morphology of the samples,NiBC50 and NiBCGS50 were examined by FESEM (Fig. 1) and compared with pure GS (Fig. 1a),prepared by direct reduction of GO-DMF suspension using N2H4·H2O as the reductant at 80 ℃ for 30 min,followed by freezing drying. The lamellar and surface folding structures of GS are observed as expected; while only nano-sized granular particles or loosely packed aggregates are present in NiBC (Fig. 1b). With regard to the samples of NiBC-GS50 (Fig. 1c and d),the GS sheets are well dispersed in the NiBC matrix and clearly identified by the arrows. As previously reported [31],two-dimensional microstructures of GO sheets with a thickness of about 1 nm and a width of several micrometers were dispersed in DMF. In this work,uniform NiBC-GO composites should be formed by quick flocculation of NiCl2-GO-DMF solution with N2H4·H2O addition,and uniform NiBC-GS50 composites finally obtained after iN-situ reduction of GO and subsequent treatment.

|
Download:
|
| Fig 1. FESEM images of GS (a),NiBC50 (b) and GS-incorporated NiBC-GS50 (c and d) powders. | |
The typical FESEM images of NiBC-GS obtained at different annealing temperatures are displayed in Fig. 2. From Fig. 2a and b, where the samples were treated by an annealing temperature at 250 ℃ or lower,NiBC-GS exhibits a loosely packed structure with GS sheets uniformly distributed in the NiBC matrix as pointed by the white narrows; while a certain degree of agglomeration was observed in the NiBC nanoparticles. When the annealing temperature was raised to 450 ℃ (Fig. 2c),a composite consisting of crumpled GS sheets and octahedral crystal particles of NiBC was obtained. With the annealing temperature further increased to 800 ℃,GS was completely decomposed,only the crystallized particles of NiBC were observed as shown in Fig. 2d. As discussed later,NiBC was transformed to NiO at this high temperature.

|
Download:
|
| Fig 2. Typical FESEM images of NiBC-GS active materials obtained at different annealing temperature (a,100 ℃; b,250 ℃; c,450 ℃; d,800 ℃). | |
Preparation of NiCl2-hydrazine complexes has been widely studied [33, 34, 35, 36, 37]. The chemical reaction in the process can be presented by the following equation:

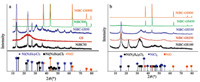
Among all the resulting complexes,dihydradrazine (Ni(N2H4)2 Cl2) and trishydrazine (Ni(N2H4)3Cl2) are the most common main products. The proportions of these complexes are highly dependent on the experimental conditions,in particular,the solvent medium used and the molar ratio of N2H4/Ni2+ [34, 35, 36, 37]. The hexahydrazine complex (n = 6) was also reportedly obtained but not isolated,since it was very unstable and decomposed to give diand trishydrazines [34]. With presence of a base in an aqueous medium,formation of hexaammonia complex was also claimed [33, 38, 39, 40]. Based on this reported information,it is rational to conclude that the products in this work basically consists of di- and trishydrazine complexes,because DMF was used as the solvent. The XRD diffractograms of samples of NiBC50 (prepared without GS) and NiBC-GS50 (prepared with the presence of GS) (Fig. 3a) provide a solid evidence for this conclusion,where one can easily see that only di- and trishydrazine complexes were present in these samples. In addition,Fig. 3a also demonstrates that both NiBC800 and NiBC-GS800 exhibit exactly identical XRD diffractograms, and both are exactly identical to that of NiO (JCPDS,no. 47- 1049),indicating both samples,regardless of the presence of GS, became NiO after annealing at 800 ℃ in air. The diffractogram of NiBC-GS50 presents practically the same profile as that of NiBC50,despite the presence of the small amount of GS. This indicates that the main components in NiBC-GS50 was basically the same as NiBC-50,i.e.,both mainly consist of Ni(N2H4)3Cl2,confirming that the presence of GO (or GS) does not interfere with the reaction of NiCl2 with N2H4·H2O.
