b. No. 1 High School of Baoding, Baoding 071001, China
Benzoylurea insecticides belong to a class of powerful insect growth regulators whose activity is based on the interference with the synthesis of chitin in target pests and blocking the molting process [1, 2]. After several decades of development,these insecticides are still widely used in agriculture [3]. However,the high employment of these insecticides has led their residues to accumulate in soil,groundwater,and surface water [4]. Because benzoylureas can cause negative effects on human health,it is desirable to develop a simple,rapid,and sensitive analytical method for monitoring these compounds in water samples.
Since benzoylureas are thermally unstable,it is unfeasible to determine them by gas chromatography (GC) unless they are derived into thermally stable derivatives [5]. The most frequently used methods for the determination of these benzoylureas in some matrices are high performance liquid chromatography (HPLC) with ultraviolet detection [6, 7],fluorescence detection (FLD) [8],and mass spectrometry (MS) [9]. Sample pretreatment procedures are often required prior to instrumental analysis and play a vital role in the whole analytical process because most of the insecticides exist at trace levels in the environmental samples. The traditional pretreatment techniques are mainly solid-phase extraction (SPE) [7, 8, 10] and liquid-liquid extraction (LLE) [7, 11],but SPE is tedious,time-consuming and relatively expensive,and LLE requires large amounts of harmful organic solvents. Recently, several novel sample pretreatment techniques,such as solid-phase microextraction (SPME) [12] or liquid-phase microextraction (LPME) [13],have been developed for the extraction and enrichment of the benzoylureas from different samples.
Recently,thin film microextraction (TFME),a new geometry for solid-phase microextraction,was introduced by Pawliszyn et al. [14]. In TFME,a thin and wide membrane is employed as a sorbent for the microextraction procedure. TFME has higher extraction efficiency and load ability due to the larger volume of its extraction phase and its higher surface-to-volume ratio. The thin films of polydimethylsiloxane membrane [15],chemically modified cellulose paper [16],polystyrene-divinylbenzene,polyacrylonitrile- phenylboronic acid [17],and octadecyl-silica glass [18] have been used as the extracting phase in TFME.
As one kind of hydrophobic membrane material,poly(vinyldene fluoride) (PVDF) has been received a great deal of attention owing to its good thermal stability,well-controlled porosity, excellent chemical resistance,and outstanding mechanical properties [19, 20]. PVDF has been applied in microfiltration,ultrafiltration, and nanofiltration for general separation purposes [21] and has also been explored as a potential candidate in the applications of membrane contactor and membrane distillation [22, 23]. Further,recent research has demonstrated the high adsorption capacity of PVDF membranes. Salehi et al. [24] applied 8- hydroxyquinoline modified PVDF membranes for removing nickel and cadmium ions from water samples. In consideration of the strong absorbent ability of PVDF,it is believed that PVDF would have good performance when used as a film absorber to extract organic pollutants from different samples. What is more,the PVDF is disposable due to its cheapness,so it completely avoids the sample carryover problem. To the best of our knowledge,it is not yet reported in the literature to use PVDF membrane as a film absorber in TFME to extract organic pollutants.
In continuation of our interest in sample preparation [25, 26], herein,PVDF membrane based thin film microextraction (PVDFTFME) was developed for the first time and used for the extraction of benzoylurea insecticides from water samples,followed by their determination with high performance liquid chromatographydiode array detection (HPLC-DAD). 2. Experimental
A stock solution containing diflubenzuron,triflumuron,hexaflumuron, and teflubenzuron each at 10.0 μg/mL was prepared in methanol. A series of standard solutions were prepared by mixing an appropriate amount of the stock solution with methanol in a 10- mL volumetric flask. All the standard solutions were stored at 4 ℃ and protected from light. The PVDF membranes of 0.22 μm pore size were purchased from Shanghai Xingya Purification Material Factory (Shanghai,China). River and lake water was obtained from Baoding (Baoding,China). Tap water was from Key Laboratory of Bioinorganic Chemistry (Baoding,China).
