The reactions with readily available and inexpensive alcohols as alkylating agents have been very attractive,because of the atom-economy [1] of the dehydration reactions. However,very few successful systems were reported due to the poor leaving ability of the hydroxyl group. Recently,some reactions of direct substitution of the hydroxyl groups in alcohols have been developed by Tian [2],Baba [3],Shibasaki [4],and others [5], with the nucleophilic agents such as N-,S-,and C-nucleophiles. As early as 1956,Wawzonek and Dufek disclosed the reaction of 9-fluorenol with 9-alkylidene fluorenes under acid catalysis to yield substituted alkenes [6]. But in the paper there were no other substrates except the reaction mentioned above. In 2008,a reaction of alcohols with alkenes was developed by Yamamoto and coworkers,catalyzed by Pd(II) with a high loading (3.0 equiv.) of Tf2O [7]. Angle’s group reported a [3 + 2] annulation reaction of benzylic carbocation with alkene,which was generated from benzylic alcohol with electron-donating groups [8a]. A similar reaction was reported by Lantaño et al.,but it also suffered from seriously limited substrate scopes [8b]. Liu group disclosed the coupling reaction between alcohols and alkenes using stoichiometric amount of FeCl3·6H2O and TsOH [9]. Herein,we wish to describe the synthesis of trisubstituted alkenes and indane derivatives from benzylic alcohols and alkenes. 2. Experimental
The 1H NMR and 13CNMR spectra were recorded on a Bruker AC-300 FT spectrometer (300 MHz and 75 MHz,respectively) or on a Bruker AC-400 FT (400 MHz and 100 MHz,respectively) using tetramethylsilane as an internal reference,and the chemical shifts (δ)and coupling constants (J) were expressed in ppm and Hz, respectively. The 2D NOESY spectra were recorded on a Bruker AC-400 FT spectrometer (400 MHz). The high resolution mass spectra were recorded on a LC-TOF spectrometer (Micromass). The melting points were uncorrected. The spectral data and spectra of all compounds can be found in Supporting information.