|
Download:
|
| Fig 3. Microstructure of the obtained samples characterized by XRD. (a) XRD patterns of GS,NiBC50,NiBC-GS50 and the remnants of their TGA analysis samples. (b) XRD patterns of the NiBC-GS samples obtained by drying NiBC-GS50 powders at different temperature. | |
To further understand the variation of composition with temperature,the GS-incorporated products (NiBC-GS series annealed at different temperature) were then subjected to XRD diffraction (Fig. 3b) and TGA (Fig. 4) testing. The first observation from Fig. 3b is that XRD patterns of NiBC-GS450 and NiBC-GS800 possess exactly the same profile,and both are identical to the standard XRD pattern of NiO (JCPDS,No. 47- 1049),indicating that all NiBC compounds were converted to NiO at 450 ℃. In addition,the narrow shape of their XRD peaks indicates a high degree of crystallization in the NiO remnant. Compared with the standard diffractograms of NiCl2 and NiO (Fig. 3b),it is obvious that NiCl2 andNiO were likely present in the sample of NiBC-GS350,along with a low content of dihydrazine complex,indicating that the reduction ofNiCl2with air took place around this temperature (we doubt that the main composition was monohydrazine complex,but are unable to conclude this from the figure because its XRD standard diffractogram was not available). As to the sample annealed at 250 ℃ (NiBC-GS250),it is very likely that NiO was not formed at this temperature; while NiCl2 was surely present along with some complexes of a lower hydrazine content (Ni(N2H4)xCl2 with x = 0.5 or 1,for instance) [37]. With regard to NiBC-GS100,Fig. 3b reveals that its XRD pattern corresponds to those of Ni(N2H4)2Cl2 instead of a mixture of Ni(N2H4)xCl2 (x = 2.3) as in NiBC50 and NiBC-GS50 (Fig. 2a). The typical and well resolved diffraction peaks for Ni(N2H4)3Cl2 (trishydrazine) at 2θ of 27°,56° and 62°,observed in Fig. 2a, disappeared; while those attributed to Ni(N2H4)2Cl2 (dihydrazine) remain,a strong indication that trishydrazine is mostly decomposed to dihydrazine or complexeswith even lower ligand numbers.

|
Download:
|
| Fig 4. TGA curves of different NiBC-GS samples at a heating rate of 5 ℃ min-1 in air flow. | |
In accordance with these changes in XRD diffractograms, significant differences were also observed in TGA tests for NiBC-GS samples annealed at different temperature (Fig. 4).
For the NiBC-GS50 sample without any annealing,loss of about 20% of its initial mass was detected up to 147 ℃,with a precipitous drop of 10% mass from 141 ℃ to 146 ℃. Similar behavior was reported by different authors and interpreted as the loss of uncoordinated water,which was assumingly dissipated prior to 150 ℃ [37, 41]. It is worthy to note that this sample,NiBC-GS50, was air flowed at 50 ℃ upon its preparation and was kept in air for several days before this TGA test,which indicates uncoordinated water molecules were likely present in this sample. Assuming that the sample consists of monohydrated trishydrazine (Ni(N2H4)3Cl2·H2O) complex,and entirely converted to dihydrazine at 150 ℃,a theoretical mass loss of 20.52% was obtained, which was practically the same as the detected mass loss (20.22%). The theoretical mass loss of 69.65% at 750 ℃ (30.65% remnant) coincided also with the detected value of 31.8% for the remnant (68.20% of mass loss). These data suggest that this sample NiBCGS50 likely consisted mainly of monohydrated trishydrazine complex. Since the sample,with temperature increase,was decomposing or transforming gradually from trishydrazine to NiO via dihydrazine and monohrdrazine as well as NiCl2 [37, 41]. As a matter of fact,it consisted of a mixture of different compositions at different intermediate temperatures,which made the identification of the remnants difficult. Nevertheless,the detected residual masses (55.02%) at 375 ℃ indicated the sample might contain a mixture of Ni(N2H4)0.5Cl2 (corresponding remnant of 59.79%) and NiCl2 (corresponding remnant of 53.22%); and similarly,the detected residual masses (46.53%) at around 580 ℃ may well indicate the sample contained a mixture of NiCl2 (corresponding remnant of 53.22%) and NiO (corresponding remnant of 31.8%).