The HPLC system,assembled from modular components (Waters,Milford,MA,USA),consisted of an in-line degasser,a 600E pump,and a diode array detection (DAD) system. A Millennium 32 workstation (Waters) was utilized to control the system and for the acquisition and analysis of the data. The injection loop volume was 20.0 mL. A Centurysil C18-BDS column (250 mm × 4.6 mm,I.D.,5.0 μm) from Dalian Johnsson Separation Science Technology Corporation (Dalian,China) was used for separations. The mobile phase was a mixture of methanol-water (85:15,v/v) at a flow rate of 1.0 mL/min. DAD monitoring wavelengths were chosen at 254 nm. The identification of the four benzoylurea insecticides was made by both their retention times and ultraviolet absorption spectra by DAD detection. The peak area of each analyte was used for quantification.
The membrane extraction procedure was carried out as follows: A piece of PVDF membrane with an area of 3.75 cm2 (1.5 cm × 2.5 cm) was washed with acetone under ultrasound for 2 min in order to remove the possible contaminants and then dried in air. Then,the PVDF membrane was put into a 30 mL screw capped glass vial containing 20.0 mL of the sample solution,1.0 g NaCl,and a magnetic stir bar. The sample was stirred for 30 min by means of a magnetic stirrer at 800 rpm. After extraction,the PVDF membrane was taken out from the vial carefully and transferred into a 0.5 mL plastic micro-vial. Then,100.0 μL of acetonitrile was added to the micro-vial and vigorously vortexed for 2 min to desorb the analytes from the PVDF membrane. Finally,an aliquot of 20.0 μL of the resultant desorption solution was injected into the HPLC system for analysis. 3. Results and discussion
In the coated fiber SPME,the volume of the extraction phase is limited by the size of the external needle. Thin-film microextraction, a new geometry for solid-phase microextraction,has become an attractive sample-preparation technique [14]. In TFME,a sheet of flat film with a high surface area-to-volume ratio is used as the extraction phase. In general,an extraction phase with a large surface area accelerates the extraction rate because the initial extraction rate is proportional to the surface area of the extraction phase. Therefore,a thin film with a large surface area-to-volume ratio results in enhancement of the extraction efficiency without sacrificing the extraction time.
In this paper,PVDF membrane is selected as the extraction phase of TFME for the first time. Fig. 1 shows the image of the PVDF-TFME. The thin film microextraction,similar to SPME and LLE in some ways,is a process of mass transfer based on partition equilibrium of the analytes between the sample solution and the membrane. The interaction force between the PVDF membrane and the target analytes might include hydrophobic interactions, dipolar interactions,hydrogen bonding,and so on. Compared to other microextraction approaches,the sensitivity of this technique might be enhanced without sacrificing the sampling time due to the high surface area-to-volume ratio together with the increase of extraction-phase volume.

|
Download:
|
| Fig. 1.The image of PVDF membrane microextraction. | |
In this experiment,in order to obtain the best extraction efficiency,a series of 20.0 mL sample solutions spiked with 50.0 ng/mL each of the four benzoylurea insecticides were used to optimize several experimental parameters that could influence the extraction efficiency. All the experiments were performed in triplicate and the means of the results were used for data analysis.
The extraction time is an important parameter for the extraction. In the present experiment,extraction time was investigated by monitoring the peak areas corresponding to extraction times from 5 min to 40 min. As shown in Fig. 2A,the peak areas increased with increasing extraction time from 5 min to 30 min,and remained almost unchanged when the extraction time was longer than 30 min. Based on the results,30 min was selected as the extraction time.