Typical procedure for the synthesis of trisubstituted alkenes and indane derivatives: To a stirred solution of alcohols 1 (0.20 mmol) in nitromethane (0.20 mL) were added successively alkenes2(0.24 mmol) and Tf2NH (11.2 mg,20 mol%). The resulting mixture was stirred at the specified temperature for a period of time under air atmosphere. After the reaction completed,which was monitored by TLC analysis,the reaction mixture was concentrated under vacuum,and the resultant crude mixture was purified by column chromatography on silica gel with petroleum ether to give the corresponding pure trisubstituted alkenes 3 and indane derivatives 4. 3. Results and discussion
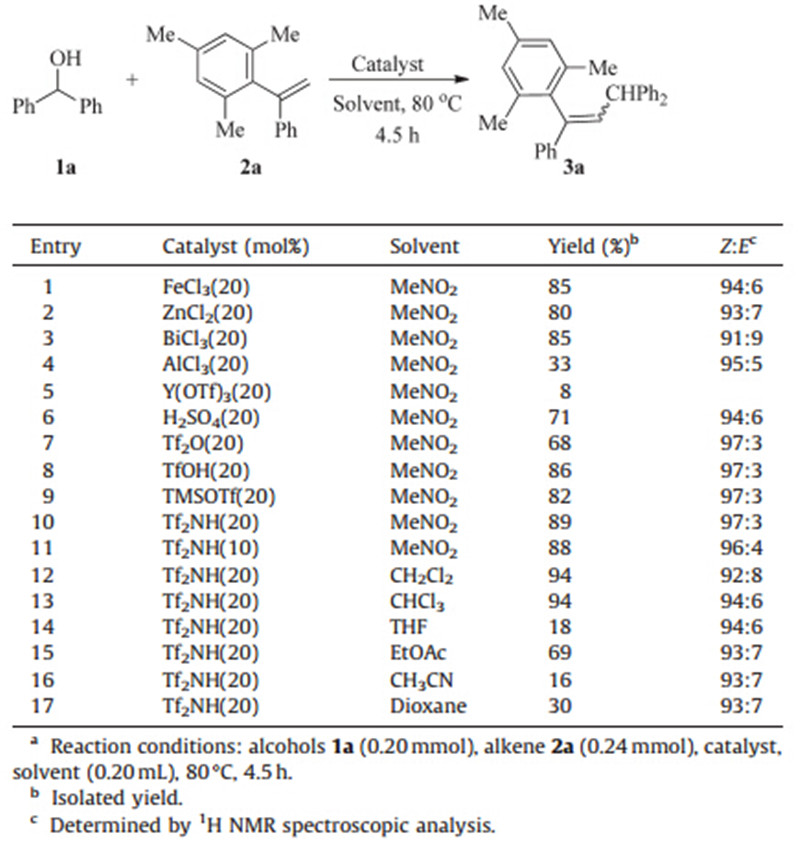
Initially,we chose benzhydrol 1a and 1,1-diarylethene 2a as model substrates to identify suitable conditions for this reaction (Table 1). Inspired by the recent work of Tian’s group [2a],the reaction was designed to be catalyzed by Lewis acid FeCl3 in nitromethane at 80℃. To our delight,the trisubstituted alkene was obtained in 85% yield and 94:6Z/Eselectivity (Table 1,entry 1). Then,several Lewis and Brønsted acids were evaluated as the catalyst (Table 1,entries 1-10),and it was found that Tf2NH was the best catalyst in this reaction with 89% yield and 97:3 Z/E selectivity (Table 1,entry 10). Reducing the catalyst loading to 10 mol% and replacing nitromethane with other organic solvents led to much lower yields and/or stereoselectivity (Table 1,entries 11-17).
| Table 1 Optimization of reaction conditions.a |
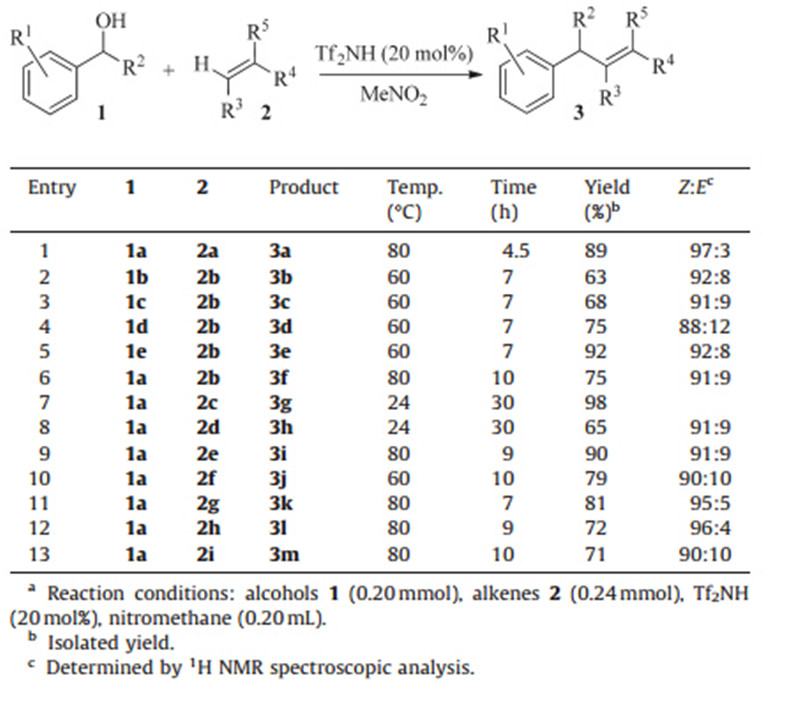
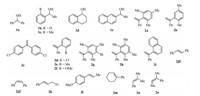
To examine the scope of this system,the reactions of benzylic alcohols with a range of alkenes were investigated,the structures of which are shown in Scheme 1. Since the reactivity of 2a is very high,we choose 2b as a suitable substrate to react with other benzylic alcohols. The reactions between benzylic alcohols and 2b proceeded well to give the trisubstituted alkenes in moderate to excellent yields andZselectivity (Table 2,entries 2-6). As can be seen,1,1-disubstituted alkenes with functional groups such as chloro,bromo,and MeO groups reacted well using this protocol to produce the corresponding trisubstituted alkenes (Table 2,entries 7-13). It is noteworthy that the present protocol also worked well for 2-(1-phenylvinyl)naphthalene 2i with benzhydrol to give the product 3m in 71% yield and with aZ/Eselectivity of 90:10 (Table 2, entry 13).