For NiBC-GS100,it is believed that the sample after 4 h of annealing at 100 ℃ lost all its water and was composed of dihydrazine (Ni(N2H4)2Cl2) and monohydrazine (Ni(N2H4)Cl2) complexes [37]. Based on their molecular weights,theoretical remnant mass of 38.56% at 750 ℃,corresponding to the transformation of dihydrazine to NiO,and that of 46.20%, corresponding to the transformation of dihydrazine to NiO were easily calculated. The detected remnant mass of 42.3% suggests that this sample consists of the mono- and dihydrazine complexes, each at about 50%. The dihydrazine complex was known to fully transform to monohydrazine before 290 ℃,which gave a theoretical weight loss of about 8.5%,in good agreement with the detected mass loss of about 8% at 290 ℃. The last part of the TGA curve (290 ℃ to 730 ℃) displayed an evolution in sample mass from 80% to 43.1% for the remnant. This represents a mass loss of 53.8% (43.1/80 = 53.8%) based on the weight of the remnant at 290 ℃,which represents exactly the theoretical mass loss (of 53.80%) for the monohydrazine complex to become NiO.
For the sample NiBC-GS250,it is believed that all the dihydrazine (Ni(N2H4)2Cl2) complex was transformed to monohydrazine (Ni(N2H4)Cl2) complex [37],and a considerable amount of NiCl2 was also formed after 4 h annealing at 250 ℃,as one can conclude also from XRD diffractograms shown in Fig. 2b (NiBCGS250). For the monohydrazine to become NiO (up to 730 ℃),the theoretical mass loss is 46.20% (remnant of 53.80%); for NiCl2 to NiO,the theoretical mass loss is 57.59% (remnant of 42.41%). The detected mass loss for the sample was 54.80%,which led to a portion of NiCl2 of 75.86% (monohydrazine of 24.14%). For the sample NiBC-GS350,there appears only one single mass loss, which was most likely attributed to the transformation of NiCl2 to NiO. The theoretical mass loss for this transformation is 37.2% (remnant of 62.8%). The low value (57.59%) for the remnant indicates that a low portion of the sample (~8.3%) was decomposed to NiO with 4 h annealing at 350 ℃,which was also supported by the XRD diffractogram of the sample NiBC-GS350 shown in Fig. 3b.
These data show clearly that the NiBC complexes,as prepared in this work,were a mixture of different components depending on the experimental conditions and the subsequent annealing also imposed an important effect on their structure and composition. The XRD test gives quite confirmative information about their composition and the evolution with temperature. TGA analysis also provides reliable information in good agreement with XRD test results. 3.3. Influence of annealing temperature on specific capacitance of active materials
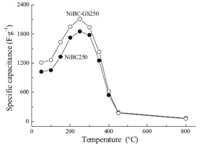
In order to find an optimal annealing temperature,the specific capacitances of the active materials NiBC-GS and NiBC were tested by galvanostatic charge/discharge process with the discharge current density of 1 A g-1 at the 10th cycle. In Fig. 5,the curve of specific capacitance as a function of annealing temperature for NiBC-GS and NiBC electrodes indicates that the specific capacitance of NiBC-GS apparently exceeded that of NiBC,due probably to the electronic conductive effect of graphene sheets dispersed uniformly in NiBC-GS. On the other hand,the specific capacitances of NiBC-GS and NiBC were increased to a maximum (2111 F g-1 for NiBC-GS and 1853 F g-1 for NiBC) and then followed by a decrease with further increase in annealing temperature. Namely,the samples annealed at 250 ℃ reached the highest specific capacitance for the corresponding electrode materials. And 250 ℃ was likely the optimal annealing temperature for both samples regardless of GS presence. Analogously,the NiOx xerogels,formed by the sol-gel method and the following heat-treatment process under ambient pressure,also exhibited the same trend; namely,its specific capacitance initially increased with the increase of calcination temperature from 110 to 250 ℃,and then reached a maximum value of 696 F g-1 at 250 ℃ and decreased as the temperature continuously increased [19]. Therefore,this annealing temperature for the samples (NiBC-GS250 and NiBC250) was used in the subsequent tests.