|
Download:
|
| Fig. 2.Effect of extraction conditions on PVDF membrane extraction efficiency. (A) Effect of the extraction time. (B) Effect of the stirring rate. (C) Effect of the sample solution pH. (D) Effect of the ionic strength. | |
The extraction can be accelerated by stirring the sample solution. So sample agitation is another important parameter affecting extraction efficiency. Different stirring rates from 200 rpm to 1000 rpm were investigated and the result was shown in Fig. 2B. According to Fig. 2B,the peak areas increased correspondingly with increasing the stirring rate from 200 rpm to 800 rpm,and thereafter remained almost constant. Therefore, 800 rpm was selected as the optimum stirring rate in the subsequent experiments.
The pH of the sample solution plays an important role in efficient extraction because the existing forms (neutral or ionic form) of the analytes could be changed by changing the pH of the sample solution. Therefore,the pH of the sample solution should be adjusted to guarantee that the analytes exist in the proper molecular state so that they can be efficiently adsorbed. In this work,the effect of sample solution pH on extraction efficiency was studied in the range from 2.0 to 9.0. Fig. 2C shows that the peak areas of the four benzoylurea insecticides increased slowly in the pH range from 2.0 to 6.0 and then significantly decreased. Generally,the amide bonds in the benzoylurea were more easily degraded in alkali conditions. The four benzoylurea insecticides could be ionizable at pHs higher than their corresponding pKa values,which were in the range from 8.16 to 9.79,so the pH of the sample solution was adjusted to 6 for the extraction.
Generally,the addition of NaCl in the sample solution decreases the solubility of the analytes,thereby enhancing the extraction efficiency. Different concentrations of NaCl (0-20%,w/v) were used to evaluate the influence of ionic strength on the extraction efficiency. The results (Fig. 2D) revealed that an increase in extraction efficiencies for all the analytes was achieved when the concentration of NaCl was increased up to 5%,and the extraction efficiencies decreased when the concentration of NaCl was higher than 5%. This may be due to high concentrations of NaCl increasing the viscosity of the solution,which may retard the mass-transfer process and reduce the diffusion coefficient. In this case,the extraction was performed with the addition of NaCl at 5%.
Complete desorption of the analytes from the PVDF membrane would improve the detection sensitivity. In this work,different organic solvents (methanol,acetonitrile and acetone) were tested to desorb the analytes from the PVDF membrane. The results are shown in Fig. 3. Among the three organic solvents,acetonitrile provided the best desorption results. Thus,acetonitrile was selected as the desorption solvent.

|
Download:
|
| Fig. 3.Effect of the type of desorption solvent on the extraction efficiency. | |
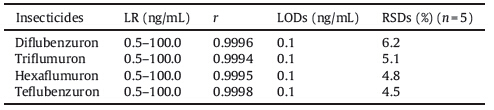
In order to evaluate the analytical performance of the proposed method,quality factors including linear range (LR),limits of detection (LODs),and repeatability were investigated under the above-mentioned optimal conditions. The linearity of the method was obtained using a series of standard solutions containing each of the four benzoylurea insecticides at seven concentration levels of 0.1,0.5,1.0,5.0,10.0,50.0 and 100.0 ng/mL,respectively. Five replicate extractions and determinations were performed for each concentration,and the results are summarized in Table 1. Good linearity was achieved for all the analytes over the studied concentration ranging from 0.5 to 100.0 ng/mL with correlation coefficient (r) higher than 0.9994. Based on S/N = 3,the limits of detection (LODs) of the four benzoylurea insecticides were 0.1 ng/mL. The relative standard deviations (RSDs,n = 5) were lower than 6.2% (spiked level of 10.0 ng/mL). These results indicated that the method had good performance for the extraction of benzoylurea insecticides.
| Table 1 The linear ranges (LR),correlation coefficients (r),limits of detection (LODs) and relative standard deviations (RSDs) of the method. |
To evaluate the applicability of the proposed method,three types of water samples (lake,river,and tap water) were subjected to the PVDF membrane based thin film microextraction procedure. The results showed that no residues of the four benzoylurea insecticides were found in tap and lake water,but triflumuron was found at 1.06 ng/mL in river water. Fig. 4 shows the typical chromatograms of the extracted analytes from river water sample before and after being spiked at 6.0 ng/mL with each of the four benzoylurea insecticides.