|
Download:
|
| Scheme 1.Structures of benzylic alcohols and alkenes. | |
| Table 2 Alkenylation of benzylic alcohols with alkenes.a |
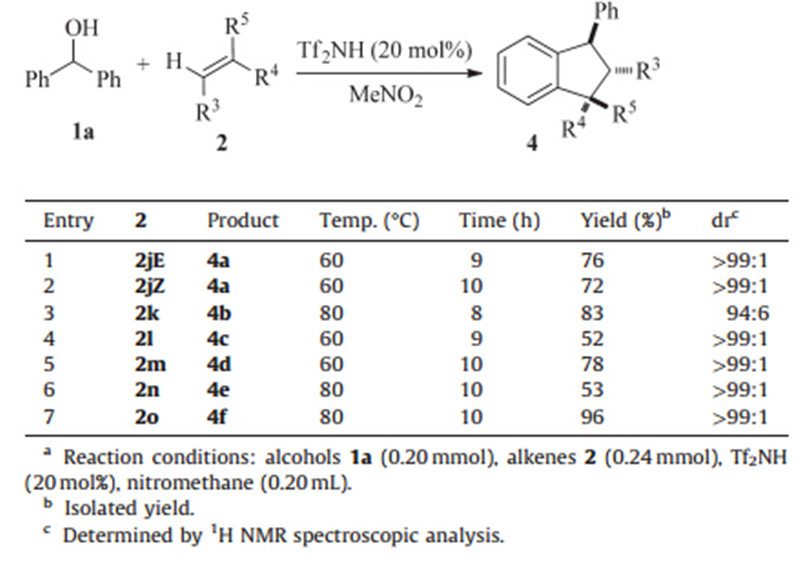
Subsequently,we investigated some other types of alkenes such as 1,2-disubstituted and trisubstituted aryl alkenes,and found that the major products were polysubstituted indanes instead of polysubstituted alkenes. Most of the reactions proceeded smoothly to give good to excellent yields with good region- and stereo-selectivity as summarized in Table 3. It is worth emphasizing that the use of eithertrans-stilbene or cis-stilbene gave the same indane product 4a with atrans/transconfiguration at the C1-C3 positions (Table 3,entries 1-2). In addition, trisubstituted alkene 2o (1:1 E/Z) reacted with benzhydrol resulting in an indane product as a singletrans/transdiastereomer. The relative stereochemistry of indane 4f was assigned by 2D NOSEY spectroscopy and comparing to the results of Tian’s group [10].
| Table 3 [3 + 2] Annulation of benzylic alcohols with alkenes.a |
According to the work of Tian’s group [10],a plausible mechanism for this reaction is depicted in Scheme 2. A carbocation intermediate5,generated from the benzylic alcohol under the acid condition by Tf2NH,is attacked by alkene2from the less sterically hindered face,which means the conformation A is more favorable than the conformation B,to give a new carbocation intermediate 6. When R3 is H,6 is prone to deprotonation to give trisubstituted alkene 3 with a thermodynamically stable configuration. On the other hand,when R3 is a non-hydrogen group it undergoes an intramolecular Friedel-Crafts reaction to give polysubstituted indane 4. The internal aromatic rings approached the carbocation away from R3 ,resulting in the observed relative stereochemistry of indane 4 with R5 being trans to R3 (it is assumed that R5 is a bulkier group than R4 in Scheme 2).
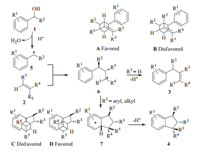
|
Download:
|
| Scheme 21.Proposed reaction pathways. | |
In summary,a reaction of benzylic alcohols with alkenes has been developed in the presence of Tf2NH for the synthesis of polysubstituted alkenes and indane derivatives with high regionand stereo-selectivity. In general,benzylic alcohols react with terminal alkenes to afford trisubstituted alkenes,and the reaction with 1,2-disubstituted and trisubstituted alkenes affords indane derivatives.