|
Download:
|
| Fig 5. Variation of specific capacitance of NiBC-GS250 and NiBC250 electrodes at a current density of 1 A g-1 with different annealing temperature. | |
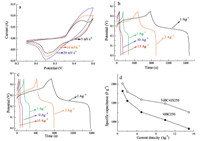
The electrochemical performance of the active materials prepared without GS (NiBC250) and with GS (NiBC-GS250),as the electrode materials for supercapacitors,was evaluated through CV and galvanostatic charge/discharge measurements in a threeelectrode system in 2 mol L-1 KOH aqueous solution. The results, shown in Fig. 6. Fig. 6a,presents typical CV curves of the NiBCGS250 electrode at scan rates of 5,10 and 20 mV s-1 in the potential range from -0.1 V to 0.6 V (vs. Ag/AgCl). A pair of broad redox peaks,located between 0.1 V and 0.5 V,was observed, indicating that the measured capacitance was mainly based on a redox mechanism. The symmetry of the peaks indicates high reaction reversibility [16]. With increased scan rate,clear shifts towards anodic and cathodic directions for the oxidation-reduction peaks were observed,likely owing to the fact that the effective ion transport interaction between the ions and the electrode is greatly reduced with increased scan rate [17, 32]. In practice,the ability to discharge at a high rate is crucial in capacitors. The galvanostatic charge-discharge of the samples NiBC-GS250 and NiBC250 was also characterized at constant current densities of 1, 2,5,10 and 15 A g-1 as shown in Fig. 6b and c. In Fig. 6b,a clear plateau between 0.20 V and 0.30 V is detected,the characteristic of pseudocapacitance resulted from the electrochemical adsorption/ desorption or redox reaction at an interface between electrode and electrolyte [17]. From Fig. 6b and c,it is also seen that the galvanostatic curve was more symmetrical with a lower current density,indicating a high reversibility of this redox reaction. Based on Eq. (1),the specific capacitances of the electrodes at different current densities were calculated,and the dependence of specific capacitance on current density is shown in Fig. 6d. Obviously,the specific capacitances decreased with the increase in the current density for both samples,which could be explained based on the pseudocapacitance mechanism and that part of the ions did not participate in the Faradiac redox reaction due to slower diffusion and electron transfer at higher current density [18].
|
Download:
|
| Fig 6. Electrochemical properties of NiBC-GS and NiBC electrode in 2 mol L-1 KOH solution: (a) CV curves of NiBC-GS250 electrode at different scan rates. (b) Charge- discharge curves of NiBC-GS250 at different current densities. (c) Charge-discharge curves of NiBC250 at different current densities. (d) Variation of specific capacitances of NiBC-GS250 and NiBC250 with different current densities electrode. | |
As reported by Wu et al. [42],the specific capacitance of the Ni(OH)2 electrode featuring the hollow nanotube arrays was 760 F g-1 at a high current density of 10 A g-1,corresponding to 57.2% of the value (1328 F g-1) at 1Ag-1. Moreover,Cheng et al. [19],reported that the specific capacitance of the NiOx xerogel treated at 250 ℃ was 479 F g-1 at the current density of 0.67 A g-1, which was about 68.8% of the value (696 F g-1) at the low current density of 0.067 A g-1. In our case,the specific capacitances of NiBC-GS250 and NiBC250 were 1588 F g-1 and 1238 F g-1 at the high current density of 10 A g-1,which was about 78.9% and 66.8% of the values (2011 F g-1 and 1853 F g-1) at the low current density of 1 A g-1,revealing a better rate performance than the above works. In addition,compared with 66.8% for NiBC250,the higher capacitance retention of 78.9% for NiBC-GS250 indicates a better performance of diffusion rate,making the GS-incorporated material,i.e.,NiBC-GS250,an attractive candidate for practical application.