|
Download:
|
| Fig. 4.The typical chromatograms for river water sample (A) and the river water sample spiked with each of the four benzoylurea insecticides at 6.0 ng/mL. (B) Peak identification: (1) diflubenzuron,(2) triflumuron,(3) hexaflumuron,(4) teflubenzuron. | |
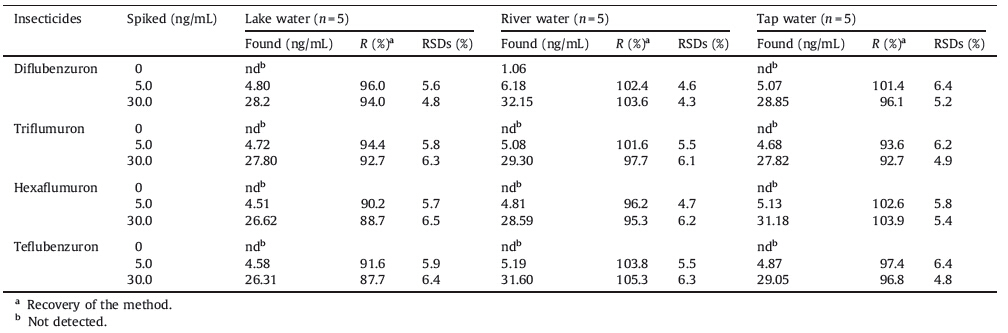
For the recovery experiment,water samples were spiked with standards of the four benzoylurea insecticides at concentrations of 5.0 and 30.0 ng/mL,respectively. The results are summarized in Table 2. As can be seen,relative recoveries were between 87.7% and 103.9%,and RSDs varied from 4.3% to 6.5%. These results proved that the present method is feasible for the determination of phenylurea pesticides in water samples.
| Table 2 Recoveries obtained for the determination of the four benzoylurea insecticides in spiked water samples. |
To highlight the extraction efficiency of PVDF-TFME method,a comparison of the important features (i.e. linearity,LOD and RSD) of the proposed method with other sample preparation methods was made. As shown in Table 3,the current PVDF-TFME method has comparable or even better LODs and reproducibility compared with other reported methods. In addition,the PVDF membrane is disposable due to its cheapness,so it can completely avoid the possible carry-over effect. In conclusion,the current method proved to be efficient and sensitive for the analysis of the benzoylurea insecticides in environmental water samples.
| Table 3 Comparison of PVDF-TFME method with other sample preparation methods for the determination of the benzoylurea insecticides. |
In the present work,a PVDF membrane based thin film microextraction method was developed and used to extract benzoylurea insecticides from water samples followed by determination with HPLC-DAD. Under the optimum conditions, outstanding performances such as good linearity,accuracy, repeatability,and low LODs were observed. What is more,the procedure is low cost and easy to operate. Therefore,the developed method is expected to open new avenues for the enrichment of organic pollutants in environmental samples.
AcknowledgmentsFinancial supports from the National Natural Science Foundation of China (No. 31171698),the Innovation Research Program of Department of Education of Hebei for Hebei Provincial Universities (No. LJRC009),the Scientific and Technological Research Foundation of Department of Education of Hebei Province (No. ZD20131033),and the Natural Science Foundation of Hebei (No. B2012204028) are gratefully acknowledged.