Appendix A. Supplementary dataSupplementary data associated with this article can be found,in the online version,at http://dx.doi.org/10.1016/j.cclet.2015.01.028
| [1] | (a) B.M. Trost, The atom economy -a search for synthetic efficiency, Science 254 (1991) 1471-1477; (b) B.M. Trost, On inventing reactions for atom economy, Acc. Chem. Res. 35 (2002) 695-705. |
| [2] | (a) H.H. Li, Y.H. Jin, J.Q. Wang, S.K. Tian, Controllable stereoselective synthesis of trisubstituted alkenes by a catalytic three-component reaction of terminal alkynes, benzylic alcohols, and simple arenes, Org. Biomol. Chem. 7 (2009) 3219-3221; (b) H.H. Li, D.J. Dong, Y.H. Jin, S.K. Tian, An expeditious entry to benzylic and allylic sulfones through byproduct-catalyzed reaction of alcohols with sulfinyl chlorides, J. Org. Chem. 74 (2009) 9501-9504; (c) H.H. Li, D.J. Dong, S.K. Tian, Three-component synthesis of amine derivatives using benzylic and allylic alcohols as N-alkylating agents in the absence of external catalysts and additives, Eur. J. Org. Chem. (2008) 3623-3626. |
| [3] | (a) T. Saito, Y. Nishimoto, M. Yasuda, A. Baba, Direct coupling reaction between alcohols and silyl compounds: enhancement of Lewis acidity of Me3SiBr using InCl3, J. Org. Chem. 71 (2006) 8516-8522; (b) Y. Nishimoto, Y. Onishi, M. Yasuda, A. Baba, a-Alkylation of carbonyl compounds by direct addition of alcohols to enol acetates, Angew. Chem. Int. Ed. 48 (2009) 9131-9134. |
| [4] | H. Qin, N. Yamagiwa, S. Matsunaga, M. Shibasaki, Bismuth-catalyzed direct substitution of the hydroxy group in alcohols with sulfonamides, carbamates, and carboxamides, Angew. Chem. Int. Ed. 46 (2007) 409-413. |
| [5] | (a) Z.P. Zhan, J.L. Yu, H.J. Liu, et al., A general and efficient FeCl3-catalyzed nucleophilic substitution of propargylic alcohols, J. Org. Chem. 71 (2006) 8298-8301; (b) S. Shirakawa, S.Kobayashi, Surfactant-type Brønsted acid catalyzed dehydrative nucleophilic substitutions of alcohols in water, Org. Lett. 9 (2007) 311-314. |
| [6] | S. Wawzonek, E. Dufek, The acid-catalyzed reaction of 9-fluorenol with 9-alkylidenefluorenes, J. Am. Chem. Soc. 78 (1956) 3530-3533. |
| [7] | H. Narahashi, I. Shimizu, A. Yamamoto, Synthesis of benzylpalladium complexes through C-O bond cleavage of benzylic carboxylates: development of a novel palladium-catalyzedbenzylationofolefins, J.Organomet.Chem.693 (2008)283-296. |
| [8] | (a) S.R. Angle, D.O. Arnaiz, Formal [3 + 2] cycloaddition of benzylic cations with alkenes, J. Org. Chem. 57 (1992) 5937-5947; (b) B. Lantaño, J.M. Aguirre, E.A. Ugliarolo, M.L. Benegas, G.Y. Moltrasio, Scope of the formal [3 + 2] cycloaddition for the synthesis of substituted 3-arylindanes and related compounds, Tetrahedron 64 (2008) 4090-4102. |
| [9] | Z.Q. Liu, Y.X. Zhang, L.X. Zhao, et al., Iron-catalyzed stereospecific olefin synthesis by direct coupling of alcohols and alkenes with alcohols, Org. Lett. 13 (2011) 2208-2211. |
| [10] | H.H. Li, X. Zhang, Y.H. Jin, S.K. Tian, Brønsted acid-catalyzed regio-and stereoselective alkenylation and [3 + 2] annulation of N-benzylic sulfonamides with alkenes, Asian J. Org. Chem. 2 (2013) 290-293." |