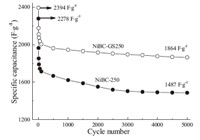
Data in Fig. 7 show the specific capacitance of NiBC-GS250 and NiBC250 as a function of cycle number at current density of 1 A g-1 in a voltage range of 0.0 V-0.45 V for up to 5000 cycles. For NiBCGS250, the initial specific capacitance was 2394 F g-1,and it was lowered to 1864 F g-1 after 5000 cycling times,i.e.,77% of its specific capacitance was retained; Whereas for NiBC250 electrode, only 65% of the specific capacitance remained through the same tests,which demonstrates an obvious higher cyclic stability for NiBC-GS250 electrode in comparison with NiBC250. In addition, the relative high values (1864 F g-1 and 1487 F g-1) of specific capacitance,for the two materials after 5000 cycles,suggest strongly that the synthesized NiBC-GS250 and NiBC250 powders are good electrode materials for supercapacitors. These results are much better than the reported values for the loose-packed nickel hydroxides [43],which exhibited a maximal specific capacitance of 2055 F g-1 in 2 mol L-1 aqueous KOH with the potential range of 0 to 0.4 V (vs. the saturated calomel electrode) at the discharging current of 0.62 A g-1; however,this value was greatly decreased to 641 F g-1 after 1500 cycles,i.e.,only 31.2% level was remained. In a word,considering the high specific capacitance,good rate retention,and long cyclic life,the as-prepared NiBC-GS250 or NiBC250 composites represent good candidates as novel electrode materials for supercapacitors.
|
Download:
|
| Fig 7. Cyclic stability of NiBC-GS250 and NiBC250 electrode tested using the galvanostatic charge-discharge with current density of 1 A g-1. | |
A series of NiBC-GS composites have been prepared by a simple flocculation from a solution of intercalated nickel dichloride- graphene oxide in DMF by addition of hydrazine hydrate,followed by a subsequent annealing process. The typical FESEM images of NiBC-GS demonstrated that the GS sheets were dispersed uniformly in a NiBC matrix. By adding a few GS sheets,the specific capacitance and the cyclic stability of the NiBC electrode materials can be greatly improved. In addition,through studying the specific capacitances of the NiBC-GS and NiBC electrodes with the variation of annealing temperature,it was found that the optimal annealing temperature was 250 ℃. After 5000 cycles,a comparison of the specific capacitance of NiBC-GS250 at 1864 F g-1 exceeded by 25% to that of NiBC250 at 1487 F g-1 at a current density of 1 A g-1,and the specific capacitance retentions of NiBC-GS250 and NiBC250 were 77% and 65%,respectively. In a word,the NiBC-GS250 composite is a good candidate as electrode material for supercapacitors. The specific capacitance and the cyclic stability of this material may be enhanced through addition of a second transition metal,cobalt for example. This will be our next research subject.
AcknowledgmentThis study has been financially supported by National Natural Science Foundation of China (No. 51075384).
| [1] | M. Winter, R.J. Brodd, What are batteries, fuel cells, and supercapacitors, Chem. Rev. 104 (2004) 4245-4269. |
| [2] | L.L. Zhang, X.S. Zhao, Carbon-based materials as supercapacitor electrodes, Chem. Soc. Rev. 38 (2009) 2520-2531. |
| [3] | M.X. Liu, L. Gan, Y. Li, et al., Synthesis and electrochemical performance of hierarchical porous carbons with 3D open-cell structure based on nanosilicaembedded emulsion-templated polymerization, Chin. Chem. Lett. 25 (2014) 897- 901. |
| [4] | Y. Xiao, C. Long, M.T. Zheng, et al., High-capacity porous carbons prepared by KOH activation of activated carbon for supercapacitors, Chin. Chem. Lett. 25 (2014) 865-868. |