| [1] | J.K. Zhou, R.Y. Liu, G. Song, M.C. Zhang, Determination of carbamate and benzoylurea insecticides in peach juice drink by floated organic drop microextractionhigh performance liquid chromatography, Anal. Lett. 42 (2009) 1805-1819. |
| [2] | F. Matsumura, Studies on the action mechanism of benzoylurea insecticides to inhibit the process of chitin synthesis in insects: a review on the status of research activities in the past, the present and the future prospects, Pestic. Biochem. Physiol. 97 (2010) 133-139. |
| [3] | A.C. Gerecke, S. Canonica, S.R. Mü ller, M. Schä rer, R.P. Schwarzenbach, Quantification of dissolved natural organic matter (DOM) mediated phototransformation of phenylurea herbicides in lakes, Environ. Sci. Technol. 35 (2001) 3915-3923. |
| [4] | G. García, M. Martínez Galera, D. Barranco Martínez, J. Gisbert Gallego, Determination of benzoylureas in ground water samples by fully automated on-line preconcentration and liquid chromatography-fluorescence detection, J. Chromatogr. A 1103 (2006) 271-277. |
| [5] | J. Mensah, E. Lundanes, T. Greibrokk, B. Holen, Determination of diflubenzuron in apples by gas chromatography, J. Chromatogr. A 765 (1997) 85-90. |
| [6] | A. Valenzuela, R. Lorenzini, M. Redondo, G. Font, Matrix solid-phase dispersion microextraction and determination by high-performance liquid chromatography with UV detection of pesticide residues in citrus fruit, J. Chromatogr. A 839 (1999) 101-107. |
| [7] | G.E. Miliadis, N.G. Tsiropoulos, P.G. Aplada-Sarlis, High-performance liquid chromatographic determination of benzoylurea insecticides residues in grapes and wine using liquid and solid-phase extraction, J. Chromatogr. A 835 (1999) 113-120. |
| [8] | A.M. de la Pena, M. Mahedero, A. Bautista-Sánchez, Monitoring of phenylurea and propanil herbicides in river water by solid-phase-extraction high performance liquid chromatography with photoinduced-fluorimetric detection, Talanta 60 (2003) 279-285. |
| [9] | A.G. Frenich, M.G. García, F. Arrebola, et al., Determination of parts per trillion levels of benzoylurea pesticides in groundwater by high-performance liquid chromatography-electrospray ionization mass spectrometry, Chromatographia 52 (2000) 569-574. |
| [10] | R. Carabias-Martínez, E. Rodríguez-Gonzalo, E. Herrero-Herná ndez, J. Hernández-Mé ndez, Simultaneous determination of phenyl-and sulfonylurea herbicides in water by solid-phase extraction and liquid chromatography with UV diode array or mass spectrometric detection, Anal. Chim. Acta 517 (2004) 71-79. |
| [11] | R. Dommarco, A. Santilio, L. Fornarelli, M. Rubbiani, Simultaneous quantitative determination of thirteen urea pesticides at sub-ppb levels on a Zorbax SB-C18 column, J. Chromatogr. A 825 (1998) 200-204. |
| [12] | J.C. Wu, C. Tragas, H. Lord, J. Pawliszyn, Analysis of polar pesticides in water and wine samples by automated in-tube solid-phase microextraction coupled with high-performance liquid chromatography-mass spectrometry, J. Chromatogr. A 976 (2002) 357-367. |
| [13] | J.F. Zhang, Z. Liang, S. Li, et al., In-situ metathesis reaction combined with ultrasound-assisted ionic liquid dispersive liquid-liquid microextraction method for the determination of phenylurea pesticides in water samples, Talanta 98 (2012) 145-151. |
| [14] | R.F. Jiang, J. Pawliszyn, Thin-film microextraction offers another geometry for solid-phase microextraction, Trends Anal. Chem. 39 (2012) 245-253. |
| [15] | Z.P. Qin, L. Bragg, G.F. Ouyang, V.H. Niri, J. Pawliszyn, Solid-phase microextraction under controlled agitation conditions for rapid on-site sampling of organic pollutants in water, J. Chromatogr. A 1216 (2009) 6979-6985. |