| [5] | H.M. Zhang, X.H. Wang, Eco-friendly water-borne conducting polyaniline, Chin. J. Polym. Sci. 31 (2013) 853-869. |
| [6] | G.P. Wang, L. Zhang, J.J. Zhang, A review of electrode materials for electrochemical supercapacitors, Chem. Soc. Rev. 41 (2012) 797-828. |
| [7] | L. Kang, S.X. Sun, L.B. Kong, J.W. Lang, Y.C. Luo, Investigating metal-organic framework as a new pseudo-capacitive material for supercapacitors, Chin. Chem. Lett. 25 (2014) 957-961. |
| [8] | P. Miró,M. Audiffred, T. Heine, An atlas of two-dimensional materials, Chem. Soc. Rev. 43 (2014) 6537-6554. |
| [9] | Y. Wang, Z.Q. Shi, Y. Huang, et al., Supercapacitor devices based on graphene materials, J. Phys. Chem. C 113 (2009) 13103-13107. |
| [10] | H.M. Sun, L.Y. Cao, L.H. Lu, Bacteria promoted hierarchical carbon materials for high-performance supercapacitor, Energy Environ. Sci. 5 (2012) 6206-6213. |
| [11] | J.T. Zhang, J.W. Jiang, H.L. Li, X.S. Zhao, A high-performance asymmetric supercapacitor fabricated with graphene-based electrodes, Energy Environ. Sci. 4 (2011) 4009-4015. |
| [12] | C.X. Guo, C.M. Li, A self-assembled hierarchical nanostructure comprising carbon spheres and graphene nanosheets for enhanced supercapacitor performance, Energy Environ. Sci. 4 (2011) 4504-4507. |
| [13] | B.H. Kim, K.S. Yang, H.G. Woo, Boron-nitrogen functional groups on porous nanocarbon fibers for electrochemical supercapacitors, Mater. Lett. 93 (2013) 190-193. |
| [14] | W.F. Wei, X.W. Cui, W.X. Chen, D.G. Ivey, Manganese oxide-based materials as electrochemical supercapacitor electrodes, Chem. Soc. Rev. 40 (2011) 1697-1721. |
| [15] | V. Khomenko, E. Raymundo-Pinero, F. Beguin, Optimisation of an asymmetric manganese oxide/activated carbon capacitor working at 2 V in aqueous medium, J. Power Sources 153 (2006) 183-190. |
| [16] | F.P. Zhao, Y.Y. Wang, X.N. Xu, et al., Cobalt hexacyanoferrate nanoparticles as a high-rate and ultra-stable supercapacitor electrode material, ACS Appl. Mater. Interf. 6 (2014) 11007-11012. |
| [17] | L.B. Kong, J.W. Lang, M. Liu, Y.C. Luo, L. Kang, Facile approach to prepare loosepacked cobalt hydroxide nano-flakes materials for electrochemical capacitors, J. Power Sources 194 (2009) 1194-1201. |
| [18] | H. Jiang, T. Zhao, C.Z. Li, J. Ma, Hierarchical self-assembly of ultrathin nickel hydroxide nanoflakes for high-performance supercapacitors, J. Mater. Chem. 21 (2011) 3818-3823. |
| [19] | J. Cheng, G.P. Cao, Y.S. Yang, Characterization of sol-gel-derived NiOx xerogels as supercapacitors, J. Power Sources 159 (2006) 734-741. |
| [20] | J. Chang, J. Sun, C.H. Xu, H. Xu, L. Gao, Template-free approach to synthesize hierarchical porous nickel cobalt oxides for supercapacitors, Nanoscale 4 (2012) 6786-6791. |
| [21] | Y. Wang, I. Zhitomirsky, Electrophoretic deposition of manganese dioxide-multiwalled carbon nanotube composites for electrochemical supercapacitors, Langmuir 25 (2009) 9684-9689. |
| [22] | H. Chen, S.X. Zhou, L.M. Wu, Porous nickel hydroxide-manganese dioxide-reduced graphene oxide ternary hybrid spheres as excellent supercapacitor electrode materials, ACS Appl. Mater. Interf. 6 (2014) 8621-8630. |
| [23] | Y. Huang, X.L. Huang, J.S. Lian, et al., Self-assembly of ultrathin porous NiO nanosheets/graphene hierarchical structure for high-capacity and high-rate lithium storage, J. Mater. Chem. 22 (2012) 2844-2847. |
| [24] | J. Yan, W. Sun, T. Wei, et al., Fabrication and electrochemical performances of hierarchical porous Ni(OH)2 nanoflakes anchored on graphene sheets, J. Mater. Chem. 22 (2012) 11494-11502. |
| [25] | Z.H. Tang, B.C. Guo, L.Q. Zhang, D.M. Jia, Graphene-rubber nanocomposites, Acta Polym. Sin. (7) (2014) 865-877. |
| [26] | X. Huang, X.Y. Qi, F. Boey, H. Zhang, Graphene-based composites, Chem. Soc. Rev. 41 (2012) 666-686. |
| [27] | H.W. Wang, Z.A. Hu, Y.Q. Chang, et al., Design and synthesis of NiCo2O4-reduced graphene oxide composites for high performance supercapacitors, J. Mater. Chem. 21 (2011) 10504-10511. |
| [28] | Y. Cao, Q.M. Su, R.C. Che, G.H. Du, B.S. Xu, One-step chemical vapor synthesis of Ni/graphene nanocomposites with excellent electromagnetic and electrocatalytic properties, Synth. Met. 162 (2012) 968-973. |
| [29] | S.B. Yang, X.L. Wu, C.L. Chen, et al., Spherical α-Ni(OH)2 nanoarchitecture grown on graphene as advanced electrochemical pseudocapacitor materials, Chem. Commun. 48 (2012) 2773-2775. |
| [30] | H.L. Wang, H.S. Casalongue, Y.Y. Liang, H.J. Dai, Ni(OH)2 nanoplates grown on graphene as advanced electrochemical pseudocapacitor materials, J. Am. Chem. Soc. 132 (2010) 7472-7477. |
| [31] | L.B. Zhang, J.Q. Wang, H.G. Wang, et al., Preparation, mechanical and thermal properties of functionalized graphene/polyimide nanocomposites, Compos., A: Appl. Sci. Manuf. 43 (2012) 1537-1545. |
| [32] | L.B. Zhang, J.Q. Wang, S.R. Yang, X.Z. Kong, Preparation and characterization of graphene sheet-polyimide nanocomposite films, Acta Polym. Sin. (2014) 1472- 1478. |
| [33] | J.W. Park, E.H. Chae, S.H. Kim, et al., Preparation of fine Ni powders from nickel hydrazine complex, Mater. Chem. Phys. 97 (2006) 371-378. |
| [34] | D. Nicholls, R. Swindells, Hydrazine complexes of nickel(II) chloride, J. Inorg. Nucl. Chem. 30 (1968) 2211-2217. |
| [35] | C. Furlani, G. Mattogno, A. Monaci, F. Tarli, Ligand field spectra of hydrazine complexes of Ni(II) and the spectrochemical position of hydrazine, Inorg. Chim. Acta 4 (1970) 187-191. |
| [36] | G.Y. Huang, S.M. Xu, G. Xu, L.Y. Li, L.F. Zhang, Preparation of fine nickel powders via reduction of nickel hydrazine complex precursors, Trans. Nonferrous Met. Soc. China 19 (2009) 389-393. |
| [37] | B. Banerjee, P.K. Biswas, N.R. Chaudhuri, Thermal studies of nickel(II) hydrazine complexes in solid state, Bull. Chem. Soc. Jpn. 56 (1983) 2509- 2517. |
| [38] | A. Leineweber, H. Jacobs, Preparation and crystal structures of Ni(NH3)2Cl2 and of two modifications of Ni(NH3)2Br2 and Ni(NH3)2I2, J. Solid State Chem. 152 (2000) 381-387. |
| [39] | K.S. Rejitha, S. Mathew, Thermal behaviour of nickel(II) sulphate, nitrate and halide complexes containing ammine and ethylenediamine as ligands, J. Therm. Anal. Calorim. 106 (2011) 267-275. |
| [40] | L. Guo, C.M. Liu, R.M. Wang, et al., Large-scale synthesis of uniform nanotubes of a nickel complex by a solution chemical route, J. Am. Chem. Soc. 126 (2004) 4530- 4531. |
| [41] | S. Kulaksizoğlu, C. Gökçe, R. Gup, Asymmetric bis(bidentate) azine ligand and transition metal complexes: synthesis, characterization, DNA-binding and cleavage studies and extraction properties for selected metals and dichromate anions, J. Chil. Chem. Soc. 57 (2012) 1213-1218. |
| [42] | M.S. Wu, K.C. Huang, Fabrication of nickel hydroxide electrodes with open-ended hexagonal nanotube arrays for high capacitance supercapacitors, Chem. Commun. 47 (2011) 12122-12124. |
| [43] | J.W. Lang, L.B. Kong, W.J. Wu, et al., A facile approach to the preparation of loosepacked Ni(OH)2 nanoflake materials for electrochemical capacitors, J. Solid State Electrochem. 13 (2009) 333-340. |