| [16] | M. Saraji, B. Farajmand, Chemically modified cellulose paper as a thin film microextraction phase, J. Chromatogr. A 1314 (2013) 24-30. |
| [17] | F.S. Mirnaghi, J. Pawliszyn, Development of coatings for automated 96-blade solid phase microextraction-liquid chromatography-tandem mass spectrometry system, capable of extracting a wide polarity range of analytes from biological fluids, J. Chromatogr. A 1261 (2012) 91-98. |
| [18] | F.S. Mirnaghi, M.R.N. Monton, J. Pawliszyn, Thin-film octadecyl-silica glass coating for automated 96-blade solid-phase microextraction coupled with liquid chromatography-tandem mass spectrometry for analysis of benzodiazepines, J. Chromatogr. A 1246 (2012) 2-8. |
| [19] | S. Deshmukh, K. Li, Effect of ethanol composition in water coagulation bath on morphology of PVDF hollow fibre membranes, J. Membr. Sci. 150 (1998) 75-85. |
| [20] | Y. Chen, L. Ying, W. Yu, E. Kang, K. Neoh, Poly(vinylidene fluoride) with grafted poly(ethylene glycol) side chains via the RAFT-mediated process and pore size control of the copolymer membranes, Macromolecules 36 (2003) 9451-9457. |
| [21] | Y. Chang, Y.J. Shih, R.C. Ruaan, et al., Preparation of poly (vinylidene fluoride) microfiltration membrane with uniform surface-copolymerized poly (ethylene glycol) methacrylate and improvement of blood compatibility, J. Membr. Sci. 309 (2008) 165-174. |
| [22] | S.R. Chae, H. Yamamura, K. Ikeda, Y. Watanabe, Comparison of fouling characteristics of two different poly-vinylidene fluoride microfiltration membranes in a pilot-scale drinking water treatment system using pre-coagulation/sedimentation, sand filtration, and chlorination, Water Res. 42 (2008) 2029-2042. |
| [23] | B. Wu, K. Li, W.K. Teo, Preparation and characterization of poly (vinylidene fluoride) hollow fiber membranes for vacuum membrane distillation, J. Appl. Polym. Sci. 106 (2007) 1482-1495. |
| [24] | E. Salehi, S. Madaeni, F. Heidary, Dynamic adsorption of Ni (Ⅱ) and Cd (Ⅱ) ions from water using 8-hydroxyquinoline ligand immobilized PVDF membrane: isotherms, thermodynamics and kinetics, Sep. Purif. Technol. 94 (2012) 1-8. |
| [25] | X.F. Yin, Z. Li, S.H. Zhang, et al., Determination of strychnine and brucine in traditional Chinese medicine preparations by micelle to solvent stacking in capillary zone electrophoresis, Chin. Chem. Lett. 22 (2011) 330-333. |
| [26] | C.X. Wu, Q.H. Wu, C. Wang, Z. Wang, A novel method for the determination of trace copper in cereals by dispersive liquid-liquid microextraction based on solidification of floating organic drop coupled with flame atomic absorption spectrometry, Chin. Chem. Lett. 22 (2011) 473-476. |
| [27] | P.P. Vázquez, A.R. Mughari, M.M. Galera, Solid-phase microextraction for the determination of benzoylureas in orange juice using liquid chromatography combined with post-column photochemically induced fluorimetry derivatization and fluorescence detection, J. Sep. Sci. 31 (2008) 56-63. |
| [28] | L. Pareja, V. Cesio, H. Heinzen, A.R. Fernandez-Alba, Evaluation of various QuEChERS based methods for the analysis of herbicides and other commonly used pesticides in polished rice by LC-MS/MS, Talanta 83 (2011) 1613-1622. |
| [29] | J.H. Zhang, M. Li, M.Y. Yang, et al., Magnetic retrieval of ionic liquids: fast dispersive liquid-liquid microextraction for the determination of benzoylurea insecticides in environmental water samples, J. Chromatogr. A 1254 (2012) 23-29. |
| [30] | Q.X. Zhou, X.G. Zhang, Combination of ultrasound-assisted ionic liquid dispersive liquid-phase microextraction and high performance liquid chromatography for the sensitive determination of benzoylureas pesticides in environmental water samples, J. Sep. Sci. 33 (2010) 3734-3740. |